Abstract
We report the case of a 23-year-old woman who presented to our Psychiatry Unit with a complex psychiatric symptomatology, 6 years after suffering from a form of encephalopathy which was retrospectively and hypothetically labeled as autoimmune limbic encephalitis. Over the years, several psychopharmacological therapies had been initiated, but none of them led to substantial remission of symptomatology. During the first visit, symptoms were characterized by dysphoric mood with suicidal ideation, anxiety, delusional thoughts. Self-harm and psychogenic seizures with daily frequency were also reported. A therapy with slow-release lithium sulfate, lurasidone, and lorazepam was prescribed. After 6 months of treatment, psychopathological manifestations significantly improved.
Introduction
Limbic encephalitis (LE) is an inflammation of the limbic area, particularly the hippocampus and amygdala, that could also involve the cerebellum and brainstem. It can occur across all ages and may show a subtle and heterogeneous presentation; therefore, its diagnosis is extremely difficult and can be delayed for weeks. 1
Depending on the etiology, LE can be broadly divided into 2 groups: paraneoplastic limbic encephalitis (PLE), when caused by cancer or tumor, and non-paraneoplastic limbic encephalitis (NPLE), not associated with cancer and typically caused by infections, autoimmunity, or other unknown conditions. Among NPLE, the autoimmune variant may be associated with a wide range of antibodies, for example, anti-NMDAR, anti-AMPAR, anti-LGI1, anti-Caspr2, anti-GABA-B, anti-GAD. 2 However, in a small proportion of cases (2 cases per 100 000 people), no specific antibodies are detected, making even more challenging the diagnosis, with a subsequent delay in initiating an appropriate therapy. 1
For a definite diagnosis of autoimmune LE, 4 diagnostic criteria have to be met: (1) Subacute onset of working memory deficits, seizures, or psychiatric symptoms suggesting the involvement of the limbic system; (2) Bilateral brain abnormalities on T2-weighted fluid-attenuated inversion recovery magnetic resonance imaging (MRI) restricted to the medial temporal lobes; (3) At least 1 between cerebrospinal fluid (CSF) pleocytosis or electroencephalography (EEG) with epileptic or slow-wave activity involving the temporal lobes; (4) Reasonable exclusion of alternative causes. 3
LE presents with subacute (days to a few weeks) progressive cognitive alterations, involving in particular short-term memory, and a decrease in the level of consciousness, often with fluctuations. Seizures and movement abnormalities may be also characteristics of LE. Psychiatric symptoms, such as irritability, anxiety, delusions, and catatonia are frequent, and their presence may cause a diagnostic delay.1,2 Nevertheless, up to date, literature has mainly focused on the acute psychiatric manifestations of LE, while little is known about the long-term outcome and the therapeutic response. In the present paper, we will discuss the management of the case of a young woman who manifested long-term psychiatric sequelae after suffering from a form of encephalopathy which was retrospectively identified as a probable autoimmune LE.
Case Presentation
In October 2019, a 23 years-old young woman presented to our outpatient service with a complex psychiatric symptomatology, characterized by mood alterations, anxiety, psychogenic non-epileptic seizures, self-directed aggressive behaviors, and delusional thinking.
Medical history
In May 2013, while staying in Russia for a study exchange, the patient suddenly developed a symptomatology characterized by nausea, vomit, and abdominal pain. After referring to the local hospital, she was found to have a mild fever (37.2°C) and a slight increase in white blood cells (5.7 × 109/L), for which she was diagnosed with appendicitis and underwent an appendectomy. After the surgical intervention, she suddenly developed an altered state of consciousness with a feverish rise (40°C) and generalized tonic-clonic seizures. The patient was thus admitted to the Intensive Care Unit (ICU) of the local hospital and treated with valproic acid 1000 mg twice daily. The electroencephalography (EEG) showed focality in the frontal region (Table 1). The brain computerized tomography (CT) did not show any significant alterations. The lumbar puncture detected a clear and colorless cerebrospinal fluid (CSF), with a protein concentration of 8.3 mg/dL.
