Abstract
Introduction:
Cervical cancer remains a major public health concern in low- and middle-income countries, including Nepal. The International Federation of Gynecology and Obstetrics (FIGO) 2018 staging system for cervical cancer introduced stage IIIC for tumors with lymph node metastasis, reflecting the prognostic significance of nodal involvement. This study aimed to evaluate stage migration from FIGO 2009 to FIGO 2018 and to validate the prognostic performance of the revised staging system in locally advanced cervical cancer.
Methods:
In this retrospective cohort study, we consecutively included 155 patients with histologically confirmed cervical cancer treated with definitive radiotherapy or chemo-radiotherapy at Kathmandu Cancer Center between August 2016 and June 2019. Patients were initially staged according to the FIGO 2009 criteria and retrospectively restaged according to the FIGO 2018 criteria. Recurrence patterns were documented, and overall survival (OS) and disease-free survival were estimated using the Kaplan-Meier method with log-rank tests for group comparisons. All patient identifiers were removed, and the study conformed to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. Ethical approval was obtained from the Nepal Health Research Council (approval number: 655/2020P), and the requirement for written informed consent was waived.
Results:
Under FIGO 2009, most patients were classified as stage IIB (47.7%) and IIIB (36.7%), whereas under FIGO 2018, this led to 34.2% in stage IIB and 31% in stage IIIC1. Stage migration occurred in 38.7% of patients, predominantly from IIIB (58.3%) and IIB (35.4%) to IIIC1. Both staging systems demonstrated decreased OS with advanced stages; however, stage IIIC1 patients had better OS than stage IIIB patients under the FIGO 2018 staging, likely reflecting heterogeneity in tumor extent and treatment factors. Five-year OS rates were not significantly different between the 2 staging systems. Recurrence was observed in 29.2% of patients, with distant metastasis being the most common pattern.
Conclusions:
The FIGO 2018 staging system results in substantial stage migration and highlights heterogeneity among node-positive (stage IIIC1) patients. Consideration of tumor extent and nodal burden is important for accurate prognostication. These findings support the need for further studies to refine the staging system and improve its prognostic precision in cervical cancer.
Keywords
Introduction
Cervical cancer is the fourth most common cause of cancer-related mortality worldwide, with more than 300,000 deaths occurring annually. 1 More than 90% of these deaths occur in low- and middle-income countries (LMICs). 2 In the context of Nepal, cervical cancer is the second most common cancer and the most common cause of cancer-related mortality in females. 3 The International Federation of Gynecology and Obstetrics (FIGO) system is the most commonly used method for cervical cancer staging. It is known for its convenience and reliability, which incorporates key prognostic factors.
The FIGO 2009 staging system was based on clinical staging and correlated cervical cancer survival with tumor extent at diagnosis. 4 In 2018, FIGO revised the cervical cancer classification guidelines to include imaging and pathological findings when available. 5 Changes to the FIGO 2018 staging system included removing horizontal spread, subcategorizing stage IB by tumor size, and adding a separate stage IIIC for cancers with lymph node metastases. 6 While stage IIIC1 includes cases with pelvic lymph node metastasis, stage IIIC2 includes cases with para-aortic lymph node metastasis. 6
The modified FIGO 2018 staging system is still undergoing validation, especially for stage IIIC cancers. Patients with stage IIIC1 disease have been reported to present heterogeneous characteristics with variable survival outcomes. 7 These patients often have better survival than patients with stage IIIA-B disease, implying the role of multiple factors in addition to nodal status in patient survival. 8
Many validation studies of the FIGO 2018 staging system have been published from different parts of the world; however, limited data are available from Southeast Asia and Africa, where most cervical cancers are diagnosed. This study compares the FIGO 2009 and 2018 staging systems with respect to stage migration and survival outcomes in LMICs.
