Abstract
Background:
Our aim was to demonstrate the efficacy and safety of pembrolizumab monotherapy as second-line treatment in Vietnamese nonsmall cell lung cancer (NSCLC) patients.
Methods:
We performed a single-center retrospective study of patients with advanced NSCLC who received pembrolizumab as second-line therapy at the Vietnam National Cancer Hospital between January 2017 and October 2023. The primary endpoints were overall survival (OS) and tumor response.
Results:
A total of 52 patients were included. Adenocarcinoma was observed in 43 of 52 cases (82.7%), and 40.4% of patients had PD-L1 expression in at least 50% of tumor cells. Median body weight was 57 kg (range, 45-80), and 73.1% of patients received pembrolizumab at a dose of 100 mg/3 weeks, with a median dose of 1.9 mg/kg/3-weeks (range, 1.3-4.3). The overall response rate and disease control rate were 26.9% and 51.9%, respectively. Median PFS was 6.5 months (95% CI, 3.3-9.7), and 1-year and 2-year PFS rates were 28.5% and 15.9%, respectively. Median OS was 12.0 months (95% CI, 9.8-14.2); 1-year, 2-year, and 3-year OS rates were 53.1%, 22.3%, and 8.5%, respectively. ECOG status and number of organs metastases were significantly associated with PFS and OS in the multivariate analysis. No adverse events of grades 3 to 4 were reported during the treatment.
Conclusions:
Second-line pembrolizumab has a good disease control rate and prolonged survival and is a viable option for the treatment of Vietnamese NSCLC patients. Further clinical studies are necessary to determine the effectiveness of administering a low dose of pembrolizumab in this setting, particularly in Asian population.
Keywords
Introduction
Among all cancers, lung cancer accounts for the highest number of deaths worldwide. 1 In Vietnam, it is one of the most frequently diagnosed cancers and is associated with high morbidity and mortality rates among newly identified cases. 2 According to the World Health Organization (WHO) classification, lung cancer is divided into 2 major histological categories: small cell lung cancer and nonsmall cell lung cancer (NSCLC), with the latter accounting for approximately 85% of all cases. 3 Among NSCLC subtypes, adenocarcinoma and squamous cell carcinoma are the most prevalent. 3 In terms of race, EGFR and ALK alterations occur more frequent in Asian than non-Asian populations.4-8 Patients harboring these mutations typically benefit from treatment with tyrosine kinase inhibitors. 9 However, such genetic alterations are absent in most NSCLC cases, in whom immune checkpoint inhibitors have emerged as the standard of care in both first- and second-line therapeutic settings.9,10
Pembrolizumab, a monoclonal antibody targeting the PD1 pathway, has shown clinical benefit in pretreated patients with NSCLC through randomized trials. Compared with docetaxel, pembrolizumab monotherapy led to notably longer progression-free survival (PFS) and overall survival in pretreated NSCLC patients, as shown in the KEYNOTE-010 study. The Keynote-010 study enrolled Asian patients but not Vietnamese patients. 11 One should keep in mind that barriers to access to immunotherapy are a major issue in the nations classified as low- or middle-income, where lung cancer is often diagnosed at a late stage and mortality rates are higher compared with high-income countries.12-15 Under these circumstances, first-line immunotherapy use remained limited, and platinum-based chemotherapy was commonly offered to patients with NSCLC lacking actionable mutations. Although pembrolizumab was approved in Vietnam in 2017 for NSCLC, few patients received the immunotherapy. Therefore, this has led to a lack of real-world data on the efficacy of pembrolizumab for NSCLC, especially in the Vietnamese population. We here conducted a retrospective study to demonstrate the treatment outcome of pembrolizumab monotherapy in previously treated Vietnamese NSCLC patients.
Methods
This retrospective study included stage IV NSCLC patients treated with pembrolizumab monotherapy as a second-line treatment from January 2017 to October 2023 at the Vietnam National Cancer Hospital. This study followed the STROBE reporting guidelines 16 (supplementary file). The eligibility criteria were (1) age over 18 years; (2) histological diagnosis confirmed of NSCLC; (3) Eastern Cooperative Oncology Group (ECOG) 17 performance status of 0 or 1, 2, or 3; and (4) confirmation of progressive disease according to Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 after systemic therapies. We excluded patients who had previously received PD-1 inhibitors, carcinomatous meningitis, or were allergic to pembrolizumab or any of its excipients or accompanied with other malignant neoplasms. In addition, patients who had changed to other treatment without experiencing progressive disease or unacceptable toxicities were excluded. Using a 22C3 PharmDx immunohistochemistry assay (Dako), PD-L1 was measured before the treatment. EGFR mutation and/or ALK rearrangement were observed in 7 patients, all of whom were treated with pembrolizumab after progressing to standard TKIs.
