Abstract
Background:
Transarterial chemoembolization (TACE) is an established therapeutic modality for malignant hepatic tumors. This study assessed the clinical efficacy, adverse events, and factors associated with treatment outcomes of drug-eluting bead transarterial chemoembolization (DEB-TACE) in the treatment of hepatic malignancies.
Methods:
Clinical data were collected from 94 patients with hepatic malignancies who underwent DEB-TACE. Treatment response was assessed in accordance with the modified Response Evaluation Criteria in Solid Tumors (mRECIST). Postoperative complications and relevant hematological parameters were analyzed by reviewing clinical symptoms and laboratory/examination findings. Logistic regression analysis was used to identify factors associated with treatment outcomes.
Results:
Median follow-up duration was 29.0 months (range: 3.0-51.0 months). Treatment response assessment post-DEB-TACE revealed that 2 patients (2.1%) achieved a complete response (CR), 53 (56.4%) achieved a partial response (PR), 27 (28.7%) had stable disease (SD), and 12 (12.8%) developed progressive disease (PD). The overall objective response rate (ORR) was 58.5%, and the disease control rate (DCR) was 87.2%. Median progression-free survival (PFS) was 5.0 months (95% confidence interval [CI]: 3.7-6.3 months), and median overall survival (OS) was 15.0 months (95% CI: 7.6-22.4 months). Multivariate logistic regression analysis demonstrated that hematological parameters—including D-dimer levels, neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and C-reactive protein (CRP) levels—were associated with patient prognosis and survival outcomes.
Conclusions:
Drug-eluting bead transarterial chemoembolization is an effective treatment for various liver malignancies, associated with few manageable postoperative complications. In addition, d-dimer, the neutrophil-to-lymphocyte ratio (NLR), the platelet-to-lymphocyte ratio (PLR), and C-reactive protein (CRP) levels may predict DEB-TACE efficacy.
Keywords
Introduction
The incidence and mortality of malignant hepatic tumors, including primary liver cancer and hepatic metastases, continue to rise globally. The liver is also the most common site for distant tumor metastases, with hepatic metastases accounting for 30% to 40% of all clinically diagnosed malignant hepatic lesions; the most common subtypes are colorectal liver metastases (CRLM) and hepatic metastases from gastric cancer. 1 The clinical management of these heterogeneous hepatic tumors poses significant challenges to both clinical practice and public health.2,3 Despite the widespread use of systemic therapies—including chemotherapy, targeted therapy, and immune checkpoint inhibitors—in the management of primary liver cancer and hepatic metastases, their overall efficacy remains suboptimal, highlighting the need for further refinement. 4 Transarterial chemoembolization (TACE) has established itself as a well-validated, effective therapeutic modality for localized malignant hepatic tumors. 5 Initially developed for the management of hepatocellular carcinoma (HCC), TACE has seen substantial expansion in its clinical utility in recent years, with 2 major technical variants: conventional transarterial chemoembolization (c-TACE) and drug-eluting bead transarterial chemoembolization (DEB-TACE).6-8
However, conventional transarterial chemoembolization (cTACE) is associated with inherent limitations that restrict its clinical utility and efficacy. 9 The high fluidity of lipiodol results in rapid elution of encapsulated chemotherapeutic agents, thereby reducing the duration of therapeutic drug exposure and potentially compromising treatment outcomes. 10 Furthermore, the simple admixture of lipiodol with chemotherapeutic drugs results in rapid phase separation, which impairs the controlled, sustained release of therapeutic agents.11,12 In contrast to cTACE, drug-eluting bead transarterial chemoembolization (DEB-TACE) uses drug-eluting beads (DEBs) as embolic agents, a delivery system that prolongs intratumoral drug release, enhances local antitumor activity, and reduces chemotherapeutic toxicity to non-target tissues. 13 In recent years, a robust body of evidence has validated the clinical value of DEB-TACE for both primary and metastatic hepatic malignancies, particularly in achieving durable local tumor control in patients with intrahepatic cholangiocarcinoma (iCCA) and colorectal liver metastases (CRLM). 14
Although the clinical utility of DEB-TACE in malignant hepatic neoplasms is supported by a growing body of evidence, robust data remain limited regarding 3 critical unmet needs: heterogeneity in therapeutic efficacy, subtype-specific safety profiles, and standardized predictive/prognostic biomarkers across distinct histological subtypes of malignant hepatic neoplasms—specifically major subtypes including hepatocellular carcinoma (HCC), intrahepatic cholangiocarcinoma (iCCA), colorectal liver metastases (CRLM), and hepatic metastases from other solid tumors.15-17 Most existing studies have focused solely on single tumor subtypes, failing to provide a holistic analysis of unresectable malignant hepatic neoplasms across all histological categories. This research gap has hindered the establishment of a unified evidence-based framework to guide DEB-TACE regimen selection, prognostic stratification, and adverse event management in patients with different subtypes, thereby impeding the translation of precision medicine strategies into clinical practice.18,19 The present study therefore aims to systematically assess the efficacy, safety, and prognostic determinants of DEB-TACE in the management of diverse malignant hepatic neoplasms, with the objective of providing high-quality evidence-based guidance for the standardized and personalized treatment of patients with distinct histological subtypes.