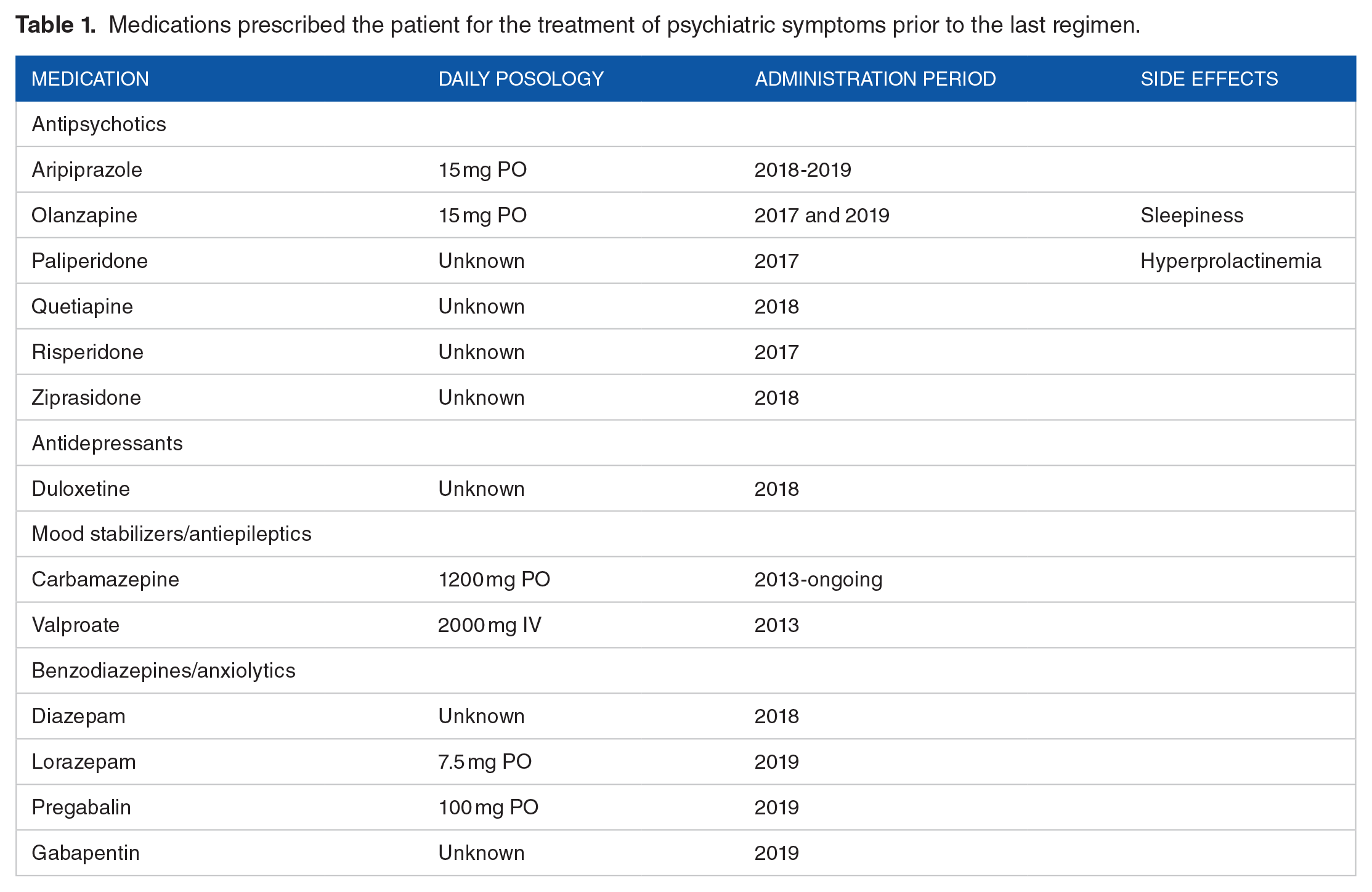
Medications prescribed the patient for the treatment of psychiatric symptoms prior to the last regimen.
After a few days, the young woman was transferred to the ICU of an Italian university hospital in a comatose state. At the admission, she was mechanically ventilated and presented with clonic movements of the right arm and tongue. Brain CT was performed in urgency and no significant lesions were detected. During the hospitalization, the patient was constantly monitored through EEG. EEG tracings are presented in Figure 1 (reports in Legend).
