Abstract
Accumulating evidence demonstrates that the tumor microenvironment (TME) drives immune suppression through complicated regulations including host-microbe interactions, which poses vaginal microbiome as one of vital regulators of immune microenvironment. This narrative review examined the composition and dynamic changes of vaginal microbiota during carcinogenesis, focusing on mechanistic insights linking microbial dysbiosis to tumor immunity. Notably, commensal bacteria exhibit diverse immunoregulatory functions that can either potentiate or inhibit anti-tumor responses. Clinical evidence further reveals that CST IV microbiota associates with significantly elevated cancer risk, while probiotic interventions show promise in restoring immune surveillance. Critical gaps in standardization of microbiota-based therapies are addressed, emphasizing the need for strain-specific characterization and optimized delivery systems. Collectively, deciphering vaginal microbiome-immune crosstalk opens new avenues for precision interception against cervical cancer.
Keywords
Introduction
Cervical cancer (CC), a malignant tumor arising from the female cervix, characterized by the infection of human papilloma virus (HPV) and abnormal uterine bleeding (AUB). The latest figures from the American Cancer Society show that cervical cancer ranks second in diagnosed gynecological malignancies, with a notable rise in cases among younger individuals. 1 In 2020, the World Health Organization (WHO) and the International Agency for Research on Cancer (IARC) reported nearly 604 000 global cases of cervical cancer, leading to 342 000 fatalities. This data highlights the persistent and substantial health risk that cervical cancer poses to women globally.
The female reproductive system, akin to the gastrointestinal system, supports a diverse array of unique microbiomes. A healthy vaginal microbial environment is dominated by Lactobacillus species, which are essential for preserving female health and homeostasis. A comprehensive analysis has revealed that the vagina harbors more than 50 species, with bacterial counts ranging from 108 to 109. 2 Recent studies have investigated the associations between cervical cancer and distinct microbial communities. A hallmark of vaginal microbiota alteration during cervical malignancy progression is the reduction of Lactobacillus species. 3 Women with HPV infection or high-grade cervical epitheliopathy (HSIL) exhibit a non-Lactobacillus dominated microbial system, accompanied by an increase in bacterial diversity.
Recent studies have highlighted similarities and close connections between the female gastrointestinal and vaginal system (Figure 1), which can be attributed to their anatomical proximity and shared microbial communities. 3 Considering the ecological continuum facilitated by the exchange of microbes along the gut–vagina axis, it is expected that over 30% of the microbiome species are shared by both areas, with numerous prevalent species being identical. 4 While these bacteria largely retain their physiological and biochemical properties in the vaginal environment, the metabolites trigger distinct immune responses. 4 Within the gut, the intestinal microbiota predominantly manifest anti-inflammatory properties, a role crucial for anaerobic bacteria in preserving gut homeostasis. Conversely, cervical epithelial lesion patients similarly exhibit a high abundance of anaerobic bacteria in vagina as well, including Gardnerella, Prevotella, Atopobium, Sneathia, and Megasphaera. These bacteria have been linked to chronic inflammation and an elevated risk of cervical epithelial neoplasia. 3 Moreover, certain strains of microflora have been implicated in regulating the tumor immune response. 5

Interactions of the microbiome within the female reproductive system: Bacteria migrate among the urinary, reproductive, and gastrointestinal tracts through both direct and indirect pathways.
Although direct studies of specific bacterial species in cervical cancer and immune modulation have not been as extensively conducted as those in gastrointestinal tumors, insights based on the gut microbiome offer valuable perspectives. Findings from these studies highlight the immunomodulatory roles of specific bacteria and their metabolites, which are likely to have parallels in the vaginal environment. This narrative review aims to bridge the gap by discussing the potential implications of gut microbiome research for understanding cervical cancer, focusing on bacterial compositional changes, potential immunomodulatory effects, and therapeutic value in cervical cancer. The search strategy for this review is as follows. Electronic searches of PubMed, Embase and Web of Science were conducted to retrieve eligible papers published since 2005. This study was restricted to English-language studies. Keywords including “vaginal microbiome,” “cervical cancer,” “cervical intraepithelial neoplasia,” “microbial dysbiosis,” “immune modulation,” “tumor microenvironment,” “Lactobacillus,” “probiotics,” and “short-chain fatty acids, SCFAs” were used to build the search query. Specific microbial species and keywords mentioned above were constructed into queries to capture the latest research developments. From the initial over 500 retrieved articles, we restricted the literature types to review articles, original research, and clinical trials. Highly relevant studies with sufficient data were selected for further analysis.
Vaginal Microbial Composition in Healthy Woman
Diseases affecting the female reproductive tract are among the most urgent global health concerns. 6 The microbial communities residing in the female reproductive tract hold the potential to provide new insights and treatment approaches for diagnosing and managing these conditions.7,8 A balanced vaginal environment is essential for minimizing the risk of gynecological disorders, including vaginitis, urinary tract infections, and sexually transmitted infections.9-11 The vaginal microbiota, a key element of the vaginal environment, is instrumental in preserving microbial balance through interactions between bacteria and the host. In healthy women of reproductive age, Lactobacillus species typically predominate this ecosystem. A seminal paper 12 published in 2011 systematically categorized the vaginal microbiota into 5 distinct community state types (CSTs), distinguished by their dominant microbial species. Respectively, CST I, II, III, and V are dominated by Lactobacillus crispatus, Lactobacillus gasseri, Lactobacillus iners, and Lactobacillus jensenii. In contrast, CST IV is marked by a high bacterial diversity rather than by a single Lactobacillus species, featuring a mixture of anaerobic bacteria including Gardnerella, Atopobium, Prevotella, and Finegoldia. Subsequent researches have built upon this foundational work, further refining the CST classification into 7 main types and 13 subtypes. 13 A recent study led by Sarah 14 suggests that the microbial composition of the female vagina may be more intricate than previously thought. The study suggests vaginal microenvironment of healthy women is not consistently led by a single Lactobacillus species. For instance, it may be co-dominated by L. crispatus and L. iners, showing traits of both CST I and CST III. This highlights the complexity and dynamism of the vaginal microbiome, which should be thoroughly accounted for in statistical models that examine lifestyle factors and long-term health trajectories.
