Abstract
Background:
Neoadjuvant chemoradiotherapy (nCRT) followed by total mesorectal excision (TME) is the current standard for locally advanced rectal cancer. This study aimed to evaluate the need for adjuvant chemotherapy and clinical outcomes in patients with ypT3-4N0M0 rectal cancer.
Methods:
This is a retrospective cohort study. We reviewed the patients with locally advanced rectal cancer who had undergone radical resection after nCRT between 2010 and 2016. A total of 69 patients with ypT3-4N0M0 rectal cancer were included. The prognostic factors affecting disease-free and overall survival were analyzed.
Results:
With clinical stage II–III and pathological stage II disease, the overall survival in the adjuvant chemotherapy group (n = 38) was better than that of the group without adjuvant chemotherapy (n = 31) (86.8%, vs 74.2%, P = 0.016). The disease-free survival was better in 2 groups, but the difference was not statistically significant (73.7%, vs 67.4%, P = 0.193).
Conclusions:
In patients with clinical stage III rectal cancer downstaged to ypStage II after nCRT and TME, adjuvant chemotherapy may improve overall survival. nCRT combined with postoperative adjuvant chemotherapy remains an effective strategy for patients unsuitable for complete total neoadjuvant therapy protocols.
Introduction
Neoadjuvant chemoradiotherpy (nCRT) following total mesorectal excision (TME)1-3 is currently the standard treatment for locally advanced rectal cancer. Neoadjuvant chemoradiotherpy can downstage tumors prior to surgery, but the benefits of adjuvant chemotherapy (AC) for patients who had undergone nCRT are still unclear. According to the National Comprehensive Cancer Network (NCCN) guidelines, all patients with locally advanced rectal cancer should receive AC, regardless of their final tumor pathological stage. 1 The standard treatment for locally advanced rectal cancer involves nCRT followed by TME, whereas all patients should receive AC according to the NCCN guidelines. The 2017 European Society of Medical Oncology (ESMO) Clinical Practice Guidelines for Rectal Cancer suggest that adjuvant chemotherapy is indicated only for stage III patients or stage II patients with high-risk factors. 4 Many studies have supported that AC may not be necessary for patients with ypT0-2N0 tumors.5,6 However, in patients with ypT3-4N0 tumors receiving AC, study results are variable. Some have shown a trend of improved outcomes without significant difference, while others have demonstrated an overall survival benefit.7-10
Although total neoadjuvant therapy (TNT) has emerged as a promising strategy offering improved disease control and opportunities for organ preservation, nCRT followed by TME remains a cornerstone in the management of locally advanced rectal cancer. This is particularly crucial for patients who show inadequate response to TNT, experience intolerable toxicity resulting in premature treatment discontinuation, or develop urgent complications such as bleeding or obstruction necessitating timely surgical intervention. In these circumstances, nCRT plus TME continues to provide a reliable and effective treatment pathway.11,12
Because different guidelines offer different recommendations, and there is no definitive clinical trial to provide clear treatment efficacy, there is no established chemotherapy protocol for clinical stage III patients who have been downstaged to stage II after completing nCRT. The aim of this study was to evaluate the need for AC and clinical outcomes in patients with ypT3-4N0M0 rectal cancer.
Subjects and methods
Patients
This is a retrospective cohort study. We retrospectively reviewed the clinicopathological data of patients with locally advanced rectal cancer (cT3-4 or N positive, with tumor location < 10 cm from the anal verge) who had radical resection after nCRT between January 2010 and December 2016. All patients received nCRT, followed by TME in Taipei Veterans General Hospital. The exclusion criteria were those with synchronous colon cancer, squamous cell carcinoma, or gastrointestinal stromal tumor (GIST), those receiving neoadjuvant short-course radiation therapy (RT), or those under the watch-and-wait strategy after nCRT. Our primary end-point is the prognostic factor affecting disease-free and overall survival. Secondary outcomes were the prognostic factors of survival and recurrence pattern. The reporting of this study conforms to the STROBE guidelines for observational studies, 13 and the completed checklist is available as Supplementary File 1.