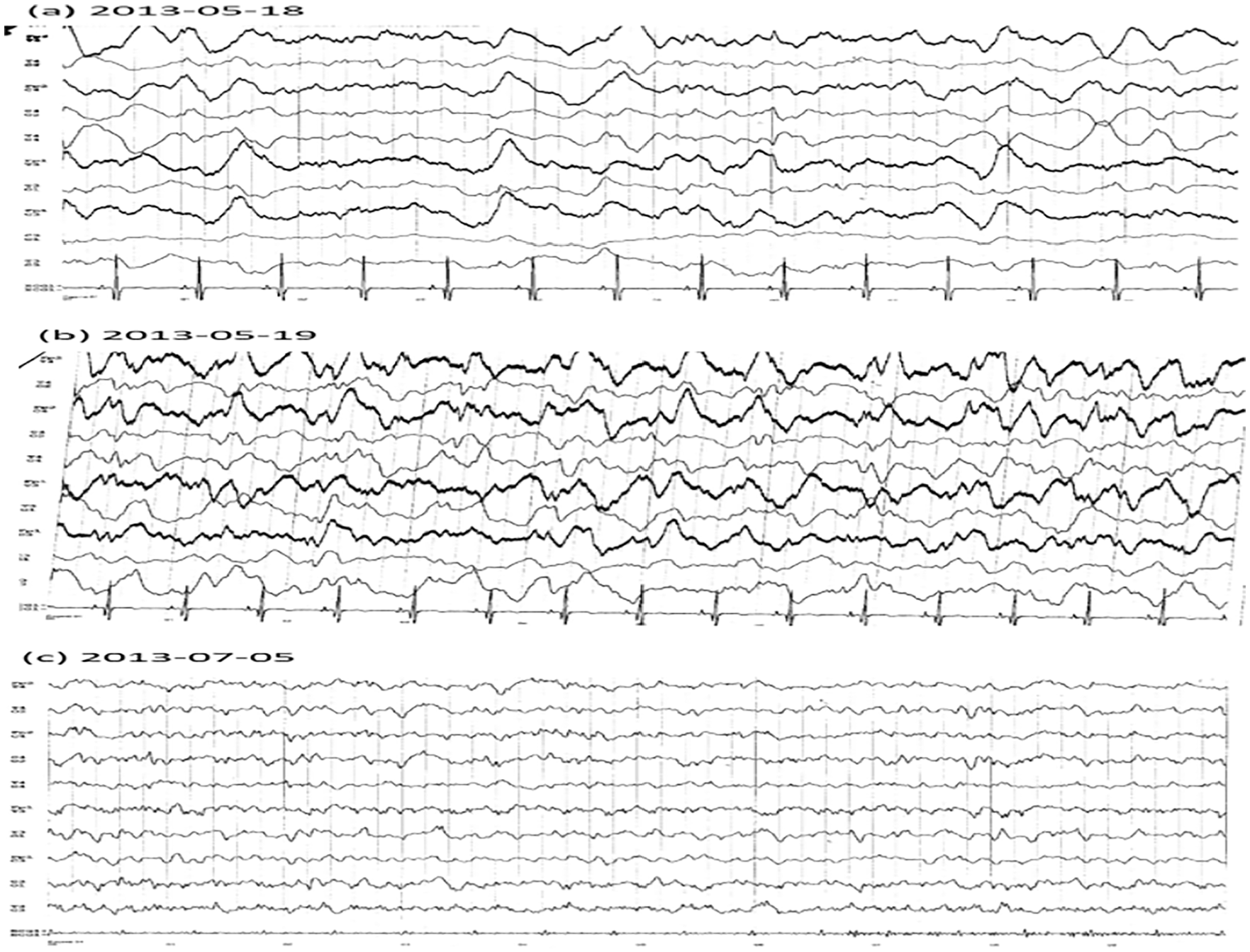
Electroencephalograms performed at the beginning (a, b) and at the end (c) of the hospitalization for suspected autoimmune limbic encephalitis. (a) Diffuse slow activity at 3 to 5 s and isolated slow spike-wave complex on the right derivation with contralateral diffusion. (b) Diffuse epileptic activity and myoclonic movements of face and lips. Incoming electrical critical episodes with asynchronous appearance on the 2 hemispheres. (c) Slow background activity in alpha band (8 Hz) on the posterior regions, slightly represented, symmetric. Presence of slow delta-theta activity more represented on the posterior regions, without clear epileptic significance.