Ethnicity, Hormones and Lifespan Effect on Vaginal Microbiota
It should be noted that vaginal microbiome exhibits compositional fluctuations influenced by various factors (Figure 2), including but not limited to race, ethnicity, age, hormone level, sexual activity, health status and genetics.15-17 From an ethnogeographic perspective, the vaginal microbiota composition differs across various states and regions, each exhibiting unique characteristics. L. crispatus and L. iners are more commonly found in white and Asian women, whereas African women typically have a greater prevalence of anaerobic bacteria associated with bacterial vaginosis (BV).12,18,19 This pattern corresponds with the higher risk of sexually transmitted infections (STIs) and adverse pregnancy outcomes faced by Black women, both of which are closely associated with vaginal dysbiosis.19,20

Dynamic factors influencing vaginal microbiota composition: Multiple factors can influence the composition of the vaginal microbiota throughout a woman’s life.
Lactobacillus levels vary throughout a woman’s life stages, positively correlating with estrogen and glycogen levels. 16 The initial colonization and long-term dominance of Lactobacillus in female newborns can be attributed to the transfer of placental estrogen and maternal Lactobacillus species. 21 This process is largely due to maternal vertical transmission. 21 During puberty, there is a temporary decrease in Lactobacillus abundance, coinciding with a rapid drop in estrogen and vaginal glycogen levels. As women enter reproductive age, Lactobacillus regains dominance in the vaginal microbiota. However, with the gradual aging and decline in sex hormone levels, Lactobacillus abundance reaches its nadir during menopause. 22
In women of reproductive age, menstruation and pregnancy are the most impactful events on hormonal levels, which dynamically regulate glycogen production and pH levels in the epithelium, resulting in alternating dominance between lactobacilli and opportunistic pathogens.23,24 During menstruation, when estrogen and progesterone levels are at their lowest, there is a corresponding decrease in lactobacillus abundance, increasing the risk of vaginal infections. In the follicular phase, a surge in estrogen levels allows lactobacilli to predominate within the microbial community. A transient fluctuation in vaginal pH during ovulation can cause temporary microbial community disturbances. During the luteal phase, lactobacillus levels decrease once more, and some women may enter a precursor state for bacterial vaginosis, facing an elevated risk for BV and yeast infections.23,24
In addition to the menstrual cycle, pregnancy significantly influences vaginal microbiota composition. Early in pregnancy, there is a notable shift toward a Lactobacillus-dominant microbiome, accompanied by a substantial decrease in bacteria linked to vaginal dysbiosis. 18 This pattern is particularly prevalent among African-American and Hispanic women, likely due to their inherently higher microbial diversity. 18 However, the Lactobacillus-rich microbial community experiences a sharp decline during the postpartum period, accompanied by an increase in bacterial diversity. 25 In addition, a study focusing on reproductive-age Chinese women indicated that CST IV, which is deficient in Lactobacillus, correlates with a higher incidence of adverse pregnancy outcomes. 26
Flora Structure Shifts in Cervical Cancer
Among a multitude of pathogenic bacteria, Helicobacter pylori is the sole microorganism confirmed to directly induce malignant tumors. Beyond specific strains of pathogenic microorganism, imbalances within the microbiota are also suspected to play a role in carcinogenesis. 27 A colon cancer model employing germ-free mice has shown that a reduction in the presence of exogenous bacteria significantly alleviates tumor burden and retards the proliferation of tumor cells. 28 Flora associated with colorectal cancer (CRC), including pks + E. coli and Clostridium difficile, interact with host cells through specific adhesins and release toxins that cause DNA damage, thereby inducing mutations that elevate the risk of epithelial cancer.29,30 In addition, chronic infections resulting from bacterial-host interactions or the production of bacterial metabolites are likely contributors to tumorigenesis. 31 The collective efforts of global researchers have enabled the summarization of the molecular mechanisms of bacterial carcinogenesis into 5 principal categories: chronic toxin-induced damage, epithelial-mesenchymal transition, DNA damage, cell cycle regulation, and modulation of apoptosis. 29 ,32-34
HPV infection is widely acknowledged as a primary etiological factor in cervical cancer. However, among patients infected with high-risk HPV types, only approximately 10% progress to develop cervical intraepithelial neoplasia (CIN) or invasive cervical carcinoma (ICC), with the majority experiencing self-resolving infections. 35 This discrepancy suggests that additional factors influence cervical cancer progression. Recent research has increasingly implicated vaginal dysbiosis as closely linked to HPV infection and as a significant contributor to cervical cancer development.3,5,36,37 A cross-sectional study in China explored the interplay between HPV infection, vaginal microbiota, and vaginal intraepithelial neoplasia (VAIN), revealing that high viral load of HPV 16, 52, and 58 correlate with a heightened risk of vaginal epithelial lesions. In addition, anaerobic bacteria including Atopobium, Gardnerella, Allobaculum, Enterococcus and Clostridium, were shown to be positively associated with epithelial neoplasia. 38 Consistently, studies across diverse regions and timeframes reported a marked decrease in Lactobacillus among HPV-infected female patients, which coincides with a substantial rise in vaginal pH, thereby diminishing resistance to pathogenic microorganisms.39,40 Concurrently, patients with HPV infection or cervical intraepithelial lesions exhibit significantly greater vaginal microbial diversity compared with healthy individuals (Figure 3). Notably, anaerobic bacteria including Sneathia sanguinegens, Anaerococcus tetradius, and Peptostreptococcus anaerobius are observed in increased abundance.41-43 I Intriguingly, research indicates that distinct Lactobacillus species are associated with varying HPV clearance rates and survival outcomes, with L. crispatus demonstrating the most favorable profile and L. iners the least protective effect. This variation may be attributed to the biological attributes of L. crispatus, which includes a high capacity for H₂O₂ production, thereby enhancing pathogen elimination. 42 Collectively, these insights suggest that microbiota composition of the microbiota evolves throughout cervical neoplasm progression, marked by a pronounced reduction in Lactobacillus abundance and an expansion in microbial diversity.