No sample size calculation was conducted prior to the study, as all eligible patients during the study period were included in this retrospective cohort. All patients were to receive pembrolizumab intravenously over 30 minutes every 3 weeks. The response to therapy was assessed after every 3 cycles of pembrolizumab using abdomino-pelvic and/or chest computed tomography (CT), or when patients had deteriorated clinical symptoms, imaging evaluation was performed immediately by clinical physicians. Pembrolizumab was given until disease progression or occurrence of intolerable toxicity.
Endpoints and evaluation
Follow-up for all patients continued until October 15, 2024, with primary endpoints evaluating overall survival (OS) and tumor response. OS was defined as the time interval between the date of starting pembrolizumab treatment and the date of death from any cause or last follow-up. The tumor response was divided into complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD) according to RECIST 1.1. 18 Radiologic evaluations were performed by board-certified radiologists, and progression was defined based on imaging findings of target and nontarget lesions in accordance with RECIST guidelines. Clinical judgment was only considered in cases where radiologic data were not available or ambiguous.
Secondary endpoints were PFS and toxicity. PFS was defined as the duration from the initiation of pembrolizumab therapy to the first documented disease progression at any site or death from any cause. Adverse events (AEs) were identified through a retrospective review of medical records, including laboratory findings and physical examination data obtained during treatment, and were graded based on the National Cancer Institute’s Common Terminology Criteria for Adverse Events (CTCAE), version 4.03.
Medical records were reviewed to collect information on patient characteristics (age, sex), smoking status, clinical symptoms, ECOG, and tumor characteristics (histological type, PD-L1 expression, EGFR mutations, ALK gene rearrangement), previous treatments (number of previous chemotherapy used, interval time between their advanced diagnosis and initiation of pembrolizumab administration). The patient’s performance status was assessed according to the ECOG score. The response to pembrolizumab was evaluated based on RECIST v1.1 by regular imaging examinations every 3 cycles of immunotherapy or a suspected clinical progression by clinical physicians. The endpoints of the study were recorded from the initiation of pembrolizumab to the patient’s events or the end of the study on October 15, 2024.
Data analysis
Statistical analyses were performed using SPSS Statistics software version 22.0 (IBM Corp., Armonk, NY, USA). Percentages were used to describe categorical variables, and continuous variables were summarized by mean ± standard deviation (SD) or mean and range based on their distribution normality. Chi-square and Fisher’s exact tests were used to assess the difference between categorical variables. Survival outcomes were assessed using Kaplan-Meier analysis. Subgroup comparisons were conducted with the log-rank test, and hazard ratios (HRs) were calculated using Cox proportional hazards regression models. All statistical tests were 2-sided, and a P-value of <.05 was considered statistically significant.
Results
Patient demographics and therapeutic information
A total of 52 patients with histologically confirmed advanced NSCLC were retrospectively included in the study at the Vietnam National Cancer Hospital between January 2017 and October 2023 (Figure 1). The demographic information, clinical characteristics, and treatment features are summarized in Table 1. Of the 52 patients in our study, the majority were men (84.6%) with a mean age of 60.7 ± 7.1 (range, 43-77); 73.1% of participants were former or current smokers. The patient’s performance status was ECOG 0-1 in 78.8% of cases. Eighteen patients (34.6%) had prior brain metastasis and were previously treated with stereotactic radiation (SRS) for 12 patients, and whole-brain radiotherapy (WBRT) for 6 patients. Adenocarcinoma was observed in 43 of 52 cases (82.7%), and PD-L1 expression (TPS) ⩾ 50% of tumor cells was in 40.4%. There were 7 patients with genetic alterations, including 6 with EGFR mutations (del19 or L858R) and 1 with ALK rearrangement, and all of them were diagnosed of progressive diseases with standard care of targeting TKIs before being enrolled into our study. Prior treatment regimens for patients with driver mutations are summarized in Table 1, under abbreviations. About one-third of patients had undergone at least 3 prior systemic treatment regimens (30.8%), and 51.6% participants had interval time between advanced diagnosis and pembrolizumab administration more than 12 months. The median total body weight of 57 kg (range, 45-80), and 38 patients who received pembrolizumab at a dose of 100 mg/3-weeks in our study (73.1%), with a median dose of pembrolizumab per body weight of 1.9 mg/kg/3 weeks (range, 1.3-4.3). Pembrolizumab was prescribed at a dose of <2 mg/kg/3 weeks in 51.9%.

Study algorithm of Vietnamese patients included in the study. NSCLC, nonsmall cell lung cancer.
Baseline characteristics of Vietnamese patients with advanced NSCLC treated with second-line pembrolizumab.
