Abstract
Background:
Understanding the patient experience, including symptoms and impacts, of cancer-related cachexia is an important step in developing and validating patient-reported outcome measures that can support the subsequent development of new treatments. The Functional Assessment of Anorexia-Cachexia Therapy–5-item anorexia-related symptoms scale (FAACT-5IASS) is an outcome measure that may be suitable for the assessment of cachexia in patients with cancer. The objective of this analysis was to better understand the patient experience of cachexia based on qualitative interviews with patients.
Methods:
Concept elicitation interviews were conducted with patients with cancer and cachexia to understand key symptoms and their impacts. Outputs from the interviews were also used to confirm the relevance and content validity of the FAACT-5IASS for characterizing the identified cachexic symptoms.
Results:
Patients (N = 20) with non–small-cell lung (n = 8), pancreatic (n = 5), or colorectal (n = 7) cancer participated in interviews. Patients identified lack of appetite, nausea (and vomiting when describing nausea), and fatigue as key symptoms, although nausea and vomiting were primarily attributed to chemotherapy rather than specific to cachexia. These symptoms were reported as the most bothersome with negative impacts on appearance, physical function, social activities, and moods/emotions. Post-hoc analysis of the interviews supported the relevance of 4 out of 5 concepts in the FAACT-5IASS (having good appetite, satiety, food tasting unpleasant, and interest in food when eating).
Conclusions:
This qualitative research identified the importance of assessing key symptoms and impacts of cachexia using patient-reported measures that are conceptually relevant.
Keywords
Introduction
Cachexia is an underestimated, underdiagnosed, and undertreated feature of many chronic medical conditions, including cancer. 1 Cancer-related cachexia is characterized by body weight loss, specifically of skeletal muscle and sometimes adipose tissue, metabolic dysfunction, and reduced appetite, and is highly associated with cancers of the pancreas, esophagus, stomach, lung, liver, and bowel.2,3 The incidence of cachexia is dependent on tumor location and stage and varies from approximately 20% in breast and prostate cancers up to 60% to 70% in gastroesophageal and pancreatic cancers.4-6 Overall, cachexia may be directly responsible for up to 20% to 30% of cancer deaths.3,4
Addressing cachexia is vital to improving patient outcomes. As well as being a direct cause of death, cachexia also negatively impacts cancer prognosis, effective anti-cancer treatment, including both surgery (increased complications) and drug therapy (shorter treatment times, lower response rates), activities of daily living, and patient quality of life.7-10 Treatment of cachexia requires a multitargeted and multidisciplinary approach to meet the physiological and psychological needs of the patient. 10 These may include nutritional and metabolic interventions and pharmacological therapies, as well as support to counter any dysfunction in the emotional or social aspects of eating. 10 However, no fully effective interventions are currently available, and no pharmacological interventions have been established as a current standard of care.11,12 Therefore, the management of cachexia remains an unmet need in the overall treatment of cancer.
Describing and understanding the patient’s experience of cachexia is a key step in capturing the patient perspective and subsequently developing interventions that improve patient outcomes.13,14 Concept elicitation, a series of semi-structured, open-ended questions delivered in an interview setting, captures items of importance to patients, either spontaneously or with additional probing.15,16 Insights from the patient experience can be used to develop patient-reported outcome measures (PROMs) that document key features or symptoms, their impact, and how these change over time.17,18 After content validation, the PROM can be used in the clinical trial setting as a measure of efficacy to aid in the development of novel pharmacological therapies. 15 Indeed, this aligns with the United States (US) Food and Drug Administration (FDA) Patient-Focused Drug Development guidance documents 1 to 4, as well as FDA recommendations for the use of PROMs.19,20 The Functional Assessment of Anorexia-Cachexia Therapy—anorexia cachexia subscale (FAACT ACS) is a PROM consisting of 12 items,21,22 of which 5 items directly assess the symptoms of anorexia/cachexia (FAACT 5-item anorexia-related symptoms scale, or FAACT-5IASS). 22 The FAACT ACS has been validated in different cancer types23,24 and used in clinical trials to assess novel treatments for cachexia, including the FAACT-5IASS subscale alone.25-27
Here, we report the results of research conducted to better understand the patient experience of cachexia. A targeted literature and instrument review and clinician interviews were conducted to identify potential concepts of interest, inform a preliminary conceptual model, and support development of the concept elicitation interview guide. Concept elicitation interviews were then used to characterize the key symptoms and their impacts in patients with cachexia across different tumor types. Finally, we conducted a post-hoc analysis of the output of the concept elicitation interviews to confirm the relevance and content validity of the concepts in the FAACT-5IASS.
