Abstract
Background:
Colonic gastrointestinal stromal tumors (cGISTs) represent an exceptionally rare subtype of gastrointestinal stromal tumors (GISTs) and exhibit distinct clinicopathological features. Nonetheless, limited research has systematically assessed the prognostic differences between cGISTs and GISTs originating from other anatomical sites under comparable baseline and oncological conditions. Current prognostic stratification systems for GISTs have significant limitations in evaluating colonic subtypes.
Methods:
A retrospective case-control study. Surgical cGIST cases (2012-2024) from 4 tertiary hospitals were analyzed, along with 676 contemporaneous gastric GISTs (gGISTs) controls. Additional individual patient data were extracted from published articles, reviews, letters, and comments identified through a systematic literature search. Propensity score matching (1:4) was performed to balance baseline variables before comparing survival outcomes.
Results:
Compared with gGISTs, cGISTs exhibited distinct clinicopathological characteristics, including older average age (P = .001), larger tumor diameter (P = .003), higher mitotic index (P = .007), and lower positive expression rates of CD117 (79.25% vs 98.81%, P < .001), CD34 (80.43% vs 98.81%, P < .001), and DOG-1 (76.67% vs 98.35%, P < .001). After propensity score matching successfully eliminated baseline differences, gGIST patients demonstrated superior survival outcomes, with higher 1-, 3-, and 5-year recurrence-free survival (RFS) rates of 100.0%, 98.76%, and 94.13%, compared with 95.50%, 86.69%, and 86.69% in matched cGIST cases. Similarly, gGIST patients showed higher 1-, 3-, and 5-year overall survival (OS) rates of 100.0%, 99.39%, and 97.06%, compared with 95.50%, 86.58%, and 82.45% in matched cGIST cases. Of note, the difference in OS was statistically significant (P = .04), whereas the difference in RFS did not reach significance (P = .31).
Conclusions:
The cGISTs exhibit distinct clinicopathological characteristics and are associated with a significantly poorer prognosis compared with gGISTs. Therefore, diagnostic and therapeutic strategies for cGISTs warrant further exploration and refinement.
Introduction
Gastrointestinal stromal tumors (GISTs) are the most common mesenchymal neoplasms of the gastrointestinal tract, with an annual incidence ranging from 0.6 to 2.2 per 100 000 individuals. Notably, GISTs occur predominantly in the stomach, while cases in the esophagus (<1%) and colorectum (5%) are rare.1,2 Among these, colonic GISTs (cGISTs) account for only 1% to 2% of all GISTs and mainly arise in the sigmoid colon of elderly patients.3,4
The cGISTs exhibit aggressive features, including larger tumor size, higher recurrence risk, and poorer outcomes compared with gastric GISTs (gGISTs). 5 Although tumor size and mitotic index are established prognostic factors, 6 only a limited number of retrospective studies6-11 and reviews5,12,13 have directly compared cGISTs and gGISTs with matched pathological parameters, hindering the development of conclusive evidence on site-specific biological behavior. Current prognostic stratification systems for GISTs have significant limitations in evaluating colonic subtypes. The modified National Institutes of Health (NIH) risk classification distinguishes only between gastric and non-gastric tumors, while the Armed Forces Institute of Pathology (AFIP) Miettinen criteria address gastric, jejunoileal, duodenal, and rectal tumors only. Thus, neither system provides validated risk stratification parameters for cGISTs.
To address this knowledge gap, we conducted a propensity score-matched cohort analysis to elucidate the clinicopathological distinctions between cGISTs and gGISTs. By standardizing baseline and oncological variables, the present study aims to provide evidence to inform site-specific management strategies.
Methods
Inclusion and exclusion criteria
Patient data were retrospectively collected from 4 tertiary medical centers: Peking University People’s Hospital, Peking University Shenzhen Hospital, Shandong Provincial Hospital, and The First Affiliated Hospital of Shandong First Medical University. The cohort included surgically resected, pathologically confirmed cGIST cases diagnosed between January 2012 and October 2024. The study analyzed 15 cGIST cases from the 4 tertiary hospitals. A contemporaneous control group consisted of 676 gGIST cases collected from the same institutions. Extracted parameters included age, sex, tumor location, size, growth pattern (intraluminal/transmural), cell morphology (spindle/epithelioid), mitotic index (/5 mm2), tumor rupture status, modified NIH risk category, immunohistochemical markers (CD117, CD34, DOG-1, Ki-67, SMA, S-100, desmin, CK), presenting symptoms, preoperative imaging (computed tomography [CT]/magnetic resonance imaging [MRI]), surgical approach, adjuvant/neoadjuvant-targeted therapy, and follow-up data (recurrence patterns, survival). All cases underwent diagnostic and therapeutic standardization through a multidisciplinary team (MDT) consensus. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 14 This study was conducted in accordance with the principles of the Declaration of Helsinki, as revised in 2024.