Brain MRI was performed 3 and 24 days after the admission and at follow-ups. The first neuroimaging examination revealed an “enlarged hippocampi with hyperintense T2-weight signal. Similar cortical alterations are present in the posterior region of the insula, the external portion of putamina, and bilaterally in the subinsular white matter. In the aforementioned regions, the parenchyma shows diffusion-weighted imaging (DWI) hyperintensity, more evidence in correspondence with the putamina, and the adjacent white matter, where the apparent diffusion coefficient (ADC) maps show reduced diffusion values. Gradient echo sequence can exclude previous parenchymal hemorrhages. After contrast medium injection, no pathological enhancements or blood-brain barrier damages could be detected. The morphology and dimensions of the ventricular system and CSF spaces appear normal. No signs of intracranial hypertension. The MR angiography does not document any flow alteration in the circle of Willis. The results of the MRI cannot be univocally interpreted. The enlarged hippocampi are compatible with post-critical alterations. The involvement of the putamen and subinsular white matter may also be compatible with hypoxic-ischemic encephalopathy, even if an inflammatory hypothesis cannot be excluded.” Unfortunately, no images of brain scans are available.
Infections, bleeding, and tumors were excluded. Of note, the analysis of the cerebrospinal fluid showed a concentration of glucose of 82 mg/dl, proteins of 23 mg/dl, and no cells. Blood analysis was negative for the following antigens: Toxoplasma gondii, HIV, Rickettsia conorii, Borrelia burgdorferi, Salmonella, Mycoplasma pneumoniae, Chlamydia trachomatis, Legionella pneumophila, Mycobacteriaceae, Candida albicans, Streptococcus pneumoniae, Treponema pallidum. Also, blood culture was negative for both Gram+ and Gram- bacteria and fungi. Serological analysis reported negativity for the following antibodies: anti-LTG1, anti-AMPA, anti-NMDA, anti-GABA-B. Only anti-GAD antibodies were weakly positive, as they were detected at the concentration of 21 U/mL (normal range 0-5 mg/dL). Analyses in the CSF showed the absence of acid-fast bacteria, neurotropic virus, Cryptococcus, Cytomegalovirus, Epstein-Barr virus, Herpesvirus, Varicella-zoster virus, Rubivirus, and Borrelia burgdoferi, Unfortunately, specific antibodies were not searched in the CSF.
Given the clinical presentation, the neuroimaging, the typical EEG, and the substantially negative laboratory results, after excluding neoplastic and infectious etiologies, the medical staff hypothesized that the patient could be affected by an autoimmune LE. The woman was thus treated with immunoglobulin infusion and indeed clinical remission was obtained in 1 month, with consciousness full recovery. During the hospitalization, she presented with daily epileptic seizures for which she was prescribed the following medications: valproic acid/sodium valproate 400 mg IV 6 times per day and levetiracetam 1000 mg IV 3 times per day. Moreover, she was monitored and had to be sedated with propofol and intravenous midazolam during seizures. Once recovered from coma, she presented several cognitive deficits, such as aphasia, anomia, difficulties in text comprehension, and deficits in executive functions. The MRI reported a “disappearance of the encephalic alterations in the nucleobasal and temporo-mesial cortex, with residual punctiform cortical hyperintensity in T2-weighted images in the bilateral frontal regions. Reduction of hippocampi enlargements and resolution of signals alterations in T2-weighted images in correspondence of the anterior portion of hippocampal structures and linear alteration sin the nucleo-capsular portion between putamen and insula. Persistence of the focal areas of altered signal - hyperintense in the long repetition time (RT) sequences, without significant restriction of the protonic diffusion and without significant “contrast enhancement” after contrast medium injection - localized in the subcortical left regions of the posterior portion of fusiform gyrus and superior lingual gyrus. Another similar alteration is now present in the subcortical regions of the left medium occipital gyrus, in absence of blood-brain barrier discontinuations or significant enlargements of the involved convolutions. No signs of hemorrhages. The morphology and dimensions of the ventricular system and CSF spaces appear normal. No signs of intracranial hypertension.” The EEG was partially, but not completely, normalized (see Figure 1).
After being discharged from the hospital (July 2013), the patient spent 6 months in a Rehabilitation Unit for motor and cognitive functional recovery. Moreover, she continued to be regularly followed by the Neurology Unit, taking antiepileptic drugs, such as carbamazepine and phenobarbital, with progressive remission of epileptic seizures. Of note, even if some attentional deficits and working memory difficulties still remains, the patient has successfully completed an academic course and recently graduated.