Methods
This article followed the EQUATOR Network Standards for Reporting Qualitative Research (SRQR) guidelines. 28 The interviews were conducted between January 2019 and February 2019, and the study duration was from June 2018 to February 2020.
Targeted literature and instrument review
A search of PubMed was conducted on June 25, 2018, to identify literature of interest since 2007 on key symptoms and impacts of cachexia or instruments used to assess cachexia (Supplementary Table 1). The titles and abstracts of returned articles were reviewed to determine eligibility (no eligibility criteria were predefined), followed by a full review of identified articles. The purpose of this search was to collate information to be used to develop a conceptual model to guide the patient concept elicitation interviews.
Clinician interviews
To participate in the interviews, clinicians had to be a board-certified oncologist, have been practicing as a board-certified oncologist for at least 2 years; involved in the direct care of cancer patients, including non–small-cell lung (NSCLC), pancreatic, colorectal, or ovarian cancer, at least 50% of the time; and be treating at least 8 patients with cachexia in a typical month. In total, 3 clinicians were identified and recruited. Semi-structured interviews were conducted by telephone by 2 interviewers and lasted approximately 60 minutes. The clinicians were asked to provide verbal consent to record the interview before being asked general questions to get them talking about their experiences of treating patients with cachexia. These general questions were followed by more targeted questions regarding the key symptoms and impacts of cachexia based on the clinicians’ experience treating this patient population.
Concept elicitation
Individual telephone interviews were conducted with patients with clinician-confirmed cachexia to obtain information on key symptoms and their impact. Patients were identified via physician referrals as well as patient databases and evaluated for eligibility. Inclusion criteria were aged ⩾18 years; diagnosis of NSCLC, pancreatic, or colorectal cancer; diagnosis of cachexia based on either unintentional weight loss ⩾5% over the past 6 months, or body mass index (BMI) < 20 kg/m2 and any degree of weight loss >2%; and ability to provide verbal consent, speak English, and participate in a 60-minute telephone interview. The exclusion criteria were weight loss due to simple starvation or physical challenges with eating or food absorption, such as bowel obstruction, or due to malnutrition caused by discomfort in eating, such as stomatitis from chemotherapy. In addition to the inclusion and exclusion criteria above, the following targets were used to ensure a representative and demographically diverse sample: at least 5 patients with each type of cancer; at least 4 males; at least 4 patients whose highest education level was a high school degree; and at least 4 patients who were not Caucasian/White.
To ensure consistency across interviews, a semi-structured interview guide (Supplementary material) was developed based on the results of the literature and instrument review and the clinician interviews. Interviews were conducted by 2 interviewers, lasted approximately 60 minutes, and were recorded and subsequently transcribed. Patients were asked open-ended questions to identify key spontaneously reported symptoms, followed by more targeted questions to identify the relative importance of symptoms. In addition, probe questions informed by the literature or clinician interviews were asked to identify other symptoms that were not spontaneously reported. Finally, patients were asked to report their most bothersome symptoms and to provide descriptions of the impacts of cachexia on daily life.
Analysis of qualitative data
The analysis of the qualitative data from the clinician interviews and concept elicitation interviews with patients was performed with researcher impartiality and a systematic approach. A deductive framework aligned with the themes outlined in the interview guides was utilized to accurately reflect the findings. Two analysts analyzed the data, one to spearhead the transcript analysis with a second to validate and confirm the accuracy of the results. Data tables were constructed to capture the content of individual interviews and to compare findings across all interviews. To protect patient privacy, illustrative quotes from the patient interview were anonymized.