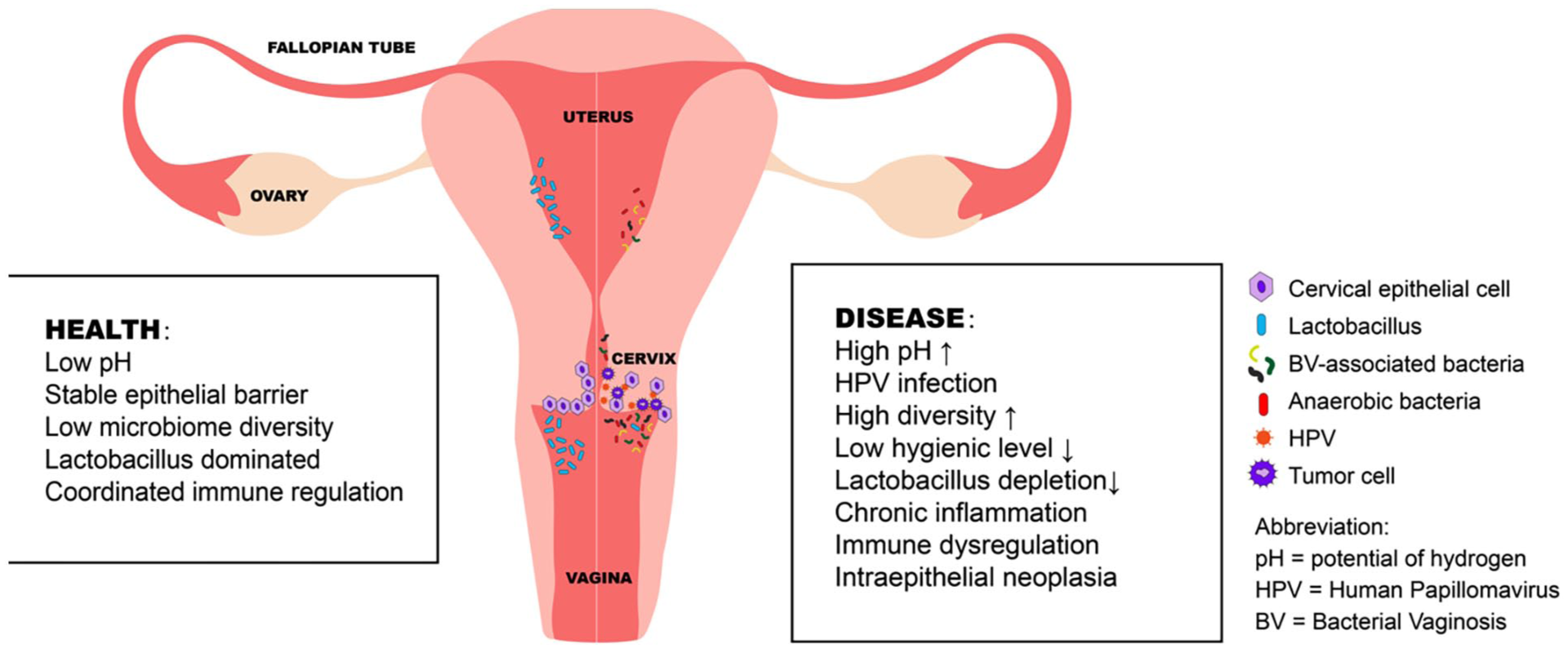
Vaginal microbiota composition in health and disease: Under healthy conditions, the vaginal microbiota is predominantly composed of Lactobacillus species, which play a crucial role in maintaining vaginal homeostasis and providing effective protection against pathogenic microorganisms.
Microbial Dysbiosis and Immunological Environment of Cervical Cancer
Lactobacillus, as a representative probiotic, has an antagonistic relationship with BV-associated bacteria. In healthy individuals, a vaginal microenvironment dominated by Lactobacillus suppresses the infection and colonization of pathogens. However, when probiotics no longer hold a dominant position, BV-associated bacteria proliferate extensively, leading to microbial dysbiosis. The occurrence of microbial dysbiosis is accompanied by fluctuations in various cytokines, activation of inflammatory pathways, cervical epithelial damage, persistent HPV infection and cervical epithelial neoplasia (Figure 4).

The antagonistic interaction between probiotics and pathogenic bacteria: Probiotics exert a protective effect on cervical health by maintaining microbial balance, producing lactic acid and hydrogen peroxide, reducing HPV colonization, lowering levels of pro-inflammatory cytokines, and preserving the integrity of the epithelial barrier.
Microbial dysbiosis and HPV infection
A Lactobacillus-dominated microenvironment exerts immunomodulatory effects against HPV. The acidic environment effectively inhibits the colonization of pathogenic microorganisms or viruses.
H2O2-producing lactobacilli stimulate epithelial cell secretion of antimicrobial substances while increasing the antibacterial activity of protective factors. 44 For instance, L. crispatus was shown to induce the expression of the anti-inflammatory cytokine IL-10 while inhibiting the expression of other pro-inflammatory cytokines such as IL-6, IL-8, and TNF-α. This immunomodulatory effect helps to maintain a healthy and functional vaginal tract and prevent excessive inflammation associated with HPV infection. 45 Lactobacilli secrete various metabolites and surfactants, such as exopolysaccharides, phosphorylated polysaccharides, and peptidoglycans, which inhibit harmful microorganisms and cervical carcinogenesis. 46 Lactobacillus supernatants have also been shown to reduce the expression of crucial HPV oncogenes, E6 and E7, which are essential for the transformation of infected cells into cancerous cells. 47 Associations between microbial dysbiosis with immune activation have been shown by both in vivo and in vitro studies. 3 ,48-50 According to the analysis of cervical fluid obtained from African women, BV-associated bacteria were highly correlated with pro-inflammatory cytokines, including IL-1α, IL-1β, TNF-α, IFN-γ, IL-10, and IL-8 which are closely associated with genital tract inflammation. 51 BV-associated bacteria, such as Prevotella amnii and Mobiluncus mulieris, induced the secretion of pro-inflammatory cytokines by vaginal epithelial cells, 51 which may cause tissue injury and epithelial remodeling. Transcriptional analysis has revealed that antigen-presenting cells (APCs) were activated by microbial products, mediated through the activation of the NF-kB signaling pathway via Toll-like receptor 4 (TLR4) signaling, which in turn promotes the secretion of pro-inflammatory cytokines and the recruitment of lymphocytes. 51 However, chronic inflammation caused by microbial dysbiosis persistence may gradually deteriorate the functionality of immune cells over time. Specifically, the activity of effector T cells and natural killer cells may be diminished, impairing their ability to effectively clear pathogens. 52 Certainly, the vaginal epithelial tissue could be vulnerable to HPV infection since HPV requires access to basal cells for cell entry usually through inflammation or injury. Although drivers of transition states between HPV acquisition, clearance, persistence and progression to precancer are poorly understood, several cellular phenomena have been observed. In a longitudinal study of women who acquired HPV 16 and cleared, elevated cytokine levels were observed in the immediate post-clearance visit, specifically for IFN-γ, IL-4, IL-5, IL-10, IFNα-2, MIP-1α and tumor necrosis factor 53 which returned to baseline months later. HPV infection further alters the immune microenvironment favoring the persistence of microbial dysbiosis and the onset of cancer.3,54 For instance, high-risk HPV infection can lead to a shift in the cervical microecology from L. crispatus dominance to a prevalence of Atopobium vaginae. 55 As the condition progresses to squamous intraepithelial lesions (SIL), there is an increase in microbial diversity, with species such as Sneathia and Fusobacterium becoming dominant.3,56
Microbial dysbiosis and cervical intraepithelial neoplasia