Materials and Methods
Study design and patients
This study was approved by the Ethics Committees for Clinical Research of Jinling Hospital Affiliated to Nanjing University and the First Affiliated Hospital of Soochow University. All patients provided written informed consent for the use of their clinical data in this retrospective study. This study was conducted in accordance with the relevant guidelines of the EQUATOR Network. 20 A total of 94 patients with hepatic malignancies who underwent DEB-TACE at Jinling Hospital and the First Affiliated Hospital of Soochow University between January 2016 and October 2020 were enrolled. The inclusion criteria were as follows: (1) confirmed diagnosis of primary liver cancer, intrahepatic cholangiocarcinoma, or various types of secondary hepatic metastases based on clinical data, pathological findings, and imaging studies; (2) age 18 to 80 years; (3) Eastern Cooperative Oncology Group (ECOG) performance status 0 to 2 21 ; (4) Child–Pugh liver function class A or B 22 ; and (5) provision of written informed consent. The exclusion criteria were as follows: (1) history of liver transplantation; (2) severe hepatic or renal insufficiency or life-threatening infections; (3) uncorrectable coagulation disorders; (4) hypersensitivity to contrast media; (5) main portal vein obstruction without significant collateral circulation; (6) significant arteriovenous fistula; (7) pregnancy or lactation; (8) estimated life expectancy of less than 3 months; and (9) unmeasurable baseline lesions per the modified Response Evaluation Criteria in Solid Tumors (mRECIST). 23
Baseline data collection
Baseline data were collected for all patients, including the following: (1) demographic characteristics, such as age and sex; (2) medical history, including hepatitis B virus (HBV) infection, hepatitis C virus (HCV) infection, alcohol use history, and cirrhotic history; (3) prior treatment history, including conventional transarterial chemoembolization (cTACE), surgical resection, systemic chemotherapy, radiotherapy, and targeted therapy; (4) clinical features, including pathological subtype, number of DEB-TACE cycles, tumor location, size and distribution, venous invasion status, Child–Pugh liver function class, and Eastern Cooperative Oncology Group (ECOG) performance status; (5) routine hematological parameters, such as platelet count (PLT), D-dimer, C-reactive protein (CRP), neutrophil–lymphocyte ratio (NLR), and platelet–lymphocyte ratio (PLR); (6) hepatic function parameters, including albumin (ALB), total bilirubin (TBIL), alanine aminotransferase (ALT), and aspartate aminotransferase (AST); (7) renal function parameters, such as serum creatinine (Scr) and blood urea nitrogen (BUN); (8) tumor markers, including alpha-fetoprotein (AFP), carcinoembryonic antigen (CEA), and carbohydrate antigen 19-9 (CA 19-9); and (9) drug-eluting microsphere diameter and drug-loading dose.
Procedure of DEB-TACE treatment
Drug-loaded microspheres were prepared using the following methodology. Microsphere solution was aspirated with a 20-mL syringe, and the supernatant was discarded. Sterile water (3 mL) was used to reconstitute the injectable chemotherapeutic agent, which was then thoroughly mixed with the microspheres—with gentle agitation of the drug-loaded microspheres every 5 minutes. Following a 20-minute drug-loading incubation period, the final drug-loaded microsphere mixture was combined with contrast medium at a 1:1 volume ratio prior to use. For patients with hepatocellular carcinoma (HCC), microspheres were loaded with doxorubicin (40-80 mg). For those with intrahepatic cholangiocarcinoma (iCCA), microspheres were loaded with gemcitabine (200-600 mg). For patients with colorectal liver metastases (CRLM), microspheres were loaded with irinotecan (100-300 mg). Chemotherapeutic dosages were calculated based on each patient’s body surface area (BSA). For patients with an Eastern Cooperative Oncology Group (ECOG) performance status of 2, chemotherapeutic dosage was reduced to 50% of the standard dose. No dosage adjustment was required for patients with Child–Pugh class B hepatic function.
The specific operational procedure for DEB-TACE was as follows. Patients were placed in the supine position and received local anesthesia. Percutaneous femoral artery puncture was performed via the Modified Seldinger technique. With guidewire guidance, a 5-Fr RH catheter was selectively inserted into the hepatic artery for angiography to determine the location, size, and number of tumor-feeding arteries within the liver. If hepatic angiography failed to clearly demonstrate the tumor-feeding arteries, careful angiographic evaluation of the superior mesenteric artery, subphrenic artery, and right renal artery was performed until the tumor-feeding arteries were identified. Under digital subtraction angiography (DSA) guidance, the prepared drug-loaded microsphere suspension was slowly injected into the tumor-feeding arteries through a 2.4-Fr Progreat microcatheter (Terumo) using a pulsed injection method, until the embolization endpoint was achieved. Finally, repeat angiography was performed to confirm the post-embolization tumor staining status and ensure complete occlusion of the tumor-feeding arteries. Notably, a maximum of 4 DEB-TACE sessions were performed within 6 months. After the procedure, pressure dressing was applied to the arterial puncture site. Postprocedural complications were monitored and documented, and timely symptomatic treatment was administered as needed.