Post-hoc content validation of item concepts in the FAACT-5IASS
Concept elicitation interview transcripts were used to confirm the relevance and validity of the FAACT-5IASS items. Transcripts were analyzed by 2 trained analysts. Fifteen percent of transcripts were reviewed by both analysts, and a third analyst confirmed analytical consistency and adjudicated over any conflicting analysis. Applying the deductive framework, transcripts were analyzed for relevance to the following 5 items: “I have a good appetite”; “My interest in food drops as soon as I try to eat”; “I have difficulty eating rich or ‘heavy’ foods”; “When I eat, I seem to get fully quickly”; and “Most food tastes unpleasant to me.” Patients could produce relevant responses either spontaneously or upon probing. Analysts recorded whether items were supported by interview transcripts and extracted supporting quotes.
Statistical analysis
This study was qualitative, and no statistical analyses were conducted.
Results
Targeted literature and instrument review and clinician interviews
The literature and instrument review of PubMed identified 38 articles for full review, of which 19 were excluded as they did not contain relevant information. Of these, 4 studies appeared to describe key symptoms and impacts of cachexia from the patient perspective and 15 identified clinical outcome assessment measures of symptoms or health-related quality of life in patients with cancer cachexia. An additional 4 articles describing key symptoms and impacts of cachexia were identified from the review of the bibliographies of included articles. These additional 4 articles were included. The most commonly reported food or appetite-related symptoms were change in appetite (8/8), including loss or reduction in appetite (8/8), and early satiety (4/8). Other symptoms included food aversion (4/8), change in food preference (2/8), change in taste (1/8), and change in food experience (3/8). Weight loss (4/8) and change in physical appearance (4/8) were the most commonly reported physical issues symptoms. Impacts of cachexia included physical impacts (inability to perform usual daily activities [2/8] and loss of independence [1/8]), psychological impacts (distress/worry about physical appearance [4/8], change in appetite [2/8], and prognosis [4/8]), and social impacts (isolation [4/8], limitation in social activities [1/8], and limited social interaction due to changes in food preferences or difficulty eating [1/8]). Similar to the literature review, interviewed clinicians (n = 3) all reported loss of appetite, weight loss, loss of muscle mass, anorexia, and early satiety as key symptoms. Lack of appetite was reported as the most important symptom with the negative impact of cachexia on activity levels and activities of daily living considered the most important. Combined results from the literature and instrument review and clinician interviews were used to develop a conceptual model to guide the patient concept elicitation interviews (Figure 1).

Conceptual model of cancer cachexia. The model was developed following the literature and instrument review and clinician interviews to guide the conduct of the patient interviews in the concept elicitation.
Concept elicitation patient characteristics
A total of 20 patients were interviewed from across the US (California, Arizona, Georgia, Colorado, Illinois, Pennsylvania, Utah, and Florida; Table 1). Mean age was 50.9 years (range 37-68 years), and most (60%) were female. Eight (40%) participants had NSCLC, 5 (25%) had pancreatic cancer, and 7 (35%) had colorectal cancer. The most common physician-reported cancer stage was stage III (40%), and most patients had Eastern Cooperative Oncology Group (ECOG) performance score 2 (45%). Of these 20 patients, 85% were receiving chemotherapy for their cancer, and 95% were receiving nutritional support for their cachexia.
Characteristics of the study population, including BMI classification and weight loss by BMI.

BMI: body mass index; ECOG PS: Eastern Cooperative Oncology Group performance status; NSCLC: non–small-cell lung cancer.
Patients had NSCLC, pancreatic, or colorectal cancer.
Patients were able to select more than 1 option.
Mean BMI at time of cancer diagnosis was 28.5 kg/m2, and current mean BMI was 22.7 kg/m2 (Table 1). At the time of cancer diagnosis, most patients (55%) were overweight (BMI 25.0-29.9 kg/m2), and 20% were obese (BMI ⩾ 30.0 kg/m2). Mean weight loss since cancer diagnosis was 20%, with the highest average weight loss (23%) in those classed as overweight at diagnosis. Based on the Weight Loss Grading System, which determines weight loss from the current BMI and amount of weight loss since cancer diagnosis, 29 85% of patients were classified in the most severe weight loss category (grade 4) at the time of interview.