Although the definitive and propelling role of vaginal dysbiosis in cervical cancer is not as widely recognized as that of HPV, relevant studies have suggested its significance in facilitating cervical epithelial lesion. Among cervical cancer patients, changes in the vaginal microbial composition were observed, with a dramatic decrease in the Lactobacillus genus and significant increases in pathogenic bacteria, including the Anaerococcus, Peptostreptococcus, Porphyromonas, Prevotella, Gardnerella vaginalis, and Sneathia genera. 57 These pathogens produce a variety of enzymes and toxins that can disrupt the integrity of cervical epithelial cells. For instance, Gardnerella vaginalis produces a cholesterol-dependent cytotoxin known as vaginolysin, leading to cell lysis of cervical epithelia. 58 In addition, pathogenic bacteria, such as Gardnerella vaginalis and Prevotella species are known to alter both local and systemic immune regulation. 46 Notably, Gardnerella vaginalis correlates with higher levels of IL-17 and lower levels of IL-10 and IFN-γ in peripheral blood when compared with vaginal microbiota dominated by Lactobacillus. The inverse relationship between IL-17 and IFN-γ implies that bacteria linked to vaginal dysbiosis may undermine the Th1 immune response. By fostering the differentiation of Th17 cells and elevating IL-17 levels within the immune milieu, microbial dysbiosis consequently leads to a less effective antiviral immune response. 59 The presence of Gardnerella is positively associated with the expression of Th2 cytokines and inversely associated with Th1 cytokine levels in cervicovaginal secretions. 60 Furthermore, elevated levels of TLR 7, TLR9, IFNL2, and IFNLR1 have been observed in cervical cells of women infected with HPV who also test positive for bacterial vaginosis. 61 The activation of these TLR triggers the production of interferons and inflammatory cytokines, which are crucial for immune responses. 46 Persistent production of inflammatory factors leads to chronic inflammation, which in turn facilitates the development of cervical dysplasia. The local cervical microenvironment of cervical cancer patients is characterized by an increase in the number of immature dendritic cells (iDCs), regulatory T cells (Tregs), PD-L1–positive cells, and macrophages, while the number of Langerhans cells (LCs), CD4⁺ T cells, and CD8⁺ T cells is reduced.3,46,62,63 Recent studies have proposed that vaginal microbial dysbiosis may influence the vaginal epithelium through the modulation of estrogen metabolism. Dysbiosis in the vaginal microbiome may disrupt the normal metabolic pathways of estrogen, leading to an abnormal elevation. 64 Elevated estrogen levels are known to promote the development of estrogen-dependent cancers, such as ovarian and endometrial cancers. Li’ group reported the association between estrogen-related receptor β (ESRRB) and cell proliferation in cervical cancer. ESRRB activated transcription of SMAD7, a TGF-β pathway inhibitor, which blocked phosphorylation and nuclear translocation of SMAD2/3 to the nucleus, thereby downregulating CDKN1A and upregulating CCNA2 and MYC. In turn, MYC transactivated ESRRB and upregulated SMAD7, thus forming a positive feedback loop with ESRRB. 65 However, the regulatory role of estrogen in cervical cancer remains to be further validated.
Probiotics and Their Regulatory Effects on Immune Cells in Tumor Microenvironment
Lactobacilli maintain vaginal health through the secretion of various compounds, including lactic acid, bacteriocins, polysaccharides, peptidoglycan and hydrogen peroxide. These secretions inhibit pathogenic microbial colonization and modulate both innate and adaptive immune responses, 3 thereby significantly contributing to the prevention of cervical carcinogenesis.47,66 The immunomodulatory action of these bacteria is manifested through the enhancement of the epithelial barrier, the polarization of immune cells, and the reprogramming of immune responses (Figure 5).
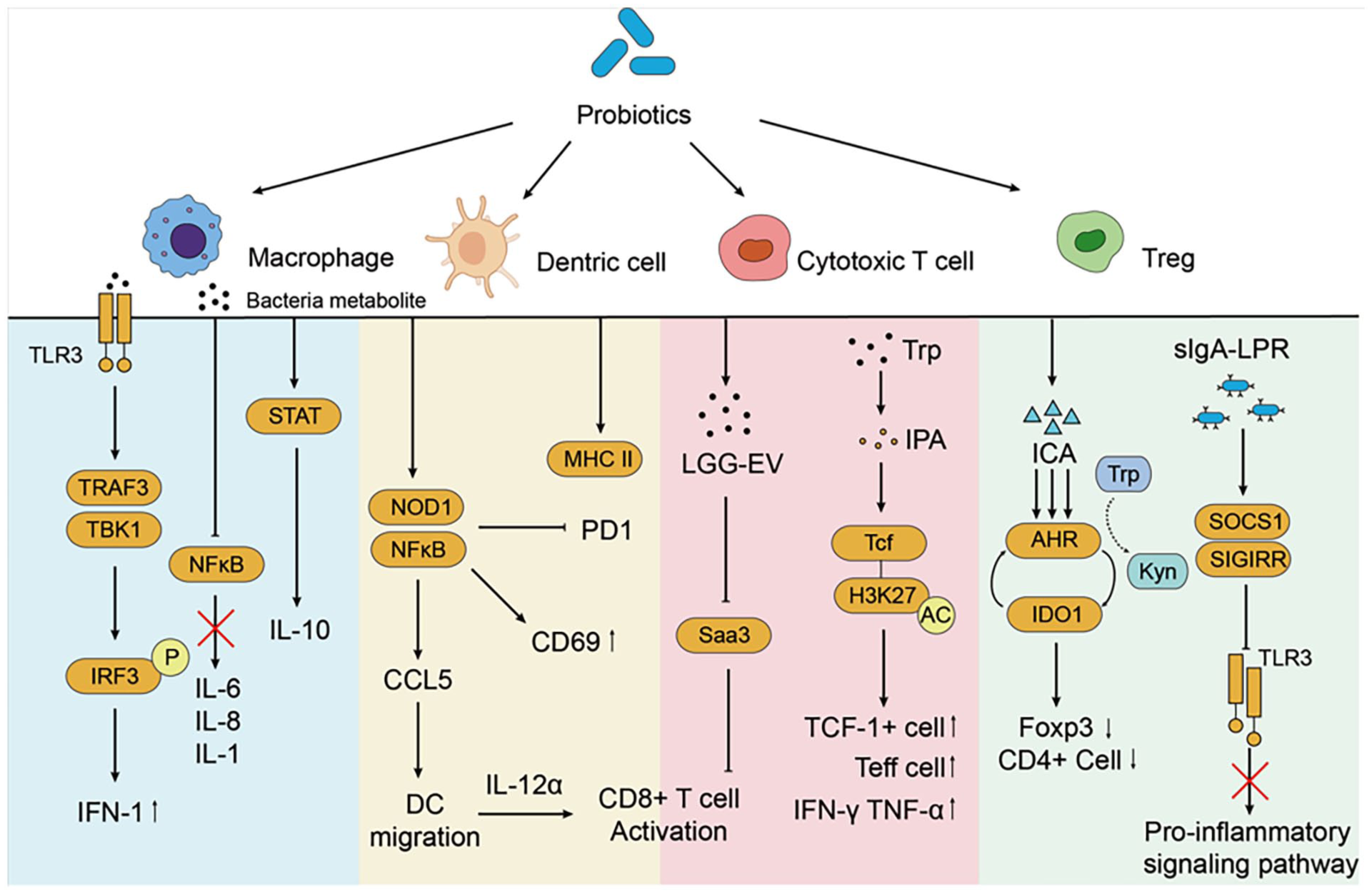
Probiotics’ regulatory impact on immune cells: Probiotics exert a modulatory effect on various immune cell types through intricate regulatory mechanisms that enhance both innate and adaptive immune responses.
Lactobacillus and innate immune modulation
The positive impact of Lactobacillus on mucosal epithelia is extensively documented. Research by Benyacoub’s group has previously highlighted its role in regulating epithelial barrier function. It has been established that Lactobacillus enhances the integrity of the human epithelial barrier by diminishing skin reactivity and modulating the host immune system. 67 Lactobacillus gasseri, a prevalent species within the vaginal lactobacilli community, has been the subject of recent investigations. A study focusing on LGV03, derived from the vaginal flora of individuals with cervical cancer, demonstrated its potential to facilitate HPV clearance and bolster host epithelial immunity in HPV-positive human cervical cancer cells. 68 LGV03 was found to increase TLR3 expression in epithelial cell, thereby promoting IRF3 phosphorylation and activating downstream signaling molecules TRAF3 and TBK1, which are instrumental in enhancing HPV clearance. This strain has also been noted to upregulate IRF3 expression in the cervical cancer cell line Ect1/E6E7, triggering the secretion of type I interferon and strengthening the antiviral immune response.
Lactobacillus and macrophage modulation
