Abstract
Background:
The incidence of testicular tumors during the COVID-19 pandemic has raised questions about the potential impact of viral infection on tumor development. This study aimed to explore the relationship between COVID-19 and testicular tumors through a retrospective analysis of 32 cases diagnosed before and during the pandemic.
Methods:
A total of 32 testicular tumors were analyzed, with distribution based on the year of diagnosis. Immunohistochemical studies were conducted to assess SARS-CoV-2 and angiotensin-converting enzyme 2 (ACE2) expression in tumor cells.
Results:
The highest frequency of tumor diagnoses was observed in 2021 (19.4%), with a notable increase in diagnoses in 2022 compared with pre-pandemic years. No significant correlation was found between COVID-19 infection and tumor types (P = .476). The distribution of seminoma and mixed germ cell tumors (MGCT) was similar in both periods. Strong SARS-CoV-2 antibody positivity was found in 11 cases, with expression primarily in Leydig cells and some in Sertoli and plasma cells. The difference in SARS-CoV-2 expression between periods was statistically significant (P = 0013). The ACE2 expression was observed in all tumor groups, but statistical analysis was not significant.
Conclusion:
The presence of SARS-CoV-2 nucleoprotein in the tumor microenvironment, particularly during the pandemic, suggests an indirect role of the virus in the development of testicular tumors. Although SARS-CoV-2 does not exhibit direct oncogenic effects, its presence could influence tumorigenesis through mechanisms like inflammation and oxidative stress. The ACE2 expression further supports the hypothesis that the virus may trigger adaptive changes in tumor cells. The SARS-CoV-2 could act as a co-factor in tumor progression, especially in individuals predisposed to testicular tumors.
Background
The development of testicular germ cell tumors (TGCTs) presents a complex challenge in understanding its underlying mechanisms. The emergence of this malignancy is typically associated with maturation arrest in embryonic germ cells, although the exact triggers of this differentiation blockage remain unclear despite extensive research. 1 Cryptorchidism, infertility, familial predisposition, single nucleotide polymorphisms, and hormonal imbalances (including androgen deficiency and inappropriate estrogen exposure) are among the significant factors contributing to this complex pathogenesis.2,3
Beyond hormonal effects, the central role of Sertoli cells in the development of germ cell tumors is particularly noteworthy. Sertoli cells are referred to as nurse cells as they protect germ cells from immune-related damage while regulating their proper differentiation within the testicular microenvironment. 4 One of the factors highlighting the importance of Sertoli cells in spermatogenesis is their production of stem cell factor (SCF), a signaling molecule present in the seminiferous tubules. The SCF interacts with the c-KIT receptor to facilitate the migration, proliferation, and survival of spermatogonia through the Kit/SCF pathway and is critical for the survival of primordial germ cells (PGCs) in both embryonic and postnatal gonads.5,6 The c-KIT is a transmembrane tyrosine kinase receptor encoded by the KIT gene, primarily expressed on the surface of germ cells, hematopoietic stem cells, and interstitial cells. On binding with SCF, c-KIT undergoes dimerization and autophosphorylation, activating downstream signaling pathways such as Phosphatidylinositol 3-kinase/Protein kinase B (PI3K/AKT), Mitogen-Activated Protein Kinase/Extracellular signal-Regulated Kinase (MAPK/ERK), and Janus Kinase/Signal Transducer and Activator of Transcription (JAK/STAT), which regulate cell survival, proliferation, and differentiation. Proper c-KIT function is essential for normal germ cell development, and its dysregulation has been implicated in various germ cell tumors and other malignancies. 6 The increase in c-KIT expression, along with pluripotency markers such as OCT3/4 and NANOG, plays an important role in maintaining PGCs in an undifferentiated state. This complex interaction suggests a delicate balance in the gonocyte microenvironment and points to a potential link between maturation arrest and tumorigenesis. 7 Viral infections have garnered significant attention as potential contributors to the development of TGCTs. Viruses such as human papillomavirus (HPV), Epstein-Barr virus (EBV), cytomegalovirus (CMV), parvovirus B-19, and human immunodeficiency virus (HIV) are under investigation for their interactions with germ cells or the surrounding microenvironment.8,9 Traditionally linked to other diseases, recent studies suggest these viruses may play a role in TGCT pathogenesis. The HPV, eg, is known for its association with cervical cancer and is hypothesized to impact TGCTs through effects on male reproductive health. 10 Similarly, EBV, CMV, parvovirus B-19, and HIV are drawing interest due to their ability to infect germ cells or alter testicular tissues.11 -13 However, the mechanisms by which these infections might contribute to TGCT development remain poorly understood.
