Abstract
Background:
The integration of clinicopathological parameters and molecular biomarkers holds significant prognostic value for endometrial carcinoma (EC) recurrence assessment. The purpose of this study is to combine clinical pathological parameters with simple molecular features to predict 3-year recurrence of EC.
Methods:
Based on a retrospective cohort with 136 patients assessing recurrence risk within 3 years postoperatively, we developed PM-TERR (clinicopathology and simplified molecular markers-based EC tailored 3-year recurrence risk). All patients were randomly assigned to the training set (96 patients) and the test set (40 patients). The nomogram comprised 4 input variables (Stage, Grade, Pathology Type, and P53 status) for calculating a PM-TERR score.
Results:
Kaplan-Meier analysis demonstrated a significant association between PM-TERR scores and disease-free survival (DFS) in both the training and test sets (both P < .001). The PM-TERR model outperformed the ESMO stratification in predicting DFS. The likelihood value of the PM-TERR model increased by 6.88 in the training set, and by 2.20 in the test set, both sets P < .05. Time-dependent ROC analysis further confirmed the PM-TERR enhanced predictive accuracy, with consistently higher AUC values across all time points than ESMO in both cohorts.
Conclusion:
In summary, the PM-TERR model integrates stage, grade, histology, and p53 status to predict 3-year recurrence risk. This retrospective, single-center study demonstrates its potential utility; however, future prospective, multi-center validation is required to confirm its robustness and clinical applicability before broader implementation.
Introduction
Endometrial carcinoma (EC) is one of the most prevalent malignancies in women and is increasing in incidence, imposing a substantial disease burden worldwide. 1 Although EC exhibits a favorable prognosis, recurrent cases are associated with significantly diminished survival outcomes. Recurrence rates among EC patients across different FIGO stages range from 10% to 70% following initial treatment. 2 Accurate identification of high-risk populations for recurrence constitutes the cornerstone of personalized therapeutic strategies. The gynecologic oncology community has devoted substantial efforts to predicting recurrence and prognostic risk based on pathological characteristics (for example, histological subtype, grade, lymphovascular space invasion (LVSI), FIGO (International Federation of Gynaecology and Obstetrics) tumor stage).3-6
Following the establishment of The Cancer Genome Atlas (TCGA) molecular classification, the European Society of Gynaecological Oncology (ESGO), the European SocieTy for Radiotherapy & Oncology (ESTRO) and the European Society of Pathology (ESP) jointly updated the guideline recommendations for EC.7,8 Then European Society of Medical Oncology (ESMO) has revised novel risk stratification schemas, recommending molecular profiling for all patients, establishing a management paradigm for EC that integrates traditional clinicopathological factors with molecular classification. 9 However, in practice, challenges remain as a result of the complexity of combining an increasing number of factors, and costs of molecular testing. Moreover, the high financial burden of molecular subtyping severely limits the implementation of precision oncology approaches for EC risk stratification in resource-constrained settings, exacerbating global disparities in cancer care.10,11 More efforts should be made to combine clinicopathological and molecular factors, to optimize clinical management.
In the present study, we conducted a retrospective analysis of EC recurrence patients, and the most common time for recurrence is within 3 years after surgery. Non-recurrent patients were incorporated for comparative analysis. We reported the development and evaluation of PM-TERR (clinicopathology and simplified molecular-based EC tailored 3-year recurrence risk), to predict 3-year recurrence from clinicopathology and simplified molecular markers for postsurgical women with stage I-III EC. Leveraging PM-TERR risk stratification, we systematically evaluated the therapeutic benefits of postoperative adjuvant treatment modalities.
