Abstract
Background:
Immune checkpoint inhibitors (ICIs) have revolutionized advanced lung cancer treatment, but immune-related adverse events (irAEs) remain a significant challenge. This study aimed to identify peripheral blood indicators associated with Immune-Related Adverse Events (irAEs) to improve early prediction and management.
Methods:
A retrospective analysis of 1910 advanced lung cancer cases treated with ICIs (2015-2024) was conducted. Patients were categorized into 2 groups: irAEs (n = 323) and control (n = 323). Peripheral blood indicators were analyzed at baseline and during treatment. Statistical analyses included t-tests, Mann-Whitney U tests, logistic regression, and Cox proportional hazards models.
Results:
Age (odds ratio [OR] = 0.96, P < .001) and radiotherapy history (OR = 2.35, P = .004) were significant irAE risk factors. Hemoglobin, red blood cells, and lymphocyte ratios decreased, while neutrophil ratios, neutrophil-to-lymphocyte ratio (NLR), lactate dehydrogenase (LDH), and systemic inflammation markers increased in irAE patients. The NLR and LDH were independent predictors of irAEs and overall survival (P < .05).
Conclusion:
Peripheral blood indicators are valuable for irAE prediction. Specifically, elevated NLR (hazard ratio [HR] = 1.14, P = .002) and LDH (HR = 1.00, P = .03) were identified as independent risk factors for irAEs and were also significantly associated with poorer overall survival.
Keywords
Introduction
Lung cancer is one of the cancers with the highest incidence and mortality rates worldwide. According to the 2022 GLOBOCAN data, there were approximately 2.5 million new cases of lung cancer and nearly 1.8 million lung cancer-related deaths in 2022, 1 with the proportion of patients with advanced lung cancer continuing to rise. Although traditional therapies such as surgery, chemotherapy, and radiotherapy are widely used for lung cancer treatment, they may be associated with significant toxic side effects and limited long-term efficacy for patients with locally advanced and metastatic disease.2-4 Over the past decade, immunotherapy targeting immune checkpoint proteins such as programmed death receptor-1 (PD-1) and programmed death ligand-1 (PD-L1) has brought new vitality to the treatment of patients with advanced lung cancer.5,6 Overall, the use of immune checkpoint inhibitors (ICIs) in advanced lung cancer has successfully transformed the treatment landscape of this disease, providing durable remission and long-term survival for a subset of patients.7,8
Meanwhile, with the release of multiple landmark Phase III clinical trial results, the treatment landscape for advanced lung cancer has been established. For instance, based on the PACIFIC study findings, durvalumab consolidation therapy has become the standard treatment for unresectable stage III non-small cell lung cancer (NSCLC) following radiotherapy and chemotherapy. 9 Meanwhile, based on the FLAURA study results, osimertinib is the first-line standard of treatment for advanced NSCLC with estimated glomerular filtration rate (EGFR) sensitive mutations. 10 These treatment regimens have been included in authoritative clinical guidelines such as European Society for Medical Oncology (ESMO) and American Society of Clinical Oncology (ASCO). 11 However, with the widespread use of these highly effective treatments, immune-related adverse events (irAEs) have become a major challenge to patients’ quality of life and treatment continuity.
However, ICIs not only target tumor cells but may also affect multiple organ systems throughout the body. Reports of irAEs are increasing, including colitis, hypophysitis, interstitial pneumonia, thyroiditis, arthritis, and others.12,13 Although most irAEs present with mild-to-moderate signs and symptoms, severe irAEs can lead to irreversible organ failure and life-threatening conditions. 14 Consequently, within the context of current clinical practice, identifying reliable biomarkers to predict irAEs is crucial for enabling risk stratification and optimizing patient management. Numerous studies have identified various biomarkers associated with irAEs, including platelet count, neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and monocyte-to-lymphocyte ratio (MLR) in peripheral blood15-17; cytokines such as TNF-α, IL-6, IL-8, IP-10, CXCL9, CXCL10, and CXCL11, which show baseline reductions18-20; elevated levels of IL-7; and increased autoantibodies, including rheumatoid factor, anti-nuclear antibodies, anti-thyroid antibodies, anti-thyroid peroxidase antibodies, and anti-thyroglobulin antibodies.21-23 In addition, elevated gastrointestinal microbiota diversity 24 and increased serum lactate dehydrogenase (LDH) levels 25 have been associated with irAEs. Based on previously published literature, we understand that changes in certain hematological parameters can signal the onset of irAEs in patients. For instance, the NLR serves as a comprehensive inflammatory indicator: an elevated neutrophil count may reflect innate immune activation and a pro-inflammatory state, while a reduced lymphocyte count can indicate immune dysfunction. Similarly, LDH, a marker of tissue damage and cellular turnover, may suggest broader cellular destruction and inflammatory conditions when elevated, which is associated with more intense immune responses. These indicators were selected not only for their clinical relevance but also because they are routinely measured in standard blood tests, offering significant advantages such as easy accessibility, low cost, and high standardization. This practicality facilitates their potential translation into clinical practice for identifying high-risk patients without requiring additional expensive or complex testing. Therefore, this study retrospectively analyzed the relationship between peripheral blood biomarkers and the occurrence of irAEs in patients with advanced lung cancer treated with PD-1/PD-L1 inhibitors. 26
Materials and Methods
The reporting of this study follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 27 The completed STROBE checklist is provided as a supplementary file (Supplementary File 1).