Methods
All patients who underwent clinical staging for colorectal cancer were evaluated by computerized tomography (CT) or pelvic magnetic resonance imaging (MRI), as well as abdominal sonography. To exclude distant metastases, chest CT was also performed. In cases suspected of having metastatic lesions, biopsy was conducted to confirm the stage. After neoadjuvant treatment, abdominal CT or pelvic MRI was performed to evaluate the tumor shrinkage status. Approximately 65.2% of the patients received at least one MRI evaluation. All pelvic MRI or abdominal CT images were interpreted by radiologists in charge of the colorectal cancer team. The radiologists recorded the rectal wall invasion depth and presence of lymphadenopathy. At the time of diagnosis, we conducted a routine test for carcinoembryonic antigen (CEA) level.
All patients received a long-course RT (4500–5040 cGy divided into 25–28 fractions). During nCRT, patients received concurrent chemotherapy with an oral 5-fluorouracil (5FU)-based regimen. Specifically, tegafur 100 mg and uracil 224 mg (Ufur) were administered orally 3 times daily, together with folinate 45 mg once daily. No body surface area (BSA) adjustment was applied. Chemotherapy was initiated once long-course RT was confirmed and continued daily throughout the entire radiotherapy course and during the waiting period until the day before surgery. Although the total duration varied depending on radiotherapy scheduling and surgical planning, the treatment period typically ranged from 17 to 19 weeks.
After completion of radiotherapy, TME was scheduled at least 8 weeks later. Postoperatively, AC was initiated approximately 1 month after surgery whenever feasible. The adjuvant chemotherapy regimens included: continuation of oral Ufur and folinate at the same dosing schedule as during nCRT, administered continuously for at least 3 months; high-dose intravenous 5-FU infusion protocol consisting of 5-FU 2800 mg × BSA mg plus folinic acid 75 mg × BSA mg every 2 weeks for 6 cycles; FOLFOX regimen consisting of 5-FU 1500 mg × BSA mg, oxaliplatin 85 mg × BSA mg, and folinate 75 mg × BSA mg, administered every 2 weeks for 6 cycles.
The choice of adjuvant chemotherapy regimen was determined by the attending physician based on clinical judgment, but the specific rationale for regimen selection was not systematically recorded in patient charts. Pathological results including tumor stage, histological grade, and presence of mucinous components, lymphovascular emboli, perineural invasion (PNI), and cut-end, and circumferential margins were recorded. All patients were followed up every 3 months postoperatively during the first 3 years and then every 6 months thereafter.
Statistical analyses
All data were analyzed with by IBM SPSS Statistics 26. All samples underwent normality testing using Kolmogorov-Smirnov test. For categorical variables, we used chi-squared test of independence. Fisher’s exact and likelihood-ratio chi-squared test were used for small samples. For continuous variables without normality distribution, we used the Mann-Whitney U test. Survival analysis was performed using the Kaplan-Meier method (log-rank test) and Cox regression model for risk factor analysis. The proportional hazards assumption was assessed using Schoenfeld residuals analysis, and no significant deviations were observed.
Ethics approval and consent to participate
This study was approved by the institutional review board of Taipei Veterans General hospital (Ethics Code: 2023-08-007CC) and laid down in an appropriate version of the Declaration of Helsinki.
Results
We analyzed the data of 279 patients with rectal cancer receiving nCRT who had an initial clinical stage of II to III. We excluded 24 patients with synchronous tumors and 2 patients who achieved clinical complete remission but did not undergo an operation until recurrence. Of the 279 patients who received nCRT for rectal cancer, 8 did not undergo TME surgery but received local treatment after nRT. During the nCRT treatment course, 4 patients were confirmed to have stage-IV disease. We excluded 6 patients with squamous cell carcinoma or GIST and 41 patients who received short-course RT. Altogether, 194 patients with cT3–4 or cN-positive rectal cancer received nCRT followed by TME surgery (Figure 1).

Flow chart of the selection of study participants.
Following TME surgery, pathological reports showed that 28 patients (14.4%) had a complete response. Moreover, 44 (22.7%), 69 (35.6%), and 53 (27.3%) patients had T1-2N0, T3-4N0 and N+ disease, respectively. In the ypT3-4N0 group, 6 patients were originally classified as clinical stage II (cT3-4N0), and 63 patients were clinical stage III (cT3-4N+).