Psychiatric history
In 2014, the patient first started to develop symptomatology characterized by anxiety and depression. Cognitive-behavioral therapy (CBT) led to partial benefits on clinical symptomatology, but in 2016 more severe symptoms appeared. Notably, parents referred to a progressive “change of personality” with bizarre thoughts, irritability, and self-harm. Medical reports described “psychotic functioning with psychomotor agitation” and “psychogenic seizures” since 2016. The epileptic etiology of seizures was excluded after several observations by both neurologists and psychiatrists. Several tentative psychopharmacological therapies were initiated with antipsychotics at different dosages (see Table 1 for details), but benefits were modest, and a complete remission could never be obtained.
Psychiatric assessment and management of symptomatology
Six years after the clinical recovery from the suspected LE (October 2019), the patient requested an appointment in the outpatient service of our Psychiatry Unit. During the psychiatric examination, she appeared lucid, fairly oriented in time, space and person. The speech was confused, disorganized and poor in content. Several delusional thoughts, such as “fear of red color and crucifixes,” were expressed during the clinical interview. The mood was dysphoric and characterized by feelings of sadness and hopelessness. Moderate anxiety and psychomotor restlessness could be observed. Suicidal ideation was reported. Parents also referred to some episodes of self-directed aggression. According to patient and parents’ accounts, she was suffering from non-epileptic psychogenic seizures daily, with manifestations similar to tonic-clonic seizures. However, as previously mentioned, the organic origin of these seizures had been excluded by several clinical observations and by EEG tracings. Moreover, neurological symptoms indicative of a possible relapse of encephalitis, such as fever, alterations of the state of consciousness, and frank cognitive deficits, were not present. Notably, at the moment of the visit, the woman was taking carbamazepine 300 mg PO 3 times per day as maintenance therapy for epilepsy, and lorazepam 1 mg PO 3 times a day, prescribed by another psychiatrist a few weeks before the appointment.
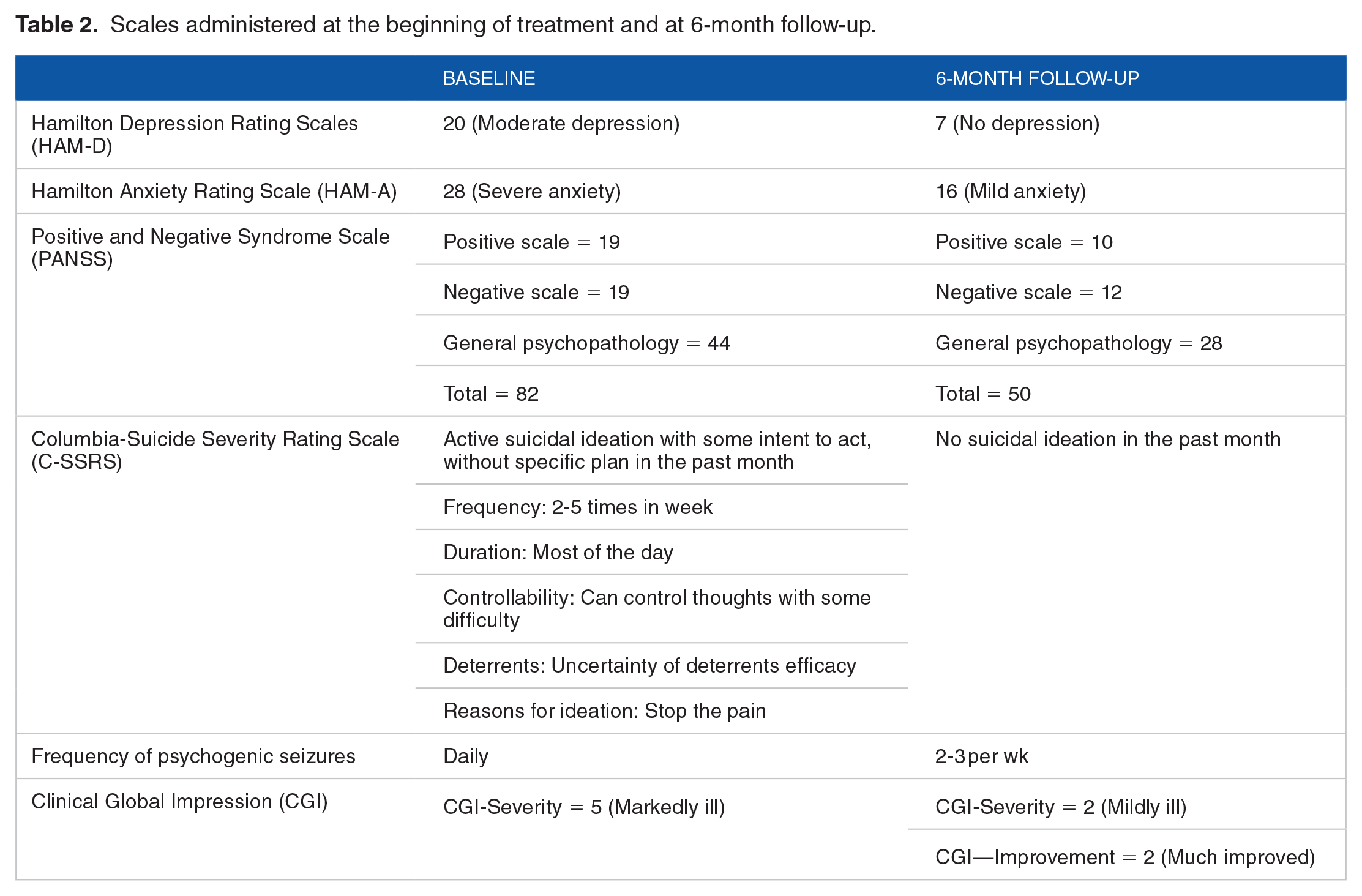
During the first visit, a clinician administered the following interviews: Hamilton Depression Rating Scale (HAM-D), 4 Hamilton Anxiety Rating Scale (HAM-A), 5 Positive and Negative Syndrome Scale (PANSS), 6 Columbia-Suicide Severity Rating Scale (C-SSRS), 7 and rated the Clinical Global Impression—Severity Scale (CGI-S). Results are reported in Table 2.
Scales administered at the beginning of treatment and at 6-month follow-up.