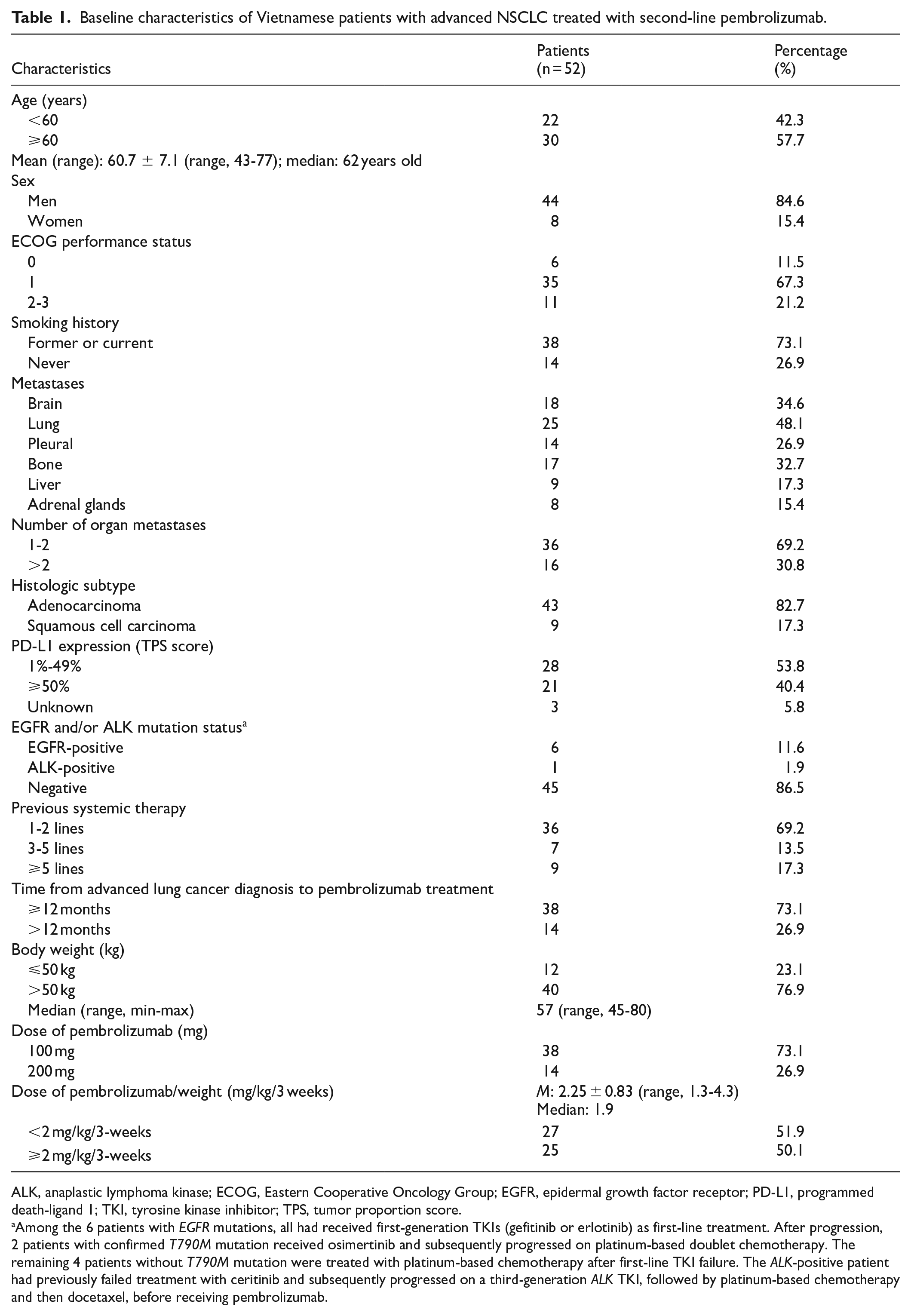
ALK, anaplastic lymphoma kinase; ECOG, Eastern Cooperative Oncology Group; EGFR, epidermal growth factor receptor; PD-L1, programmed death-ligand 1; TKI, tyrosine kinase inhibitor; TPS, tumor proportion score.
Among the 6 patients with EGFR mutations, all had received first-generation TKIs (gefitinib or erlotinib) as first-line treatment. After progression, 2 patients with confirmed T790M mutation received osimertinib and subsequently progressed on platinum-based doublet chemotherapy. The remaining 4 patients without T790M mutation were treated with platinum-based chemotherapy after first-line TKI failure. The ALK-positive patient had previously failed treatment with ceritinib and subsequently progressed on a third-generation ALK TKI, followed by platinum-based chemotherapy and then docetaxel, before receiving pembrolizumab.