Study subjects
This retrospective observational study received ethical approval from the Institutional Review Board of the General Hospital of the People’s Liberation Army (approval number: S2021-462-02), which was conducted in full compliance with the ethical principles of the Declaration of Helsinki.
We conducted a systematic review of the medical records of patients with stage III to IV lung cancer who received ICI therapy at the People’s Liberation Army General Hospital (PLAGH) from January 2015 to September 2024. The inclusion criteria were as follows: (1) histologically confirmed diagnosis of lung cancer (including both small cell lung cancer and NSCLC) with clinical stage III or IV according to the 8th edition of the American Joint Committee on Cancer (AJCC) staging system; (2) availability of complete clinical data; and (3) receipt of at least 1 treatment cycle of PD-1/PD-L1 inhibitors, either as monotherapy or in combination with chemotherapy. The exclusion criteria were (1) patients with a history of other active malignancies within the past 5 years; (2) patients with severe autoimmune diseases requiring systemic immunosuppressive therapy; and (3) patients who were lost to follow-up immediately after the first cycle of treatment, making survival data unavailable.
Diagnosis of immune-related adverse events
In this study, the Common Terminology Criteria for Adverse Events (version 5.0), as recommended by the National Cancer Institute (NCI) and the National Institutes of Health (NIH) in the United States, was employed for the evaluation, diagnosis, classification, and grading of adverse reactions.
The diagnosis of irAEs is primarily based on the clinical features associated with immune activation that emerge following the administration of ICIs. These features may encompass dermatological manifestations such as rashes, gastrointestinal symptoms like diarrhea, hepatic dysfunction including hepatitis, pulmonary complications such as pneumonitis, and endocrine abnormalities.
Processing and data collection
Clinical data were retrospectively extracted from the electronic medical records of patients, covering comprehensive demographic characteristics, histopathological confirmation of lung cancer diagnosis, clinical staging according to the 8th edition of the AJCC classification, radiotherapy parameters, and detailed treatment protocols. For analytical purposes, patients were categorized into 2 age groups: <65 years and ⩾65 years. This cutoff was employed based on its common use in pivotal immuno-oncology trials investigating irAEs, enabling direct comparability of our safety outcomes with this established body of literature.9,28
For the irAEs cohort, peripheral blood parameters were systematically collected at 2 critical time points: baseline (initial ICI administration) and irAEs onset. For the control group, routine hematological parameters, biochemical profiles, and coagulation indices were obtained at 2 time points: baseline (initial ICI administration) and the final ICI administration preceding data collection.
In addition, 3 systemic inflammatory indices were calculated: the NLR, MLR, and systemic immune-inflammation index (SIRI). The SIRI was calculated using the formula: (neutrophil count × monocyte count)/lymphocyte count.
Statistical analysis
Statistical analyses were performed using SPSS version 26.0 (IBM Corp, Armonk, New York). The initial identification of potential risk factors associated with irAEs was carried out via univariate logistic regression analysis. Variables that exhibited statistical significance (P < .05) in the univariate analysis were subsequently included in a multivariate logistic regression model to determine the independent risk factors for the development of irAEs.
For continuous variables, those with a normal distribution and homogeneity of variance were analyzed using independent samples t-tests. In contrast, non-parametric data were assessed using the Mann-Whitney U test. Survival outcomes were evaluated through Cox proportional hazards regression modeling. For the survival analysis, patients were censored at the date of loss to follow-up or the end of the study period (December 31, 2023), whichever came first. Cases with missing data for the variables of interest were excluded from the respective analyses (complete-case analysis). A 2-tailed P-value of less than .05 was deemed statistically significant for all analyses.
Results
Baseline characteristics of patients
This retrospective cohort study encompassed 1910 patients with stage III to IV lung cancer who received ICI therapy at PLAGH from January 2015 to September 2024. The study population consisted of 323 patients (16.91%) who developed irAEs and an equal number of randomly selected controls from the remaining patients (Figure 1). Although demographic characteristics were similar between the groups, significant differences were observed in immunotherapy regimens and radiotherapy history.

Flowchart of the study design and included patient information.
The irAE cohort had a median age of 65 years (range = 20-87 years), with a predominance of male patients. Notably, patients aged ⩾ 65 years exhibited a significantly higher incidence of irAEs compared with controls (51.7% vs 21.4%; P < .001). All participants received PD-1/PD-L1 inhibitors, with PD-1 inhibitors being more commonly used. Combination therapy was the primary treatment approach for advanced cases, with first-line regimens being the most frequent. Moreover, the irAE group had a significantly higher prevalence of radiotherapy history compared with controls (30.7% vs 16.1%; P < .001) (Table 1).
Basic information of patients with advanced lung cancer treated with ICIs.