In December 2019, Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), a novel RNA virus, was first identified in Wuhan, China, with the initial outbreak linked to the Huanan Seafood Wholesale Market. 14 This virus triggered a global pandemic, with over 776 million confirmed cases and more than 7 million deaths worldwide as of October 2024. 15 Coronaviruses infect humans and other vertebrates, impacting multiple organ systems, including the respiratory, cardiovascular, gastrointestinal (GI), central nervous, and genitourinary systems. 16 Research consistently indicates that men experience higher COVID-19 severity and mortality rates compared with women, emphasizing the need for focused attention from healthcare professionals specializing in men’s health. 17 A study from São Paulo highlighted that male patients, particularly those with comorbidities, tend to have more severe disease outcomes. 18 The authors declared that male sex is an independent risk factor for severe SARS-CoV-2 infection, with men showing increased susceptibility to more severe outcomes. This vulnerability is partly attributed to the higher expression of ACE2 (angiotensin-converting enzyme 2) and TMPRSS2 (transmembrane protease serine 2) in male tissues, both of which play critical roles in viral entry. The ACE2, which facilitates SARS-CoV-2 binding, is expressed at elevated levels in the testes, especially in Leydig cells, Sertoli cells, and spermatogonia.19,20 Furthermore, TMPRSS2 primes the viral spike (S) protein following ACE2 binding, a crucial step for the virus to invade host cells. This biological mechanism may contribute to the higher severity of infection observed in men. Notably, the authors also pointed out that fertile men have higher ACE2 levels than infertile individuals with severe spermatogenesis impairment, suggesting a link between ACE2 expression and male reproductive function. 18
Although ACE2 serves as a receptor for SARS-CoV-2, its primary function is within the renin-angiotensin system. Its expression is regulated by transcription factors such as Sp1, Sp3, and GATA, which bind to its gene promoter, while epigenetic modifications like DNA methylation and histone acetylation may further influence its levels.21 -23 Beyond facilitating viral entry, ACE2 converts angiotensin II into angiotensin-(1-7), a molecule with vasodilatory and anti-inflammatory properties. In the testes, this function is essential for maintaining tissue homeostasis, protecting germ cells from oxidative stress, and potentially influencing sperm quality and fertility.24,25 It is worth noting that Sertoli cells and Leydig cells contribute to this anti-inflammatory environment by modulating immune responses and secreting factors that support germ cell survival and function. 26
This study aims to evaluate the expression of SARS-CoV-2 nucleoprotein and ACE2 receptor in TGCTs diagnosed during the COVID-19 pandemic and compare these findings with germ cell tumors diagnosed before the pandemic. By analyzing the cellular distribution of SARS-CoV-2 within testicular tissue, we seek to determine whether viral presence exhibits a pattern that may be associated with tumor development or progression.
Methods
In this study, a total of 16 cases, aged between 23 and 46, who presented with a testicular mass and were pathologically diagnosed with seminoma (n = 6) or mixed germ cell tumor (MGCT; n = 10) between December 2019 and November 2023 were evaluated. It should be noted that for peer review, these 16 cases represent all the testicular tumor cases diagnosed during this period in our department. In addition, 16 testicular tumor cases, aged between 18 and 52, diagnosed between January 2014 and May 2019 from the archives of the Department of Pathology, Kırıkkale University Faculty of Medicine, were used as a control group for comparison of viral proteins and ACE2 markers. This control group also comprised all available cases during this earlier period and consisted of seminoma (n = 8) and MGCT (n = 8) cases.