To construct a literature-derived patient cohort, we conducted a systematic search of PubMed, Embase, and WoSCC databases for published articles, reviews, letters, and comments on cGISTs, by October 31, 2024. The complete search strategies for all databases are provided in Supplementary Material 1. The goal was to identify studies from which individual patient data could be extracted. Non-English publications, duplicate entries identified by EndNote (version 21), and unpublished conference proceedings were excluded. Two reviewer teams (Jianqiao Yang/Guoshuai Chen and Zhuofei Li/Dongchen Zhang) independently applied the inclusion and exclusion criteria after completing screening training. Full-text articles were retrieved and assessed when necessary. Disagreements were resolved through group discussion according to pre-specified guidelines.
Patient-level data were extracted from eligible studies according to the predefined criteria. Only studies providing comprehensive individual patient information were included, and non-English articles were excluded. The same 4 reviewers completed the selection process. The literature search and screening process was conducted systematically, and its reporting adheres to the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines to ensure transparency. A flow diagram illustrating the screening and inclusion process is shown in Figure 1.

Flow chart of included literature.
The primary endpoint was recurrence-free survival (RFS), defined as the time (in months) from definitive tumor resection to the first documented locoregional recurrence or distant metastasis. Secondary endpoints included overall survival (OS), defined as the time (in months) from curative surgery to death from any cause. For patients without documented events, RFS and OS were administratively censored at the last follow-up.
Statistical analysis
Data analysis was performed using SPSS (version 21.0) and R (version 4.3.1). Continuous variables were reported as mean ± standard deviation or median with interquartile range, while categorical variables were expressed as frequencies and percentages. Categorical variables were analyzed using the Pearson χ2 test or the Fisher exact test, depending on expected values. Skewed continuous variables were compared using the Mann-Whitney U test and reported with dual descriptors: median with interquartile range to describe distribution shape and mean ± standard deviation for central tendency. Time-to-event outcomes were analyzed using the Kaplan-Meier survival curves with comparisons between strata via the log-rank test. Multivariable Cox proportional hazards models were used to estimate hazard ratios (HRs) with 95% confidence intervals (CIs). Type I error was controlled at a .05 significance level, with the Benjamini–Hochberg adjustment applied for multiple comparisons.
Propensity score matching
To address inherent covariate imbalances in baseline demographics and tumor biological characteristics between the cGIST and gGIST cohorts, propensity score matching (PSM) was performed. A logistic regression model was used to assess associations between the cGIST cohort and relevant baseline variables. Consistent with established methodologies in the literature, categorical variables were appropriately handled throughout the analysis.
The PSM was performed using the PSMatch2 package in Stata, yielding a 1:4 matched cohort of cGIST and gGIST cases. The 1:4 ratio represents an optimal balance between statistical efficiency and bias reduction for rare subgroups. Based on case-control design principles, this configuration maintains a power of ⩾80% to detect moderate-to-strong effects (HR ⩾ 2.0) when the index group includes fewer than 100 cases. A caliper width of 0.2 was applied using the nearest-neighbor algorithm to optimize covariate balance across key prognostic variables, including age, tumor size, mitotic index, and cellular morphology. Variables that did not meet the predefined matching criteria or lacked sufficient data completeness were systematically excluded.
Survival analysis was conducted in 2 stages. First, univariable Cox proportional hazards regression identified variables with marginal significance (P < .2). These variables were subsequently entered into a multivariable Cox model to determine independent prognostic factors. The assumption analyses and assessment of PSM reporting quality are presented in Supplemental Digital Content 1 and Supplemental Digital Content 2.