The patient signed a written informed consent in which she agreed to receive on- and off-label therapies. The following psychiatric medications were prescribed: slow-release lithium sulfate was titrated up to 124.5 mg (83 plus 41.5 mg) once daily; lurasidone was titrated up to 74 mg PO once daily. Moreover, lorazepam 1 mg PO 3 times a day was confirmed. Given the potential pharmacological interactions (in particular with carbamazepine, which is a strong inducer of CYP3A4), the woman and her parents were instructed to pay attention to any possible adverse event and change in symptomatology, especially suicidal ideation. In addition to pharmacological therapy, the patient started and regularly followed a psychotherapy pathway, namely cognitive behavioral therapy (CBT). She underwent monthly follow-up psychiatric visits and plasmatic drug dosages according to current guidelines. Moreover, she was asked to periodically undergo blood tests, in order to monitor physiological parameters, and in particular thyroid and renal function. The medication was well-tolerated by the patient, who did not report any remarkable adverse event; no modifications of the prescribed posology were necessary. Only lorazepam has been gradually reduced after the improvement of anxiety.
At six-month follow-up (March 2020), the assessment was repeated by the same interviewer, who was blind to the medications prescribed. Overall, depression and anxiety much improved, and psychogenic seizures decreased in frequency. Moreover, delusional beliefs completely disappeared, while still remained slight disorganization in speech and affective flattening. Suicidal ideation and aggressiveness were reported neither by the patient or her parents. Clinical Global Impression—Improvement Scale (CGI-I) was rated as 2 (Much improved). The results are reported in Table 2.
Discussion
In the present paper, we depict the intriguing case of a young woman who presented long-term psychiatric sequelae after suffering from a suspected form of autoimmune LE. It is widely known that, in its acute phase, autoimmune encephalitis may present with progressive psychiatric symptoms, such as confusion, hallucinations, delusions, mnemonic and cognitive deficits. 8 However, even if the descriptions of the acute manifestations of the disease are numerous in literature, little is known about the long-term psychiatric sequelae.
During the onset of the encephalitis, our patient primarily presented neurological issues, such as tonic-clonic seizures, cognitive impairments, and loss of consciousness, until coma. Nevertheless, after recovering, she progressively developed a worsening pattern of psychopathological symptoms, which did not remit with the most commonly used psychotropic medications. This is not surprising, as some patients with autoimmune LE may not respond to drugs such as antipsychotics or benzodiazepines. 9 Given the complex symptomatology, lurasidone in association with slow-release lithium sulfate was prescribed.