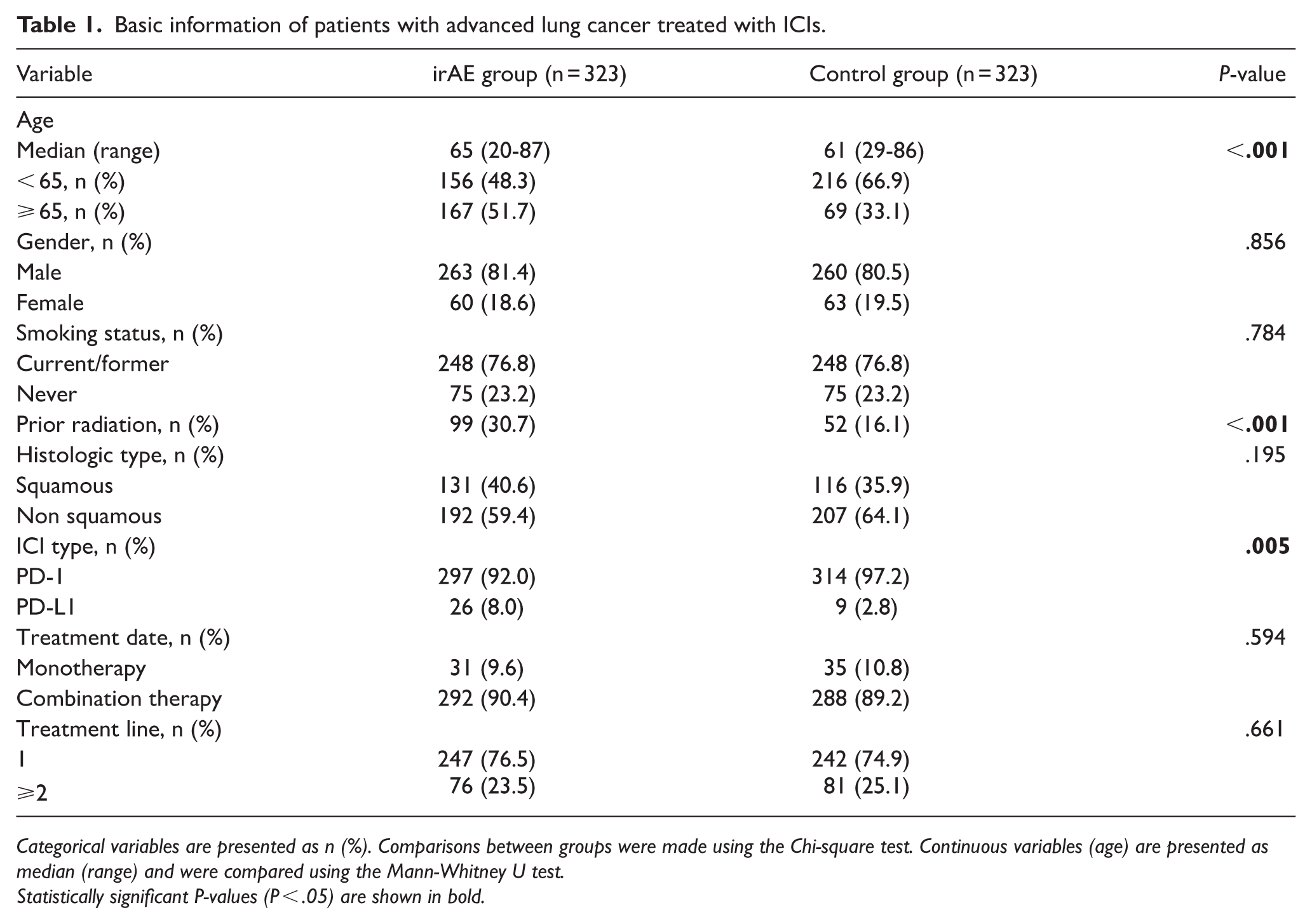
Categorical variables are presented as n (%). Comparisons between groups were made using the Chi-square test. Continuous variables (age) are presented as median (range) and were compared using the Mann-Whitney U test.
Statistically significant P-values (P < .05) are shown in bold.
Both univariate and multivariate analyses identified age (odds ratio [OR] = 0.96, 95% confidence interval [CI]: 0.95-0.98; P < .001) and radiotherapy history (OR = 2.35, 95% CI = 1.58-3.50; P = .004) as independent risk factors for the development of irAEs (Table 2 and Supplementary Tables 1 and 4).
Univariate and multivariate logistic regression analysis of risk factors for irAEs.

Odds ratios (OR) and 95% confidence intervals (CI) for the risk of developing irAEs were calculated using univariate and multivariate logistic regression analysis.
Statistically significant P-values (P < .05) are shown in bold.
Occurrence of immune-related adverse events
The study revealed that irAEs often affected multiple organ systems in patients after ICI treatment. Single-organ system involvement was most common, seen in 276 cases (84.75%). Multi-organ system involvement was less frequent, with 47 cases (14.55%). Most irAEs emerged within the first 2 months of ICI therapy, with incidence rates gradually declining over time (Figure 2). These results highlight the need for close monitoring and timely intervention early in immunotherapy to maximize treatment efficacy and improve patient outcomes.

Kaplan-Meier curves for event-free survival by organ system of the first immune-related adverse event.