Results
Patients’ selection
As shown in Figure 1, systematic literature searches of PubMed, Embase, and WoSCC databases from inception up to October 2024 identified 2915 potentially relevant records. Non-English publications, duplicates detected using EndNote (version 21), and unpublished conference abstracts were excluded. After applying the inclusion and exclusion criteria, 84 pathologically confirmed cGIST cases were initially identified, with patient-level data ultimately extracted from 38 studies. This cohort was then expanded through multicenter collaboration to include consecutively enrolled patients diagnosed and treated between January 2012 and October 2024 at 4 tertiary referral centers: Peking University People’s Hospital, Peking University Shenzhen Hospital, Shandong Provincial Hospital, and The First Affiliated Hospital of Shandong First Medical University.
To minimize bias from variability in surgical proficiency, all procedures were performed by board-certified attending surgeons with specialized expertise in gastrointestinal oncology. Following rigorous quality control, the final analytic data set comprised 99 histologically confirmed cGIST cases and 676 gGIST cases from these institutions, forming the basis for subsequent comparative analyses.
Colonic gastrointestinal stromal tumors
As summarized in Table 1, female patients comprised 48.88% of the cohort, with a mean age at diagnosis of 62.57 years; notably, 59.60% of patients were over 60 years old. Specifically, age was dichotomized using the mean as the cutoff. Tumor locations along the colon were as follows: sigmoid colon (31.52%, 29/92), cecum (28.26%, 26/92), transverse colon (15.22%, 14/92), descending colon (13.04%, 12/92), and ascending colon (11.96%, 11/92). Regarding gross morphology, extraluminal tumors (44.87%, 35/78) were more common than intraluminal lesions (30.77%, 24/78), while infiltrative growth patterns were observed in 24.36% (19/78) of cases, primarily derived from literature-based reports.
Clinical and surgical characteristics of cGIST patients.

ANED, Alive with No Evidence of Disease; AWD, Alive With Disease; DOC, Dead Of Other Causes; DOD, Dead Of Disease.
Pathologically, tumor size ranged from 0.2 to 22.0 cm, with a mean diameter of 6.89 cm; more than half of the tumors (56.18%, 50/89) exceeded 5 cm in diameter. The spindle cell phenotype predominated, accounting for 93.13% (82/89) of cases. Assessment of mitotic activity showed that most tumors had either <5 mitoses/50 high-power fields (HPFs) (54.43%, 43/79) or >10 mitoses/50 HPF (34.18%, 27/79). According to the NIH risk stratification system, 64.52% (60/93) of patients were classified as high-risk. Clinically, the most frequently reported symptoms were abdominal pain (32.47%, 25/77) and gastrointestinal bleeding (16.88%, 13/77). However, 28.57% (22/77) of patients were asymptomatic at presentation. Surgically, most patients (61.02%, 36/59) underwent local resection, while radical anatomical resection was performed in 37.29% (22/59), and 1 patient (1.69%) received palliative surgery. Among patients with available data on surgical approach, open surgery (69.77%, 30/43) was more common than laparoscopic resection (30.23%, 13/43). Adjuvant therapy was administered to 20 patients, including 8 from the institutional cohort and 12 from published literature, while 7 patients received neoadjuvant treatment. During follow-up, tumor recurrence occurred in 7 patients, and distant metastases were documented in 17 patients.
Propensity score matching cohort
As presented in Table 2, prior to PSM, significant differences were observed between the cGISTs and gGISTs cohorts in key variables, including patient age (P = .001), tumor size (P = .003), mitotic index (P = .007), and cellular morphology (P < .001). After PSM, these variables were well balanced between the groups, with no statistically significant differences. Notably, the NIH risk classification, determined by tumor site, size, and mitotic index, was intentionally excluded from the matching process to avoid collinearity and potential confounding. The Kaplan-Meier survival analysis revealed significant differences in RFS and OS between the cohorts prior to matching (P < .0001; Figure 2A and B). In the gGISTs group, the 1-, 3-, and 5-year RFS rates were 100.0%, 98.70%, and 95.74%, respectively, with OS rates of 100.0%, 99.65%, and 97.61%. By contrast, the cGIST cohort had lower RFS rates of 86.79%, 73.49%, and 70.29%, and OS rates of 88.08%, 74.54%, and 65.89% over the same intervals. After PSM (Figure 3A and B), survival differences persisted, although attenuated. In the matched cohort, the 1-, 3-, and 5-year RFS rates for gGISTs were 100.0%, 98.76%, and 94.13%, compared with 95.50%, 86.69%, and 86.69% for cGISTs. Corresponding OS rates were 100.0%, 99.39%, and 97.06% for gGISTs, and 95.50%, 86.58%, and 82.45% for cGISTs. In the matched cohorts, OS differed significantly between the cGIST and gGIST groups (P = .04), whereas the difference in RFS was not statistically significant (P = .31). These findings indicate that cGISTs are associated with poorer long-term outcomes, particularly in terms of OS, even after adjustment for major prognostic factors.