Postoperative assessment
One month after treatment, all patients underwent contrast-enhanced magnetic resonance imaging (MRI) to assess treatment response per the modified Response Evaluation Criteria in Solid Tumors (mRECIST). 24 The evaluation criteria were as follows: (1) Complete Response (CR): complete disappearance of arterial phase enhancement in all target lesions; (2) Partial Response (PR): ⩾ 30% reduction in the sum of the diameters of target lesions (assessed in the arterial phase); (3) Progressive Disease (PD): ⩾ 20% increase in the sum of the diameters of target lesions (assessed in the arterial phase) or the appearance of new lesions; and (4) Stable Disease (SD): reductions not meeting PR criteria or increases not meeting PD criteria. The objective response rate (ORR) was defined as the sum of CR and PR cases, while the disease control rate (DCR) was defined as the sum of CR, PR, and SD cases. Two experienced radiologists performed independent evaluations. Monthly follow-up was conducted, including assessments of relevant clinical symptoms, imaging studies, and hematological test results. The study follow-up concluded on December 31, 2023. Overall survival (OS) was calculated from the date of initial diagnosis to the date of death from any cause, while progression-free survival (PFS) was calculated from the date of DEB-TACE procedure to the date of disease progression.
Prior to the first DEB-TACE session and 1 month post-treatment, patients’ hepatic function—including serum albumin (ALB), total bilirubin (TBIL), alanine aminotransferase (ALT), and aspartate aminotransferase (AST) levels—was assessed to evaluate the impact of DEB-TACE on hepatic function. Adverse events (AEs), including pain, fever, nausea, vomiting, and myelosuppression, occurring intraoperatively and within 1-month post-treatment were documented. All AEs were graded using the validated National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE), Version 5.0. Adverse event grading was independently conducted by 2 senior interventional radiologists; any discrepancies were resolved via consensus discussion with a third experienced clinician to ensure the accuracy of the findings.
Statistics
Statistical analyses were performed using SPSS 22.0 software (IBM, USA) and GraphPad Prism version 7.00. Data were presented as means ± standard deviations, percentages (%), and medians (P25-P75). X-tile software was used to determine the optimal cutoff values for NLR and PLR, which were then used to stratify patients into 2 subgroups. Comparisons of outcomes at each follow-up visit were conducted using the Wilcoxon rank-sum test or McNemar’s test. Kaplan-Meier (K-M) curves were generated, and log-rank tests were employed to compare overall survival (OS) and progression-free survival (PFS). Univariate and multivariate logistic regression analyses were used to identify factors associated with objective response rate (ORR), while univariate and multivariate Cox proportional hazards regression models (stepwise forward selection) were used to evaluate factors related to OS. Two-tailed P < .05 was considered statistically significant.
Results
Selection and study descriptions