The system-specific analysis (Figure 3) revealed that the respiratory system was the most frequently affected site, accounting for 26.01% of cases. Checkpoint inhibitor pneumonitis (CIP) was identified as the primary manifestation within this category. The CIP commonly presents with symptoms such as dyspnea, hypoxemia, and cough. In severe cases, it could progress to respiratory failure, emphasizing the necessity for enhanced respiratory monitoring throughout the course of ICI therapy. In comparison, dermatological manifestations were the second most common, comprising 19.81% of cases. These were typically characterized by erythema, pruritus, or maculopapular eruptions. Fortunately, these cutaneous reactions were generally mild, easily detectable, and manageable. Importantly, they did not typically necessitate the discontinuation of ongoing immunotherapy.

Distribution of irAEs by organ system.
Other systems that were frequently affected included the endocrine system (15.79%), gastrointestinal system (15.48%), and hematologic system (10.22%). Less frequent involvement was noted in the neurological system (4.64%), renal system (3.14%), musculoskeletal system (2.79%), and cardiovascular system (1.86%). Notably, cardiovascular irAEs predominantly manifested as myocarditis, which typically presented with symptoms such as fatigue, chest pain, and palpitations. In severe cases, myocarditis could progress to acute heart failure. This underscores the critical importance of rigorous cardiac monitoring during ICI therapy and the need for immediate intervention when indicated to optimize clinical outcomes (Supplementary Table 2).
Correlation between peripheral blood biomarkers and immune-related adverse events
This study utilized comprehensive hematological profiling to explore the temporal changes in peripheral blood parameters in relation to the onset of irAEs. Both univariate and multivariate logistic regression analyses were employed to evaluate the predictive value of these parameters. Comparative analysis showed that the irAE cohort experienced significant hematological alterations. Specifically, hemoglobin (HB) levels, red blood cell (RBC) counts, and lymphocyte ratios exhibited a pronounced downward trend at the time of irAE onset compared with baseline measurements. Although the control group also displayed similar reductions in these parameters, the magnitude of change was substantially less pronounced than that observed in the irAE group.
Univariate analysis revealed significant associations between multiple hematological parameters and the occurrence of irAEs. Hemoglobin levels were inversely correlated with irAE development, with an OR of 0.96 (95% CI = 0.950-0.969; P < .001). In contrast, several parameters exhibited positive correlations with irAEs, including neutrophil ratio (OR = 7.14, 95% CI = 1.917-26.56; P = .003), mean corpuscular volume (MCV; OR = 1.06, 95% CI = 1.033-1.089; P < .001), LDH (OR = 1.00, 95% CI = 1.002-1.007; P = .001), NLR (OR = 1.15, 95% CI = 1.088-1.224; P < .001), MLR (OR = 2.13, 95% CI = 1.241-3.662; P = .006), and SIRI (OR = 2.82, 95% CI = 1.438-5.541; P = .003). Conversely, significant negative correlations were observed for lymphocyte ratio (OR = 0.00, 95% CI = 0.000-0.013; P < .001) and hematocrit (OR = 0.00, 95% CI = 0.00-0.00; P < .001).
Multivariate analysis pinpointed several key hematological parameters as independent predictors of irAE occurrence. Hemoglobin levels were identified as a significant predictor, with an OR of 0.96 (95% CI = 0.95-0.97; P < .001). The lymphocyte ratio also emerged as a strong predictor, with an OR of 0.00 (95% CI = 0.00-0.17; P = .008). Other significant predictors included MCV (OR = 1.06, 95% CI = 1.03-1.09; P < .001), LDH (OR = 1.00, 95% CI = 1.00-1.00; P < .001), and NLR (OR = 1.14, 95% CI = 1.03-1.27; P = .013). It is worth noting that RBC count, neutrophil ratio, and hematocrit did not retain statistical significance in the multivariate model (P > .05) (Figure 4). In addition, parameters such as monocyte ratio, platelet count, albumin, and D-dimer did not show significant associations in the univariate analysis and were therefore excluded from the multivariate modeling (Table 3 and Supplementary Table 3).

Bar charts of peripheral blood parameters at different time points in the irAE group and the control group. (A) Hemoglobin, (B) red blood cells, (C) neutrophil ratio, (D) lymphocyte ratio, (E) lactate dehydrogenase, (F) neutrophil-to-lymphocyte ratio, (G) monocyte-to-lymphocyte ratio, and (H) systemic immune-inflammation index.
Univariate and multivariate logistic regression analysis in the irAE group.

Odds ratios (OR) and 95% confidence intervals (CI) were calculated using univariate and multivariate logistic regression analysis. Continuous variables were included in the regression model using their original numerical values.
Variables with P < .001 in the univariate analysis were included in the multivariate model. Statistically significant P-values (P < .05) are shown in bold.
Clinical factors associated with overall survival in patients with immune-related adverse events
This study conducted a comprehensive evaluation of clinical prognostic factors associated with overall survival (OS) in patients experiencing irAEs, using both univariate and multivariate Cox regression analyses. The univariate analysis identified 3 significant predictors of reduced OS: advanced age (> 65 years; hazard ratio [HR] = 1.04, 95% CI =1.00-1.08, P = .013), elevated NLR (⩾4.28; HR = 1.12, 95% CI = 1.03-1.22, P = .006), and increased LDH levels (⩾204.45 U/L; HR = 1.00, 95% CI = 1.00–1.00, P = .013). The multivariate analysis confirmed that an NLR ⩾ 4.28 (HR = 1.14, 95% CI = 1.05–1.24, P = .002) and LDH ⩾ 204.45 U/L (HR = 1.00, 95% CI = 1.00-1.01, P = .03) were independent prognostic factors for OS. However, age did not retain statistical significance in the multivariate model (P = .07).