Patient baseline characteristics and follow-up results in unmatched and matched cohorts.
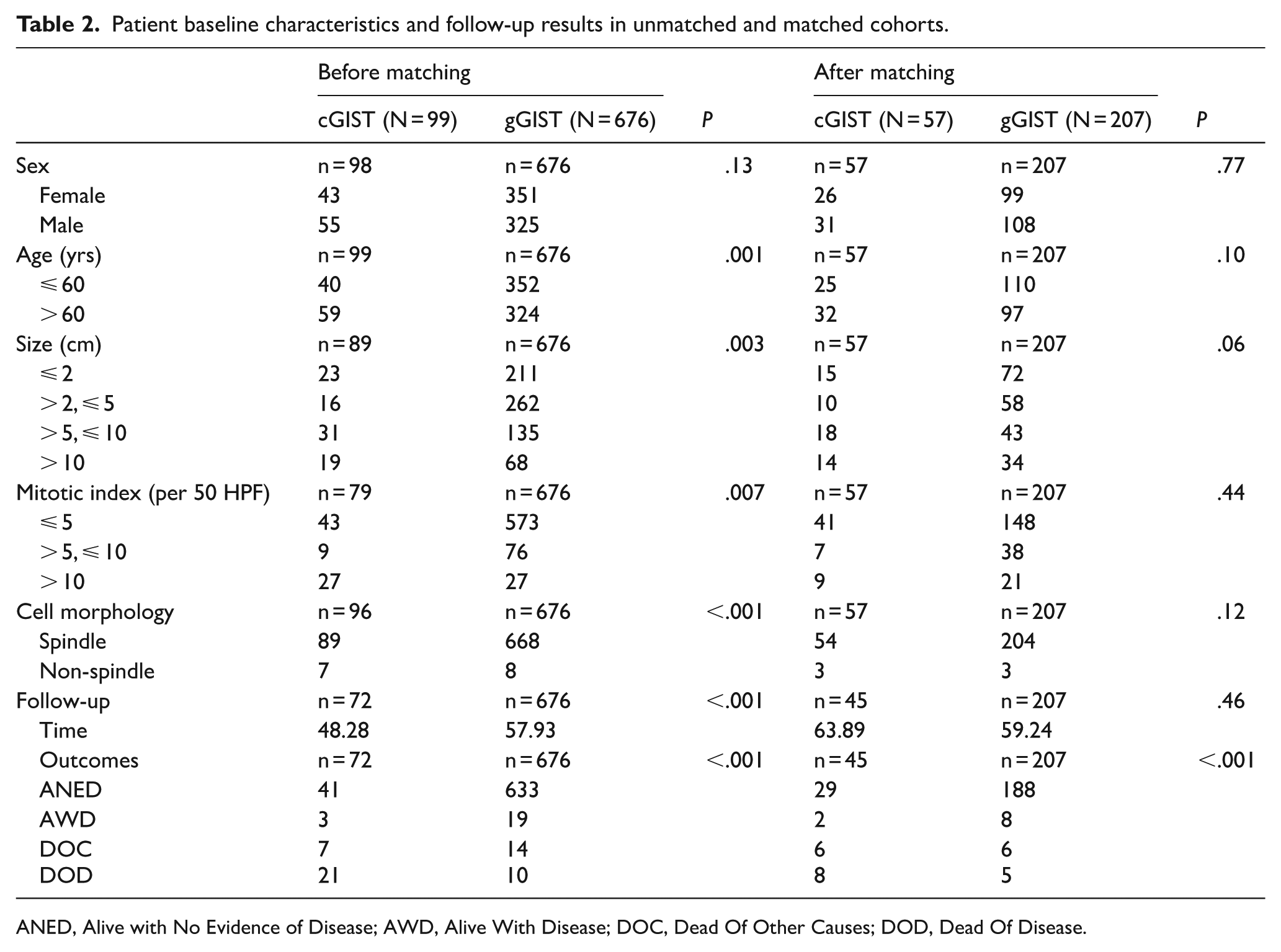
ANED, Alive with No Evidence of Disease; AWD, Alive With Disease; DOC, Dead Of Other Causes; DOD, Dead Of Disease.