Most (n = 12) patients were familiar with the term “cancer-related cachexia” but rarely used “cachexia” when describing their weight loss. The most common preferred terms were “weight loss,” “weight loss due to chemotherapy” or “weight loss due to cancer.” Eleven of the 20 patients experienced weight loss prior to cancer diagnosis or before starting cancer treatment but described it as insignificant or barely noticeable. All patients reported noticeable weight loss upon initiation of chemotherapy and described it as “accelerated,” “drastic,” or “noticeable.” Most patients reported initiating discussions with their physician about their cachexia. All but one of the patients reported using nutritional support to manage cachexia, but that even when combined with prescribed medications it was not effective in controlling their weight loss.
Reporting of cachexia symptoms
Patients consistently reported nausea (n = 20; 19 spontaneously and 1 after probing), change in appetite (n = 20; 15 spontaneously and 5 after probing), and fatigue (n = 19; 11 spontaneously and 8 after probing) as key symptoms of cachexia and treatment-related effects (Figure 2A). Illustrative quotes from patients on the description, severity, timing, and attribution to cancer treatment of nausea, change in appetite, and fatigue are shown in Table 2. Reports of vomiting were often included when reporting nausea, and 8 patients explicitly reported this symptom spontaneously. Notably, patients overwhelmingly attributed their nausea (and vomiting) to cancer treatment, which changed in response to the timing and duration of chemotherapy. Other reported symptoms included taste alteration (n = 14; 5 spontaneously and 9 after probing), early satiety (n = 14; 1 spontaneously and 13 after probing), changes in the experience of eating (n = 14; 1 spontaneously and 13 after probing), and changes in food preferences (n = 11; all after probing). Physical issues and physical difficulties with eating were much less commonly reported or were not reported to be a significant contributor to cachexia. Nausea was reported as the most bothersome symptom, followed by change in appetite, and then fatigue (Table 3). Illustrative quotes on the degree of bothersome for the key symptoms are shown in Table 4.
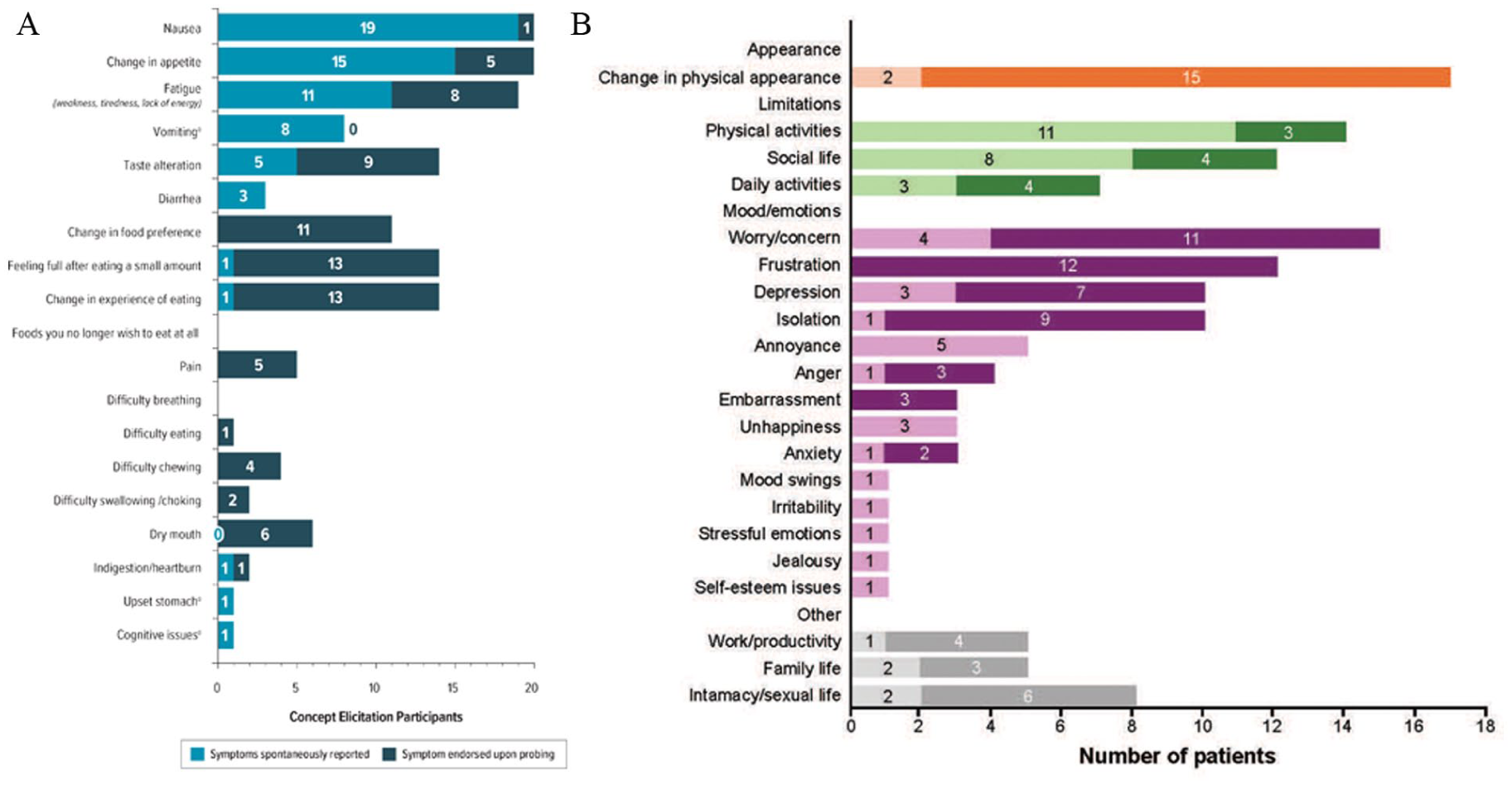
Symptoms and impacts reported by study participants. (A) Symptoms and (B) impacts were reported either spontaneously or upon probing. Panel B: Lighter colors are impacts reported spontaneously, darker colors are those reported upon probing. For the symptoms of vomiting, upset stomach, and cognitive issues, and the impacts of unhappiness, mood swings, irritability, stressful emotions, jealousy, and self-esteem issues, specific concepts were not included in probes; therefore, all reports were spontaneous. Change in physical appearance was probed in the discussion of symptoms but is reported as an impact.