Hematoxylin and eosin-stained slides from paraffin blocks were re-evaluated by 2 pathologists, and slides representing both the tumor and surrounding tissues were selected. The antibody used for SARS-CoV-2 nucleoprotein was rabbit monoclonal [EPR24334-118] (Abcam, USA), diluted at 1:75. The antibody for ACE2 was rabbit monoclonal antibody (Boster Bio, USA), diluted at 1:200. The staining procedures were carried out using the Benchmark XT Ventana machine. First, deparaffinization and rehydration steps were performed, followed by the application of an antigen retrieval solution. The standard Ventana protocol was followed for staining, and specific antibodies were used with appropriate primary and secondary antibodies. The results were visualized using DAB (3,3’-Diaminobenzidine) chromogen, and counterstaining was performed with hematoxylin for contrast. Lung tissue from known COVID-19-infected patients was used as a positive control. The SARS-CoV-2 immunostaining was subjectively evaluated using a 2-tier scale, categorizing the staining intensity as either strong or weak to moderate. 26
Histological evaluations were performed using a Nikon Eclipse Ni light microscope, and representative digital images were captured with a Nikon DS-Fi1c camera. Image acquisition and processing were conducted using NIS-Elements software (Nikon Corporation, Tokyo, Japan).
Immunohistochemistry (IHC) was preferred in this study due to its ability to provide cell-specific localization of target proteins, allowing for the differentiation of staining patterns among various cell types within the tumor microenvironment. This method enables the evaluation of both positively and negatively stained cell populations, offering insights into the heterogeneity of tumor and stromal interactions. In contrast, molecular techniques, such as polymerase chain reaction (PCR) or sequencing-based methods, analyze pooled nucleic acid content from entire tissue sections, encompassing both tumor and non-tumor components. While molecular approaches provide a comprehensive overview of genetic and epigenetic alterations driving tumor and normal tissue pathogenesis, they lack the spatial resolution necessary to assess cellular distribution and microenvironmental context. Therefore, IHC serves as a valuable tool for correlating protein expression with histopathological features, facilitating a more detailed interpretation of tumor biology at the cellular level.
Descriptive statistics were presented as mean, standard deviation, median, minimum, maximum, frequency, and percentage. Normality was assessed using the Kolmogorov-Smirnov and Shapiro-Wilk tests. The independent sample t-test was used for normally distributed quantitative data, and the Mann-Whitney U-test for non-normally distributed data. Chi-square or Fisher’s exact test was applied for qualitative data. Analyses were performed using SPSS 28.0. The reporting of this study adheres to the STrengthening the Reporting of OBservational Studies in Epidemiology (STROBE) guidelines. 27
Results
The tumor types, age distribution, tumor sizes, focality, and their distribution over the years in the 32 cases included in our study are summarized in Table 1-Figure 1. The highest frequency of tumor diagnoses was observed in 2021, accounting for 19.4% of the cases. In addition, an increase in tumor diagnoses was detected in 2022 compared with the pre-pandemic period. However, no significant correlation was found between COVID-19 infection and tumor types (P = .476). The incidence of seminoma and mixed germ cell tumors (MGCTs) showed a similar distribution across both periods (Table 2-Figure 2-3). Furthermore, no statistically significant differences were observed between the 2 periods in terms of tumor size or multifocality.
The tumor types, age distribution, tumor sizes, focality, and their distribution over the years in the 32 cases included in our study.
