Abstract
In intensive care unit (ICU) patients, delirium contributes to prolonged hospitalization, long-term cognitive impairment and increased mortality. Sleep disturbance, a risk factor for delirium, has been attributed to impaired melatonin secretion in critically ill patients. Ramelteon, a synthetic melatonin receptor agonist, is indicated for insomnia; there is limited, but growing evidence, to support its use for the prevention of delirium. The primary objective of this study is to describe the use of ramelteon and the incidence of delirium, assessed by Confusion Assessment Method for the ICU (CAM-ICU) scores, in adult surgical ICU patients from May 22, 2016 to June 30, 2018. The primary endpoint is the number of delirium free days in the week prior to and post first ramelteon administration. A total of 231 patients were included in the study with 201 (87%) positive for delirium at least once during the study timeframe. The median number of CAM-ICU negative days in the week pre-ramelteon administration was 4 days (IQR 2-7 days) compared to 6 days (IQR 3-7 days) in the week post-first ramelteon administration (P < .05). The time to CAM-ICU positive increased slightly to 3 days (IQR 1-7 days) following ramelteon initiation compared to 2 days (IQR 1-5 days) from initial ICU admission. Additionally, the median number of antipsychotic doses per patient decreased from 4 doses (IQR 1.25-14 doses) prior to ramelteon to 2 doses (IQR 1-4 doses) after ramelteon. Ramelteon administration was associated with a greater number of CAM-ICU negative days in surgical ICU patients. These findings describe a potential role for ramelteon in mitigating delirium in this patient population.
Background
Delirium is defined as a change in cognition and/or disturbance of consciousness which fluctuates with time and can be directly attributed to a medical condition or medication. Delirium in the ICU contributes to increased mortality, prolonged hospitalization, and long-term cognitive impairment. This acute behavioral disturbance can manifest in 60% to 80% of medical and surgical intensive care unit (ICU) patients supported by mechanical ventilation. Among medical ICU patients not requiring mechanical ventilation, that number remains as high as 50% to 70%.1,2
The pathophysiology of delirium is poorly understood. Multiple hypotheses exist including imbalances in neurotransmitters, neuroinflammation, oxidative stress, large amino acids, network connectivity, and diurnal variation. Sleep disturbance, a risk factor for delirium, has been attributed to impaired melatonin secretion in critically ill patients. 3
Melatonin is a biological hormone synthesized from tryptophan and secreted by the pineal gland in response to darkness.3,4 Melatonin binds to MT (1) and MT (2) receptors in the pituitary gland and retina, respectively. This cascade of events promotes nocturnal sleep and regulates the sleep-wake cycle. It is hypothesized that exogenous melatonin may help mitigate delirium through a negative feedback mechanism that decreases the breakdown of melatonin and serotonin’s precursor tryptophan. 5 Currently, no pharmacological agents are guideline recommended for the prevention or treatment of delirium. 6
Ramelteon, a synthetic melatonin agonist with higher affinity for melatonin receptors than endogenous melatonin, is FDA approved for the treatment of insomnia. 7 To date, only 2 randomized controlled trials (RCTs) examining ramelteon for the prevention or treatment of ICU delirium have been published.8,9 Hatta et al evaluated elderly patients in both ICU (35.8%) and acute care (64.2%) settings and found ramelteon to be associated with a lower risk of delirium in comparison to placebo (3% vs 32%). Nishikimi et al demonstrated a reduction in ICU length of stay (LOS) (4.56 vs 5.86 days) among patients receiving ramelteon.
As evidenced above, there is now data to suggest ramelteon could have a role in delirium prevention for both the acute care and medical ICU settings. However, there are no ramelteon trials to date evaluating surgical ICU patients who arguably carry a higher risk of developing delirium. The objective of this study is to describe the effects of ramelteon on delirium in surgical ICU patients.
Methods
A single center, retrospective, descriptive cohort study was conducted at a large-academic medical center in the United States from May 22, 2016 through June 30, 2018. The protocol was approved by the institutional review board of the hospital and informed consent was waived due to the retrospective nature of the study.