Macrophages are crucial in innate immune response, 69 primarily phagocytosing pathogens and cellular debris, while acting as antigen-presenting cells that bridge innate and adaptive immunity. Certain strains of Lactobacillus were shown to modulate macrophage differentiation and reduce inflammatory responses. 70 A study indicated that L. johnsonii, commonly found in the vagina, promoted the differentiation of naïve macrophages into CD206⁺ macrophages via the STAT pathway, thereby reducing inflammatory injury in a colitis mouse model. This effect is partly attributed to increased IL-10 production, which alleviates inflammatory damage. Macrophages typically differentiate into 2 subtypes: M1 and M2. M1 macrophages are primarily involved in pro-inflammatory responses, supporting Th1 immune responses and strengthening the protection against infections and tumors. Conversely, M2 macrophages display anti-inflammatory properties, often associated with tumor progression in various human solid tumors through the secretion of cytokines IL-10 and TGF-β. With the secretion of anti-inflammatory cytokines such as IL-10 and TGF-β, suppressed inflammatory reactions are generally associated with tumor progression in various human solid tumors. Notably, different strains of Lactobacillus may drive macrophage differentiation toward either the M1 or M2 phenotype, underscoring the genus’s functional diversity.70-72
Lactobacillus and adaptive immune modulation
The adaptive immune system represents a sophisticated defense mechanism capable of identifying and responding to specific pathogens. Through the collaborative efforts of various immune cells, it provides long-lasting protection and immunological memory.
Lactobacillus and dendritic cells
Antigen presentation, a cornerstone of adaptive immunity, is crucial for the human immune system to identify and eliminate tumor cells. The potential of Lactobacillus-derived metabolites to activate dendritic cells (DCs) has been previously reported. 73 Foodborne supplementation with Lactobacillus and its metabolites a been found to enhance DC activity and strengthen local immune responses.74-77
Lactobacillus intestinalis, a bacterium that resides in both the gut and the vagina, significantly increased immune cell infiltration within an AOM/DSS-induced mice CRC model. It was reported the activation of NOD1/NF-kB signaling pathway in tumor cells led to the secretion of chemokine CCL5, facilitating the migration of dendritic cells while activating CD8⁺ T cells, characterized by an elevated CD69 and reduced PD-1 expression. 78 CD69 serves as an early activation marker for T cells, and its upregulation signifies increased T cell activity and immune responsiveness. 79 In contrast, PD-1 functions as an inhibitory receptor on T cells. The downregulation of PD-1 suggests a decrease in T cell exhaustion and an enhanced ability to maintain effector functions. These changes collectively indicate that T cells are in a more active and functional state, which is beneficial for robust anti-tumor immunity. Another study explored the potential of foodborne probiotics to enhance the efficacy of PD-1 immunotherapy. Lactobacillus rhamnosus is one of the normal components of the vaginal microbiota in healthy women. Researchers successfully isolated extracellular vesicles derived from Lactobacillus rhamnosus (LGG-EV). After the application of LGG-EV in a mouse CRC model, an increased ratio of CD8⁺ T cells to CD4⁺ T cells were observed in the mesenteric lymph nodes, indicating enhanced cytotoxic capacity. Concurrently, elevated levels of MHC II⁺ dendritic cells, CD4⁺ T cells, and CD8⁺ T cells were observed, indicating a potential shift toward a more robust defense mechanism against pathogens and tumor cells. 80
Lactobacillus and cytotoxic CD8+ T cells
Recent research into Lactobacillus metabolites has provided pivotal insights, demonstrating that certain strains of Lactobacillus may modulate immune cell differentiation and function via their metabolites. For instance, indole-3-lactic acid (ILA) is produced by Lactobacillus plantarum, which can be found in both the gut and the vagina. ILA enhanced the IL-12 secretion by DCs 81 in patients with rheumatoid arthritis, which led to the activation of CD8⁺ cytotoxic T cells. In addition, ILA was shown to remodel the metabolic state of tumor-infiltrating CD8⁺ T cells by inhibiting Saa3 expression, thereby enhancing their survival and cytotoxic capacity in vitro and in a mouse CRC model. 82 While Lactobacillus has demonstrated the ability to enhance anti-tumor immunity by modulating CD8⁺ T cell function, it has also been observed to mitigate CD8⁺ T cell-mediated inflammatory responses in non-oncological diseases. This suggests that Lactobacillus plays a dual role in the regulation of CD8⁺ T cells, implying its immunomodulatory effects are more intricate than previously acknowledged. 81 In 2024, a study published in Cell offered a novel perspective. Lactobacillus johnsonii is a part of gastrointestinal and vaginal mucosal microbiota. It was revealed that oral supplementation with L. johnsonii or tryptophan-derived metabolite indole-3-propionic acid (IPA) enhanced the stemness of CD8⁺ T cells in human pan-cancer through epigenetic regulation mechanisms. 83 Specifically, IPA significantly enhanced the sustained anti-tumor capacity of T cells by elevating H3K27 acetylation levels within the Tcf7 gene’s super-enhancer region. This enhancement improves chromatin accessibility and fosters the differentiation and proliferation of precursor-depleted T cells (Tpex). Tpex cells are a subset of T cells that lack expression of essential transcription factors for maturation into effector or memory T cells, remaining in a state of functional quiescence and metabolic dormancy. Single-cell multi-omics analysis has confirmed that IPA treatment significantly boosts the presence of TCF-1⁺ positive Tpex cells and aids their maturation into effector T cells (Teff). Validated across various human cancer models, 83 this study pioneeringly reveals the molecular mechanism through which microbial metabolites modulate T cell stemness via epigenetic reprogramming. This discovery lays a theoretical groundwork and presents a novel avenue for personalized immunotherapy leveraging bacterial community interventions.