Methods
Patient selection
We retrospectively reviewed the clinical records of the patients diagnosed with cervical cancer and treated with definitive radiotherapy or chemo-radiotherapy at Kathmandu Cancer Center, Bhaktapur, Nepal. Patients treated between August 1, 2016, and June 30, 2019, were included. The inclusion criteria of the patients in the study were (1) histologically confirmed cervical cancer; (2) FIGO stage I, II, and III treated with primary radiotherapy or chemo-radiotherapy; and (3) no evidence of distant metastasis prior to treatment. The exclusion criteria were (1) postoperative radiotherapy, (2) palliative treatment, (3) stage IV disease, and (4) incomplete radiation therapy.
Patients were selected consecutively from the institutional records. All patient identifiers were removed to ensure confidentiality. The study conformed to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines.
Ethical approval
This retrospective study was approved by the Ethical Review Board of Nepal, that is, the Nepal Health Research Council (NHRC) (Approval number 655/2020P; date of approval: April 29, 2021). The study was conducted in accordance with the Declaration of Helsinki (1975, revised 2024). The requirement for written informed consent from patients was waived by the NHRC due to the retrospective nature of the study.
Staging
All patients underwent clinical examinations by gynecologists and radiation oncologists. Proctoscopy, cystoscopy, and ultrasonography (for hydronephrosis) were performed when indicated. Final staging was documented according to FIGO 2009 staging criteria.
Patients undergoing radiotherapy with or without chemotherapy underwent contrast-enhanced computed tomography (CECT)-based planning. For patients unable to undergo CECT, magnetic resonance imaging (MRI) was performed, and images were fused with noncontrast computed tomographic (CT) scans for treatment planning.
Patients were retrospectively restaged according to the FIGO 2018 criteria. A team of a radiation oncologist and a radiologist reviewed all imaging. Lymph node positivity was determined based on CT/MRI criteria: short-axis ⩾1 cm or morphologically suspicious nodes. 9
Treatment
All patients underwent image-guided high-dose-rate brachytherapy with CT-based planning. During the study period, dose prescription was uniformly performed using a conventional point A–based approach in all patients. Volume-based brachytherapy prescription, as well as interstitial or hybrid brachytherapy techniques, was not routinely implemented at our center until after 2020.
The patient cohort received external beam radiation with either 50 Gy delivered in 25 fractions or 46 Gy in 23 fractions. Initially, the departmental protocol prescribed 50 Gy in 25 fractions, which was later revised to 46 Gy in 23 fractions. Patients who received chemotherapy were treated with weekly cisplatin at a dose of 40 mg/m2.
Treatment planning adhered to established cervical cancer contouring guidelines for patients undergoing definitive chemo-radiotherapy or radiotherapy.10,11 Patients with positive para-aortic nodes (FIGO stage IIIC2) or common iliac node involvement received extended-field radiation encompassing the para-aortic lymphatic drainage region. 12 Based on institutional treatment guidelines, radiologically positive pelvic lymph nodes received a nodal boost.
High-dose-rate brachytherapy was delivered following completion of External Beam Radiation Therapy (EBRT) using an iridium-192 afterloader (Gammamed, Varian Inc). The prescribed regimen consisted of 8 Gy per fraction delivered in 3 fractions at weekly intervals. Dose prescription and organ-at-risk constraints were based on following the recommendations of the International Commission on Radiation Units and Measurements (ICRU) guidelines. 13 Patients were followed up at 3 months after treatment completion for initial response assessment, every 6 months for the subsequent 2 years, and annually thereafter for up to 5 years.
Recurrence assessment
Initial treatment response was classified as a complete response or residual disease based on clinical examination and imaging (CT or MRI). Recurrence was evaluated only in patients with an initial complete response and categorized as local, nodal, or distant metastasis.
Data collection
Patients were prospectively followed to determine their disease status. Collected data included age, tumor type, FIGO 2009 stage, nodal status, recurrence status, and initial follow-up status. Stage distribution and survival outcomes were assessed and compared between FIGO 2009 and FIGO 2018 systems. Recurrence patterns were analyzed according to FIGO 2018 criteria.