Altogether, 69 patients with clinical stage II to III disease who were reported to have ypT3-4N0 disease showed no significant differences in demographic characteristics, including age, sex, tumor location, American Society of Anesthesiologists (ASA) score, Eastern Cooperative Oncology Group (ECOG) performance status, and so on (Table 1). In the TME-only group and TME with AC group, 28 (90.3%) and 36 (94.7%) patients, respectively, received concurrent chemotherapy. The main protocol used was Ufur plus folinate, which was used by 27 (77.4%) and 33 (86.8%) patients in the TME only group and TME with AC groups, respectively (Table 2). There were no significant differences in the operative characteristics, except for the distal margin, between the 2 groups (Table 1). The average distal margins of the TME only and TME with AC groups were 2.00 cm [interquartile range (IQR) 1.00 cm] and 3.00 cm (IQR 1.73 cm), respectively, showing significant differences (P = 0.026). No significant differences in the operative time and complication rate were found between patients who underwent low anterior resection and abdominal perineal resection, and between those who underwent high ligation or not, operative times, or complication rates.
Patients’ demographic and operative characteristics.
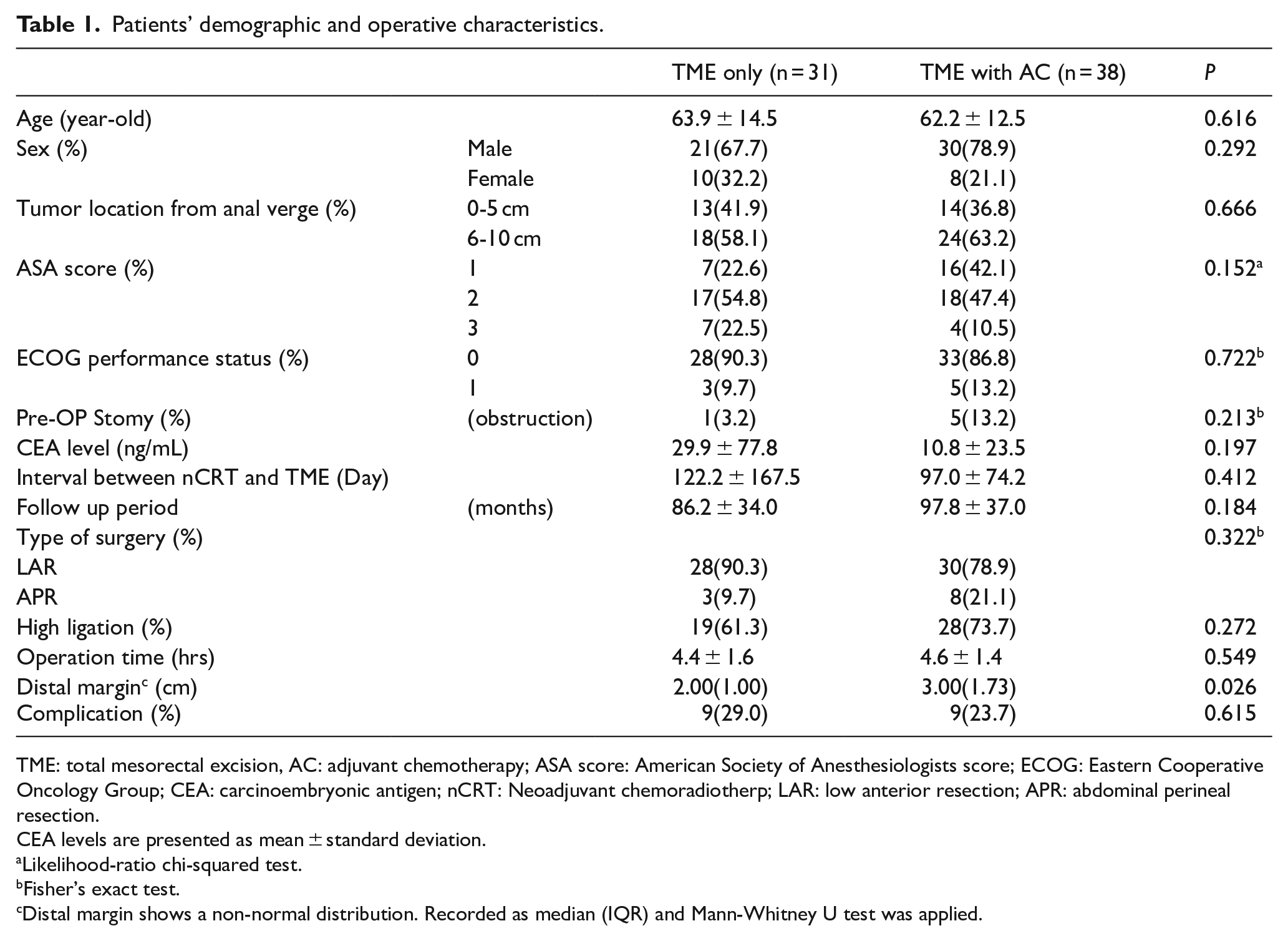
TME: total mesorectal excision, AC: adjuvant chemotherapy; ASA score: American Society of Anesthesiologists score; ECOG: Eastern Cooperative Oncology Group; CEA: carcinoembryonic antigen; nCRT: Neoadjuvant chemoradiotherp; LAR: low anterior resection; APR: abdominal perineal resection.