Recurrence-free survival (A) and overall survival (B) of cGISTs and gGISTs before propensity score matching.

Recurrence-free survival (A) and overall survival (B) of cGISTs and gGISTs after propensity score matching.
Discussion
As a rare subset with limited clinical data, previous studies have shown that cGISTs differ from GISTs at other anatomical sites.3,5,9,15 However, none of these studies compared outcomes while controlling for comparable clinicopathological characteristics. Given that imbalanced covariates can artificially exaggerate or mask true biological differences between cGISTs and gGISTs, we employed PSM to balance baseline characteristics, enabling a statistically robust comparison of long-term outcomes. Regarding sex distribution, previous research indicates that GISTs affect both sexes equally,1,16 and earlier studies specifically on cGISTs also found no significant sex differences (P > .05).17-19 Consistently, our cohort showed no statistically significant sex difference in either group (P > .05), suggesting that the sex distribution of cGISTs aligns with that of GISTs overall.
Previous studies have reported the most common locations of cGISTs as the sigmoid and transverse colon.9,20 Herein, the most frequent sites were the sigmoid colon (31.52%, 29/92) and the cecum (including the appendix, 28.26%, 26/92), showing a slight discrepancy from earlier reports. We hypothesize that GISTs originating from the appendix, which may clinically mimic appendicitis, could contribute to publication bias and thus increase the observed proportion of cecal GISTs in our cohort. Notably, Hagger et al 21 demonstrated that Cajal cells are primarily located in the myenteric plexus of the transverse colon rather than the sigmoid colon, making the distribution of cGIST difficult to explain. Gomez-Pinilla et al 22 reported that the number of Cajal cells declines with age throughout the colon, by approximately 13% per decade, while this decline occurs more slowly in the sigmoid colon, which may partly explain the higher frequency of sigmoid cGISTs in older patients. Compared with gGISTs, cGISTs exhibited larger tumor diameters and higher mitotic indices, resulting in a greater proportion of high-risk cases according to established risk stratification systems (eg, NIH or AFIP criteria). This elevated risk profile has important clinical implications, as patients with high-risk GISTs are strongly recommended to receive adjuvant imatinib therapy to reduce the risk of recurrence and metastasis. Therefore, accurate identification of these adverse pathological features is crucial for prognostic assessment and tailoring postoperative therapeutic strategies. 23 Cell morphology is a critical factor in the diagnosis of GISTs. The spindle cell type is typically associated with gastric location, v-kit Hardy-Zuckerman 4 feline sarcoma viral oncogene homolog (KIT) negativity, and platelet-derived growth factor receptor alpha (PDGFRA) mutation. Conversely, non-spindle cell types were observed in 7.87% of cGISTs and 1.18% of gGISTs in our cohort and were associated with larger tumor size, a higher mitotic index (>5/50 HPF), and increased tumor-related mortality. 3 Regarding immunohistochemical features, cGISTs exhibited significantly lower positive expression rates for CD117 (79.25%, 42/53 vs 98.81%, 663/671; P < .001), CD34 (80.43%, 37/46 vs 98.81%, 661/669; P < .001), and DOG-1 (76.67%, 23/30 vs 98.35%, 657/668; P < .001) compared with gGISTs, highlighting their distinct characteristics. Clinically, abdominal pain, bleeding, and intestinal obstruction are common presenting complaints in GIST patients. 24 Nonetheless, 15% to 30% of GIST cases are asymptomatic and are often discovered incidentally during surgery for unrelated conditions. 25 In this multicenter cohort, the most common symptoms among cGIST patients were abdominal pain (32.47%, 25/77) and gastrointestinal bleeding (16.88%, 13/77), while 28.57% of patients were asymptomatic. Currently, diagnostic approaches for cGISTs rely primarily on CT and colonoscopy. However, both modalities are costly, and colonoscopy is invasive, limiting their widespread use. Improving the detection of asymptomatic cGIST patients thus remains an important area for future research.