The study flowchart is illustrated in Figure 1. A total of 125 patients were initially screened; however, 2 patients with renal cell carcinoma, 2 with missing laboratory data, and 27 with incomplete imaging or treatment response assessment records were excluded. Ultimately, 94 patients with malignant hepatic tumors were included in the efficacy analysis, comprising 49 with hepatocellular carcinoma (HCC, 52.1%), 20 with intrahepatic cholangiocarcinoma (iCCA, 21.3%), and 25 with secondary hepatic malignancies (including colorectal, gastric, pancreatic, and lung cancer metastases, 26.6%). A total of 105 DEB-TACE procedures were performed to analyze treatment efficacy, safety, and treatment-related influencing factors.
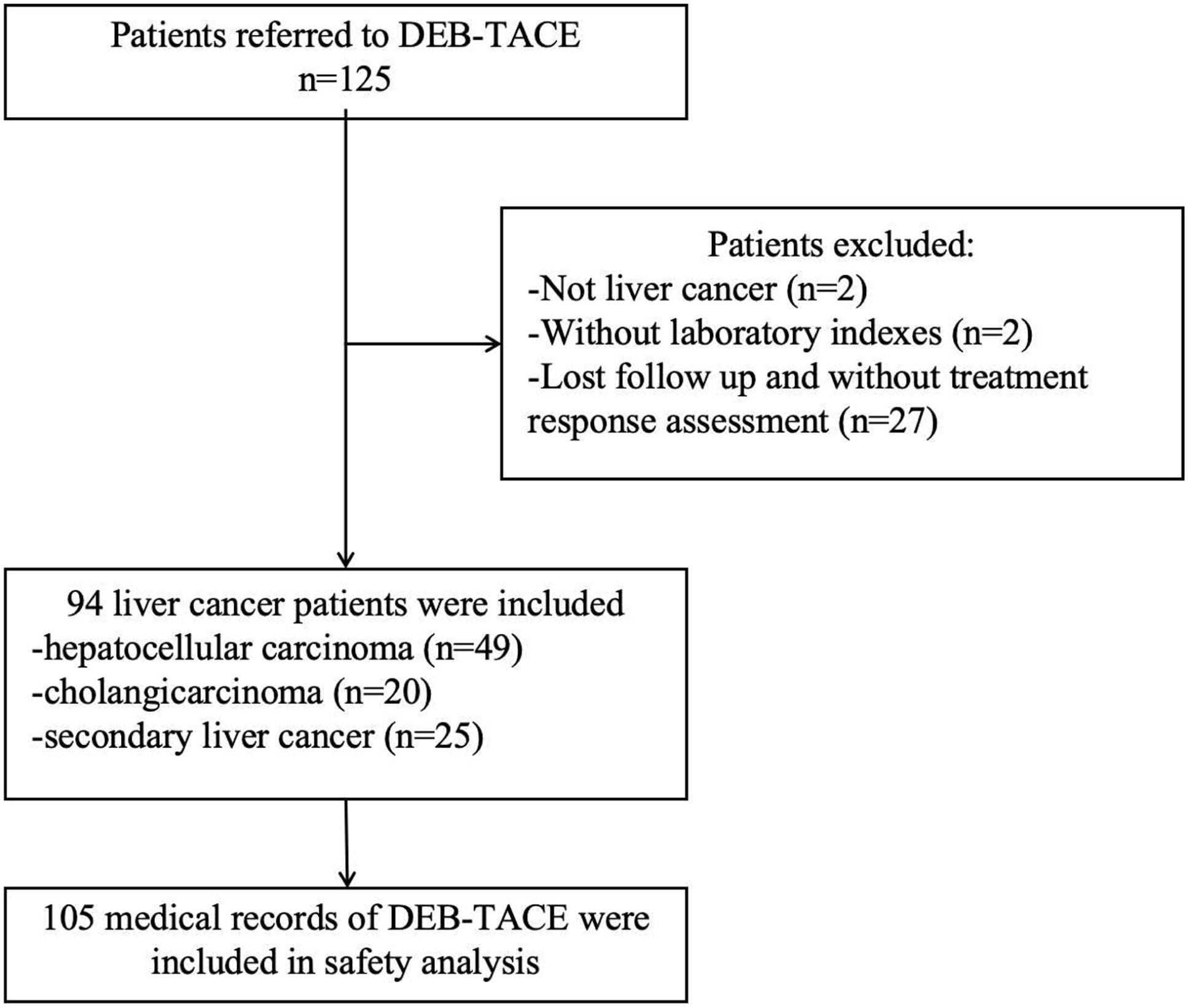
Summary flow chart of the study (DEB-TACE: drug-eluting bead transarterial chemoembolization).
Baseline characteristics of patients
Among the 94 patients with malignant hepatic tumors, the mean age was 60.86 ± 11.28 years, including 22 females (23.4%) and 72 males (76.6%). Of these patients, 69 had primary hepatic malignancies (49 with HCC and 20 with iCCA), while 25 had secondary hepatic malignancies. In addition, 50 patients (53.2%) had a history of HBV infection. Patients with a NLR ⩾ 3.1 and a PLR ⩾ 147.5 were stratified into the high-NLR and high-PLR groups, respectively. Baseline characteristics of all patients are summarized in Table 1, including demographic data, hepatic function classification, portal vein invasion status, intrahepatic lesion distribution, medical history, tumor markers, and other relevant indicators. Notably, this cohort included 1 patient (1.1%) with an Eastern Cooperative Oncology Group (ECOG) performance status of 2 and 11 patients (11.7%) with Child–Pugh class B hepatic function.
Analysis of baseline characteristics of patients in the study.

Data are presented as mean ± standard deviation, median (p25-p75), or percentage (%). Abbreviations used are as follows: HBV, hepatitis B virus; HCV, hepatitis C virus; HCC, hepatocellular carcinoma; ICC, intrahepatic cholangiocarcinoma; ECOG, Eastern Cooperative Oncology Group; PLT, platelets; CRP, C-reactive protein; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; ALB, albumin; TBIL, total bilirubin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BCr, blood creatinine; BUN, blood urea nitrogen; AFP, alpha-fetoprotein; CEA, carcinoembryonic antigen; CA19-9, carbohydrate antigen 19-9; DEB-TACE, drug-eluting bead transarterial chemoembolization; cTACE, conventional transarterial chemoembolization.
Efficacy: tumor response
Treatment response assessments per mRECIST are presented in Figure 2. Following DEB-TACE, 2 patients (2.1%) achieved complete response (CR)—including one with gastric cancer liver metastases and one with intrahepatic cholangiocarcinoma (iCCA) (Figure 3)—53 patients (56.4%) achieved partial response (PR), 27 patients (28.7%) had stable disease (SD), and 12 patients (12.8%) developed progressive disease (PD). The overall objective response rate (ORR) and disease control rate (DCR) were 58.5% and 87.2%, respectively. Additional prognostic analysis results are shown in Figure 4. Kaplan-Meier curves revealed a median progression-free survival (PFS) of 5.0 months (95% confidence interval [CI]: 3.7-6.3 months) and a median overall survival (OS) of 15.0 months (95% CI: 7.6-22.4 months). Subgroup analyses of treatment responses were further performed based on tumor subtypes, with detailed results summarized in Table 2.

Efficacy assessment results after DEB-TACE treatment for all patients. CR indicates complete response; PR, partial response; ORR, overall response rate; SD, stable disease; DCR, disease control rate; PD, progressive disease.