Patients were eligible for inclusion in the study if they were 18 years of age or older, admitted to the surgical-liver ICU (SLICU) or cardiovascular surgery ICU (CVICU), and received at least 3 doses of ramelteon 8 mg within a 7-day period while in the ICU. Patients were excluded with an ICU LOS less than 3 days, those who received ramelteon prior to ICU admission, and any patient without documentation of CAM-ICU scores.
Model for end-stage liver disease (MELD) and sequential organ failure assessment (SOFA) scores were calculated for patients with laboratory and hemodynamics data available within 48 hours of the first ramelteon dose. The intent was to assess the patient’s severity of illness in closest proximity to ramelteon initiation as opposed to status at admission due to the anticipated LOS of the patient population.
CAM-ICU scores were assessed for the presence (positive) or absence (negative) of delirium and converted to CAM-ICU positive or negative days. This validated bedside nursing delirium assessment tool was completed at minimum once per 12-hour shift. A positive score trumped a negative score; thus, if a patient had a positive CAM-ICU score at any time during the day, it was considered a CAM-ICU positive day. CAM-ICU negative or positive days, while not specific to prevention or treatment effect, was utilized in order to capture any potentially meaningful effect ramelteon may have on delirium. As ICU patients often have fluctuating delirium status, determining if the primary use was for prevention versus treatment was not feasible given the retrospective nature of the study and lack of requirement to link the medication order to an indication.
The primary endpoint was the number of CAM-ICU negative days in the week prior to and following the first day of ramelteon administration. Secondary endpoints included time to CAM-ICU positive, ICU LOS, mortality at discharge, days of mechanical ventilation, and number of as needed (PRN) antipsychotic doses per patient prior to and after ramelteon initiation. Secondary evaluations were also conducted of CAM-ICU negative days based on MELD scores, SOFA scores and total number of ramelteon doses received.
Data was extracted from patient electronic medical records (EMR) via electronic query. The data collected was analyzed using descriptive statistics for continuous variables and counts or percentages for categorical variables. The primary outcome was further assessed by the Wilcoxon signed-rank test with the use of Minitab 16 software. The 2-sided alpha for significance was set at 0.05.
Results
A total of 610 patients were eligible for inclusion; 231 met inclusion criteria as depicted in Figure 1. Patients included had a median age of 64 years, were more likely to be male (60.2% male vs 39.8% female), predominantly hospitalized in the SLICU (86.1%) and received a median of 4 ramelteon doses in the week immediately following initiation (Table 1).