Statistical analysis
Descriptive statistics were used to summarize demographic and clinical characteristics. Overall survival (OS) and disease-free survival (DFS) were estimated using the Kaplan-Meier method, and group comparisons were performed using the log-rank test. All statistical analyses were conducted using R software version 4.3.2. A P value <.05 was considered statistically significant. No sample size calculation was performed due to the retrospective nature of the study.
Results
During the study period, a total of 181 cervical cancer patients received treatment at our hospital. Of 181 patients, 26 did not meet the inclusion criteria and were excluded; the remaining 155 were included in the final analysis.
Demographic and clinical characteristics of the patients
Among the 155 patients, 138 (89%) had squamous cell carcinoma, and 17(11%) had adenocarcinoma. The characteristics of the patient cohort are presented in Table 1. The median age of the study cohort was 53 years (range, 29-85 years). All patients in our study received planned radiotherapy, and 110 (71%) patients also received chemotherapy. The median follow-up duration after the treatment was 54 months (range, 3-88 months).
Demographic and clinical characteristics of the patients.

Stage distribution
The stage distribution of patients among FIGO 2009 and FIGO 2018 classifications is depicted in Figure 1. According to FIGO 2009 staging, stage distribution comprised predominantly stage IIB (47.7%) and stage IIIB (36.7%). When the patients were restaged according to the FIGO 2018 criteria, most common stage was IIB (34.2%) and IIIC1 (31%).

Sankey diagram depicting stage distribution and migration among FIGO 2009 and FIGO 2018 staging.
Stage migration
Stage migration from FIGO 2009 to FIGO 2018 staging occurred in 60 of 155 (38.7%) patients. A decrease in the proportions of FIGO 2009 stages, primarily IIB and IIIB, contributed to the stage migration to stages IIIC1 and IIIC2. Patients with stage IIIC1 mainly migrated from former stages IIIB (58.3%) and IIB (35.4%), and a small number from stage IB (6.25%) (Figure 1). Patients with stage IIIC2 migrated from former stages IIIB (67%) and IIB (33%).
Survival