According to current guidelines and expert consensus on GISTs, cGISTs should be surgically excised regardless of tumor size or risk category. 5 En bloc segmental resection is generally sufficient, with radical resection to achieve negative margins indicated only when adjacent organ involvement is suspected.15,26 One study reported a rate of local excision for cGISTs of 8.4%. 27 Meanwhile, a previous study found that the most commonly used surgical method for cGISTs was radical resection (35.34%), followed by local resection (31.33%). In contrast, for GISTs at other sites, local resection was the most frequently adopted approach (44.45%). 19 In our cohort, 61.02% (36/59) of cGIST patients underwent local resection, while 37.29% (22/59) underwent radical resection. The relatively high rate of radical resection in our cohort may be attributed to several factors: (1) some patients had GISTs of the appendix, which increases the likelihood of radical resection; (2) cGISTs are rare, and the accuracy of preoperative diagnosis is relatively low, leading surgeons to favor radical surgery; and (3) cGISTs are often large, making it difficult to achieve a tension-free anastomosis after local resection, so surgeons may prefer radical resection to allow for more extensive tissue mobilization.
Most colonic/rectal GISTs (crGISTs) are classified as high-risk for recurrence, emphasizing the need for long-term follow-up and targeted therapy.28,29 For rectal GISTs (rGISTs), guidelines recommend neoadjuvant targeted therapy when baseline assessment indicates potential need for combined organ resection or abdominoperineal resection (APR). 30 On the contrary, preoperative treatment is not routinely recommended for cGISTs. One study reported that postoperative adjuvant therapy was used significantly more often in cGISTs patients than in rGIST patients (93.6% vs 51.6%, P < .05). 31 However, some studies suggest that tyrosine kinase inhibitor (TKI) therapy does not improve OS in cGISTs patients and may even be associated with worse OS in tumors smaller than 5 cm. 26 In the present study, the rate of neoadjuvant targeted therapy was significantly higher in cGISTs than in gGISTs (79.55% vs 13.49%, P < .05), primarily due to the larger tumor size and deep-seated anatomical location of cGISTs, which necessitate tumor downsizing to achieve R0 resection, as well as potential variations in clinical practice between institutions. In contrast, the rate of postoperative adjuvant therapy was similar between the 2 groups (23.38% vs 28.52%, P > .05), reflecting adherence to standardized risk-stratified protocols for post-resection treatment decisions. Nonetheless, both RFS and OS were worse in cGISTs compared with gGISTs. After PSM, cGISTs continued to demonstrate inferior OS (P = .04), although not all clinicopathological variables were included in the matching, leaving the possibility of residual confounding. Together with previous findings, 26 our results suggest that cGISTs patients may respond poorly to current therapies and exhibit distinct biological behavior related to their primary anatomical site.
Furthermore, our study suggests that patients with cGISTs might be at risk of overtreatment in terms of surgical approach and targeted therapy. Consequently, current diagnostic and treatment strategies require optimization. Greater emphasis on preoperative needle biopsy and improved diagnostic accuracy could help guide the selection of more appropriate surgical procedures. In addition, whether TKI is being overused remains to be determined through rigorous prospective studies.
Nevertheless, the present study has several limitations. First, the relatively small sample size, particularly within the cecal GISTs subgroup, may limit statistical power and generalizability. Second, potential confounding effects from combined therapies, such as concurrent use of imatinib with other treatments, were not systematically adjusted for, which could have influenced outcomes. Third, the absence of detailed molecular data, including KIT/PDGFRA mutation status, restricted our ability to perform molecular subtype-specific analyses or explore genotype-phenotype correlations. Although PSM improved comparability between cGISTs and gGISTs for baseline characteristics, key prognostic variables, such as tumor rupture status and duration of adjuvant therapy, were not included due to inconsistent documentation. Moreover, the low number of outcome events precluded Cox multivariate regression analysis, which would have allowed more robust adjustment for confounding factors. Finally, including only English-language publications may introduce selection bias, potentially affecting the comprehensiveness of our literature-based cohort.
Conclusion
Colonic GISTs exhibit distinct characteristics, including an older age at presentation, a low incidence, large tumor size, high mitotic activity, a poor prognosis, and potential resistance to TKI therapy, reflecting their unique biological behavior. The current prognostic stratification systems for GISTs could potentially enable further risk stratification of cGISTs.