These findings firmly establish the NLR and LDH levels as robust prognostic biomarkers in patients experiencing irAEs. The clinical utility of these biomarkers is significant, as they can be used for risk stratification and to optimize personalized treatment strategies. The study underscores several critical research priorities. These include large-scale validation of the predictive value of these biomarkers to confirm their reliability across diverse patient populations. In addition, there is a need for mechanistic investigations to elucidate the role of NLR and LDH in the pathogenesis and progression of irAEs. Such research will enhance our understanding of the underlying biological processes and potentially lead to more effective interventions (Table 4 and Figure 5).
Cox proportional hazards regression analysis of clinical factors associated with OS in patients with irAEs.

The table presents the results of univariate and multivariate Cox regression analyses, reporting the “Hazard Ratio (HR)” with its “95% Confidence Interval (CI)” for each clinical variable. A multivariate model was constructed only for variables that showed a potential association (P < .1) in the univariate analysis; a dash (-) indicates that a variable was not included in the multivariate model. An “HR > 1” indicates a factor associated with worse survival, while an “HR < 1” indicates a protective factor. Statistically significant P-values (P < .05) are shown in bold.
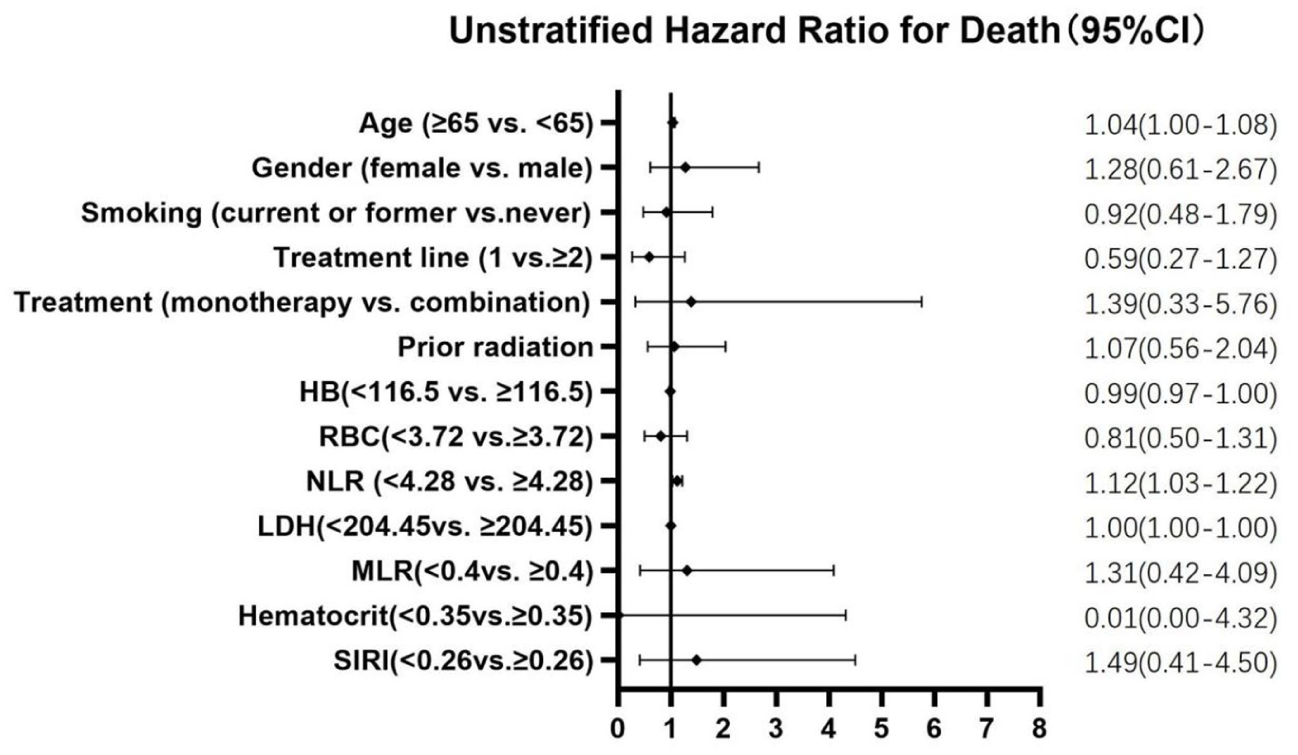
Forest plot of prognostic factor analysis for the overall survival rate of irAEs.
Discussion
This real-world study utilized a comparative analysis framework, juxtaposing the irAE cohort against a control group to meticulously scrutinize the multifaceted etiology of irAEs. The analysis unveiled a triad of significant associations with irAE occurrence: patient age, prior radiotherapy exposure, and discernible changes in hematological parameters. The forthcoming discourse will meticulously dissect these findings, employing a multifarious analytical lens to elucidate their implications and interconnections.
Incidence and risk factors of immune-related adverse events
The crude incidence rate of irAEs reported in this study was 16.91%, which falls within the ranges reported in some previous literature. However, due to the limited sample size and selected population, the generalizability of this rate should be interpreted with caution. Multivariable analysis indicated that advanced age (OR = 0.96, 95% CI = 0.95-0.98; P < .001) and a history of radiotherapy (OR = 2.35, 95% CI = 1.58-3.50; P < .001) were significantly associated with the development of irAEs.