Methods
This is a retrospective cohort study, and the methodology includes an explanation of cohort information and modeling methods. The reporting of this study adheres to the STROBE guidelines. 12
Cohort
The study was approved by the Institutional Review Board of Shandong Provincial Hospital and was performed in compliance with the Declaration of Helsinki. This retrospective cohort study utilized data obtained from medical records, with all patients having provided consent for the use of their treatment-related information for scientific research upon hospital admission. As the study involved no disclosure of private patient information or conflicts of interest, an exemption from informed consent was sought and approved by the ethics committee (Approval No. 2025-112). Patients with recurrent EC treated at Shandong Provincial Hospital between January 2021 and December 2024 were retrospectively enrolled. Data on the time of initial diagnosis, clinicopathological characteristics, and molecular subtyping were systematically retrieved for analysis. Inclusion criteria were (1) histopathologically confirmed stage I-III EC with surgical staging (with or without lymphadenectomy) at initial diagnosis; (2) well-documented adjuvant therapy regimen postoperatively; (3) recurrence within 3 years postoperatively, with histopathological confirmation via biopsy or surgical resection of the recurrent lesions; (4) availability of molecular subtyping data at initial diagnosis. Concurrently, a control group comprising patients with primary EC treated at Shandong Provincial Hospital during the same period was established. Inclusion criteria for the control group were (1) histopathologically confirmed stage I-III EC; (2) well-documented adjuvant therapy regimen postoperatively; (3) no recurrence documented during 3 years of follow-up; (4) availability of molecular subtyping data at initial diagnosis. In total, 43 patients experiencing recurrence within 3 years and 93 recurrence-free patients were included in the final cohort.
Variables
The extracted patient data included age at initial diagnosis, depth of myometrial invasion, lymphovascular space invasion (LVSI), FIGO stage, cervical stromal invasion, tumor grade, histological type, ESMO risk stratification, and medical comorbidities such as diabetes mellitus, hypertension, and history of cardiovascular/cerebrovascular complications. Endometrial carcinomas are molecularly classified into four subtypes: (1) POLE ultramutated, characterized by mutations in the exonuclease domain of the POLE gene and an excellent prognosis; (2) Mismatch Repair-deficient (MMRd), exhibiting microsatellite instability due to defective DNA mismatch repair; (3) p53-mutant (p53abn), defined by aberrant p53 immunohistochemical expression indicative of TP53 mutation and associated with poor outcomes; and (4) No Specific Molecular Profile (NSMP), which lacks the defining features of the other groups and represents a heterogeneous category. Age at the time of primary diagnosis was treated as a continuous variable in the statistical analysis. Histologically, tumors were described as two types: endometrioid adenocarcinoma and non-endometrioid carcinoma, including mucinous adenocarcinoma, serous adenocarcinoma, clear cell adenocarcinoma and carcinosarcoma. Grade was recorded with a two-grade system: low grade (well differentiated or moderately differentiated) and low grade (poorly differentiated, also called grade 3). LVSI was classified into three categories: no LVSI, mild LVSI, and extensive LVSI. The intermediate-risk, intermediate-high-risk, and high-risk categories in the ESMO risk stratification system were consolidated into a unified intermediate-to-high risk group, thereby transforming the original classification into a binary variable (low-risk vs. intermediate-to-high risk). 9 Furthermore, DFS was calculated as the number of months from cancer diagnosis to recurrence.
PM-TERR Development and Validation
All patients were randomly assigned to the training set and test set in a 7:3 ratio. First, single-factor Cox regression was used to screen the relevant characteristics and select variables significantly related to survival (P < .05). Then, in the multivariable Cox regression method, independent predictors for constructing the final model were selected using stepwise regression based on the Akaike information criterion (AIC). The PM-TERR model is developed by selected independent predictors, and a PM-TERR score was calculated; the visual graph is generated simultaneously.
To estimate the accuracy of the PM-TERR model in predicting EC 3-year recurrence after initial treatment, Harrell’s concordance index (C-index), hazard ratio (HR), and the area under the time dependent receiver operating characteristics curve (Time-AUC) were calculated. The PM-TERR score was dichotomized by the optimal cutoff value identified by using maximally selected rank statistics. The association between the PM-TERR and the DFS was evaluated by Kaplan–Meier survival analysis both on the train set and test set.