Efficacy analysis
Table 2 shows the results of tumor response. The ORR was observed in 26.9% of patients (14 of 52) and DCR was 51.9% with 13 patients experiencing SD. The subgroup analyses of ORRs and DCR are shown in Table 3. In the univariate analysis, the ORR was significantly higher in the patients with ECOG 0-1 (P = .02), and the DCR was significantly higher in the patients with ECOG 0-1 (P = .013), in the patients with ⩽2 organ metastases (P = .002), and in the group with ⩽2 previous lines of systemic therapy (P = .045). The multivariate analysis revealed a significant difference in ORR associated with ECOG (P = .009). In addition, ECOG status (P = .01) and number of organ metastases (P = .002) were significantly associated with DCR according to the multivariate analysis.
Treatment outcome to pembrolizumab according to RECIST v1.1.
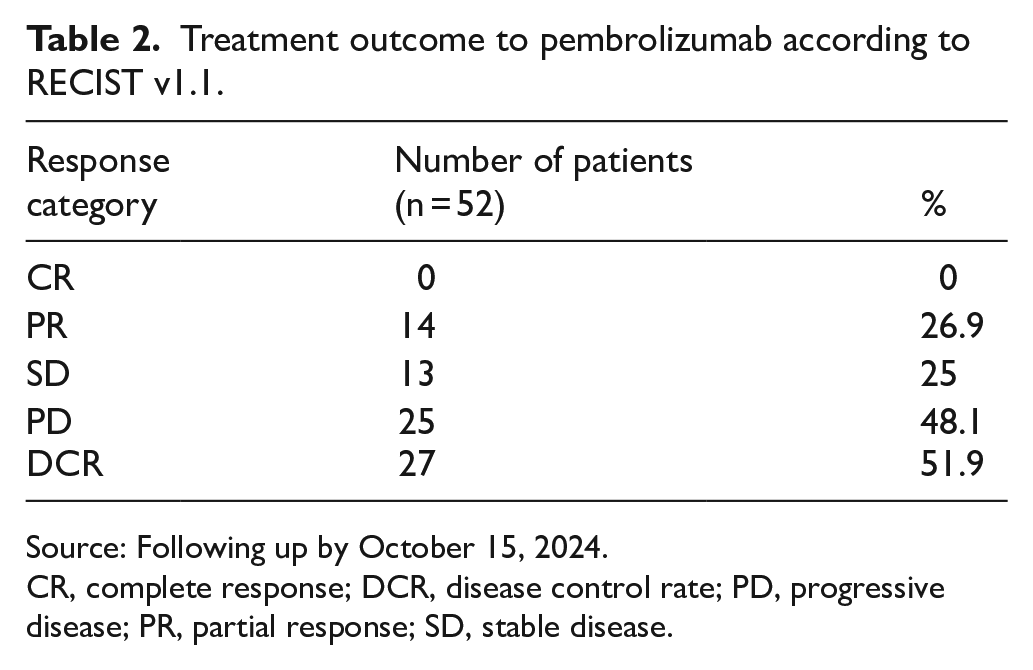
Source: Following up by October 15, 2024.
CR, complete response; DCR, disease control rate; PD, progressive disease; PR, partial response; SD, stable disease.
Univariate and multivariate analyses of ORR and DCR according to clinical and subclinical features.
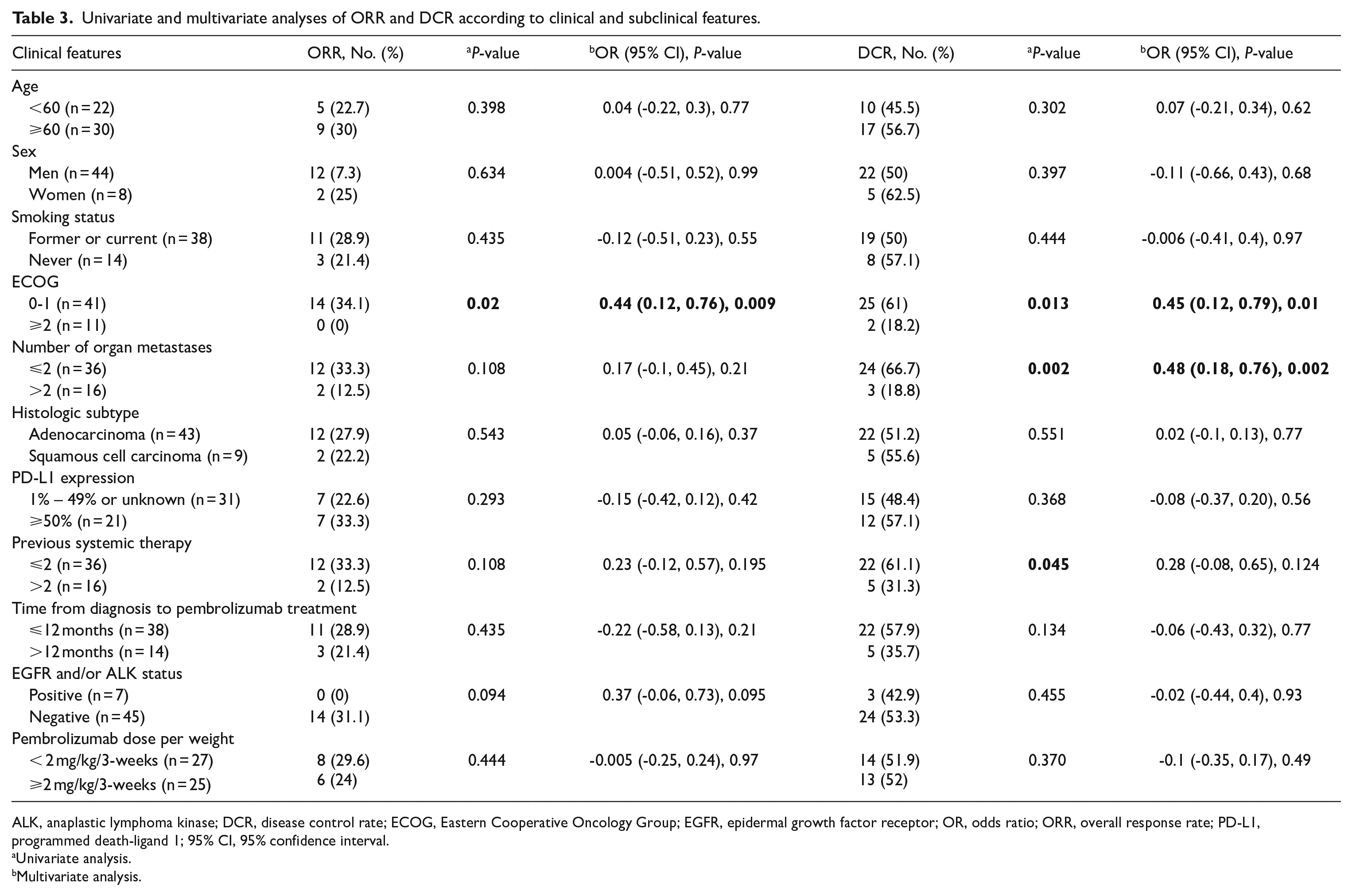
ALK, anaplastic lymphoma kinase; DCR, disease control rate; ECOG, Eastern Cooperative Oncology Group; EGFR, epidermal growth factor receptor; OR, odds ratio; ORR, overall response rate; PD-L1, programmed death-ligand 1; 95% CI, 95% confidence interval.
Univariate analysis.
Multivariate analysis.