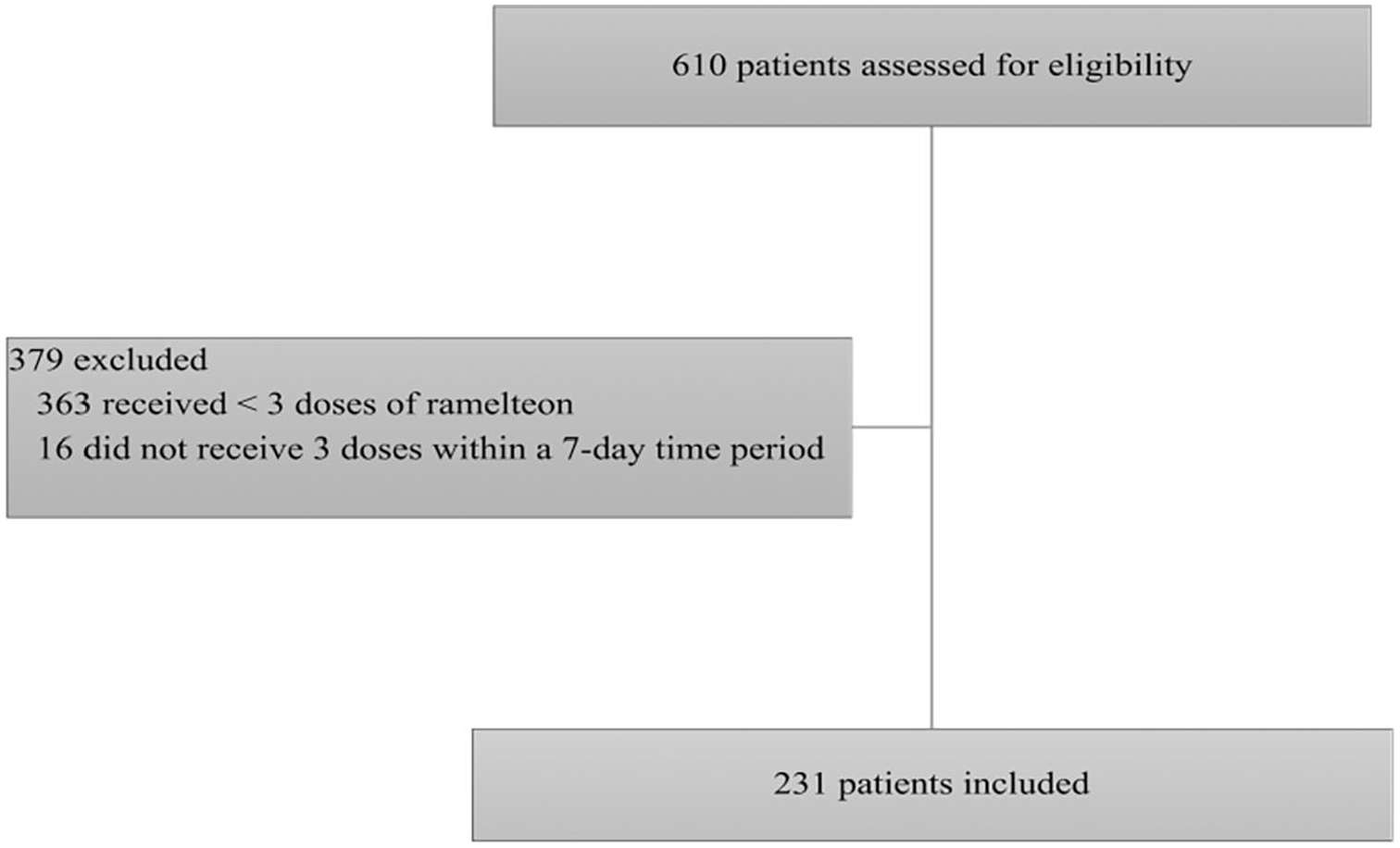
A total of 610 patients received a dose of ramelteon while in the ICU during the study time-frame and 231 patients met inclusion criteria. There were 379 patients excluded, 363 patients received < 3 doses of ramelteon and 16 patients did not get 3 doses within 7 days.
Baseline patient characteristics.
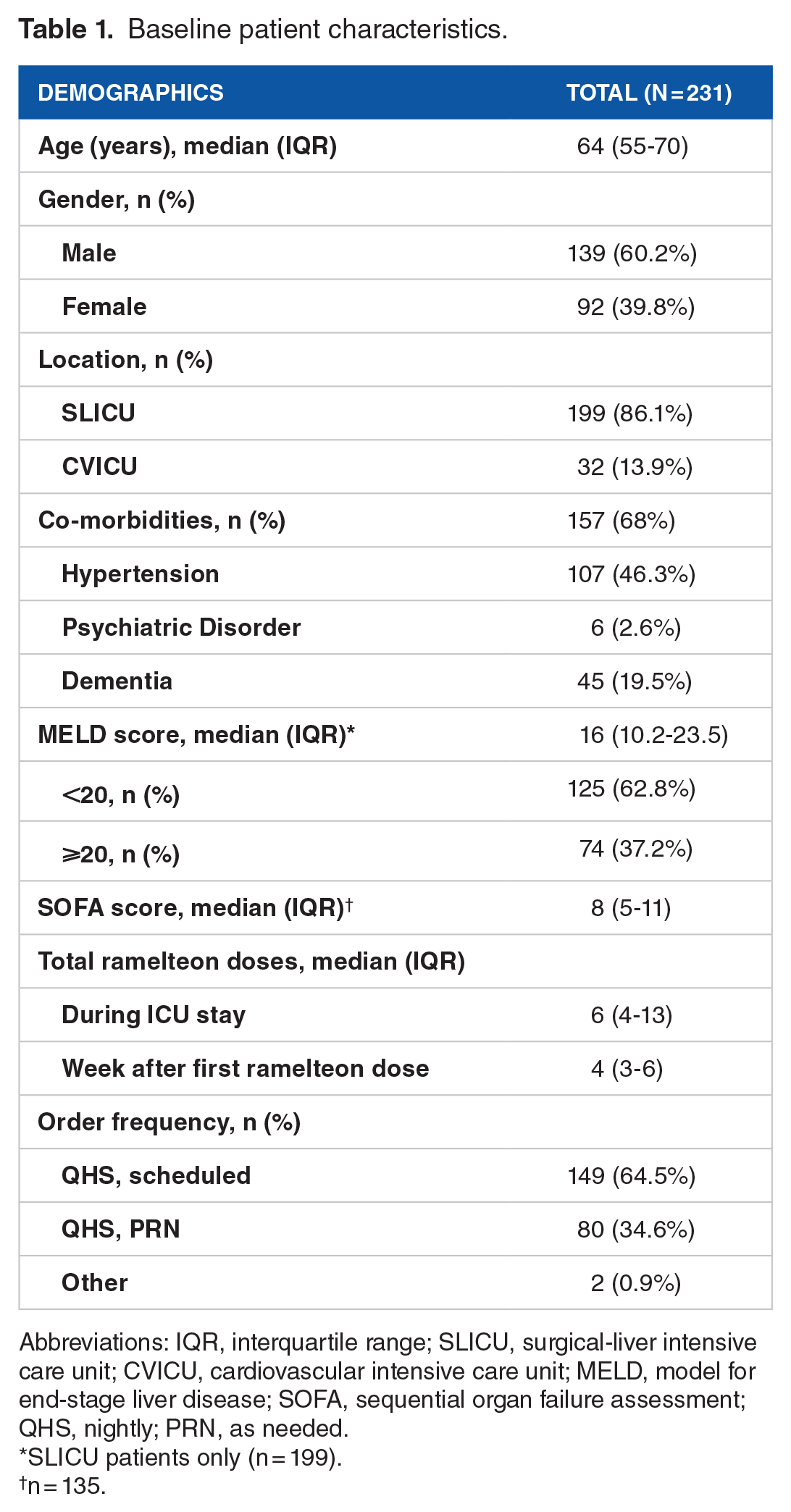
Abbreviations: IQR, interquartile range; SLICU, surgical-liver intensive care unit; CVICU, cardiovascular intensive care unit; MELD, model for end-stage liver disease; SOFA, sequential organ failure assessment; QHS, nightly; PRN, as needed.
SLICU patients only (n = 199).
n = 135.
Delirium developed in 201 (87%) patients during their ICU stays with 151 (65.4%) with at least 1 CAM-ICU positive day after ramelteon administration. In the week prior to and including the first day of ramelteon administration, the median number of CAM-ICU negative days per patient was 4 days (IQR 2-7 days). In the week following the first ramelteon dose, the median number of CAM-ICU negative days per patient increased to 6 days (IQR 3-7 days) (P < .05) (Figure 2). Sixty patients (26%) evaluated had a least 1 CAM-ICU positive day prior to ramelteon initiation and zero CAM-ICU positive days after ramelteon. Nine (3%) patients demonstrated zero CAM-ICU positive days in both the week before and after first ramelteon administration.