Statistical Analysis
R statistical software (Version 4.4.0, https://www.rproject.org) was used to conduct baseline statistics. Categorical variables are expressed as frequencies (percentages), while continuous variables are presented as mean ± standard deviation or median (interquartile range) based on their distribution characteristics. Categorical variables are analyzed using chi-square tests or Fisher’s exact tests, while continuous variables with normal distributions are analyzed using t-tests and those with non-normal distributions are analyzed using Mann-Whitney U tests. Cox regression analysis, survival analysis, C-index and AUC analysis were performed using “survminer,” “survival,” and “timeROC” packages.
Results
Cohort with endometrial cancer
In 51 cases of recurrent EC, molecular classification was conducted at the time of primary diagnosis, prior to disease recurrence. The median disease-free survival (DFS) time was 21.9 (Q1-Q3: 13.0 ~ 33.0) months. DFS intervals, measured in years, demonstrated a characteristic distribution pattern as depicted in Supplementary Figure 1. Notably, 43 (84.3%) of cases experienced recurrence within the 3-year postoperative window. Those recurrent cases in 3 years with molecular classification testing were enrolled in the cohort, along with 93 controls who remains recurrence-free throughout the 3-year surveillance period with molecular classification testing. Clinical data from the initial diagnosis and treatment were retrospectively tracked. Among 43 recurrent women in 3 years, no POLEmut cases were identified, with molecular subtypes distributed as follows: 7 cases (6.9%) of MMRd, 26 cases (25.7%) of NSMP, and 18 cases (17.8%) of p53abn subtypes. The cohort information is detailed in Figure 1. The clinical and pathological characteristics of 136 patients used for model construction are shown in Supplementary Table 1.
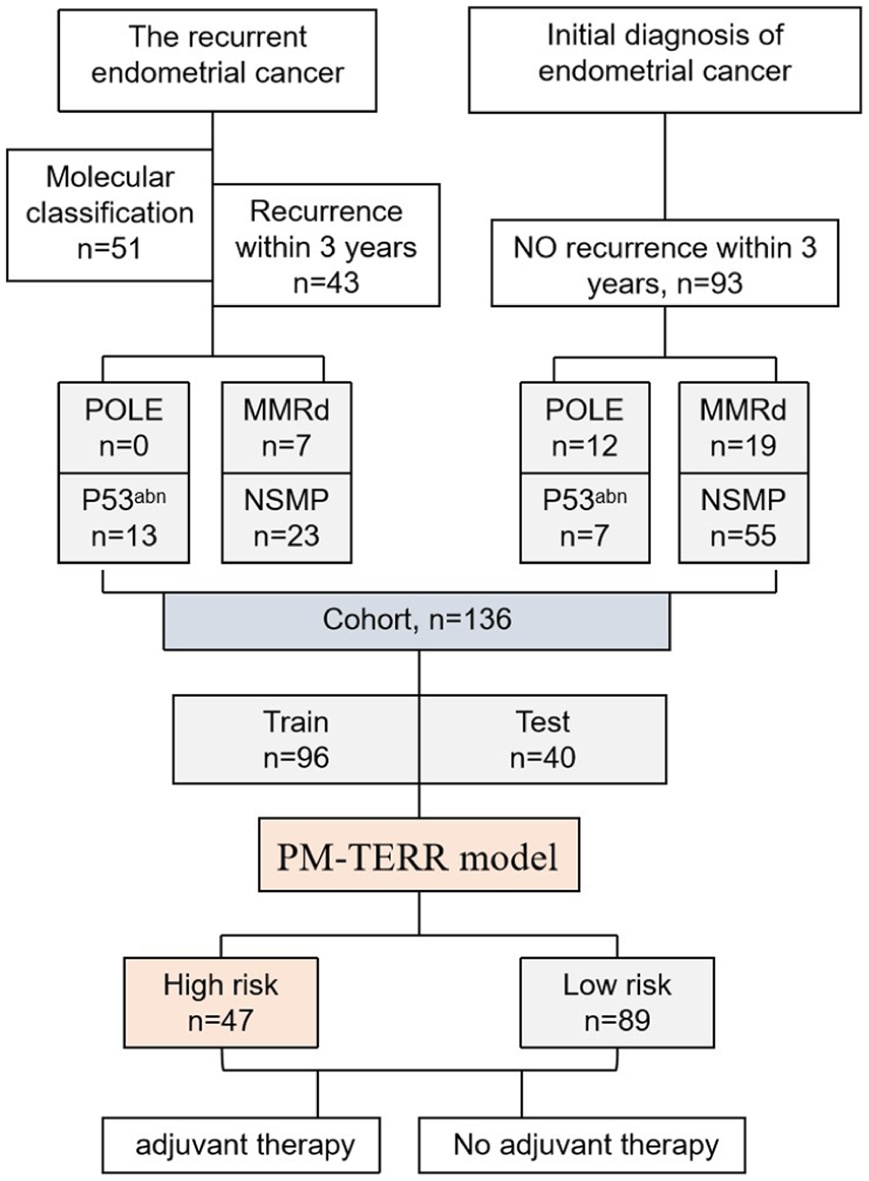
Flowchart of the study cohort, molecular classification, recurrence outcomes, and PM-TERR risk stratification.
PM-TERR development and validation