By the end of the study period, 9 patients were progression-free and still alive, particularly 1 patient who experienced 35 pembrolizumab cycles with completed response. However, this patients had disease relapse after 1 year of follow-up. One patient had an SD for 18 months with an oligo hepatic lesion on CT scan, however, we determined that he had an SD with pembrolizumab of NSCLC and had a second cancer of HCC during the treatment according to liver biopsy. Median PFS was 6.5 months (95% CI, 3.3-9.7), 1- and 2-year PFS rates were 28.5% and 15.9%, respectively (Figure 2). During the follow-up, 10 patients were alive and median OS was 12.0 months (95% CI, 9.8-14.2); 1-, 2-, and 3-year OS rates were 53.1%, 22.3%, and 8.5%, respectively (Figure 3). Univariate and multivariate analyses of median PFS and OS by subgroups are presented in Tables 4 and 5. Univariate analysis showed significantly longer median PFS and OS in patients with ECOG 0-1, in patients with ⩽2 organ metastases, and in patients with time from initial diagnosis to pembrolizumab treatment ⩽12 months (P < .05). In addition, patients without EGFR mutations or ALK rearrangements showed significantly longer OS in univariate analysis compared with those with EGFR/ALK alterations. However, this difference was not maintained in the multivariate Cox regression model. No significant differences in PFS were observed between the EGFR/ALK-negative and -positive groups. Cox regression analysis revealed a significant difference in PFS and OS associated with ECOG and number of organ metastases (P < .05).

Kaplan-Meier estimates of progression-free survival.

Kaplan-Meier estimates of overall survival.
Univariate and multivariate analyses of PFS according to clinical and subclinical features.