Although patients aged ⩾65 years exhibited a significantly higher incidence of irAEs (51.7% vs 21.4%; P < .001), this association may not only stem from biological mechanisms such as immunosenescence but could also be influenced by confounding factors, including a higher burden of comorbidities and polypharmacy commonly observed in older adults. Similarly, while a history of radiotherapy was identified as a significant risk factor—potentially mediated through radiation-induced alterations in the tissue microenvironment and immune dysregulation—these findings require further validation in large prospective cohorts incorporating detailed radiation dosimetry and organ-specific toxicity assessments.
Organ distribution and temporal characteristics of immune-related adverse events
The irAEs are characterized by their multi-organ system involvement. Among these, the respiratory system is most frequently affected, accounting for 26.01% of cases, with CIP being the primary manifestation. Although the exact pathogenesis of CIP remains to be fully elucidated, it is primarily attributed to T-cell-mediated immune responses activated by ICIs, which induce pulmonary inflammation and tissue damage. Given its severity and complexity, early detection and immediate intervention for CIP are crucial to optimizing clinical outcomes. Other commonly affected organ systems include the dermatological system (19.81%), endocrine system (15.79%), gastrointestinal system (15.48%), 29 and hematological system (10.22%). In contrast, the neurological, renal, musculoskeletal, and cardiovascular systems exhibit lower frequencies of involvement. Notably, cardiovascular irAEs, particularly myocarditis, although rare (1.86%), 30 represent potentially fatal complications that require vigilant monitoring and prompt therapeutic intervention. Temporal analysis revealed that irAEs predominantly manifest within the initial 2-month window following ICI initiation, with a subsequent reduction in incidence rates. These findings underscore the critical importance of intensive monitoring during the early immunotherapy period, especially for high-risk populations, including elderly patients and those with prior radiotherapy exposure. Proactive surveillance facilitates early detection and management of irAEs, ultimately improving patient prognosis. 31
The NLR is closely related to the body’s immune status and inflammation expression. Neutrophils indicate non-specific inflammation, while lymphocytes have a defensive and regulatory role in the inflammatory response. The combination of the 2 can more comprehensively reflect the body’s inflammatory status. Due to its comprehensive analysis of neutrophils and lymphocytes in white blood cell components, NLR has the advantages of simplicity and easy accessibility. The NLR refers to the ratio of neutrophils to lymphocytes in peripheral blood and is one of the effective indicators of systemic inflammatory response. Elevated NLR may indicate angiogenesis or pro-inflammatory status of tumor tissue, reflecting the balance between neutrophils and lymphocytes and reflecting the patient’s immune status. High NLR may be related to elevated neutrophils or decreased lymphocytes. After the occurrence of a tumor, the tumor area of the body is in an immunosuppressive state with low levels of lymphocytes. The elevation of NLR could reflect a lower lymphocyte-mediated immune function, resulting in a poor prognosis.15,32,33
Correlation between peripheral blood indicators and immune-related adverse events
The existing literature has firmly established the NLR as a significant immunological biomarker in patients experiencing irAEs. Specifically, patients who exhibit an initial elevation in NLR followed by a rapid decline tend to have superior progression-free survival and OS outcomes compared with those with persistently elevated NLR levels. Moreover, extensive research has supported the prognostic utility of NLR and LDH as predictive markers for both clinical outcomes and the development of irAEs in patients undergoing PD-1 inhibitor therapy. In the current study, comprehensive univariate and multivariate logistic regression analyses were conducted. These analyses identified LDH (OR = 1.00, 95% CI = 1.00-1.00, P < .001) and NLR (OR = 1.14, 95% CI = 1.03-1.27, P = .013) as independent predictors of irAE occurrence. These alterations in hematological parameters likely reflect systemic inflammatory responses and immune homeostasis dysregulation induced by immunotherapy. As a result, they offer promising biomarkers for early detection and risk stratification of irAEs.
Our study identified elevated baseline NLR as an independent risk factor for irAEs. This finding suggests that a pre-existing chronic inflammatory microenvironment is a key driver of excessive immune activation following treatment. Elevated neutrophil levels may prime the immune system through the release of pro-inflammatory cytokines such as IL-6 and IL-8, while lymphocytopenia potentially impairs the suppressive function of regulatory T cells (Tregs). Together, these mechanisms may contribute to uncontrolled immune activation after ICI therapy, leading to targeting of normal organs. Furthermore, this observation carries substantial clinical implications. It indicates that calculating the NLR from routine complete blood counts before initiating immunotherapy could help identify patients at high risk of irAEs. For these individuals, clinicians may implement intensified monitoring strategies—such as more frequent follow-ups, patient education, and baseline organ function assessments—as well as low-threshold intervention criteria. Such an approach may facilitate earlier detection and management of irAEs and has the potential to improve patient outcomes.
In addition, there have been no prospective or exploratory studies that have clearly identified any biomarkers related to the occurrence, exacerbation, and prognosis of CIP, up to now. In clinical practice, oncologists rely more on a comprehensive analysis of imaging and relevant blood indicators to diagnose and treat CIP. This indicates that our retrospective study has a certain degree of foresight and predictability in clinical practice and can predict the occurrence, development, and outcome of CIP to a certain extent through simple and feasible hematological indicators.