All patients are randomly assigned to the training set (96 patients) and the test set (40 patients). The distributions of clinical characteristics are balanced between the training and test sets (Supplementary Table 2). Figure 2 presents a PM-TERR model for predicting DFS probability. The PM-TERR model comprises 4 input independent predictors (Stage, Grade, Pathology Type and P53 status) for calculating a patient’s PM-TERR score. This score is then used to predict the probability of 1-year, 2-year, and 3-year DFS on the corresponding axes. The hazard ratios (HRs) for the variables in the univariable and multivariable Cox proportional hazards model are presented in Supplementary Tables 3 and 4. The optimal cutoff value for the PM-TERR score is determined to be 0.755. Using this optimal cutoff, patients were stratified into high and low PM-TERR score groups. Kaplan-Meier analysis demonstrated a significant association between PM-TERR score and DFS in both the training and test sets (Figure 3A and B, both log-rank P < .001). C-index showed both the training set and the test set have good predictive performance (0.758 and 0.706).

Nomogram for predicting disease-free survival (DFS) probability in endometrial cancer.

Disease-free survival (DFS) analysis stratified by PM-TERR risk groups. (A) Training set (n = 96): Kaplan-Meier curves comparing DFS between low-risk and high-risk groups (P < .0001). Number at risk tables are shown below the curves. (B) Test set (n = 40): Kaplan-Meier curves comparing DFS between low-risk and high-risk groups (P < .001). Number at risk tables are shown below the curves.
Superior predictive performance of the PM-TERR compared with ESMO criteria
The PM-TERR model significantly outperformed the ESMO stratification in predicting DFS. In the training cohort (n = 96), the PM-TERR demonstrated a higher hazard ratio (HR = 11.16, 95% CI: 4.54-27.41, P < .001) and C-index (0.758 vs. 0.706 for ESMO). This superiority persisted in the test cohort (n = 40), where the PM-TERR maintained strong statistical significance (HR = 6.20, 95% CI: 1.85-20.81, P = .003), while ESMO showed only borderline significance (HR = 4.50, 95% CI: 0.98-20.58, P = .052). The likelihood ratio test revealed that the PM-TERR model was significantly superior to the ESMO model. The likelihood value of the PM-TERR model increased by 6.88 in the training set, and by 2.201 in the test set, both of sets P < .05. Time-dependent ROC analysis further confirmed the PM-TERR’s enhanced predictive accuracy, with consistently higher AUC values across all time points compared with ESMO in both cohorts (Figure 4A and B).