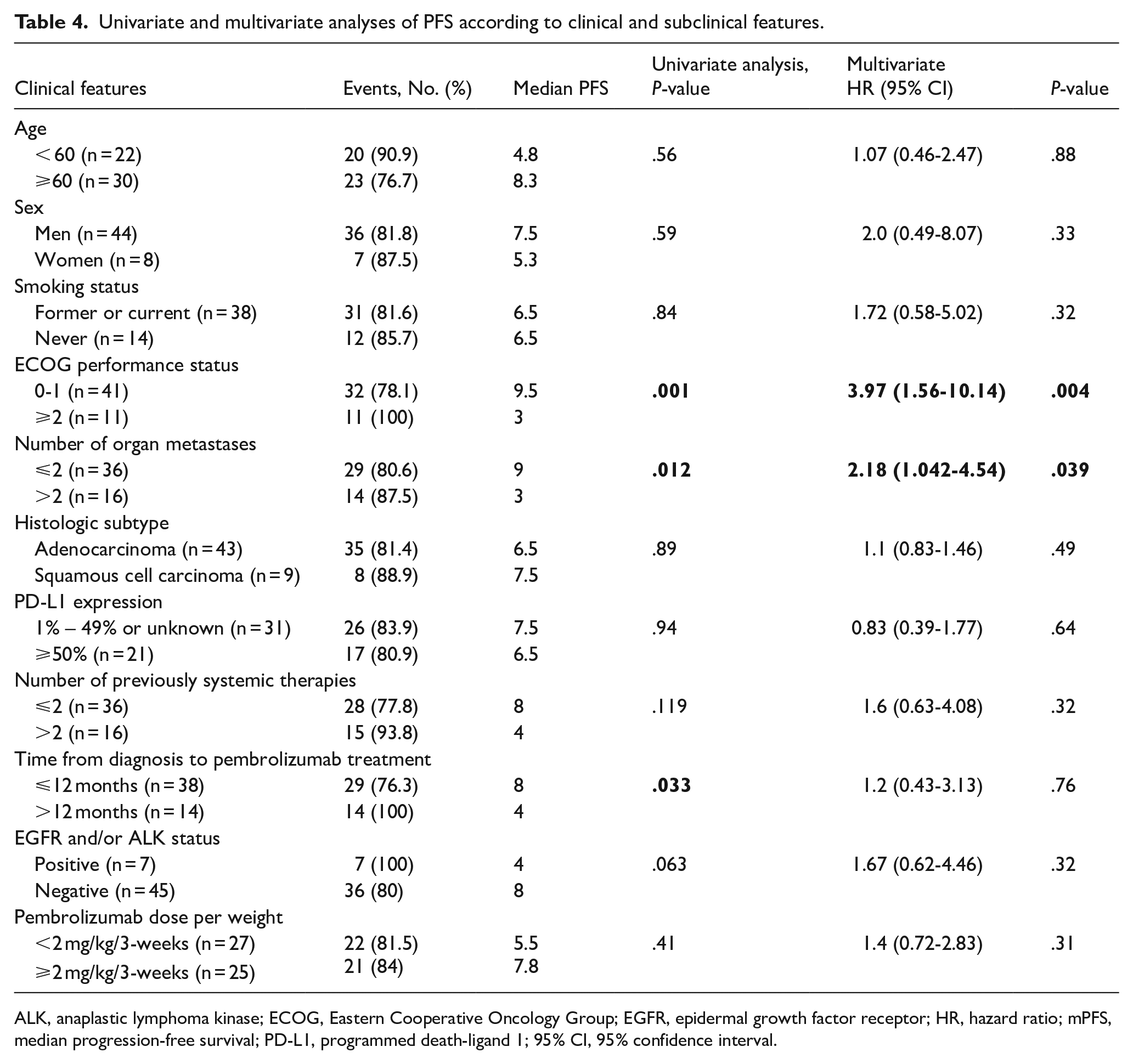
ALK, anaplastic lymphoma kinase; ECOG, Eastern Cooperative Oncology Group; EGFR, epidermal growth factor receptor; HR, hazard ratio; mPFS, median progression-free survival; PD-L1, programmed death-ligand 1; 95% CI, 95% confidence interval.
Univariate and multivariate analyses of OS according to clinical and subclinical features.

ALK, anaplastic lymphoma kinase; ECOG, Eastern Cooperative Oncology Group; EGFR, epidermal growth factor receptor; HR, hazard ratio; mOS, median overall survival; PD-L1, programmed death-ligand 1; 95% CI, 95% confidence interval.
Toxicity
The AE profile of pembrolizumab presents in Table 6. The frequent AEs were rash (13.5%), diarrhea (5.8%), nausea (5.8), and hypothyroidism (5.8%). Pneumonitis was reported in 2 patients (3.8%), with 1 case of grade 1 and 1 case of grade 2 severity. No AEs of grade 3-4 were reported during the treatment. No patients were required to be discontinued or excluded due to unacceptable toxicity.
Adverse events of treatment according to CTCAE 4.03.