CEA levels are presented as mean ± standard deviation.
Likelihood-ratio chi-squared test.
Fisher’s exact test.
Distal margin shows a non-normal distribution. Recorded as median (IQR) and Mann-Whitney U test was applied.
The chemotherapeutic regimens.
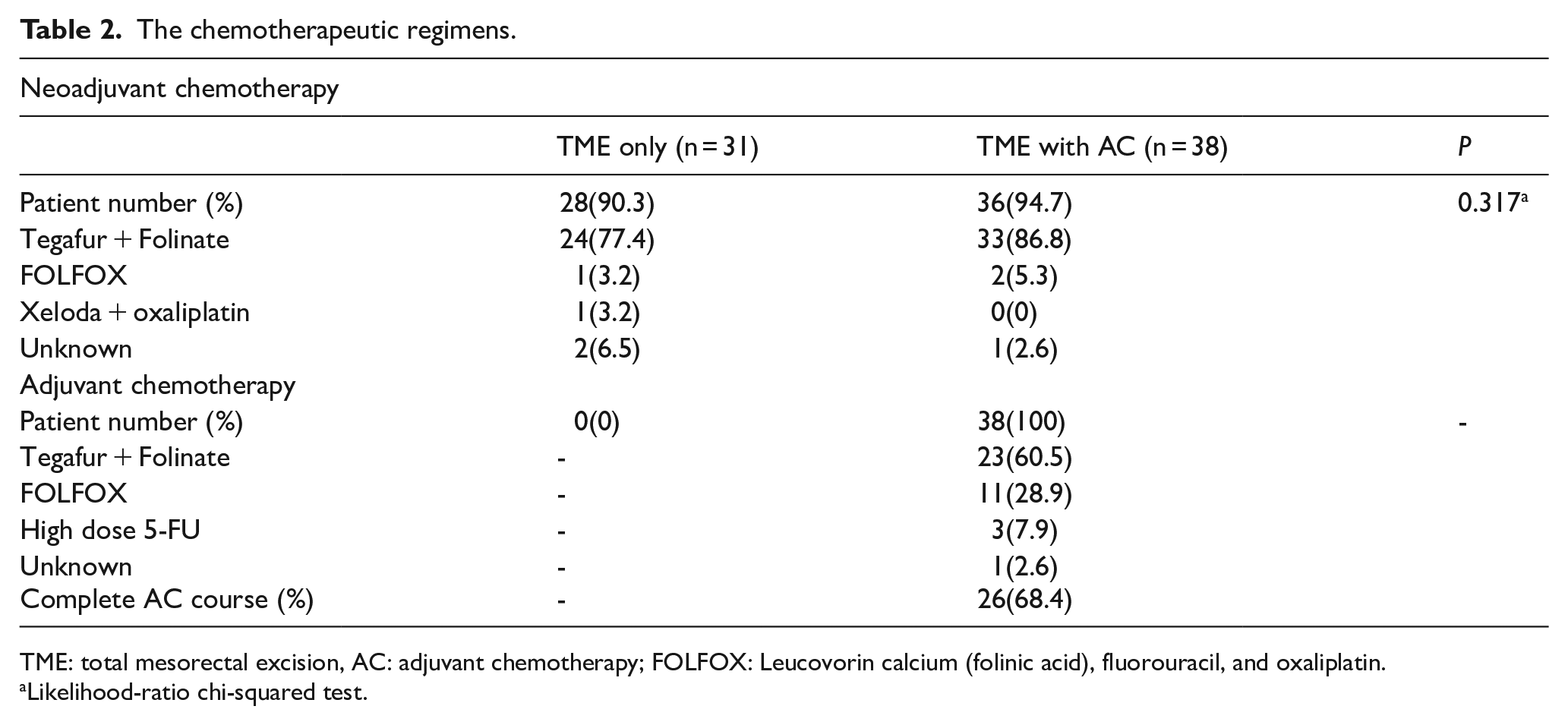
TME: total mesorectal excision, AC: adjuvant chemotherapy; FOLFOX: Leucovorin calcium (folinic acid), fluorouracil, and oxaliplatin.