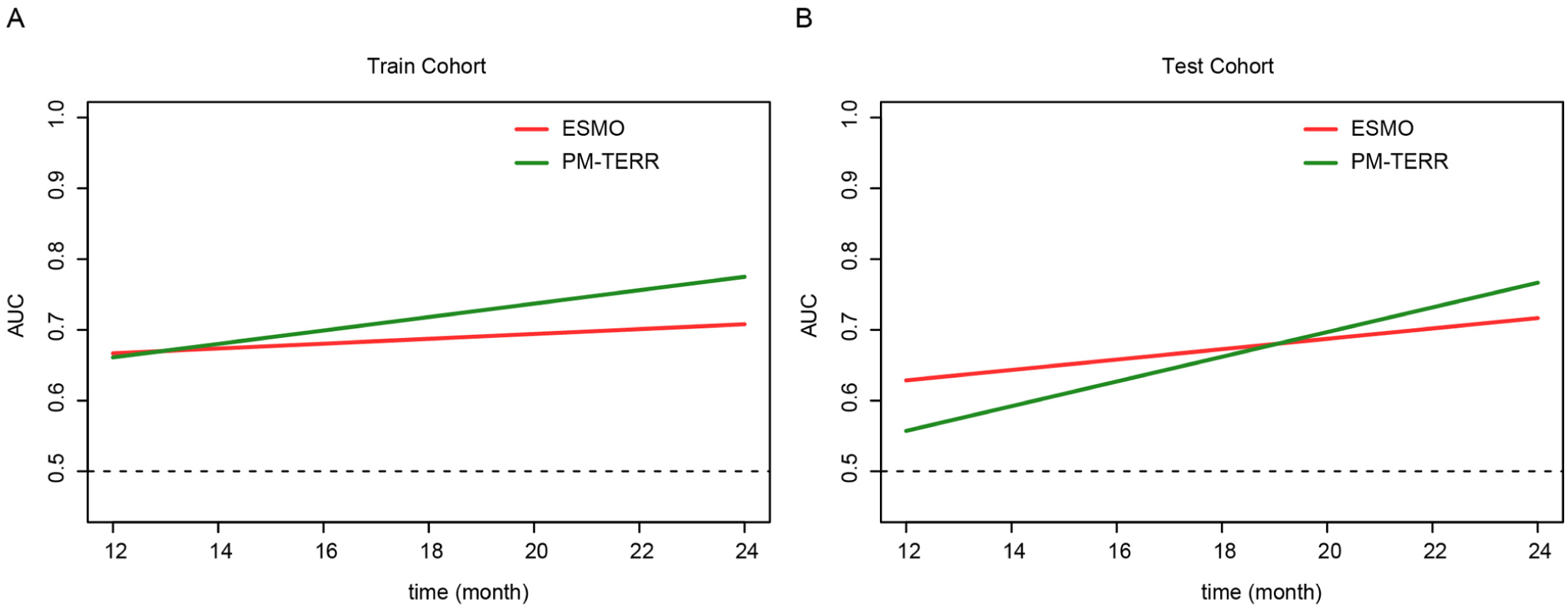
Comparison of ESMO and PM-TERR risk stratification models over time. Performance metric (AUC) is shown for both the (A) Train Cohort and (B) Test Cohort across monthly intervals from 12 to 24 months.
Lack of benefit from radiotherapy ± chemotherapy in low-risk patients
Eighty-nine patients were classified into the PM-TERR low-risk group, and forty-seven patients were categorized into the PM-TERR high-risk group. Prognostic outcomes were compared between patients who received adjuvant radiotherapy and those who did not within each risk group. In the high-risk group, radiotherapy ± chemotherapy significantly reduced the incidence of recurrence within 3 years postoperatively (Figure 5A, P < .01). Conversely, in the low-risk group, radiotherapy ± chemotherapy was associated with an increased risk of postoperative recurrence (Figure 5B, P < .01). Chemotherapy alone neither increased the 3-year recurrence risk nor conferred clinical benefit.