In the Supplementary Figures 1 and 2, it was reflected that during the progressive aggravation of CIP in 2 patients with advanced tumors, NLR and PLR also showed a trend of progressive increase. Similarly, in Supplementary Figures 3 and 4, it was reflected that 2 patients with advanced tumors showed a corresponding decrease in NLR and PLR after CIP was relieved with symptomatic treatment. This provides further evidence of evidence-based medicine for the practical application of our retrospective study.
Prognostic factors in patients with immune-related adverse events
Through comprehensive univariate and multivariate Cox regression analyses, this study identified 3 significant predictors of reduced OS: advanced age (>65 years), elevated NLR (⩾4.28), and increased LDH (⩾204.45 U/L). Multivariable analysis confirmed NLR and LDH as independent prognostic factors for OS, providing substantial clinical evidence for risk stratification in irAE patients.
The strong association between elevated NLR and reduced OS underscores the potential role of systemic inflammation in irAE prognosis. This finding aligns with existing literature demonstrating NLR’s prognostic value across various malignancies, potentially reflecting tumor-mediated immunosuppression. Similarly, elevated LDH levels, indicative of increased tumor burden and metabolic dysregulation, demonstrated significant prognostic value in this study, further supporting its utility in irAE management and outcome prediction. 34
Significance and limitations of the study
This study has generated several clinically significant findings. First, age and a history of radiotherapy serve as valuable parameters for risk stratification of irAEs. This enables clinicians to identify high-risk patients and implement personalized surveillance protocols. Second, dynamic alterations in hematological parameters, such as HB, NLR, and LDH, may facilitate the early detection of irAEs, especially in cases where imaging findings are ambiguous. Third, intensive monitoring and proactive intervention during the initial period of immunotherapy for high-risk patients may help to mitigate the incidence and severity of irAEs. Furthermore, NLR and LDH have emerged as significant prognostic biomarkers, aiding in the identification of high-risk patients and the planning of individualized treatment. Specifically, patients with an NLR of ⩾ 4.28 or an LDH level of ⩾ 204.45 U/L may require enhanced surveillance and early intervention to optimize outcomes. These biomarkers also provide valuable insights into the pathogenesis of irAEs, particularly in relation to inflammatory responses and metabolic dysregulation.
However, several limitations must be acknowledged. Although we mitigated confounders through matching, the retrospective nature of this study introduces the possibility of selection bias and residual confounding. Most notably, a significant imbalance in prior radiotherapy history existed between the irAEs and control groups. This discrepancy may introduce potential bias. It is well-established that radiotherapy, particularly to the thorax, can cause radiation-induced lymphopenia (RIL), a condition associated with immune dysfunction and unfavorable prognosis. 35 Therefore, the higher incidence of irAEs in patients with radiotherapy history may be partly attributed to radiotherapy-induced alterations in the immune microenvironment, rather than solely to the hematological parameters examined in this study. Although multivariate analysis was performed to adjust for this factor, the possibility of residual confounding cannot be fully excluded. Future studies should ensure balanced radiotherapy exposure in prospective designs or perform stratified analyses comparing patients with and without prior radiotherapy to more accurately clarify the independent contributions of each factor. Furthermore, ongoing debates regarding the optimal modality, dose, and volume of radiotherapy in advanced NSCLC remain unresolved. With the increasing use of stereotactic body radiotherapy (SBRT) in oligometastatic disease and growing exploration of its combination with immunotherapy in patients with a higher metastatic burden (eg, up to 10 lesions), 36 evolving treatment strategies may influence the spectrum and incidence of irAEs. Our study did not analyze these specific radiotherapy parameters in depth, which represents an important direction for future research.
Incomplete serial monitoring of hematological parameters across all patients may impact the precision of the results. In addition, the diagnosis of irAEs primarily relied on clinical and imaging assessments, without histopathological confirmation. Future investigations should incorporate larger prospective cohorts and multi-omics approaches to validate these findings.
In conclusion, this study identifies age and a history of radiotherapy as independent risk factors for irAEs, while dynamic changes in hematological parameters (eg, HB, NLR, LDH) offer predictive value for early irAE detection. Our study identifies elevated pre-treatment NLR (⩾ 4.28) and LDH (⩾ 204.5 U/L) as robust and readily available biomarkers for predicting the development of irAEs and poorer survival in patients with advanced lung cancer receiving ICIs. We propose a practical risk stratification strategy incorporating these 2 parameters to guide more vigilant monitoring and pre-emptive management strategies in high-risk individuals. Future prospective studies are warranted to validate the efficacy of this model in improving patient outcomes.