Supplemental Material
sj-docx-2-onc-10.1177_11795549251399463 – Supplemental material for Colonic Gastrointestinal Stromal Tumors Demonstrate Unique Biological Characteristics: A Retrospective, Propensity Score-Matched Cohort Study Pooling Multicenter and Literature-Derived Individual Patient Data
Supplemental material, sj-docx-2-onc-10.1177_11795549251399463 for Colonic Gastrointestinal Stromal Tumors Demonstrate Unique Biological Characteristics: A Retrospective, Propensity Score-Matched Cohort Study Pooling Multicenter and Literature-Derived Individual Patient Data by Zhuo-Fei Li, Guo-Shuai Chen, Han Li, Chen Li, Hao Wu, Ke-Wei Jiang and Ying-Jiang Ye in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-3-onc-10.1177_11795549251399463 – Supplemental material for Colonic Gastrointestinal Stromal Tumors Demonstrate Unique Biological Characteristics: A Retrospective, Propensity Score-Matched Cohort Study Pooling Multicenter and Literature-Derived Individual Patient Data
Supplemental material, sj-docx-3-onc-10.1177_11795549251399463 for Colonic Gastrointestinal Stromal Tumors Demonstrate Unique Biological Characteristics: A Retrospective, Propensity Score-Matched Cohort Study Pooling Multicenter and Literature-Derived Individual Patient Data by Zhuo-Fei Li, Guo-Shuai Chen, Han Li, Chen Li, Hao Wu, Ke-Wei Jiang and Ying-Jiang Ye in Clinical Medicine Insights: Oncology
Supplemental Material
sj-jpg-1-onc-10.1177_11795549251399463 – Supplemental material for Colonic Gastrointestinal Stromal Tumors Demonstrate Unique Biological Characteristics: A Retrospective, Propensity Score-Matched Cohort Study Pooling Multicenter and Literature-Derived Individual Patient Data
Supplemental material, sj-jpg-1-onc-10.1177_11795549251399463 for Colonic Gastrointestinal Stromal Tumors Demonstrate Unique Biological Characteristics: A Retrospective, Propensity Score-Matched Cohort Study Pooling Multicenter and Literature-Derived Individual Patient Data by Zhuo-Fei Li, Guo-Shuai Chen, Han Li, Chen Li, Hao Wu, Ke-Wei Jiang and Ying-Jiang Ye in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
The authors thank the library of Peking University for database accessing and acquiring full texts and Hong-Chen Zheng, Quan Wang, Ke-Lu Yang, Jianqiao Yang, and Dongchen Zhang providing help with the statistical analysis. Finally, the authors give their respect to all patients involved in the study.
Ethical considerations
Ethical approval for this retrospective analysis of anonymized data was obtained from the Ethics Committees of all participating centers: Peking University People’s Hospital (Approval No: 2024PHB210-001) on July 2, 2024; Peking University Shenzhen Hospital (Approval No: 2025(254)) on October 13, 2025; The First Affiliated Hospital of Shandong First Medical University (Approval No: 2025(S919)) on October 9, 2025; and Shandong Provincial Hospital (Approval No: SWYX:NO.2021-035) on February 4, 2021. The committees provided retrospective approval for the secondary use of these data and waived the requirement for informed consent.
Consent to participate
The requirement for informed consent was waived by the Institutional Review Board (IRB) due to the retrospective nature of the study and the use of anonymized data.
Consent for publication
Not applicable.
Author contributions
CL: Conceptualization (lead); Data curation (equal); Formal analysis (lead); Visualization (supporting); Writing—original draft (supporting); Writing—review and editing (supporting). G-SC: Conceptualization (equal); Data curation (supporting); Visualization (lead); Writing—original draft (supporting). HL: (supporting); Data curation (lead); Investigation (supporting); Methodology (lead); Supervision (lead). Z-FL: Supervision (supporting); Validation (supporting); Data curation (lead). HW: Data curation (equal); Formal analysis (equal); Visualization (supporting). KWJ: Funding acquisition (lead); Methodology (equal); Supervision (equal); Writing—review and editing (supporting). Y-JY: Methodology (equal); Supervision (equal); Writing—review and editing (lead). CL, G-SC, and HL contributed equally to this work. Z-FL, K-WJ, and Y-JY contributed equally to this work. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Scientific Center Project (No. 62088101).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed during this study are included in this published article. Further information can be obtained from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