Likelihood-ratio chi-squared test.
Regarding the pathological characteristic, no significant differences were found between TME only and TME with AC (Table 3). Although patients with circumferential resection margin involvement or lymphovascular invasion (LVI) received AC, this did not have a significant impact on the analysis. The distribution of AC is listed in Table 2, with patients receiving AC for over 6 cycles or 3 months considered to have completed the full AC course. This was achieved by 26 patients (68.4%).
The pathological characteristics of the study participants.
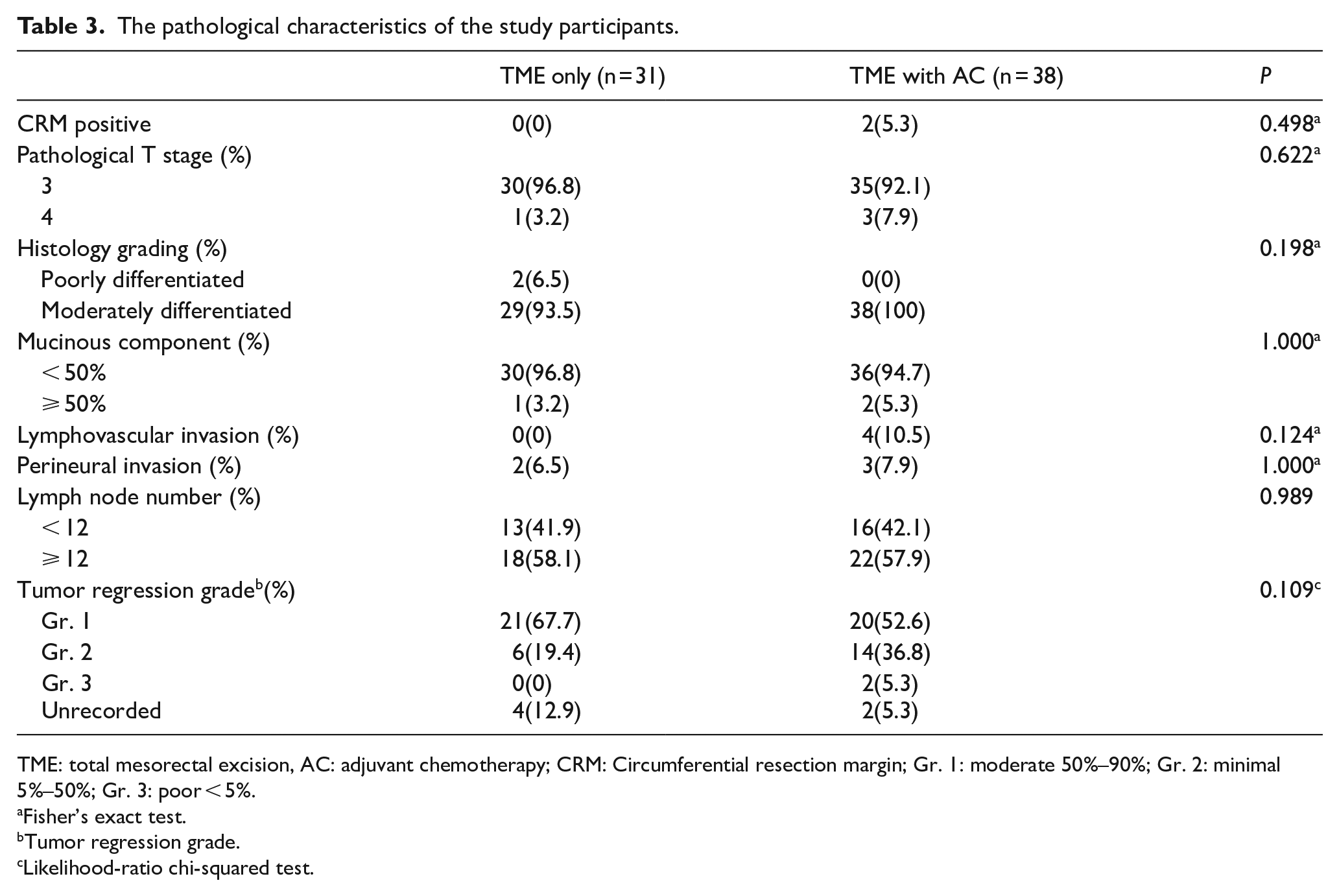
TME: total mesorectal excision, AC: adjuvant chemotherapy; CRM: Circumferential resection margin; Gr. 1: moderate 50%–90%; Gr. 2: minimal 5%–50%; Gr. 3: poor < 5%.