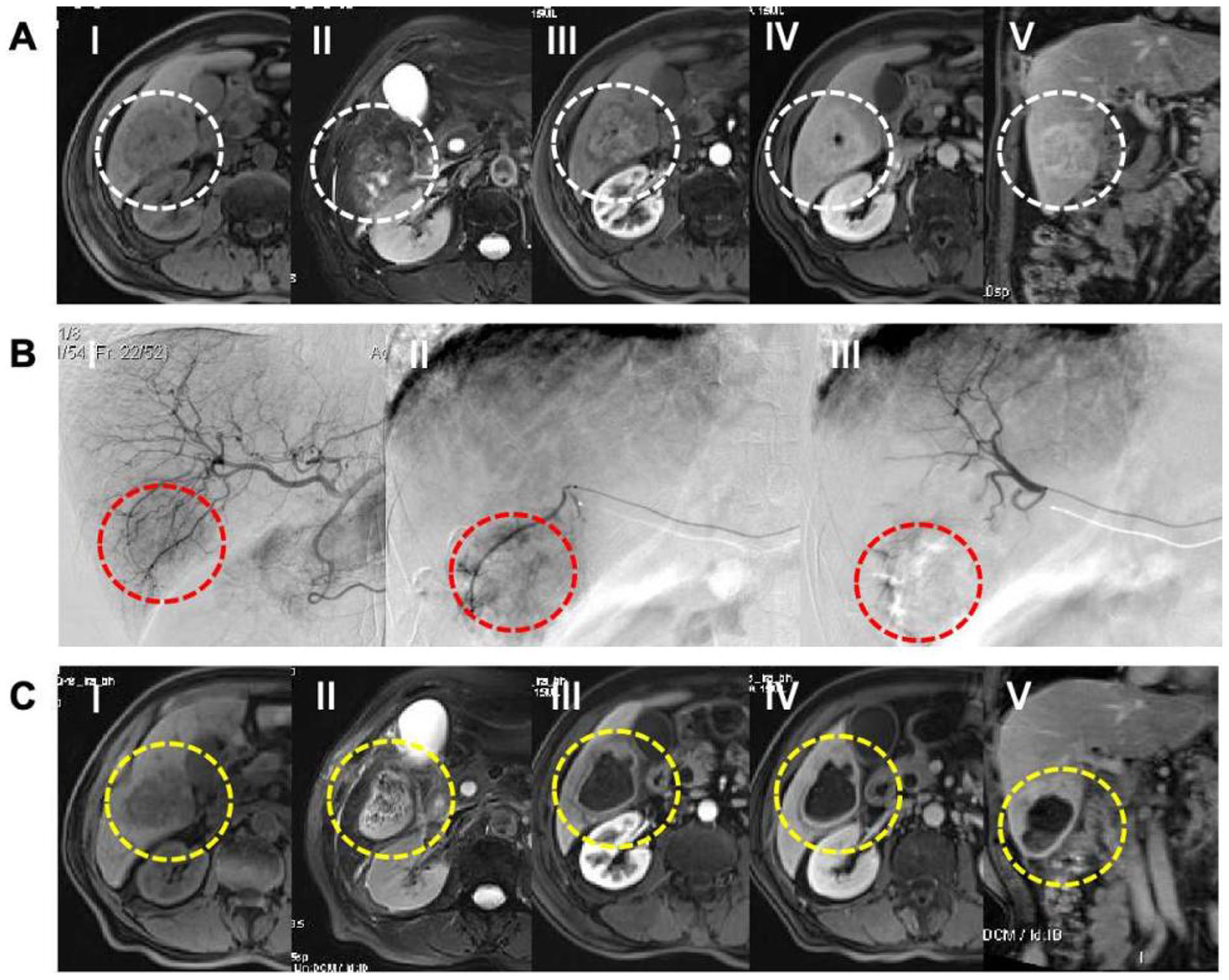
(A) Magnetic resonance imaging (MRI) of an intrahepatic cholangiocarcinoma prior to DEB-TACE treatment (I: T1-weighted imaging, II; T2-weighted imaging, III: MRI with contrast enhancement during the arterial phase, IV: MRI with contrast enhancement during the venous phase, V: MRI with contrast enhancement during the delayed phase). (B) Treatment of DEB-TACE for the intrahepatic cholangiocarcinoma. (I: Hepatic arteriography reveals the intrahepatic lesion located at the inferior margin of the right lobe of the liver. II: Super-selective hepatic arteriography shows the arterial supply to the tumor in the right lobe of the liver. III: Post-DEB-TACE treatment angiography shows the disappearance of blood supply to the intrahepatic lesion in the right lobe. (C) MRI after 1 month of DEB-TACE treatment for the intrahepatic cholangiocarcinoma (I: T1-weighted imaging, II: T2-weighted imaging, III: MRI with contrast enhancement during the arterial phase, IV: MRI with contrast enhancement during the venous phase, V: MRI with contrast enhancement during the delayed phase).

Kaplan-Meier survival curves for PFS and OS in all patients. The median PFS for all patients was 5.0 months [95% CI: 3.71 to 6.29 months], while the median OS was 15.0 months [95% CI: 7.57 to 22.43 months].
Comparison of therapeutic efficacy responses to DEB-TACE in different subtypes of liver malignancies (n = 94, %).