Lurasidone is a novel atypical antipsychotic, which has been approved in Europe only for the treatment of schizophrenia in adults; in the United States, it is also used for the treatment of bipolar depression in adults, as monotherapy and as adjunctive therapy with lithium or valproate. 10 Our patient presented with a composite clinical picture, with a wide variety of symptoms: on 1 hand, she had disorganized poor speech and delusions, which might be attributable to psychotic-like state; on the other hand, her mood was depressed with dysphoric notes, typical of bipolar depression. In fact, lurasidone seems to exert positive effects on psychotic-like symptoms, mood, and cognition. Moreover, the drug has shown a low propensity for cardiometabolic adverse events, such as QTc interval prolongation, weight gain, and hyperprolactinemia, and could thus be well-tolerated by a young woman 10 Slow-release lithium was prescribed in add-on because of the presence of suicidal ideation and self-aggression. Lithium represents an effective treatment for reducing the risk of suicide through 2 main mechanisms: first, it reduces the relapses of mood disorders; second, it decreases aggression and impulsivity. 11 Finally, the prescription of lorazepam, already taken by the patient, was initially confirmed for its anxiolytic properties; however, the benzodiazepine has been gradually reduced after amelioration of general psychopathological conditions.
It is worth mentioning that the patient was also taking carbamazepine for antiepileptic maintenance. Although the anti-aggressive effects of lithium and carbamazepine have been reported, 12 there is no report showing such effects in the long-term psychiatric manifestations of autoimmune LE. To our knowledge, only 1 paper has been published by Hirakawa et al. 9 who described the case of a 22-year-old man with seronegative LE and serious aggressiveness and anger outbursts. According to the authors, the psychiatric symptomatology did not respond to commonly used drugs, such as valproate, risperidone, or levomepromazine. However, the combination of carbamazepine and lithium appeared useful for treating mood instability and aggressive behaviors. While administering the association of lithium and carbamazepine, we had to consider the potential onset of side effects, such as the syndrome of inappropriate antidiuretic hormone (ADH) secretion (SIADH) or thyroid dysfunctions. As the long-term management of carbamazepine and lithium in combination might be challenging, it is crucial for the patient to undergo regular follow-ups to monitor both thyroid and renal function. Once the clinical situation is more stable, we could plan to reduce the posology of lithium sulfate.
None is reported in the literature regarding the usefulness of lurasidone in treating the LE, neither in the acute phase nor in the long-term. Even if considering that the strong inhibition of CYP3A4 caused by carbamazepine may have reduced the plasmatic levels of lurasidone, we have indeed observed a significant improvement of psychotic symptoms in our patient, as documented with PANSS scores. As far as concerns functional seizures, it is well-known that psychotherapy should be considered the first-line treatment, although some authors have reported the beneficial effects of low-dose neuroleptic drugs. 13 Indeed, our patient is regularly following CBT, which she has initiated just after the first visit in our Psychiatry Unit. It could be hypothesized that psychotherapy may have synergistically acted with pharmacological therapy in improving her mental conditions, particularly mood, and delusional thoughts, favoring in turn a significant decrease in non-epileptic crises.
It is important to acknowledge that the present case report has several limitations. First, autoimmune LE was a diagnosis of exclusion and was only retrospectively formulated; as clinical and instrumental investigations were somehow ambiguous, other forms of encephalopathy cannot be completely excluded. Furthermore, most of the original documentation has been lost, and some crucial clinical documents, such as brain images, could not be reported in the paper. Also, the posology of the medications prescribed prior to the last regimen was in some cases unknown.
However, our paper may further support the notion that autoimmune LE is on the boundary between neurology and psychiatry, and it not infrequent to get a diagnosis after a long time because of the misinterpretation of psychiatric symptoms. We do acknowledge that it is not possible to generalize a single case study, but our report suggests that people suffering from encephalitis, and more generally encephalopathies, need a regular follow-up, not only for the neurological impairments (eg, epilepsy or memory deficits) but also for the psychiatric sequelae. Finally, it is important to take into account that symptomatology may be complex and not respond to typically prescribed drugs, which may drive psychiatrists to undiscovered roads.
Footnotes
Acknowledgements
The authors would like to acknowledge the patient and his family for providing details and documentation about medical history.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
LF conceived the manuscript, participated to data collection and wrote the manuscript. AG, GB, MS, and EA contributed to the writing of the manuscript and to the critical interpretation of results. All authors reviewed and approved the final manuscript.
Disclosures and Ethics
This article has been performed in accordance with the Declaration of Helsinki. Written informed consent was signed from the patient. Ethical approval was obtained by our internal review board.