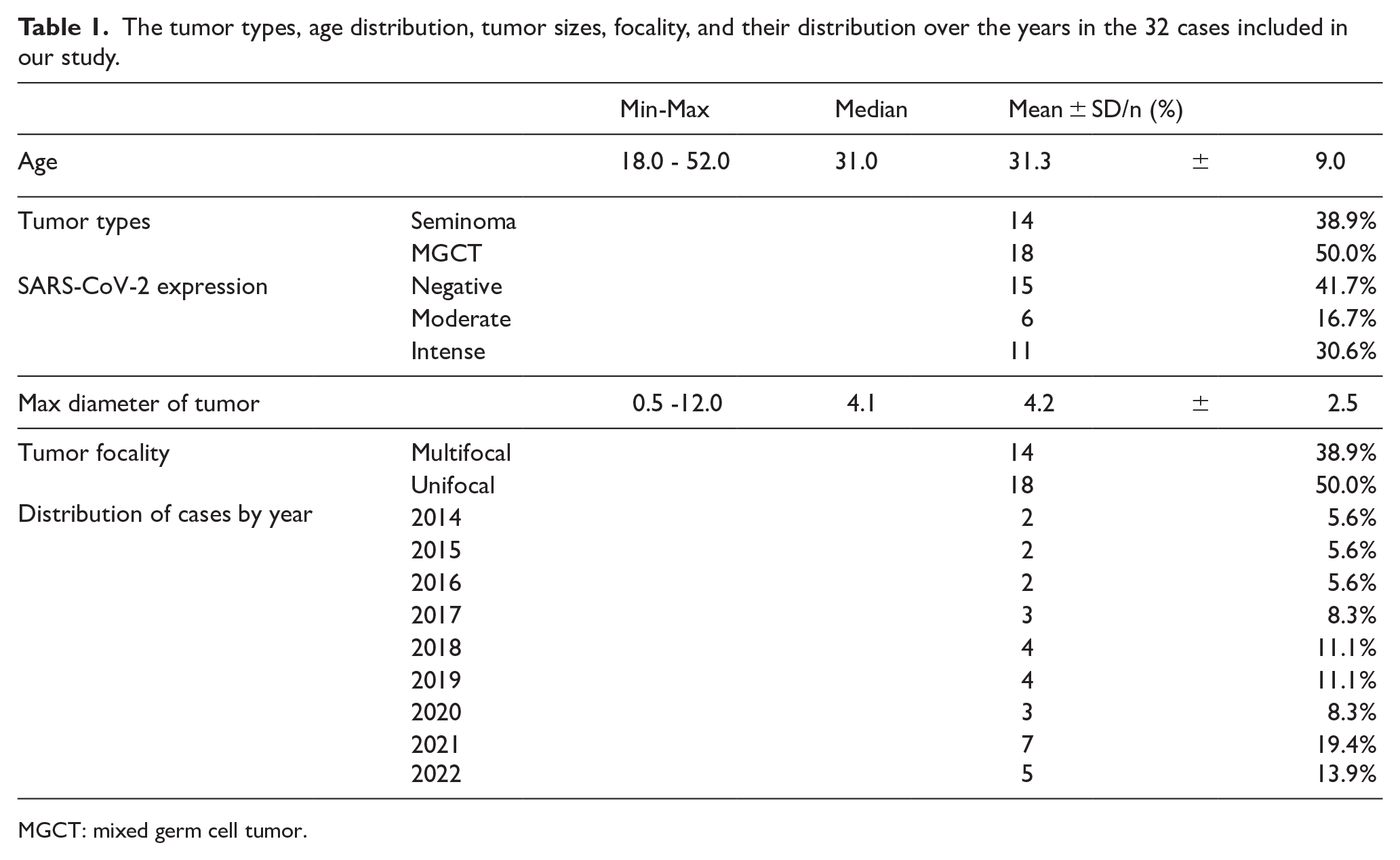
MGCT: mixed germ cell tumor.

The bar chart illustrates the annual percentage distribution of cases from 2014 to 2022. A gradual increase is observed starting in 2017, with a marked peak in 2021. The red dotted line represents the trend over time.
In the immunohistochemical analysis using the SARS-CoV-2 antibody, 15 cases were immunonegative, 11 of which were diagnosed before the pandemic, representing 68.8% of the pre-pandemic cases.

Independent sample t-test results/Mann-Whitney U-test/χ2 chi-square test (Fischer test), MGCT: mixed germ cell tumor.
Bold values indicate statistically significant differences between groups, with p < 0.05 considered significant.

(Left) The number of seminoma and mixed germ cell tumor (MGHT) cases is shown for the periods before and after SARS-CoV-2 infection. An increase in MGHT cases is observed following the pandemic. (Right) The mean age (± standard deviation) of patients was similar before and after SARS-CoV-2 infection, with no significant difference.