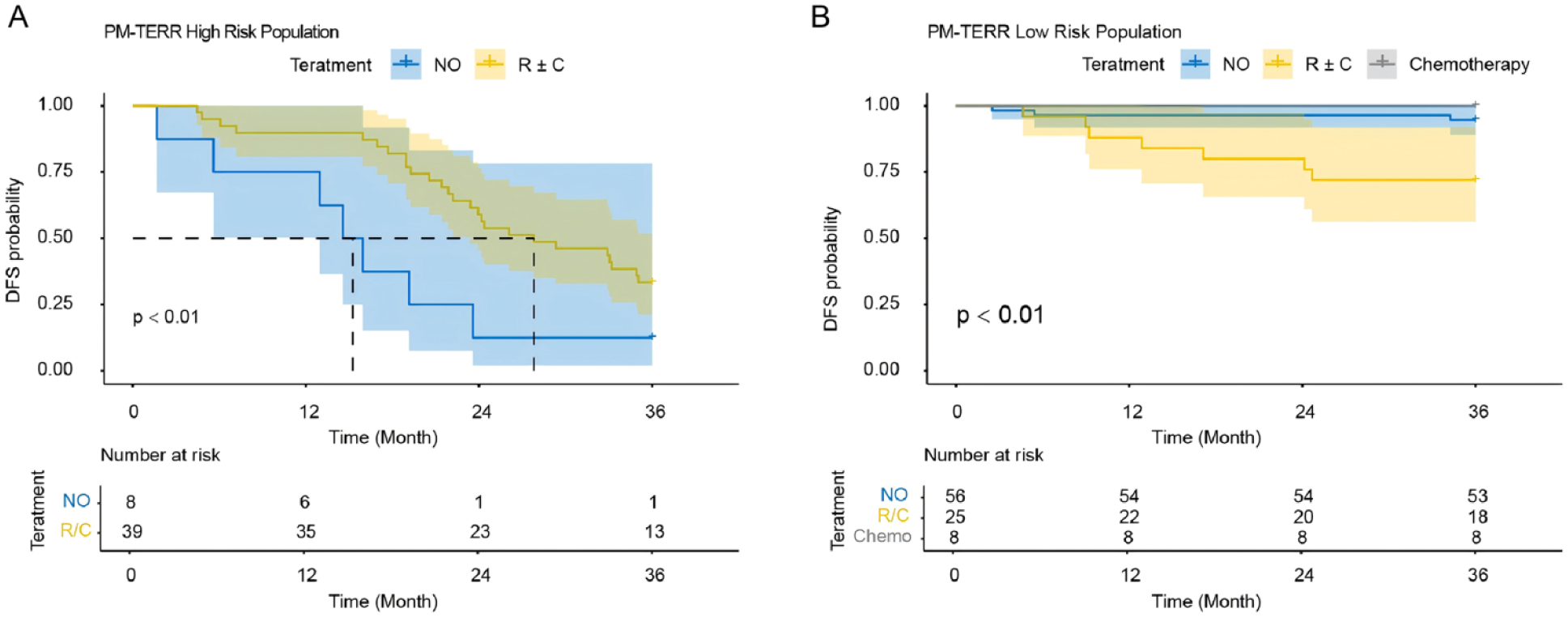
Treatment effect on disease-free survival (DFS) in PM-TERR risk-stratified populations. (A) High-risk group: Kaplan-Meier curves comparing DFS between patients receiving no adjuvant therapy (“NO”) versus radiotherapy with or without chemotherapy (“R ± C”) (P < .01). Number at risk tables are shown. (B) Low-risk group: Kaplan-Meier curves comparing DFS between patients receiving no adjuvant therapy (“NO”), radiotherapy/chemotherapy (“R/C”), or chemotherapy alone (“Chemotherapy”) (P < .01). Number at risk tables are shown. Time scale in months for all analyses.