Discussion
To our knowledge, no previous study has evaluated second-line pembrolizumab in Vietnamese patients with NSCLC, an Asian population not previously published. In this study, 16 of 52 patients had received at least 3 previous systemic treatments (30.8%), and 73.1% of patients had an interval time less than 12 months between their advanced stage diagnosis and initiation of pembrolizumab administration. In our study, pembrolizumab monotherapy was administered only to patients with PD-L1–positive tumors, except for 3 individuals with missing PD-L1 data due to insufficient biopsy material following next-generation sequencing testing. These patients had previously received multiple lines of treatment, including platinum-based doublets and other second-line chemotherapy regimens, before receiving pembrolizumab as a later-line therapy, which reflects real-world clinical practice in tissue-limited settings where PD-L1 assessment is not always possible.
Our study demonstrated a median total body weight of 57 kg (range, 45-80) and 38 patients received pembrolizumab at a fixed dose of 100 mg/3 weeks (73.1%), with a median dose of pembrolizumab per body weight of 1.9 mg/kg/3 weeks (range, 1.3-4.3). About 52% patients received doses of less than 2 mg/kg/3-weeks. In the clinical Keynote-010 trial, patients received doses of 2 mg/kg or 10 mg/kg every 3 weeks as the second-line immunotherapy treatment. 11 A study by Low et al 13 using pembrolizumab at 2 doses of 100 mg/3 weeks and 200 mg/3-weeks for the treatment of Asian NSCLC patients showed no differences in PFS and OS between the 2 doses, however, cost saving to patients and health care system were mentioned. In Vietnam, pembrolizumab was approved for the treatment of previously treated NSCLC patients in 2017. However, the main source of health care costs was likely to associate with immunotherapy, accounting for about more than 95% of total costs, particularly when patients received a fixed dose of 200 mg of pembrolizumab for the first-line treatment of advanced NSCLC. This is why the cost of immunotherapy was one of the major barriers that affected patient’s decisions in developing countries, including Vietnam. 19 In these circumstances, chemotherapy is typically proposed to patients before considering immunotherapy, and most of patients received a fixed dose of pembrolizumab of 100 mg/3-weeks for the second-line treatment due to the low total body weight of the Asian population. One should keep in mind that using the doses based on body weight (2 mg/kg/3 weeks) instead of a fixed dose of 200 mg for pembrolizumab might reduce cost and increase accessibility to immunotherapy in low- and middle-income countries, especially for patients with average or low body weight as Asian population.12-15 Beyond cost-saving and patient weight considerations, real-world studies and pharmacokinetic/pharmacodynamic modeling support the use of 100 mg every-3-week dosing of pembrolizumab. A Taiwanese cohort study found no significant differences in OS and safety between standard and low-dose groups, identifying 1.8 mg/kg as the minimum effective dose. 20 In addition, pharmacokinetic/pharmacodynamic analyses have shown that pembrolizumab achieves linear pharmacokinetics and saturates PD-1 receptor occupancy at doses ⩾1 mg/kg, indicating that lower fixed doses may be clinically effective, especially in patients with lower body weight. 21 In particular, one retrospective study in Singapore demonstrated similar survival outcomes between patients who received a low-fixed dose of 100 mg/3 weeks compared with a dose of 200 mg/3-weeks in Asian NSCLC patients, even though 80% of patients received pembrolizumab as their first-line therapy. 13 This is also true in upper and middle-income countries, there were enormous cost savings to the health care systems by using a dose of 2 mg/kg. 22 Therefore, using a low dose of 100 mg, or ideally 2 mg/kg every 3 weeks for the treatment of average-sized patients with previously treated NSCLC could be reasonable in the context of Vietnam.
In our study, 52 patients treated with pembrolizumab monotherapy showed the ORR of 26.9% and the DCR of 51.9%. The Keynote-010 study reported that the ORRs for the total population and TPS ⩾50% were 18.0% and 30.2%, respectively, in the group that used pembrolizumab 2 mg/kg; 18.5% and 29.1%, respectively, in the group that used 10 mg/kg. The median PFS and OS were 3.9 months and 10.4 months, respectively, in participants who received pembrolizumab 2 mg/kg. 11 The Keynote-001 trial using pembrolizumab 10 mg/kg for the treatment of NSCLC demonstrated an ORR of 19.4% in the general population, particularly an ORR of 18.0% in patients that had previously been treated and 24.8% in patients that had not been previously. Indeed, current or former smokers had a higher ORR than never smoked patients, 22.5% versus 10.3%, respectively. 23 In our study, the multivariate analysis revealed a significant difference in ORR associated with ECOG. In addition, ECOG and number of organ metastases were significantly associated with DCR.