Lactobacillus and regulatory T cells
Tregs are a subset of T cells marked by CD4 and CD25 expression and high levels of the transcription factor Foxp3, primarily function to maintain immune homeostasis by suppressing excessive immune responses and regulating immune tolerance. However, Tregs are also commonly implicated in tumor immune evasion. The Fong team reported indole-3-carboxylic acid (ICA) derived from Lactobacillus lactis competitively bound to the aryl hydrocarbon receptor (AHR). By modulating the IDO1-Kyn-AHR signaling axis, ICA effectively blocks Kyn-mediated activation of AHR, thereby inhibiting the differentiation and proliferation of Tregs. 84 The study emphasized that Lactobacillus species reshaped the tumor immune microenvironment in syngeneic mouse models through host-bacteriophage metabolic interactions. In addition, bacteria-derived structural substances have exhibited capacity in immunomodulation. Lactobacillus rhamnosus is one of the strains isolated from both the vagina and the gut. Mikulic’s team reported that secreted IgA (sIgA) complexes with Lactobacillus rhamnosus potentiates (LPR) enhanced dendritic cell immunomodulatory function in a tissue-specific manner. 85 The SIgA-LPR complex inhibited the TLR-mediated pro-inflammatory signaling pathway in vitro by upregulating the expression of negative regulatory cytokines in dendritic cells recovered from mouse Peyer’s patches, mesenteric lymph nodes, and spleen. Meanwhile, an elevated expression of retinaldehyde dehydrogenase 2 (RALDH2) was observed, which promoted the production of retinoic acid, favoring the differentiation of regulatory T cells.
Immunomodulatory Functions of Anaerobic Tumor-Associated Bacteria
Tumor-associated bacteria are characterized by a group of anaerobic bacteria residing in the hypoxic region with rising abundance in tumor patients. 86 Multiple studies have documented the occurrence of vaginal dysbiosis in patients with HPV infection or cervical epithelial lesions, marked by a notable rise in the prevalence of anaerobic bacteria, facultative anaerobes, and BV-associated bacteria.3,38,40 While Lactobacillus species predominantly generate lactate, anaerobic bacteria are characterized by their production of short-chain fatty acids (SCFAs), including acetic acid, propionic acid, and butyric acid. 87 Rising evidence has indicated the immunomodulatory role of bacteria-derived short-chain fatty acids in both pathological and physiological states.87-90 The immune regulatory mechanisms discussed in the subsequent text are summarized as shown (Figure 6).

Regulatory impact of anaerobes and BV-associated bacteria on immune cells: Anaerobes and bacteria associated with BV typically stimulate the proliferation of immune cells that exert negative regulatory effects.
Bacteria-derived SCFAs in immune modulation
Bacteria-derived SCFAs and macrophage polarization
Previous research focused on the impact of bacteria-derived SCFAs on macrophages has revealed significant insights. Hezaveh’s team reported that low-concentration butyrate promoted tumor metastasis by upregulating the H19-MMP15 pathway, which in turn promoted the differentiation of M2-type macrophages in a mouse model of pancreatic adenocarcinoma. 91 M2-type macrophages are known to be tumor-promoting macrophages 92 that inhibit the activity of CD8⁺ T cells and NK cells by secreting immunosuppressive factors such as IL-10 and TGF-β. Propionic acid, derived from bacteria, similarly exhibits immunomodulatory effects on macrophages, fostering an immunosuppressive environment. Treatment with propionic acid significantly reduced CD68+ macrophages infiltration and inhibited the expression of pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α in a mouse model of colitis. 93 Propionic acid curbs macrophage-mediated immune responses by inhibiting the activation of the STAT3 signaling pathway. 93 Furthermore, it has been demonstrated to stimulate tumor cell proliferation by suppressing macrophage function in a transgenic TRAMP mouse model. Liu’s team found that propionic acid enhances the migration and invasiveness of prostate cancer cells via TLR3-dependent autophagy, which subsequently activates the NF-kB and MAPK signaling pathways. Autophagy in cancer cells releases the chemokine CCL20, thereby remodeling the tumor microenvironment by attracting macrophages and inducing their polarization toward an immunosuppressive M2 phenotype. 94
Bacteria-derived SCFAs and DC maturation
Previous studies have indicated that treatment with propionic acid suppresses the activity of HDAC1 and HDAC3, leading to a significant decrease in the expression of critical transcription factors PU.1 and RelB, which are essential for dendritic cell maturation. This inhibition hinders the differentiation of myeloid precursor cells into dendritic cells. 95 Subsequent research has further demonstrated that propionic acid impedes the maturation and function of dendritic cells by downregulating the expression of co-stimulatory molecules, including CD83, CD80, and CD40, as well as reducing the secretion of cytokines such as IL-12 and IL-23 in vitro. 96 The cytokines produced by these dendritic cells are essential for the activation and maturation of CD8⁺ cytotoxic T cells. Consequently, propionic acid inhibits the activation of CD8⁺ T cells by suppressing the function of dendritic cells.