Illustrative quotes for the key symptoms of nausea, change in appetite, and fatigue reported by patients during concept elicitation.

Ranking of the most bothersome symptoms of cachexia.

Of the 20 patients in the concept elicitation interviews, 19 provided input on their most bothersome symptom, and 14 also provided input on their second most bothersome symptom.
Although not considered a sign or symptom of cancer-related cachexia, this concept has been included to reflect patient report.
Illustrative quotes on the degree of bothersome of cachexia symptoms.

The most frequently reported physical and social impacts of cachexia on patients’ lives were changes in physical appearance (n = 17; 2 spontaneously and 15 after probing), limitations in physical activities (n = 14; 11 spontaneously and 3 after probing), and limitations in social activities (n = 12; 8 spontaneously and 4 after probing) (Figure 2B). These impacts were most frequently attributed to weight loss, muscle loss, and fatigue. Cachexia also had an impact on patient moods and emotions, including worry/concern (n = 15; 4 spontaneously and 11 after probing), frustration (n = 12; all after probing), depression (n = 10; 3 spontaneously and 7 after probing), and isolation (n = 10; 1 spontaneously and 9 after probing). Nine patients described fear and lack of control over further weight loss as the hardest part of having cachexia. Illustrative quotes on the impacts of cachexia are shown in Table 5.
Illustrative quotes on the impacts of cachexia.

Content validation of the FAACT-5IASS
The results from the concept elicitation interviews (N = 20) were used to confirm the relevance and content validity of the FAACT-5IASS concepts for characterizing appetite loss related to patients’ cachexia (Table 6). Nearly all patients (n = 18; 90%) described having or previously having good appetite. In contrast, since diagnosis most patients described getting full quickly (n = 15; 75%) and food tasting unpleasant to them (n = 14; 70%). Fewer patients described their interest in food dropping as soon as they try to eat (n = 6; 30%) or having difficulty eating rich or heavy foods (n = 1; 5%). Illustrative quotes from patients for each of the concepts are shown in Table 7. Overall, these findings support the relevance of the concepts measured by FAACT-5IASS items in assessing changes in appetite due to cachexia. However, other symptoms, particularly vomiting and fatigue, are not captured in the FAACT-5IASS.