Conclusion
This study provides a comprehensive analysis of the risk factors for irAE development and their impact on patient survival following PD-1/PD-L1 inhibitor therapy. Regarding irAE risk, elevated NLR and LDH levels were identified as independent predictors. Univariate analysis also indicated older age (⩾ 65 years) and a history of radiotherapy as significant risk factors. The respiratory, skin, endocrine, and gastrointestinal systems were the most frequently involved. More critically, survival analysis (Cox regression) within the irAE cohort revealed that high NLR and high LDH were also independent factors associated with poorer OS. This indicates that these biomarkers not only signal a higher probability of developing irAEs but also help identify a subset of patients with irAEs who have a less favorable prognosis. In contrast, most other clinical variables (eg, gender, treatment line) showed no significant independent association with either irAE occurrence or subsequent survival. In conclusion, NLR and LDH serve as dual-purpose, readily available biomarkers. They are valuable for pre-therapeutic risk stratification to guide proactive monitoring for irAEs and, subsequently, for prognostic stratification among patients who develop irAEs. These findings underscore the importance of integrating these biomarkers into clinical practice to optimize the management and personalization of immunotherapy.
Supplemental Material
sj-docx-5-onc-10.1177_11795549251413304 – Supplemental material for Clinical Analysis of Peripheral Blood Indicators for Immune-Related Adverse Events (irAEs) in Patients With Advanced Lung Cancer
Supplemental material, sj-docx-5-onc-10.1177_11795549251413304 for Clinical Analysis of Peripheral Blood Indicators for Immune-Related Adverse Events (irAEs) in Patients With Advanced Lung Cancer by Yin-Min Ji, Yu-hui Qin, Ya-Hui Lv, Shu-Yan Xiao, Yi Dong, Zhi-Song Hu, Peng-Fei Cui, Tao Li, Xiao-Ran Cui and Yi Hu in Clinical Medicine Insights: Oncology
Supplemental Material
sj-jpg-1-onc-10.1177_11795549251413304 – Supplemental material for Clinical Analysis of Peripheral Blood Indicators for Immune-Related Adverse Events (irAEs) in Patients With Advanced Lung Cancer
Supplemental material, sj-jpg-1-onc-10.1177_11795549251413304 for Clinical Analysis of Peripheral Blood Indicators for Immune-Related Adverse Events (irAEs) in Patients With Advanced Lung Cancer by Yin-Min Ji, Yu-hui Qin, Ya-Hui Lv, Shu-Yan Xiao, Yi Dong, Zhi-Song Hu, Peng-Fei Cui, Tao Li, Xiao-Ran Cui and Yi Hu in Clinical Medicine Insights: Oncology
Supplemental Material
sj-jpg-2-onc-10.1177_11795549251413304 – Supplemental material for Clinical Analysis of Peripheral Blood Indicators for Immune-Related Adverse Events (irAEs) in Patients With Advanced Lung Cancer
Supplemental material, sj-jpg-2-onc-10.1177_11795549251413304 for Clinical Analysis of Peripheral Blood Indicators for Immune-Related Adverse Events (irAEs) in Patients With Advanced Lung Cancer by Yin-Min Ji, Yu-hui Qin, Ya-Hui Lv, Shu-Yan Xiao, Yi Dong, Zhi-Song Hu, Peng-Fei Cui, Tao Li, Xiao-Ran Cui and Yi Hu in Clinical Medicine Insights: Oncology
Supplemental Material
sj-jpg-3-onc-10.1177_11795549251413304 – Supplemental material for Clinical Analysis of Peripheral Blood Indicators for Immune-Related Adverse Events (irAEs) in Patients With Advanced Lung Cancer
Supplemental material, sj-jpg-3-onc-10.1177_11795549251413304 for Clinical Analysis of Peripheral Blood Indicators for Immune-Related Adverse Events (irAEs) in Patients With Advanced Lung Cancer by Yin-Min Ji, Yu-hui Qin, Ya-Hui Lv, Shu-Yan Xiao, Yi Dong, Zhi-Song Hu, Peng-Fei Cui, Tao Li, Xiao-Ran Cui and Yi Hu in Clinical Medicine Insights: Oncology
Supplemental Material
sj-jpg-4-onc-10.1177_11795549251413304 – Supplemental material for Clinical Analysis of Peripheral Blood Indicators for Immune-Related Adverse Events (irAEs) in Patients With Advanced Lung Cancer
Supplemental material, sj-jpg-4-onc-10.1177_11795549251413304 for Clinical Analysis of Peripheral Blood Indicators for Immune-Related Adverse Events (irAEs) in Patients With Advanced Lung Cancer by Yin-Min Ji, Yu-hui Qin, Ya-Hui Lv, Shu-Yan Xiao, Yi Dong, Zhi-Song Hu, Peng-Fei Cui, Tao Li, Xiao-Ran Cui and Yi Hu in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
We are grateful to all patients, their families, and all the collaborative group members in this trial.
Ethical Considerations
This study was approved by the Ethics Committee of the PLA General Hospital of China in accordance with the ethical standards of the Declaration of Helsinki and its subsequent amendments (Ethical Approval Number: S2021-462-02). Ethical approval was obtained on September 28, 2023.
Consent to Participate
All participants in this study provided written informed consent.
Patient consent for publication
Not applicable.
Author contributions
T.L. and Y.-M.J. contributed to the conceptualization and design. Y.-M.J., Y.D., and X.-R.C. contributed to the data collection and analysis. Y.Q., S.-Y.X., Y.D., and Y.-M.J. contributed to the manuscript drafting. Z.-S.H, P.-F.C., and Y.H.L. contributed to the critical revision of the article for important intellectual content. Y.H. contributed to the study supervision. All authors have approved the final version of the article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