Fisher’s exact test.
Tumor regression grade.
Likelihood-ratio chi-squared test.
The median follow-up period was 97 (range: 10-151) months. Patients who received TME plus AC had a significantly higher 5 year survival rate (86.8%) than those who received TME alone (74.2%) (P = 0.016) (Table 4). However, there was no significant difference in the 5 year disease-free survival rates between the 2 groups (73.7% for TME plus AC group vs 67.4% for TME only group; P = 0.193) (Figure 2). The univariate and multivariate analyses of risk factors for 5 year overall and disease-free survival are summarized in Table 4. In the univariate analysis, treatment type (TME plus AC vs TME only) and the presence of LVI or PNI were significantly associated with overall survival (P = 0.016 and P = 0.048, respectively). For disease-free survival, only the presence of LVI or PNI showed a significant impact (P = 0.004). Other variables, including age (< 65 vs ⩾ 65 years, P = 0.811), sex (male vs female, P = 0.652), pre-treatment CEA levels (<5 vs ⩾5 ng/mL, P = 0.318), ASA score, tumor location, and concurrent neoadjuvant chemotherapy were not significantly associated with survival outcomes. In the multivariate analysis, whether AC is performed or not and whether LVI or PNI presence is performed independently affect the 5-year survival rate.
Risk factors of the 5-year overall and 5-year disease-free survival.

TME: total mesorectal excision, AC: adjuvant chemotherapy; CEA: carcinoembryonic antigen; ASA score: American Society of Anesthesiologists score; LVI: Lymphovascular invasion, PNI: perineural invasion.

Overall survival and disease-free survival curves.
Additionally, we compared the overall survival rates between patients who received 5-FU-based AC (Ufur) and those who received a combination of 5-FU and oxaliplatin (FOLFOX regimen). No significant difference in 5-year overall survival was observed between the 2 groups (P = 0.614) (Figure 3).

Overall survival curve among patients stratified by the adjuvant chemotherapy regimen.
Discussion
According to NCCN guidelines, all patients with locally advanced rectal cancer should receive AC, regardless of their final tumor pathological stage. 1 However, it is controversial on the issue of AC in patient has pathological stage II.7-10 Based on our results, we found that patients with clinical stage II–III and pathological stage II disease who underwent TME with adjuvant AC had a significantly better overall survival rate than those who underwent TME alone.
In our study, the decision to administer adjuvant chemotherapy was primarily based on the attending physician’s clinical judgment. Some physicians recommended AC according to the pathological stage after nCRT and surgery, while others preferred to base their decision on the initial clinical staging before neoadjuvant treatment. These differing clinical practices resulted in variations in AC administration among patients, reflecting the lack of a universally accepted protocol for patients downstaged to pathological stage II after nCRT.
Moreover, some patient-related factors might have influenced the decision for AC administration. Postoperative ECOG performance status was not available in our dataset. Instead, we used the ASA score as a surrogate marker to reflect patient health status, although it cannot fully replace ECOG in evaluating patient functional capacity. Our 5-year overall survival rate is similar to those of other studies.7,9,14,15 The NCCN guideline recommends that all patients receiving nCRT should undergo AC based on the initial clinical staging. 1 However, several papers have questioned the necessity of AC in patients who achieve a good tumor response.5,6,16-18 In previous studies, patients who achieved tumor remission without lymph node involvement (ypT0-2N0) were shown to derive limited benefit from AC. However, in our study, after excluding patients with well pathological remission, we found that AC significantly improved overall survival even in patients with residual ypT3-4N0 disease following nCRT. According to previous research, distant metastasis is the main cause of treatment failure among patients with locally advanced rectal cancer. 19 In our analysis, the recurrence patterns showed no significant difference between the TME with AC group and the TME only group. However, the improvement in overall survival may suggest that AC prolongs survival after recurrence.