Efficacy was evaluated based on RECIST 1.1 criteria. Pearson’s chi-square test was used for overall group comparisons with a significance level of α = 0.05. The overall treatment response grades (CR/PR/SD/PD) were compared among all groups: χ2 = 3.854, df = 6, P = 0.696. Bonferroni correction was applied for pairwise comparisons among subgroups, with the corrected significance level α′ ≈ 0.0167. All pairwise comparison P values were greater than 0.0167, indicating no statistically significant differences.
Influencing factors: prognostic factors for ORR and OS
An analysis of factors associated with treatment efficacy was performed for patients who underwent DEB-TACE. Univariate logistic regression analysis identified several factors correlated with objective response rate (ORR) (Table 3). The results indicated that age ⩾ 65 years, alcohol use (defined as daily ethanol intake ⩾ 20 g for ⩾ 5 years), portal vein invasion, elevated neutrophil-to-lymphocyte ratio (NLR), and abnormal D-dimer levels were associated with a lower ORR (P < 0.05). Further multivariate logistic regression analysis, incorporating factors with P < 0.100, identified that age ⩾ 65 years, alcohol use, and abnormal D-dimer levels were independent risk factors for a lower ORR (Table 3).
Factors affecting ORR by logistic regression model analysis.

Data are presented with P values, odds ratios (OR), and 95% confidence intervals (CI). Univariate logistic regression analysis was used to identify factors influencing the overall response rate (ORR). Factors with P < 0.100 underwent multivariate logistic regression analysis. P < 0.05 was considered statistically significant. Liver cancer was categorized as 0 for primary liver cancer and 1 for secondary liver cancer; tumor location was scored as 0 for unilobar and 1 for bilobar.
To investigate the impact of baseline factors on overall survival (OS), a Cox proportional hazards regression model was employed, with results summarized in Table 4. Univariate Cox regression analysis identified several predictors of poor OS: age ⩾ 65 years, larger drug-eluting microsphere diameter, higher Eastern Cooperative Oncology Group (ECOG) performance status, maximum tumor nodule size ⩾ 5 cm, bilobar hepatic involvement, portal vein invasion, prior systemic chemotherapy, abnormal C-reactive protein (CRP) levels, elevated neutrophil-to-lymphocyte ratio (NLR), elevated platelet-to-lymphocyte ratio (PLR), abnormal D-dimer levels, abnormal carbohydrate antigen 19-9 (CA19-9) levels, and abnormal total bilirubin (TBIL) levels. A multivariate Cox proportional hazards regression model was subsequently applied, incorporating factors with P < .05 from the univariate analysis, which revealed that age ⩾ 65 years, portal vein invasion, abnormal CRP levels, abnormal CA19-9 levels, and abnormal TBIL levels were independent risk factors for poor OS.
Factors affecting OS by Cox’s proportional hazards regression model.

Data are presented with P values, hazard ratios (HR), and 95% confidence intervals (CI). Factors influencing OS were analyzed using univariate Cox regression. Factors with P < 0.050 underwent multivariate Cox regression analysis. P < 0.050 was considered statistically significant. Liver cancer was categorized as 0 for primary liver cancer and 1 for secondary liver cancer, while tumor location was scored as 0 for unilobar and 1 for bilobar.
Liver function and AEs following DEB-TACE
Hepatic function data from 105 DEB-TACE procedures were analyzed. Among patients with normal baseline hepatic function parameters, serum albumin (ALB), alanine aminotransferase (ALT), and total bilirubin (TBIL) levels slightly increased post-treatment, but these increases were not statistically significant. Serum aspartate aminotransferase (AST) levels decreased compared with baseline values, though this difference also did not reach statistical significance (P > .05, Table 5). In addition, analysis was performed for patients with abnormal baseline hepatic function parameters. Post-DEB-TACE, AST levels decreased significantly (P = .001), whereas ALB, ALT, and TBIL levels did not differ significantly from pretreatment values (P > .05, Table 6). Subgroup analyses of hepatic function parameters before and after DEB-TACE were additionally performed based on tumor subtypes, with detailed results summarized in Table 7.
Liver function status before and after DEB-TACE treatment.

Data are presented as counts/total (%). The Wilcoxon rank-sum test was used for continuous variables, while the McNemar test was used for categorical variables. P < 0.05 was considered statistically significant.
Analysis of liver function indicators before and after DEB-TACE treatment in patients with abnormal liver function.

Data are presented as median (p25-p75). The Wilcoxon rank-sum test was used to compare differences at each visit. The P < 0.05 was considered statistically significant.
Comparison of liver function before and after DEB-TACE treatment in different subtypes of liver malignant tumors.

Adverse events (AEs) occurring intraoperatively and within 1 month postprocedure in the 94 patients are summarized in Table 8. Intraoperatively, pain was reported in 76 patients (72.3%), fever in 34 patients (32.3%), and nausea and vomiting in 22 patients (20.9%). One month postprocedure, only 16 patients (15.2%) reported pain, with no other AEs observed. Among patients with an Eastern Cooperative Oncology Group (ECOG) performance status of 2, no significant adverse reactions occurred after the procedure. Patients with Child–Pugh class B hepatic function recovered well postprocedure, with no significant deterioration in hepatic function. Overall, most AEs were mild and manageable. In addition, a comparative analysis of adverse reactions before and after DEB-TACE was performed based on tumor subtypes, with detailed results summarized in Table 9. All postoperative complications were graded in accordance with the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0.