Discussion
PM-TERR is a model trained and validated in 136 patients with stage I–III EC, with 3-year follow-up, predicts postoperative 3-year recurrence risk using grade, histological type, anatomical stage and p53 status. PM-TERR obtained C-indices of 0.758 and 0.706 in train and test sets for 3-year recurrence outcome. Its performance favorably compares with traditional prognostic assays such as ESMO risk stratification with C-index of 0.706 and 0.657 in train and test sets, respectively. In the PM-TERR high-risk and low-risk groups, radiotherapy with or without chemotherapy demonstrated contrasting roles in influencing postoperative recurrence outcomes. Our results suggest that PM-TERR may have the potential to be a highly effective tool for individualized prognostication of women with EC, while assisting in deciding postoperative treatment.
The benefit of adjuvant radiotherapy in low-risk EC patients remains controversial. Kong et al 13 reviewed randomized controlled trials (RCTs) that compared postoperative adjuvant radiotherapy in women with stage I EC to assess the efficacy of adjuvant radiotherapy following surgery for stage I EC. In the subgroup of low-risk patients (IA/B and grade 1/2), pelvic external beam radiotherapy increased the risk of EC-related deaths (517 patients; RR 2.64, 95% CI 1.05 to 6.66).14,15 One trial of vaginal intracavity brachytherapy versus no additional treatment in women with low-risk lesions reported a non-significant reduction in locoregional recurrence (RR 0.39, (95% CI 0.14 to 1.09). 16 In stage I low-risk EC, adjuvant radiotherapy not only failed to demonstrate a survival benefit but was also associated with increased treatment-related toxicity. Pelvic external beam radiotherapy was associated with significantly increased severe acute toxicity (two trials, 1328 patients, RR 4.68, 95% CI 1.35 to 16.16), increased severe late toxicity (3501 women; RR 2.58, 95% CI 1.61 to 4.11). Among PM-TERR-classified low-risk patients, radiotherapy—either alone or combined with chemotherapy—was associated with increased incidence of 3-year recurrence. Based on cumulative data from prior RCTs and this investigation, routine adjuvant radiotherapy should not be advocated for low-risk EC patients following surgery, given the absence of survival benefit and potential for increased treatment-related morbidity.
The current gold standard of combined pathological and molecular profiling is costly, which hampers its widespread implementation. Consequently, some studies have developed machine learning-based models to broaden the toolkit for prognostic assessment in endometrial cancer. For instance, Zhang et al 17 reported a machine learning-based radiomics model that contributes to predicting postoperative overall survival (OS) in EC patients. Volinsky-Fremond et al 18 developed HECTOR (Histopathology-based Endometrial Cancer Tailored Outcome Risk), a multimodal deep learning prognostic model using hematoxylin and eosin-stained whole-slide images and tumor stage as input. Although various machine learning-based models have demonstrated promising predictive performance, pathological and molecular profiling retains irreplaceable value. Our PM-TERR model aims to reduce testing costs while focusing on the early warning of high-risk patients. However, a prospective, head-to-head comparison of the PM-TERR model against the current molecular subtyping standard is warranted to formally assess its potential incremental clinical benefit and cost-effectiveness.
The primary limitation of this study was its relatively small sample size, which precluded further subgroup analyses for risk stratification, as well as the need for more granular validation of adjuvant treatment patterns. We posit that the binary risk stratification model of PM-TERR may underestimate recurrence risks and radiotherapy benefits in certain low-risk subgroups, necessitating larger cohorts for model optimization and validation. The relatively limited performance of the ESMO risk classification in our cohort should be interpreted with caution. This is likely influenced by the distinct molecular profile of our study population, which is characterized by a complete absence of POLEmut cases and a higher proportion of p53abn tumors compared to the reference distributions underlying the ESMO model. This unbalanced molecular distribution, particularly the lack of POLE-mutated cases in the recurrence group, not only affects the comparative performance of existing models but may also limit the generalizability of our own findings to populations with different molecular profiles. Furthermore, the absence of detailed clinical data (e.g., BMI, comprehensive reproductive history) represents an additional constraint, as it restricts the model’s ability to account for all potential confounding factors. In addition, this is an exploratory study with no minimum sample size limit, and the retrospective design of this study, which collected cases during a predetermined period and at a single geographic location, potentially limits the reproducibility and universality of the predictive model. Although model generalizability was evaluated through test set validation, the limited sample size may compromise translational reliability. However, PM-TERR shows promising clinical utility in identifying high-risk populations. By incorporating only grade, histological type, anatomical stage, and p53 status—as opposed to costly molecular subtyping—p53 assessment via immunohistochemistry (IHC) offers a triage strategy. This approach could significantly reduce socioeconomic burdens in healthcare systems while effectively flagging high-risk EC patients, making it particularly valuable in resource-limited settings. Finally, it should be noted that our model predicts disease-free survival (DFS) but not overall survival (OS). This distinction is primarily due to the nature of our cohort, which included patients with recent recurrence who were all alive and undergoing heterogeneous salvage therapies at the time of analysis. These varied post-recurrence treatment regimens represent a significant confounder for OS outcomes. Therefore, future studies with extended follow-up and adjustment for treatment effects are warranted to evaluate the model’s prognostic utility for OS.