Although the improvement in overall survival was observed, no significant difference in disease-free survival was noted. This discrepancy may be explained by several factors. First, AC may have contributed to a longer post-recurrence survival, as patients receiving chemotherapy could have had better general health or more effective salvage treatments after recurrence. Second, the limited sample size and lower event rate for disease-free survival may have resulted in insufficient statistical power to detect a difference, whereas overall survival outcomes were more readily distinguished. Third, although chemotherapy administration generally followed physician preference based on initial clinical or pathological stage, adjustments according to patients’ general condition were possible, leading to potential selection bias. Finally, while recurrence patterns were similar between groups, patients who received adjuvant chemotherapy may have had greater access to second-line or third-line therapies, contributing to the observed survival advantage.
Our results showed that, based on the results of our multivariate analysis, both AC and the presence of LVI or PNI had an independent influence on the overall survival. Additionally, the disease-free survival rate significantly differed between patients with either LVI or PNI, which is consistent with the results of other studies.6,20,21 Therefore, the findings of the pathological reports are highly indicated as risk factors.
For the chemotherapy regimens, our study used Ufur instead of FOLFOX or CAPOX in 2010–2016. It was thought to be less effective for distant metastasis control. 5 However, we found no characteristic difference in the concurrent treatment with nRT between the TME with AC and TME only. AC with Ufur was mainly prescribed to our study population, as it was thought to an effect similar to 5-FU infusion.22,23 In our study, the type of adjuvant chemotherapy regimen, whether based on 5-FU alone or combined with oxaliplatin, did not appear to significantly influence overall survival outcomes. This suggests that, within this patient population, the administration of adjuvant chemotherapy itself, rather than the specific regimen used, may be the key contributing factor to improved survival. This finding is consistent with prior reports suggesting that the survival benefit is more related to the receipt of systemic therapy rather than the specific agents used. 17 In our study, only 68.4% of patients completed the full AC course, defined as at least 3 months of treatment. The main reason for incomplete AC was patient refusal, mostly due to treatment-related issues such as decreased appetite and reduced physical activity, although most patients did not specify detailed reasons. In contrast to the study by Park et al, 24 where AC completion was necessary to achieve survival benefits in ypStage II rectal cancer patients, our findings demonstrated overall survival benefits even among patients who only received partial AC. In Park et al’s study, chemotherapy was typically limited to 2 short infusions of 5-FU or capecitabine used primarily as a radiosensitizer during radiotherapy. Nevertheless, in our study, most patients had already received oral 5-FU during the nCRT and interval to TME phase. This prolonged systemic exposure to 5-FU derivatives during nCRT may have contributed to earlier control of micrometastatic disease, thereby reducing the dependency on complete adjuvant chemotherapy for survival benefits. 25 This hypothesis aligns with emerging concepts in rectal cancer management, such as TNT, where early delivery of systemic therapy is prioritized to maximize treatment efficacy before surgery.
Moreover, a recent post hoc analysis of the CAO/ARO/AIO-04 phase 3 trial by Diefenhardt et al 26 demonstrated that adherence to nCRT, rather than to AC, was associated with improved outcomes. This further supports the notion that early systemic therapy during the neoadjuvant phase may play a more decisive role in long-term prognosis than the completion of postoperative AC alone. Thus, despite the suboptimal AC completion rate in our study, the administration of AC—along with prior systemic therapy during nCRT—still translated into an overall survival benefit for patients with ypT3-4N0 disease.
Although TNT has emerged as an ideal approach to maximize systemic therapy delivery, it was not a standard treatment during our study period (2010–2016). For patients unsuitable for TNT due to treatment intolerance or urgent clinical needs requiring early surgery, the combination of nCRT followed by TME remains a critical treatment strategy. In these cases, additional AC may help compensate for potentially insufficient chemotherapy exposure during the neoadjuvant phase. Although our study defined the full course of AC as 3 months, it remains uncertain whether a shorter duration could still provide comparable survival benefits. Further prospective studies are warranted to elucidate the optimal AC regimen and duration in this subset of patients.Although a short-course RT as neoadjuvant treatment is now widely accepted,15,27,28 during the time of our study, a long-course RT was the protocol generally applied in our hospital. Many studies have suggested long-course RT with 45 Gy in 25 fractions, 29 and delaying surgery for 6–8 weeks after long-course RT has been recommended. 30 However, the optimal timing of surgery after short-course RT was unclear in the early 2000s. Later on, delayed surgery followed by short-course RT was suggested due to its lower operative complications and better pathological response.31-34 Immediate surgery was conducted in our population, so we excluded the patients who received short-course RT from our analysis.