Bacteria-derived SCFAs and cytotoxic T cell regulation
Prior studies have investigated the regulatory influence of SCFAs on CD4⁺/CD8⁺ T cells. Zhu’s team reported a positive correlation between bacteria-derived butyrate and the expression levels of CD8⁺ and Vδ2⁺ T cells in human peripheral blood mononuclear cells (PBMCs). Individuals exhibiting a favorable response to anti-PD-1 therapy had significantly higher serum butyrate levels compared with non-responders. 97 Yu’s team further demonstrated that butyrate increases CD28 expression and H3K27 acetylation in the promoter regions of the Pdcd1 gene, which encodes PD-1, resulting in upregulated PD-1 and CD28 expression. 98 In addition, butyrate was shown to facilitate the cytotoxic function by strengthening T cell receptor (TCR) signaling while promoting the production of anti-tumor cytokines such as IFN-γ and TNF-α in human gastric cancer microenvironment. 98 These findings indicate that butyrate can enhance the efficacy of anti-PD-1 therapy through epigenetic regulation of PD-1/CD28 expression and modulation of the TCR signaling pathway, offering a novel biomarker and combination strategy for optimizing cancer immunotherapy.
Bacteria-derived SCFAs and Treg regulation
Tregs are typically recognized for their negative regulatory role within the immune milieu. Research focused on human autoimmune diseases has noted an upregulation of Foxp3 expression and TGF-β1 production subsequent to the application of propionic acid. 99 Although the molecular mechanisms require further confirmation. Butyrate has been reported to modulate both effector and regulatory T cell populations. Initial investigations have highlighted butyrate’s role in fostering the differentiation and expression of Tregs in serum by promoting the acetylation of H3K27 in the Foxp3 gene’s promoter region.100,101 This epigenetic alteration enhances the transcriptional activity of regulatory T cells, thereby bolstering Treg stability.100,101 Wen’s group has further revealed that bacteria-derived butyrate altered the cellular metabolic state by activating the cytosolic receptor PPARγ. The interaction suppresses the expression of the hypoxia-inducible factor HIF-1α, curtailing cellular glycolytic metabolism while enhancing mitochondrial oxidative phosphorylation. Such metabolic shifts favor the differentiation of CD4⁺ T cells into Tregs and mitigate Th17 cell polarization, as observed in a dextran sodium sulfate (DSS)-induced colitis model. 102 The activation of PPARγ further consolidates the phenotype and function of Tregs by modulating the mTOR pathway, governing metabolism-related gene expression. 102 The conclusion has also been validated in a recent study by He’s group. 103 These findings unveil that bacteria-derived butyrate is effective in promoting the differentiation and proliferation of Tregs, which suppresses immune function and reduces inflammatory responses. While offering a protective effect against inflammatory injuries, this may also potentially abet immune evasion in tumor development. 104
Tumor-associated bacteria and immunosuppressive microenvironment
Bacterial structural proteins have been reported to reshape the tumor microenvironment. Peptostreptococcus anaerobius, one of the tumor-associated bacteria found abundant in cervical cancer patients, was reported to combine to α2/β1 integrins overexpressed by tumor cells via surface protein PCWBR2. 105 The interaction activated the PI3 K-Akt pathway, which subsequently induced NF-kB signaling activation, leading to increased production of IL-10 and IFN-γ in a mouse colorectal cancer model. 105 Concurrently, P. anaerobius has been shown to significantly increase the population of immunosuppressive cells via the NF-kB signaling pathway, encompassing myeloid-derived suppressor cells (MDSCs), tumor-associated macrophages (TAMs), and granulocytic tumor-associated neutrophils (TANs) in vitro. Infection with P. anaerobius leads to persistent inflammatory damage that promotes tumor progression. 105 A key finding of this study is the demonstration of P. anaerobius’ role in the direct activation of pro-oncogenic signaling pathways and in reshaping the immunosuppressive tumor microenvironment. Nevertheless, further research is needed to assess the generalizability of these results and their potential clinical implications for translational applications.
Recent clinical studies have further analyzed the close relationships between various vaginal microorganisms and immunomodulatory cytokines under both healthy and pathological conditions. In healthy populations, decreases in Lactobacillus, particularly L. crispatus, were significantly associated with increased levels of pro-inflammatory cytokines. Conversely, the abundance of dysbiosis-associated pathogens, including Gardnerella, Prevotella, and Arthrobacter, increased in parallel with elevated inflammatory factors. 106 In patients with high-grade cervical intraepithelial neoplasia, Lactobacillus species were significantly reduced, with widely expressed L. crispatus in vagina became nearly undetectable. Concurrently, the vaginal microbial composition of patients displayed elevated diversity, marked by the characteristic CST IV profile. An increased activation of the PI3K-Akt-NF-κB signaling pathway was detected, which in turn stimulated the secretion of pro-inflammatory cytokines including IL-1β and IL-6. The activation resulted in the establishment of a chronic inflammatory microenvironment that subsequently fostered angiogenesis and immunosuppression.106,107
The Prognostic and Therapeutic Potential of Microbiome in Cervical Cancer
Indications for cervical cancer prognosis
An increasing number of studies have demonstrated that the microbiota of CIN patients influences the efficacy and prognosis of postoperative radiotherapy and chemotherapy.108-111 A 4-year study conducted in the United States has indicated that microbial diversity could be an independent prognostic factor for patients with cervical cancer who undergo chemoradiotherapy post-surgery. 109 Elevated gut microbiota diversity was correlated with enhanced survival outcomes for among cervical cancer patients. Patients exhibiting high microbial diversity experienced significantly improved recurrence-free survival (RFS) and overall survival (OS) compared with those with low diversity. Conversely, fecal samples with elevated levels of Porphyromonas, Porphyromonadaceae, and Dialister were linked to an unfavorable outcome. The study further demonstrated that high microbiota diversity was positively correlated with increased infiltration of CD4⁺ T cells into tumors, particularly characterized by an elevated expression of activated CD4⁺ T cell subsets, as indicated by Ki67⁺ and CD69⁺ markers. The presence of Ki67⁺ and CD69⁺ tumor-infiltrating T cells signifies both proliferative potential and immune activation, aligning with robust anti-tumor responses. While Lactobacilli are generally considered beneficial, recent findings reveal that some strains of Lactobacilli isolated from cervical cancer microenvironments may adversely affect patient prognosis.110,112 Interestingly, the tumor microenvironment reconfigured the vaginal microbiota at the genetic level. The cancer-associated L. iners are genetically distinct and associated with chemoradiation resistance in cervical cancer. The study further uncovered that strains of lactate-producing bacteria were linked to a less favorable prognosis in various cancer types, including non-small cell lung cancer, colorectal cancer, and squamous cell carcinoma of the head and neck. This finding suggests that the metabolic influence exerted by lactic acid bacteria within the tumor microenvironment may represent a widespread mechanism that impacts cancer progression. Notably, a non-Lactobacillus dominated gut microenvironment was shown to affect responsiveness of cervical cancer patients to pembrolizumab in another study, 111 while the cellular mechanisms require further validation.