Experience (past or current) with FAACT-5IASS concepts reported by patients.

“Y” indicates the concept measured by the item was described by the patient spontaneously or in response to a probe. “N” indicates the concept measured by the item was not described spontaneously, in response to a probe, or was not probed. This table is derived from the concept elicitation interviews and did not directly debrief FAACT-5IASS item content. FAACT-5IASS: Functional Assessment of Anorexia-Cachexia Therapy–5-item anorexia-related symptoms scale; N: No; Y: yes.
Illustrative quotes for the FAACT-5IASS items reported by patients.

FAACT-5IASS: Functional Assessment of Anorexia-Cachexia Therapy–5-item anorexia-related symptoms scale.
Discussion
This study aimed to better understand the patient experience and identify the key symptoms and impacts on concepts of cancer cachexia through qualitative interviews. Our findings identified nausea, vomiting, change in appetite, and fatigue as the key symptoms from the patient perspective. Moreover, these symptoms were considered the most bothersome. The greatest impacts were seen on patients’ appearance, physical function, and social activities. One important caveat from this list of symptoms is that patients overwhelmingly attributed nausea and vomiting to chemotherapy and not specifically to cachexia. Therefore, the connection between these symptoms and cachexia is not clear. Future longitudinal studies to monitor symptom evolution throughout cancer treatment may help to shed light on this relationship.
Our study used the output from the concept elicitation research to assess the content validity of the FAACT-5IASS. 22 A previous study by Gelhorn et al 23 validated the FAACT-5IASS and included qualitative interviews with lung cancer patients with involuntary weight loss. Our content validity results are similar to those from Gelhorn et al. 23 For our study versus Gelhorn et al, “good appetite” was reported by 90% versus 100% of patients, “getting full quickly” by 75% versus 73%, “unpleasant tasting food” by 65% versus 64%, although “interest in food drops” was 30% versus 73%, and “difficulty eating rich/heavy food” was 5% versus 55%. Some of these differences may be explained by differences in the patient characteristics between the 2 cohorts. For instance, our study included a larger number of patients (N = 20 vs 11) and more types of cancer (NSCLC, pancreatic, and colorectal vs NSCLC only). Compared with Gelhorn et al, the patients in our study had less severe cancer (stage IV: 63.6% vs 25%; stage III: 50% vs 36.4%). Moreover, all patients (100%) in the Gelhorn et al study had ECOG performance status ⩾2, compared with 80% of patients in our study. As the disease advances it may be expected that experiences related to appetite would become more severe and impact more dimensions of appetite, like interest in food or ability to eat certain types of food; evaluating the progression of appetite loss during disease advancement would help further characterize cancer cachexia.
Overall, our findings support the concepts included in the FAACT-5IASS that speak to change in appetite. Appetite loss is a multidimensional concept not only experienced by patients as a lack of desire to eat but also early satiety, food tasting unpleasant, lack of interest in food, etc. When assessing change in appetite, PROMs that include a single question on appetite loss may not therefore adequately reflect the patient experience. A more comprehensive PROM that more deeply explores change in appetite, such as the FAACT-5IASS, may be more appropriate. In addition to appetite loss, patients may also experience other symptoms that impact appetite, such as nausea and vomiting. The results from our patient interviews also identified fatigue as a key symptom of cachexia. Moreover, appetite loss may have a significant impact on patients’ health-related quality of life, particularly their physical function and social activity, as supported by our findings. In addition to assessing change in appetite using the FAACT-5IASS, PROMs that assess these additional symptoms and their impact should also be incorporated into routine clinical practice or in the clinical trial setting when evaluating the patient experience of cachexia; for example, using the PRO-CTCAE items to capture vomiting and fatigue, and the PROMIS Fatigue 7a measure to capture fatigue more comprehensively. Our assessments of the validity of the FAACT-5IASS warrant replication and further study in a larger patient population. In addition, conducting such studies in the context of future clinical trials may provide opportunities for evaluating the relationship between PROMs and objective measures of body composition, such as assessment of skeletal muscle or adipose tissue.