Our results showed that the median OS in this study was similar to previous study even though about one-third of our participants received at least 3 lines of systemic therapies before using pembrolizumab, 12.0 months compared with 10.4 months in the Keynote 010 trial using pembrolizumab 2 mg/kg. 24 In addition, the 1-year OS rate in our study was 53.1%, and the Keynote 010 showed a 1 year OS rate of 43.2%. Interestingly, the median PFS and OS in our study were nonsignificant difference in patients with a dose ⩾2 mg/kg compared with <2 mg/kg according to both univariate and multivariate analyses. This is also true in the ORR and DCR.
In our study, we observed that patients without EGFR mutations or ALK rearrangements appeared to have improved overall survival in univariate analysis compared with those with these alterations. However, this difference did not remain statistically significant in multivariate analysis when adjusted for other clinical variables. In addition, no clear difference in PFS was noted between the 2 groups. These results reflect the complexity of immunotherapy outcomes in oncogene-driven NSCLC.25,26 Although immune checkpoint inhibitors are generally considered less effective in EGFR- or ALK-positive populations, especially after TKI failure, the limited number of such cases in our cohort (n = 7) restricts the robustness of this finding. Further studies with larger sample sizes are needed to clarify the clinical benefit of immunotherapy in this subgroup.
Furthermore, pembrolizumab monotherapy was well tolerable in our study for Vietnamese patients. The common AEs were rash (13.5%), diarrhea (5.8%), nausea (5.8), and hypothyroidism (5.8%). There were no serious grade 3 or grade 4 AEs. No patients were required to be discontinued or excluded due to unacceptable toxicity. Our results were similar to those of previously published studies.11,13,15
This study had few limitations. Despite the cost-effectiveness of dosing pembrolizumab at 100 mg/3 weeks in our patients, low-fixed doses should be carefully considered in patients who are heavier, especially when overweight is becoming more common in the Vietnamese population. In our study, a median total body weight was 57 kg and there was a median dose of 1.9 mg/kg, and none of the patients received a dose that was less than 1 mg/kg. The retrospective and single-institution nature may introduce selection bias and confounding. In addition, the small sample size limited the power of the study and reduced the ability to compare the differences between subgroups by the statistical analysis.
In resource-constrained settings such as Vietnam, where access to first-line immunotherapy is limited, our data on the use of second-line pembrolizumab monotherapy provide practical and clinically meaningful insight. Although first-line immunotherapy remains the standard of care when feasible, these findings offer important guidance for alternative treatment strategies in real-world practice. In clinical practice, treatment decisions are often influenced not only by efficacy and toxicity profiles, but also by patient preferences and financial considerations. This is particularly relevant when selecting between low-dose regimens, which may be more affordable. In such settings, shared decision-making becomes crucial to align therapeutic choices with patients’ values, goals, and economic capacity. Recent literature emphasizes the importance of this approach, especially in oncology where treatment costs can be prohibitive.27,28
Conclusion
Second-line pembrolizumab has a good disease control rate and prolonged survival and is a viable option for Vietnamese patients who have previously been treated NSCLC. These results highlight the need for further research assessing the clinical effectiveness of a low-fixed-dose pembrolizumab regimen on this particular group of the Asian population.
Supplemental Material
sj-docx-1-onc-10.1177_11795549251359142 – Supplemental material for Effectiveness of Second-Line Pembrolizumab Monotherapy for the Treatment of Nonsmall Cell Lung Cancer: A Retrospective-Single Institution Study of 52 Vietnamese Patients
Supplemental material, sj-docx-1-onc-10.1177_11795549251359142 for Effectiveness of Second-Line Pembrolizumab Monotherapy for the Treatment of Nonsmall Cell Lung Cancer: A Retrospective-Single Institution Study of 52 Vietnamese Patients by Van Tai Nguyen, Hung Kien Do, Quang Van Le, Anh Tu Do, Van Chu Nguyen, Thi Bich Phuong Nguyen and Thi Nhu Hoa Nguyen in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
Not applicable.
Ethical Consideration
Ethical approval for the study was obtained from the Institutional Ethics Review Board of Vietnam National Cancer Hospital, decision number No. 566/QD-BVK dated March 18, 2022. The patient data were maintained with confidentiality, in compliance with the Declaration of Helsinki.
Consent for Publication
Informed consent was waived because of the retrospective design of the study
Author Contribution
Conception and design: Hung Kien Do, Van Tai Nguyen.
Collection and assembly of data: Hung Kien Do, Quang Van Le, Van Tai Nguyen, Anh Tu Do, Van Chu Nguyen, Thi Bich Phuong Nguyen, Thi Nhu Hoa Nguyen.
Data analysis and interpretation: Hung Kien Do, Van Tai Nguyen.
Manuscript writing (draft and revised): All authors.
Final approval of manuscript: All authors.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
The data underlying this study’s findings are included in the article and can be obtained from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