On survival analysis, there was a significant trend of the worst OS of advanced-stage patients based on FIGO 2009 (P = 0.011) (Figure 2A) and FIGO 2018 staging (P = 0.02) (Figure 2B), respectively. However, the OS of stage IIIC1 patients was better than that of stage IIIB patients, according to the FIGO 2018 scheme.
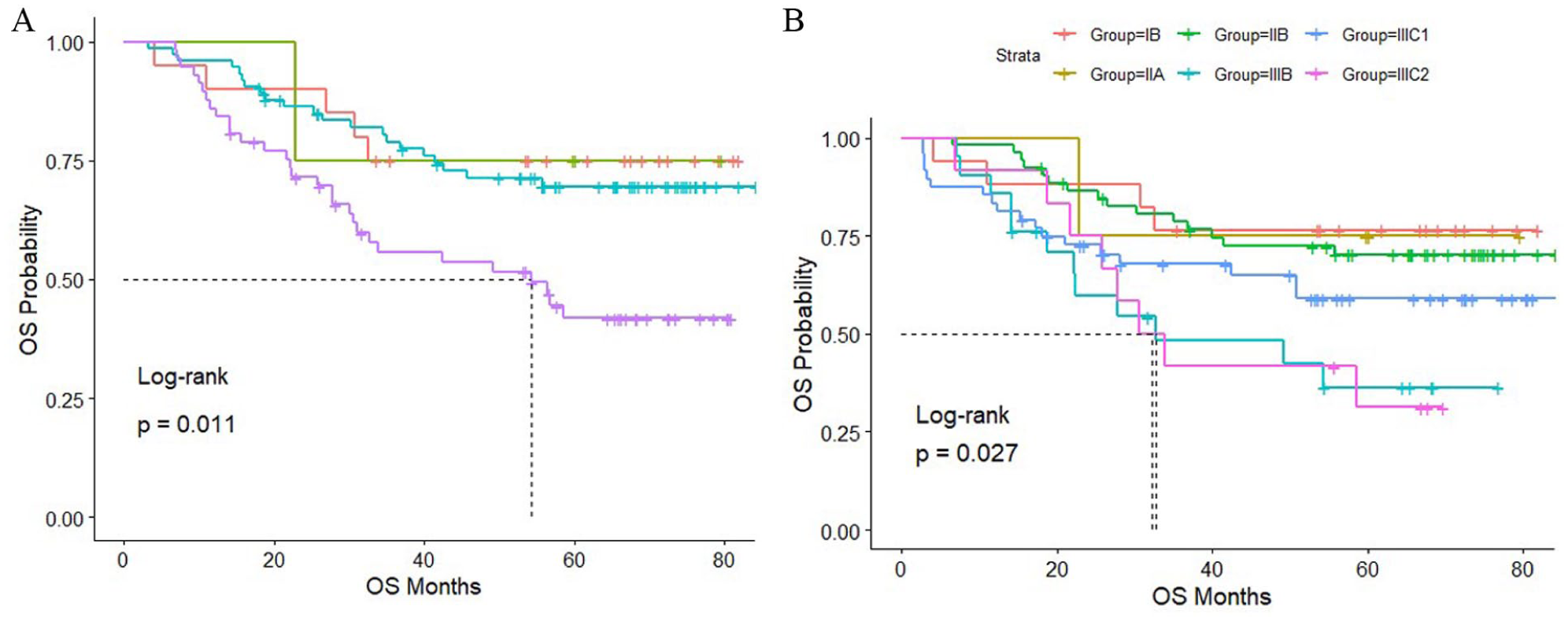
Kaplan-Meier curves for overall survival (OS) by FIGO 2009 (A) and 2018 (B) staging.
The 5-year OS for each stage was not statistically significantly different between the FIGO 2009 and FIGO 2018 staging systems, as depicted in Table 2.
Five-year overall survival of the patients according to the FIGO 2009 and FIGO 2018 systems.

Overall survival in stage IIIC stratified by former migrated stages showed a trend toward worse prognosis at the advanced stage; however, survival probabilities were not significantly different among the groups (Figure 3). The 5-year OS of prior-migrated stages was 66.7% (95% confidence interval [CI], 30%-100%), 63.9% (95% CI, 45.5%-89.7%), and 44.9% (95% CI, 30.3%-66.6%) for stages IB, IIB, and IIIB, respectively.

Kaplan-Meier curve for the overall survival (OS) of stage IIIC patients stratified by former migrated stages of FIGO 2009 staging.
Recurrence pattern
Our treatment regimen resulted in an initial complete response in 137 (88.4%) patients and residual disease in 18 (11.6%) patients. The stage distribution of complete response and residual disease is shown in Figure 4A.

Treatment outcomes (A) and recurrence status (B) based on FIGO 2018 staging.
Among patients who initially presented with a complete response, 40 (29.2%) developed recurrence. The mean recurrence time from the treatment completion was 20.34 months. Of 40 patients, 13 (32.5%), 15 (37.5%), 5 (12.5%), and 7 (17.5%) developed recurrence within 1 year, 2 years, 3 years, and 4 years of treatment, respectively.
Recurrence was high in stage IIIC2 patients (44.4%), followed by stage IIIB (37.7%), stage IIIC1 (31.7%), stage IIB (28%), stage IIA (25%), and stage IB patients (11.8%). Distant metastasis was the predominant recurrence type (65%), followed by local (27.5%) and nodal (7.5%) recurrences. The distribution of recurrence status by stage is depicted in Figure 4B.
At the end of the study period, 94(60.9%) patients were disease-free, 4(2.6%) were living with the disease, and 43(27.7%) patients died.