Intraoperative and postoperative AEs of DEB-TACE.

Data were presented as count (%).
Comparison of adverse reactions in patients with different subtypes of liver malignant tumors after receiving DEB-TACE treatment.

The efficacy assessment was based on the CTCAE 5.0 standard; the Pearson chi-square test was used for intergroup comparison, with the test level α = 0.05; “-” indicates no intergroup difference and no statistical test was required.
Discussion
This study performed a retrospective analysis of patients with unresectable malignant hepatic tumors who underwent DEB-TACE at the participating institutions between January 2016 and October 2020, with the primary objective of assessing the clinical efficacy and safety of this intervention. Our findings demonstrate that DEB-TACE is a feasible and effective therapeutic option for advanced malignant hepatic tumors across diverse histological subtypes. Furthermore, the application of drug-eluting beads in DEB-TACE can effectively control intrahepatic tumor progression while mitigating systemic chemotherapy-related toxicity, which may contribute to improved survival outcomes in this patient population.25,26
In the present study, we evaluated patients with malignant hepatic tumors who underwent DEB-TACE at the participating institutions. Analysis of 94 patients revealed an overall objective response rate (ORR) of 58.5% and a disease control rate (DCR) of 87.2%, with a median overall survival (OS) of 15.0 months (95% confidence interval [CI]: 7.6-22.4 months). These results are generally consistent with those reported in previous literature. For instance, Zhou et al 27 reported that in a study of 88 patients with unresectable intrahepatic cholangiocarcinoma (iCCA) treated with DEB-TACE using doxorubicin-loaded CalliSpheres, the 1-month ORR assessed by mRECIST criteria was 65.9%. In addition, a retrospective study involving 39 Chinese patients with malignant hepatic tumors reported an ORR of 61.5% when CalliSpheres were used for DEB-TACE. 28 These findings further confirm the efficacy of DEB-TACE in the treatment of unresectable malignant hepatic tumors, underscoring its clear clinical value. In terms of treatment response classification, our study observed a complete response (CR) rate of 2.1%, partial response (PR) rate of 56.4%, stable disease (SD) rate of 28.7%, and progressive disease (PD) rate of 12.8%. In contrast, an Italian multicenter trial investigating HepaSphere microspheres for DEB-TACE in hepatocellular carcinoma (HCC) patients reported 1 month CR, PR, and SD rates of 48%, 36%, and 16%, respectively. 29 Although the CR rate in our study was relatively lower, the high PR and SD rates coupled with a low PD rate indicate that DEB-TACE can effectively control tumor progression and enable tumor-bearing survival in patients. Regarding survival outcomes, a Hong Kong study involving 143 HCC patients reported a median OS of 12.53 months over a 9-year follow-up period in those treated with DEB-TACE. 30 Another study of 275 HCC patients in Zhejiang Province who received DEB-TACE with CalliSpheres reported a median OS of 380 days (95% CI: 389-390 days). 31 Compared with these previously reported OS data, DEB-TACE achieved comparable efficacy in treating diverse malignant hepatic tumor subtypes in our study, further highlighting its potential to improve the prognosis of patients with malignant hepatic tumors.
Further exploration of factors associated with DEB-TACE efficacy in malignant hepatic tumors may facilitate the identification of novel prognostic biomarkers and the formulation of optimized treatment strategies. Previous research has indicated that cancer patient prognosis depends not only on tumor-related factors but also on the patient’s overall immune status. 32 Various peripheral inflammatory cells and disease-related factors have potential as prognostic indicators for survival in patients with malignant hepatic tumors.33-37 In the present study, analysis of factors influencing ORR and OS revealed that multiple baseline characteristics and peripheral blood inflammatory indicators were associated with treatment response and survival duration. Specifically, age ⩾ 65 years, alcohol use, portal vein invasion, elevated NLR, and abnormal D-dimer levels were identified as predictors of poor ORR. Further multivariate analysis confirmed that age ⩾ 65 years, alcohol use, and abnormal D-dimer levels were independent risk factors for reduced ORR. For OS, univariate analysis identified several factors associated with poor prognosis: age ⩾ 65 years, larger drug-eluting microsphere diameter, higher ECOG performance status, maximum lesion diameter ⩾ 5 cm, bilobar hepatic involvement, portal vein invasion, prior systemic chemotherapy, abnormal CRP levels, elevated NLR, elevated PLR, abnormal D-dimer levels, abnormal CA19-9 levels, and abnormal total bilirubin (TBIL) levels. Among these, age ⩾ 65 years, portal vein invasion, abnormal CRP levels, abnormal CA19-9 levels, and abnormal TBIL levels were confirmed as independent risk factors for poor OS. Comprehensive analysis of these findings demonstrated that multiple peripheral blood indicators—particularly D-dimer, NLR, PLR, and CRP—are closely associated with prognosis and survival in patients with malignant hepatic tumors undergoing DEB-TACE, suggesting their potential clinical utility as prognostic tools. The correlation between inflammatory indicators and prognosis is consistent with conclusions from previous studies.38,39 In addition, larger drug-eluting microsphere diameter was identified as a predictor of poor OS, which may be attributed to the fact that larger embolic microspheres are more likely to deposit in the main branches of tumor-feeding arteries, limiting penetration to distal vessels and thus preventing complete embolization. This is particularly relevant for hypovascular hepatic metastases, where incomplete embolization may contribute to the development of DEB-TACE resistance and subsequent disease progression.