Restoration of local and systematic microenvironment
To date, a definitive treatment for HPV has not been established, however, probiotic-based therapies show considerable promise as a potentially effective strategy. According to the clinical trial involving 117 women affected by bacterial vaginosis or vaginitis with concomitant HPV-infections, antibiotic treatment plus vaginal Lactobacillus implementation exhibited significantly higher chance to solve HPV-related cytological anomalies. 113 Recent studies have identified gut-vaginal microbiome as a novel therapeutic strategy for HPV eradication, offering potential prevention of cervical cancer at its inception.114,115 Among the various Lactobacillus species, L. gasseri and L. jensenii have demonstrated notably strong antiviral effects against HPV infections. 59 Nicolò’s team demonstrated that Lactobacillus significantly boosted the production of IFN-γ from peripheral mononuclear cells through the secretion of lactic acid and SCFAs. The elevated level of IFN-γ is thought to be prone to the enhancement of Th1 immune response, which effectively supports the clearance of HPV infections. Whereas bacteria linked to vaginal dysbiosis primarily enhance the production of IL-17, thereby disrupting the balance between TH17 and TH1 cells. This disruption can lead to a predisposition for infectious and inflammatory diseases. 59 Other studies have suggested the supplement of probiotics enhance the function of innate immune cells, thereby boosting anti-tumor immune responses. For instance, vaginal usage of probiotics was shown to increase the production of anti-inflammatory cytokines in patents with cervical epithelial neoplasia, which mitigated cervical epithelial carcinogenesis by alleviating chronic inflammation caused by microbial dysbiosis. 116 Furthermore, probiotics were shown to enhance the immune system’s capacity to identify cancer cells by regulating the expression of immune checkpoint proteins. Specifically, levels of PD-L1 and LAG-3 are inversely correlated with the dominance of health-associated Lactobacillus, while TLR2 levels show a positive correlation.117,118 Considering the prevalent chemotherapeutic approaches that incorporate a combination of targeted therapies, the integration of immunotherapy with probiotics presents a promising avenue for enhancing treatment efficacy. The implications of microbial dysbiosis for cervical cancer prognosis and therapy are outlined as follows (Figure 7).

Impact of microbial dysbiosis on cervical cancer therapy: Various strains of vaginal and intestinal microbiota are color-coded to indicate their beneficial or detrimental effects. Overall, a healthy microbial environment enhances the regulatory function of T cells, improving individual immune function and leading to increased immune cell infiltration and activation within tumors.
Probiotics-based therapeutic HPV vaccines
Engineered probiotic-based vaccines are emerging as an innovative immunotherapeutic approach against HPV. 119 Traditional HPV L1 capsid protein vaccines have demonstrated significant efficacy in preventing initial HPV infection, whereas they are less effective in addressing established HPV infection. Lactobacillus species are considered superior candidates for vaccine vectors, attributed to their well-documented safety, lack of pathogenicity, and the feasibility of genetic engineering to express HPV-specific antigens. 120 These proteins can be presented on the bacterial surface for antigen delivery, utilizing a variety of signaling peptides and cell wall anchoring systems, such as PgsA and CWAM6. Therapeutic HPV vaccines enhance the immune system’s ability to eliminate existing HPV infections. By eliciting HPV-specific cytotoxic T cell responses and fostering the generation of targeted antibodies, the local epithelium constructs a highly specific defense against HPV.44,114 Despite these advancements, challenges hinder the clinical application of therapeutic HPV vaccines. These include transient immunogenicity, antigen degradation in vivo, and the determination of optimal dosing regimens. 114 The important studies reviewed in this overview, which are relevant to treatment and prognosis, are summarized in the table below (Table 1).
Impact of microbiota on immune response and therapy outcomes.

Conclusion
Increasing progress have been made in depicting the landscape of vaginal microbiota and its multifaceted role in cervical cancer. Although specific microbial strains or molecular pathways implicated in tumor progression have been identified, a holistic understanding of the intricate regulatory network remains elusive. The vaginal microbiota in cervical cancer patients is highly heterogeneous, underscoring the necessity for personalized treatment paradigms. These require standardized and rigorous evaluation through multicenter randomized controlled trials, complemented by dynamic monitoring to delineate long-term benefits and potential adverse effects. Studies on the gut microbiome provides valuable insights into the potential roles of the vaginal microbiome in cervical cancer, there is an urgent need for more direct studies focusing on cervical cancer. Future research should be directed toward unraveling the precise mechanisms by which vaginal bacteria modulate immune responses and influence tumor progression in cervical cancer, which could enhance our comprehension of the disease and provide theoretical evidence for the development of targeted and effective therapeutic strategies.
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
Not applicable.
Author Contributions
JHC, MLZ, and YXG participated in the writing of the article. JHC, ZG, and HZ, participated in revision. JHC originally created all images and tables involved in this review. YYG and FS participated in the writing and revision of the article. GNZ participated in designing and the writing of the article. JXD participated in the revision of the article. All authors read and approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no data sets were generated or analyzed during the current study.