Our study has several limitations. First, it is a single-institution retrospective analysis, which may limit the generalizability of our results. In addition, for patients who underwent neoadjuvant treatment at local hospitals close to their residences, detailed chemotherapy records were sometimes unavailable if the patients failed to transfer their external treatment records. In such cases, the chemotherapy regimen was classified as “unknown” to avoid misclassification bias. Minor missing information regarding neoadjuvant chemotherapy details is acknowledged as a limitation. Second, the decision to administer AC was made by the attending physician, which could result in bias. A prospective clinical trial, such as ACRNaCT, which published its trial protocol in 2019, 35 may provide more definitive results in determining which patients should receive AC. This trial is designed to evaluate whether patients with clinical stage II–III rectal cancer who are downstaged to stage II after nCRT and TME require AC. Third, although all patients were enrolled based on the institutional protocol of nCRT, which mandates concurrent chemotherapy during radiation, a few cases did not receive chemotherapy during neoadjuvant treatment due to clinical considerations such as treatment toxicity or physician discretion, and the regimen of neoadjuvant or AC mainly comprised Ufur plus folinate, rather than the current treatment of FOLFOX or CAPOX. We had a high variation in the interval between nCRT, and TME and between TME and AC. We excluded patients receiving short-course RT, because immediate surgery followed by nRT was applied at that time. This may have caused differences in the metastasis disease patterns and pathological responses. The median interval between neoadjuvant long-course RT and TME was 56 days (IQR 15 days). Last but not least, sample size calculation was not performed prior to this study. The limited number of patients may affect the statistical significance of the results.
Conclusion
While TNT has emerged as a preferred strategy for rectal cancer, not all patients are suitable candidates. Our study shows that in patients with clinical stage III disease downstaged to ypStage II after nCRT and TME, the addition of AC may remain beneficial for improving overall survival (86.8% vs 74.2%, P = 0.016). Neoadjuvant CRT combined with postoperative AC continues to play an important role for patients who cannot undergo full TNT protocols.
Supplemental Material
sj-docx-1-onc-10.1177_11795549251359151 – Supplemental material for The Benefits of Adjuvant Chemotherapy for ypT3-4N0M0 Rectal Cancer Following Neoadjuvant Chemoradiation and Surgery
Supplemental material, sj-docx-1-onc-10.1177_11795549251359151 for The Benefits of Adjuvant Chemotherapy for ypT3-4N0M0 Rectal Cancer Following Neoadjuvant Chemoradiation and Surgery by Chih-Hsien Chang, Hung-Hsin Lin, Shih-Ching Chang, Jeng-Kai Jiang, Huann-Sheng Wang and Yuan-Tzu Lan in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
This study was approved by the institutional review board of Taipei Veterans General hospital (Ethics Code: 2023-08-007CC) on September 12, 2023, and laid down in an appropriate version of the Declaration of Helsinki.
Consent to Participate
Consent to participate has been waived by the relevant Ethics Committee or Institutional Review Board, and has been approved with ethics code 2023-08-007CC on September 12, 2023.
Consent for Publication
Not applicable
Authors Contributions
(I) Conception and design: Hung-Hsin Lin
(II) Administrative support: Hung-Hsin Lin
(III) Provision of study materials or patients: Hung-Hsin Lin, Shih-Ching Chang, Jeng-Kai Jiang, Huann-Sheng Wang, Yuan-Tzu Lan
(IV) Collection and assembly of data: Chih-Hsien Chang
(V) Data analysis and interpretation: Chih-Hsien Chang
(VI) Manuscript writing: All authors
(VII) Validation: Hung-Hsin Lin, Shih-Ching Chang, Jeng-Kai Jiang, Huann-Sheng Wang, Yuan-Tzu Lan
(VIII) Final approval of manuscript: All authors
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability Statement
All data supporting the findings of this study are included in the manuscript and its supplementary materials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