Discussion
Our study provides important insights into stage migration and recurrence patterns in cervical cancer patients when applying the FIGO 2018 staging system. Among our cohort, 60 of 155 (38.7%) patients were reclassified to stage IIIC, primarily from stage IIIB (58.3%) and IIB (35.4%). This upward stage migration aligns with previous reports, which range from 29% to 53%,14-16 and underscores the impact of incorporating nodal status into the FIGO 2018 criteria. The distribution of stage migration in our study is also comparable to that reported by Raut et al, 16 who observed migration rates of 45% and 40% from stages IIIB and IIB, respectively.
The introduction of stage IIIC to account for lymph node involvement represents the most significant change in the FIGO 2018 staging system. 17 Nodal positivity is a well-established adverse prognostic factor associated with decreased OS and DFS in both early-stage and advanced-stage cervical cancer.16,18,19 In accordance with previous studies, poorer survival has been reported in some cohorts of stage IIIC1 patients compared with stage IIIB disease.16,20
Our study demonstrated worse prognosis in advanced stages for both FIGO systems; however, stage IIIC1 patients had better OS than stage IIIB patients (36% vs 59%). This type of paradox was also reported in previous studies.7,15,21,22 This apparent paradox may result from heterogeneity introduced by grouping nodal positivity as stage IIIC and from the interplay between nodal status and local tumor extent (T stage). In addition, treatment factors, such as dose-escalated brachytherapy for nodal disease in stage IIIC1 patients versus standard point A–based brachytherapy for large stage IIIB tumors, may have contributed to differences in survival.
Several tumor characteristics are associated with nodal positivity, including larger tumor size, locally advanced disease, squamous histology, lymphovascular space invasion, and higher tumor grade. 23 The observation of better survival in stage IIIC1 compared with stage IIIA-B suggests that grouping all lymph node–positive patients into a single stage (IIIC) creates a heterogeneous population with variable prognoses.7,22 While nodal positivity in endometrial cancer is associated with poorer outcomes in stage IIIC patients compared with stage IIIA-B, the relatively better survival observed in stage IIIC1 cervical cancer may reflect the complex interplay between nodal involvement and local tumor burden.22,24 In the present study, nodal status was primarily assessed using CT and/or MRI criteria, as positron emission tomography/computed tomography was not routinely available for most patients. We acknowledge that this imaging-based approach may have resulted in misclassification of nodal involvement, which could have influenced stage migration and survival outcomes. Therefore, the observed prognostic differences, particularly among stage IIIC patients, should be interpreted cautiously.
Our findings show that an advanced order of prior migration stage contributes to heterogeneity among stage IIIC1 patients (5-year OS 63.9% vs 44.9% in stages IIB and IIIB, respectively). Failure to account for the local extent (T stage) of the tumor has been suggested as the cause of prognostic heterogeneity in stage IIIC1 patients. 14 Shin et al 25 in the study based on data on patients with cervical cancer from the Korean Cancer Registry report statistically significantly different survival of stage IIIC patients stratified with T stages (P < .001).
Similarly, Kim et al 21 and Matsuo et al 22 demonstrated that the survival among stage IIIC1 patients is related to T stage with statistically significant 5-year survival rates (T1 = 80%, T2 = 74%, T3 = 45%, P < .0001 and T1 = 75%, T2 = 59%, T3 = 39%, P < .001, respectively) which are comparable with stage IIIA (40%) and IB2 (76%) in the study of Quinn et al. 8 Duan et al 26 further reported that stage IIIC T1 and IIIC T2a-T2b patients have better OS and DFS than stage IIIA + IIIB patients, whereas stage IIIC (T3a-T3b) patients have poorer OS than stage IIIA + IIIB.
In our treatment regimen, patients with stage IIIB disease received standard brachytherapy with a point A–based prescription, which may not be enough to cover large tumors. On the contrary, stage IIIC1 patients received dose escalation with a boost to metastatic lymph nodes, which may have contributed to better local control and survival compared with stage IIIB patients. Computed tomography–guided volume-based brachytherapy, when combined with interstitial brachytherapy, provides better dose coverage, especially in large residual tumors, which are often seen in stage IIIB tumors.27-29 The incorporation of volume-based brachytherapy into hybrid brachytherapy, when indicated, should increase local control and thus improve survival in stage IIIB patients.