This study had a number of limitations. The study may not be generalizable to all patients with cancer-related cachexia as all patients interviewed were from the US and the sample is small. In addition, most patients received chemotherapy and there is an overlap between chemotherapy side effects and cachexia symptoms such as nausea and vomiting. These symptoms were overwhelmingly attributed to cancer treatment in this study; however, the true causality may be unclear. In cases such as this, clinicians may be able to distinguish whether such symptoms are related to treatment by carefully evaluating the patient’s experience where possible. The interview guides had not been pilot-tested; however, the guides followed best practice. 15 Replication of the findings from both the concept elicitation and the FAACT-5IASS validation would benefit from more research and a more varied array of cancer types. Assessment of the validity of the FAACT-5IASS was not an original aim of the study and was based on the output of the concept elicitation interviews, and the comparison of the FAACT-5IASS with other tools was not within the scope of the study. Since the concept elicitation interviews did not directly debrief FAACT-5IASS item content, the relevance of items to the patients may be underestimated. For instance, the interviews probed for patients’ experience with appetite changes, getting full quickly, and unpleasant tasting food more consistently than for interest in food dropping as they try to eat or having difficulty eating rich or heavy foods. The FAACT-5IASS has yet to be fully validated using longitudinal data but has been used in a longitudinal, Phase 2, randomized controlled trial, 30 and although we do not consider this study to be a full validation of the measure, our findings generally support the relevance of the concepts in the FAACT-5IASS. However, as this study only enrolled patients in the US, interpreting the results in the context of non-US populations should take possible cultural influences on eating behavior and symptom perception into account.
Conclusion
In conclusion, following a targeted literature and instrument review and clinician interviews, concept elicitation interviews were conducted with patients with cancer-related cachexia to better understand the patient experience and identify the key symptoms and their impacts. These interviews identified nausea, vomiting, change in appetite, and fatigue as key symptoms of importance to these patients. Impacts included effects on physical appearance, physical and social activities, and moods/emotions. The output from the interviews was used in a post-hoc analysis to evaluate the content validity of the FAACT-5IASS PROM, with general support for the concepts included in that assessment. Given the impact of cancer cachexia on patient outcomes, identifying the key symptoms and their impacts as well as suitable assessments to evaluate changes over time are important. Further characterization of relevant PROMs should allow these measures to more accurately determine appropriate clinical interventions and may also assist in the development of suitable treatment options for the management of cancer cachexia.
Supplemental Material
sj-docx-1-onc-10.1177_11795549261430229 – Supplemental material for The Patient Experience of Cancer-Related Cachexia: Implications for Patient-Reported Outcomes Measures
Supplemental material, sj-docx-1-onc-10.1177_11795549261430229 for The Patient Experience of Cancer-Related Cachexia: Implications for Patient-Reported Outcomes Measures by Magdalena Harrington, Susan Martin, Oyebimpe Olayinka-Amao, Jarjieh Fang, Euan McLeod and Ira Jacobs in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
Medical writing support was provided by David Cope, PhD, of Engage Scientific Solutions and Tina Wasmeier, PhD, of Nucleus Global, an Inizio Company, and was funded by Pfizer.
Ethical considerations
All study materials were reviewed and approved by RTI International’s institutional review board (Ethical approval number STUDY00020336. Study approved on 10/02/2018).
Consent to participate
Prior to the concept elicitation interview, an informed consent discussion was conducted with each participant to obtain verbal consent to participate. Consent was documented by RTI-HS staff on a signed and dated informed consent form. Participants were provided an unsigned copy of the consent form for their records.
Consent for publication
Not applicable.
Author contributions
All authors were involved in the study design. Susan Martin and Oyebimpe Olayinka-Amao acquired the data. All authors had full access to the study data, were involved in data analysis and interpretation, drafted the manuscript and critically reviewed or revised each draft for intellectual content, and approved the final version for submission.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Pfizer; the sponsor had a role in research design, execution, analysis, interpretation, and reporting.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Magdalena Harrington, Jarjieh Fang, and Euan McLeod are employees of Pfizer and may have stock and/or stock options in Pfizer. Ira Jacobs was an employee of Pfizer at the time of the study and during manuscript development. Susan Martin and Oyebimpe Olayinka-Amao are employees of RTI Health Solutions.
Data availability statements
Supplemental material
Supplemental material for this article is available online.
Geolocation information
This study was conducted in the United States. All authors are based in the United States.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