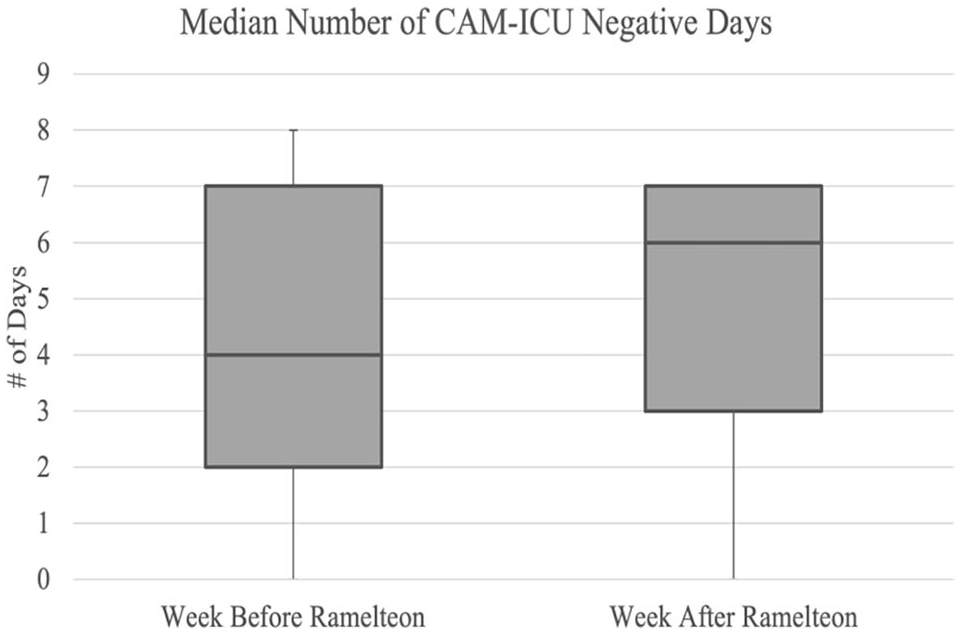
Based on confusion assessment method for the ICU (CAM-ICU) scores, the median number of delirium free days in the week prior to first ramelteon dose was 4 days (IQR 4-7) compared to 6 days (IQR 3-7 days) in the week after (P < .05).
After ramelteon initiation, the time to first CAM-ICU positive day was delayed from 2 days (IQR 1-5 days) to 3 days (IQR 1-7 days). The number of PRN antipsychotics administered, which included haloperidol, lorazepam, risperidone, and quetiapine, were higher in the week prior to ramelteon compared to the week post first dose (4 doses (IQR 1.25-14 doses) vs 2 doses (IQR 1-4 doses)) (Table 2).
Secondary outcomes.
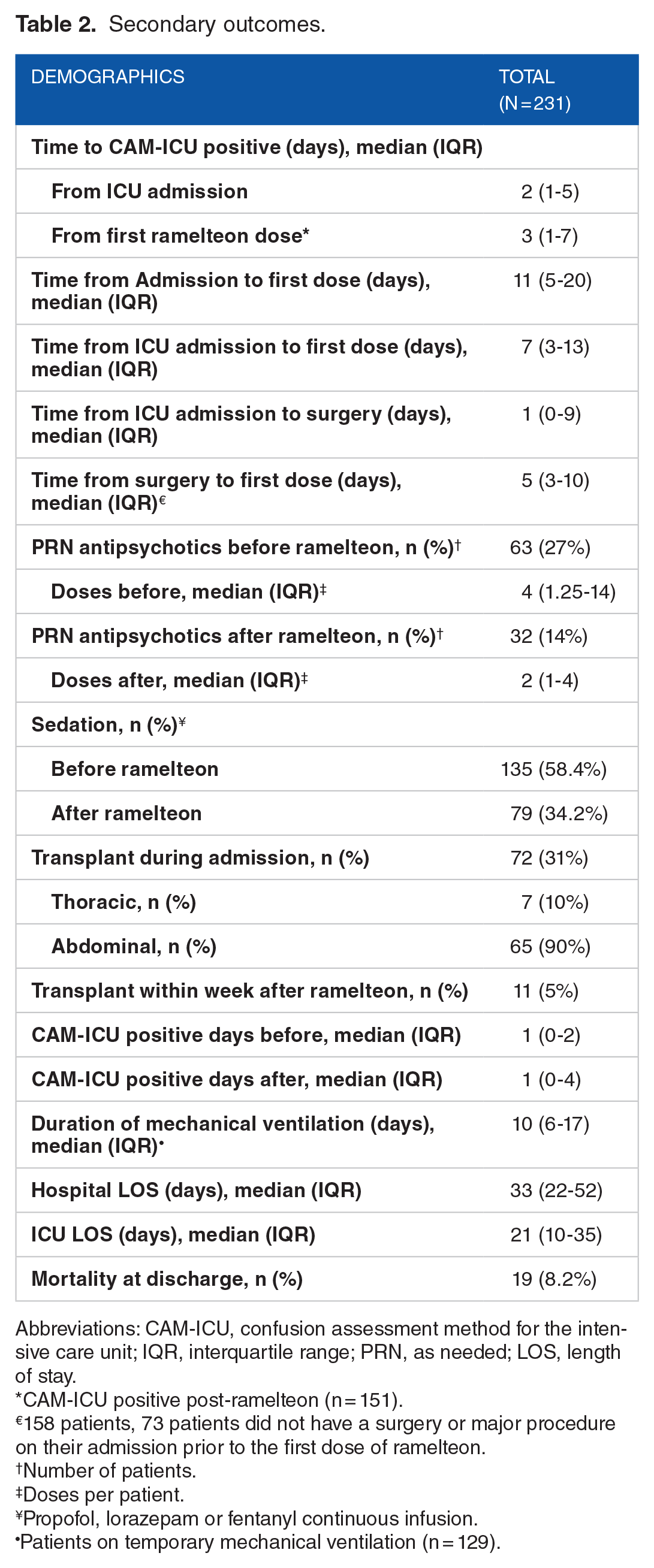
Abbreviations: CAM-ICU, confusion assessment method for the intensive care unit; IQR, interquartile range; PRN, as needed; LOS, length of stay.
CAM-ICU positive post-ramelteon (n = 151).
158 patients, 73 patients did not have a surgery or major procedure on their admission prior to the first dose of ramelteon.
Number of patients.
Doses per patient.
Propofol, lorazepam or fentanyl continuous infusion.
Patients on temporary mechanical ventilation (n = 129).
No correlation was noted between the severity of illness or hepatic dysfunction and the number of CAM-ICU negative days (Figure 3). There was also no clear dose-dependent relationship based on the number of ramelteon doses received and the number of CAM-ICU negative days in the week after initiation (Figure 4). The order frequency for ramelteon administration, scheduled versus PRN, also demonstrated no notable difference on the number of CAM-ICU negative days. The number of CAM-ICU negative days for scheduled ramelteon orders before and after ramelteon initiation was 4 days versus 5 days; for PRN orders the difference was 4 days versus 6 days.