Conclusion
In summary, the validation and refinement of PM-TERR—a risk stratification model integrating staging, grade, histological type, and p53 status—provides a tool to predict the 3-year recurrence risk in women with stage I-III EC undergoing primary surgery. Theoretically, this model provides a postoperative assessment strategy that is less resource-intensive than comprehensive molecular subtyping. Nevertheless, the validity of omitting adjuvant radiotherapy for PM-TERR low-risk patients necessitates further validation through studies with expanded cohorts to ensure robustness of this risk-stratified approach.
Supplemental Material
sj-docx-2-onc-10.1177_11795549261428372 – Supplemental material for A Clinicopathology and Simplified Molecular Marker-Based Risk Stratification Model for Predicting 3-Year Recurrence in Endometrial Cancer
Supplemental material, sj-docx-2-onc-10.1177_11795549261428372 for A Clinicopathology and Simplified Molecular Marker-Based Risk Stratification Model for Predicting 3-Year Recurrence in Endometrial Cancer by Juntao Zhang, Yichuan Zhou, Yuzhen Huang, Xiaoyan Qin, Hua Yang, Chunrun Yang and Jianbin Guo in Clinical Medicine Insights: Oncology
Supplemental Material
sj-jpg-1-onc-10.1177_11795549261428372 – Supplemental material for A Clinicopathology and Simplified Molecular Marker-Based Risk Stratification Model for Predicting 3-Year Recurrence in Endometrial Cancer
Supplemental material, sj-jpg-1-onc-10.1177_11795549261428372 for A Clinicopathology and Simplified Molecular Marker-Based Risk Stratification Model for Predicting 3-Year Recurrence in Endometrial Cancer by Juntao Zhang, Yichuan Zhou, Yuzhen Huang, Xiaoyan Qin, Hua Yang, Chunrun Yang and Jianbin Guo in Clinical Medicine Insights: Oncology
Footnotes
Author note
During the preparation of this work, the author(s) used Deepseek in order to improve readability and language. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Ethics approval
The study was approved by the Institutional Review Board of Shandong First Medical University Affiliated Provincial Hospital (Approval No. 2025-112) on March 1, 2025. The committee is located at Renhe Building, 324 Jingwu Weiqi Road, Huaiyin District, Jinan City.
Consent to participate
This retrospective study used anonymized patient data from medical records and was granted an exemption from informed consent by the ethics committee.
Consent for publication
All authors agree to the terms of the publication consent form.
Author Contributions
Z.JT. and Z.YC. analyzed data and wrote the main manuscript text. Z.JT., Z.YC., and Y.H. prepared Table and Figures. H.YZ., Y.CR., and G.JB. proposed the idea, collected data, and revised the manuscript finally. Q.XY. made contributions in revising the manuscript. All authors reviewed the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Special Foundation for Taishan Scholars Program of Shandong Province (No. tsqnz20240855, No. tstp20230657), Shandong Provincial Natural Science Foundation (ZR202211160007), National High Level Hospital Clinical Research Funding under Grant (2022-PUMCH-C-060) and Beijing Natural Science Foundation (L232074).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