Increasingly advanced nodal involvement affects patient survival, as significantly lower survival has been observed in patients with para-aortic node positivity compared with pelvic node positivity; 5-year OS in stage IIIC2 patients was 31% compared with 59% in stage IIIC1 patients in our study.
The mean time to recurrence in our study was 20.3 months, consistent with previously reported ranges of 7 to 36 months.30-32 More than half of the recurrences occurred within 2 to 3 years after treatment, in line with the existing literature.33-35 Most of the patients in our cohort experienced distant metastasis (65%), followed by local (27.5%) and nodal recurrence (7.5%). Distant metastasis was the most common recurrence pattern, likely reflecting the high proportion of advanced-stage disease in our cohort. Most patients in our cohort experienced distant metastasis (65%), followed by local (27.5%) and nodal recurrence (7.5%). Distant metastasis was the predominant recurrence in all stages. However, studies have shown that nodal metastasis is the most common site of recurrence, followed by locoregional and distant metastasis.30-32 This difference could be attributed to the higher number of advanced-stage tumors at diagnosis in our cohort.
This study has several limitations. Its retrospective, single-center design limits generalizability, and the relatively small sample size precluded detailed subgroup analyses, including FIGO 2018 stage IB substages. The lack of routine Fluorodeoxyglucose-Positron Emission Tomography (FDG-PET) imaging may have affected staging accuracy. Survival analyses were based solely on Kaplan-Meier estimates and log-rank tests without multivariable adjustment; therefore, findings should be considered exploratory. The observed survival advantage in stage IIIC1 patients over stage IIIB patients may also reflect unmeasured treatment-related factors, warranting cautious interpretation.
Despite these limitations, this study has notable strengths. Treatment was relatively uniform, with all patients receiving EBRT and brachytherapy and most receiving concurrent chemotherapy. To our knowledge, this is the first study from Nepal to evaluate stage migration and prognostic performance of the FIGO 2018 staging system. Given limited access to advanced imaging modalities in many low- and middle-income countries, our findings provide valuable real-world evidence for similar resource-constrained settings.
Conclusions
In conclusion, our study reveals a substantial stage migration and its variable impact on prognostication when implementing the FIGO 2018 staging system in locally advanced cervical cancer. While heterogeneity among pelvic node–positive patients in stage IIIC appears evident, detailed stratified analyses were limited, and the observed impact of T stage should be interpreted cautiously. These findings underscore the potential importance of the T stage in prognostic evaluation and highlight the need for further studies to refine the staging system and improve its prognostic precision.
Footnotes
Acknowledgements
The authors acknowledge the support of Kathmandu Cancer Center. Minor language assistance was used during manuscript preparation, and any AI tools used for language suggestions have been verified by the authors for accuracy.
Ethical Considerations
This study received ethical approval from the Ethical Review Board of the Nepal Health Research Council (NHRC) (Approval No.: 655/2020P; approval date: April 29, 2021). The study was conducted in accordance with the principles outlined in the Declaration of Helsinki (1975, revised 2024).
Consent to Participate
The requirement for written informed consent was waived due to the retrospective nature of the study.
Consent for Publication
Not applicable.
Author Contributions
Conceptualization: Simit Sapkota, Subhas Pandit, Abish Adhikari, Sunil Shrestha
Methodology: Simit Sapkota, Subhas Pandit, Abish Adhikari, Anjani Kumar Jha, Sunil Shrestha
Data analysis and investigation: Jeebana Bhandari, Simit Sapokota
Writing original draft: Simit Sapkota, Jeebana Bhandari
Reviews and editing: Subhas Pandit, Anjani Kumar Jha, Abish Adhikari, Sunil Shrestha
Supervision: Subhas Pandit, Sunil Shrestha, Anjani Kumar Jha
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statements
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Informed Consent
Not applicable, as the study was retrospective and consent was waived by the NHRC.