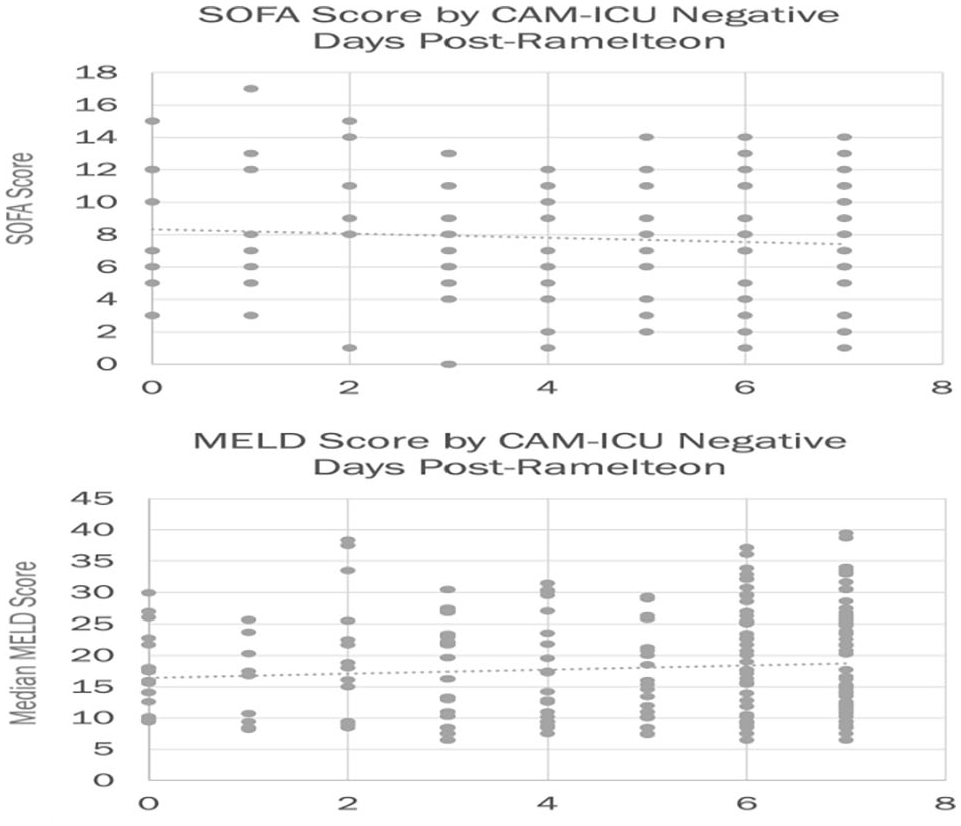
Sequential organ failure assessment (SOFA) and model for end-stage liver disease (MELD) scores were calculated for patients with laboratory and hemodynamics data available within 48 hours of the first ramelteon dose to assess for any correlation between severity of illness and delirium free days. No correlation was found.
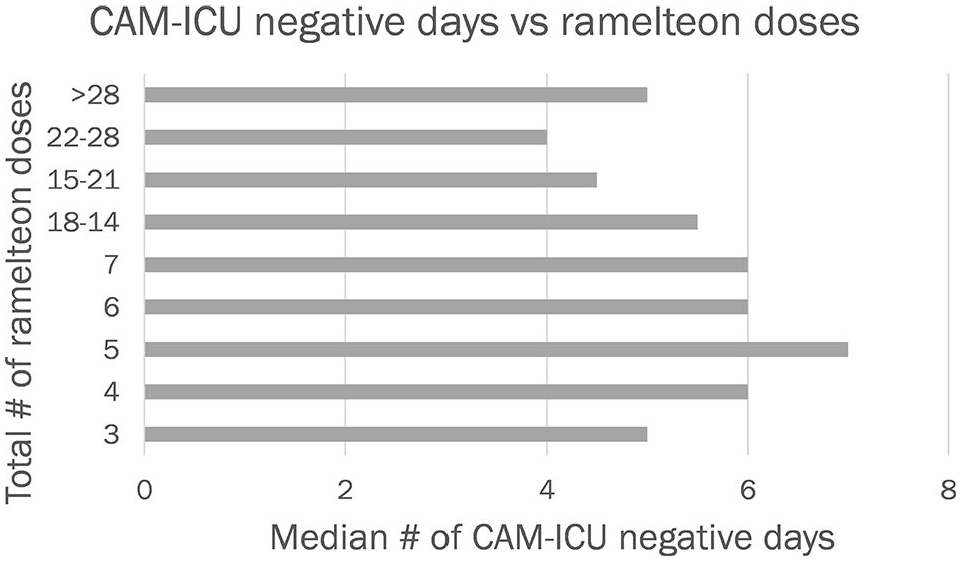
The total number of ramelteon doses received per patient was compared to the median number of confusion assessment method for the ICU (CAM-ICU) negative days in the week after first dose in order to assess for any dose-dependent correlation; none was found.