However, in the assessment of postprocedural complications, the present study found no significant differences between baseline hepatic function parameters and those measured 1 month postprocedure. Furthermore, we observed that patients with slightly elevated baseline AST levels exhibited a reduction in AST levels at the 1-month follow-up after DEB-TACE. This finding may be attributed to the selective and precise embolization of tumor-feeding arteries during the procedure, which minimizes hepatic parenchymal damage. In addition, the observed posttreatment reduction in AST levels—achieved through effective tumor control—indicates favorable hepatic tolerance to DEB-TACE. Patients not only did not experience significant hepatic function impairment postprocedure, but the effective antitumor response also appeared to contribute to the restoration of hepatic function. Regarding other postprocedural complications, only a small number of patients reported mild AEs, such as mild pain, nausea, vomiting, and fever, most of which resolved significantly following symptomatic management. This is consistent with previous studies reporting common AEs in patients with malignant hepatic tumors after DEB-TACE.40-42 All these complications are considered clinically manageable. Furthermore, in the subgroup analyses of different tumor types, there were no significant statistical differences in liver function indicators and adverse reactions among the various groups. Collectively, these findings support that DEB-TACE represents a relatively safe therapeutic option for malignant hepatic tumors, particularly in patients with compromised hepatic function. Given that only 1 patient with an Eastern Cooperative Oncology Group (ECOG) performance status of 2 and 11 patients with Child–Pugh class B hepatic function were included in the study, it was not feasible to conduct further subgroup analyses of postprocedural complications in these populations.
This study also has several limitations. First, as a retrospective study, no sample size calculation was performed due to the limited availability of historical cases. Furthermore, the relatively small sample size restricted the analysis of certain key factors in the univariate regression analysis, and the multivariate regression analysis may be susceptible to overfitting due to the large number of included variables. Thus, future studies with larger sample sizes are warranted to validate the current findings. Second, the retrospective design of this study—encompassing diverse tumor subtypes, different chemotherapeutic agents, and predominantly advanced-stage tumors—may have introduced biases into the analysis. Third, the absence of a control group precluded direct comparison of DEB-TACE efficacy and safety with other treatment modalities (eg, surgical resection or systemic therapy). Notably, this study performed comparative analyses in a heterogeneous patient cohort. Hepatocellular carcinoma (HCC) is typically hypervascular and relatively sensitive to embolization and chemotherapy, whereas intrahepatic cholangiocarcinoma (iCCA) has abundant stroma and heterogeneous vascularity, which limits drug penetration and embolization efficacy. The treatment response of hepatic metastases depends on the biological characteristics of the primary tumor. These 3 tumor types exhibit significant differences in biological behavior and vascular features, which may lead the overall efficacy data (eg, ORR and DCR) of the mixed cohort to mask the true therapeutic effects of DEB-TACE for specific subtypes when compared with single-HCC studies, failing to accurately reflect subtype-specific differences in treatment response. Therefore, conclusions from cross-sectional comparisons in this study can only serve as a reference for overall trends and cannot directly infer the relative efficacy of DEB-TACE across different tumor subtypes. Further targeted studies focusing on individual tumor subtypes are required to verify these findings.
Conclusions
In conclusion, the findings of this study demonstrate that DEB-TACE effectively controls the progression of malignant hepatic tumors, particularly in patients with advanced disease who are ineligible for surgical resection. Thus, DEB-TACE serves as a valuable therapeutic option for the clinical management of malignant hepatic tumors. In addition, the identification of poor prognostic indicators—including D-dimer, neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and C-reactive protein (CRP) levels—holds promise for clinical efficacy prediction and risk stratification, thereby offering improved treatment prospects for patients with advanced malignant hepatic tumors. Future research should focus on further exploring the utility of DEB-TACE in diverse patient cohorts and assessing its combination with other therapeutic modalities to further improve the overall survival and quality of life of patients with malignant hepatic tumors who are unable to undergo surgical resection.
Footnotes
Ethical Considerations
This study has been approved by the ethics committees of the First Affiliated Hospital of Soochow University (Ethical Review No. 708 of 2025) and Jinling Hospital Affiliated to Nanjing University (No. 2025DZKY-021-01).
Author Contributions
Li Liu, Yuling Sheng, and Xiaoli Zhu designed the study and wrote the draft of the manuscript. Bo Chen, Tantan Xu, Min Ai, Husheng Shan, Li Dong, and Jinjing Tang collected the clinical dates and follow-up. Xianxian Liang, Tao Yang, and Dazhi Gao analyzed the experimental and clinical data. Longjiang Zhang, Donghong Shi, and Xiaoli Zhu supervised the overall study and revised the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We greatly appreciate the financial support from the National Natural Science Foundation of China, China (82302321).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