Discussion
The results of this study support the hypothesis that, in addition to melatonin’s ability to improve nocturnal sleep, there may be an added benefit of delirium mitigation in surgical ICU patients. Patients who received at least 3 doses of ramelteon had more CAM-ICU negative days in the week after therapy started compared to the week prior (median 6 vs 4 days). While sleep quality was not monitored, ramelteon’s mechanism of action, including higher affinity for endogenous melatonin receptors, suggests targeting this sleep-promoting pathway may affect delirium pathophysiology as well. Other retrospective studies have shown the benefit of melatonin on prevention of ICU delirium; however, ramelteon was used in our study over other commercially available melatonin supplements as it is the only FDA approved melatonin product and is a formulary medication at our institution. 10
At our institution one of the key initiatives utilized to combat delirium is the ABCDEF bundle. 2 This acronym was created as a guide to address several fundamental activities which can help improve ICU patient outcomes. The letter “D” stands for delirium mitigation which suggests utilizing the CAM-ICU score to assess for delirium daily, removing delirium prone medications (such as benzodiazepines) and improving the patient’s daily environment, which includes promoting sleep hygiene through early mobilization, lights on in the day but off at night and minimal overnight interruptions.
Melatonin has a theoretical benefit for both prevention and treatment of delirium, as such, both indications were included in our study. Regarding prevention, only 9 patients had zero CAM-ICU positive days in both the week before and after first ramelteon dose; however, time to first CAM-ICU positive day was delayed slightly from 2 to 3 days. Additionally, the median time from ICU admission to surgery was only 1 day and subsequently the median time to first ramelteon dose was less than 1 week which may correlate more to a preventative than treatment effect. Although the time-frame to develop delirium is variable and highly patient dependent, the further removed patients are from a contributory event, such as surgery, the incidence of delirium would be expected to decline. However, given the long ICU LOS for patients in this study it is more challenging to assess.
Twenty-six percent of patients had a least 1 CAM-ICU positive day prior to ramelteon and zero positive days after, representing a possible treatment effect. Currently in ICU literature the 2 RCTs, Hatta et al and Nishikimi et al, that evaluated ramelteon focused on delirium prevention as opposed to treatment.8,9 While in theory our goal is to prevent delirium and thus not have to treat the condition, delirium remains common in ICU patients. With no proven pharmacological treatment, the promising preventative results seen with ramelteon could justify further trials investigating its use for delirium treatment as well.
The strengths of our study included a relatively large sample size compared to previously published trials. The patients included in the study were at high risk of developing delirium due to critical illness and prolonged hospitalizations, with a median ICU LOS of 3 weeks. This patient population is representative of those that would benefit most from a pharmacological option to treat or prevent delirium.
Limitations included the lack of a comparator group, potential for human error in CAM-ICU assessment and retrospective nature of the study preventing long-term assessment of cognitive effects. Additionally, the indication for ramelteon included either delirium prevention or treatment making it more challenging to determine where the medication may be most efficacious. This was done, in part, due to a lack of clear documentation of the indication and potential for overlap, such as starting the medication for prevention but then continuing it for treatment. The narrow time frame of 1 week prior to and following first ramelteon dose maximized the sample size and reduced confounding variables, but hindered a more comprehensive analysis of the magnitude of effect, or lack thereof. As the majority of patients received the first dose of ramelteon within 1 week of ICU admission, the ability to detect treatment effect is limited as there may not have been sufficient time for the patients to develop delirium in the first place. Lastly, a majority of patients in the SLICU have hepatic dysfunction resulting in encephalopathy which complicates the assessment for etiology of delirium.
Conclusion
Ramelteon administration to surgical ICU patients had a positive impact on the number of CAM-ICU negative patient days. This supports prior studies that found ramelteon reduced development of delirium in the ICU patient population. Further studies are warranted to solidify ramelteon’s role in delirium prevention and determine any added treatment effect.
Footnotes
Acknowledgements
Ranran (Christina) Xia, PharmD: project development
Yung Tran: data acquisition and analysis
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AF and JB conceived the idea. CL, AF and JB designed the analysis. AF, AD and JB provided project oversight and planning. CL collected the data and performed the analysis. CL wrote the manuscript with significant contributions from AF, AD and JB.