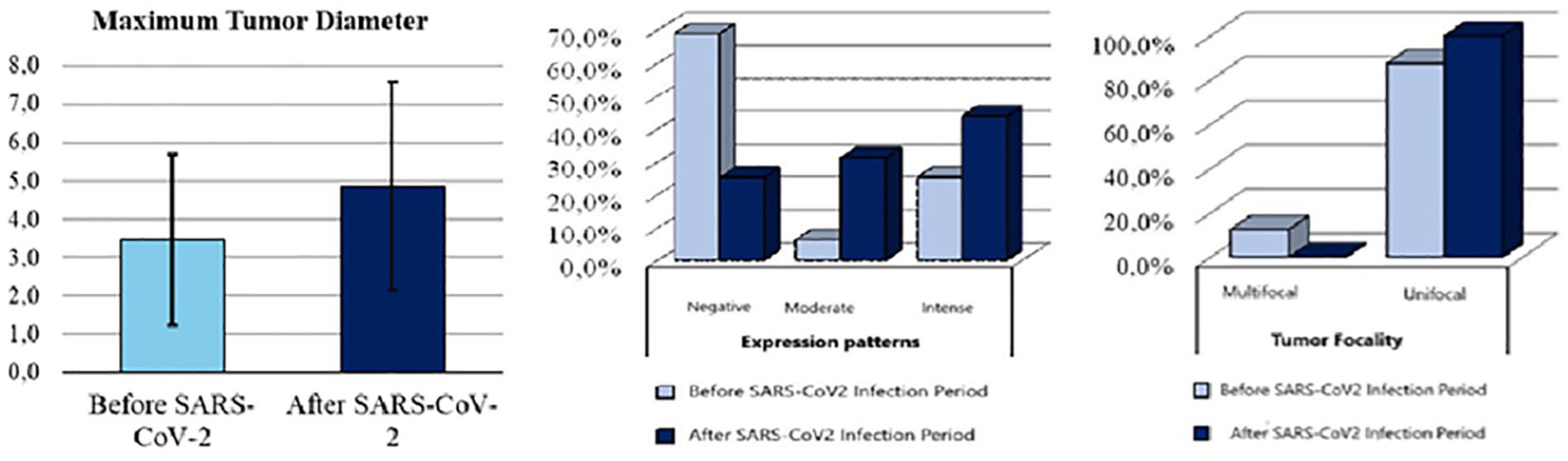
(Left) ACE expression patterns demonstrate a shift toward increased intense staining after SARS-CoV-2 infection, while negative expression became less frequent. (Middle) The mean maximum tumor diameter (± SD) was greater in the post-infection group, suggesting a trend toward larger tumor size. (Right) Tumor focality analysis shows that the majority of tumors were unifocal in both groups, with a slight increase observed after SARS-CoV-2 infection. Multifocal tumors remained rare overall.
In the immunohistochemical analysis using the SARS-CoV-2 antibody, 15 cases were immunonegative, 11 of which were diagnosed before the pandemic, representing 68.8% of the pre-pandemic cases (Tables 1-Figure 1 and Table 2-Figure 2-3). Strong positivity for the SARS-CoV-2 nucleoprotein was observed in 11 cases, primarily in Leydig cells, with some positivity also detected in Sertoli and plasma cells (Figure 4). Among these cases, 4 were diagnosed before the pandemic (2015 and 2019), while the remaining 7 were diagnosed during the pandemic. The difference in SARS-CoV-2 expression between the pre-pandemic and pandemic periods was statistically significant (P = .013). Cases showing weak-to-moderate staining for SARS-CoV-2 are presented in Figure 5. Importantly, no tumor tissue exhibited SARS-CoV-2 nucleoprotein expression.

Cases showing strong immunopositivity for SARS-CoV-2. (A) Granular cytoplasmic staining in Leydig cells (×40) (green arrows). (B) Basolateral/apical staining in Sertoli cells (×20) (black arrows). (C) Strong cytoplasmic staining observed in plasma cells surrounding the tumor (×20) (red arrows).
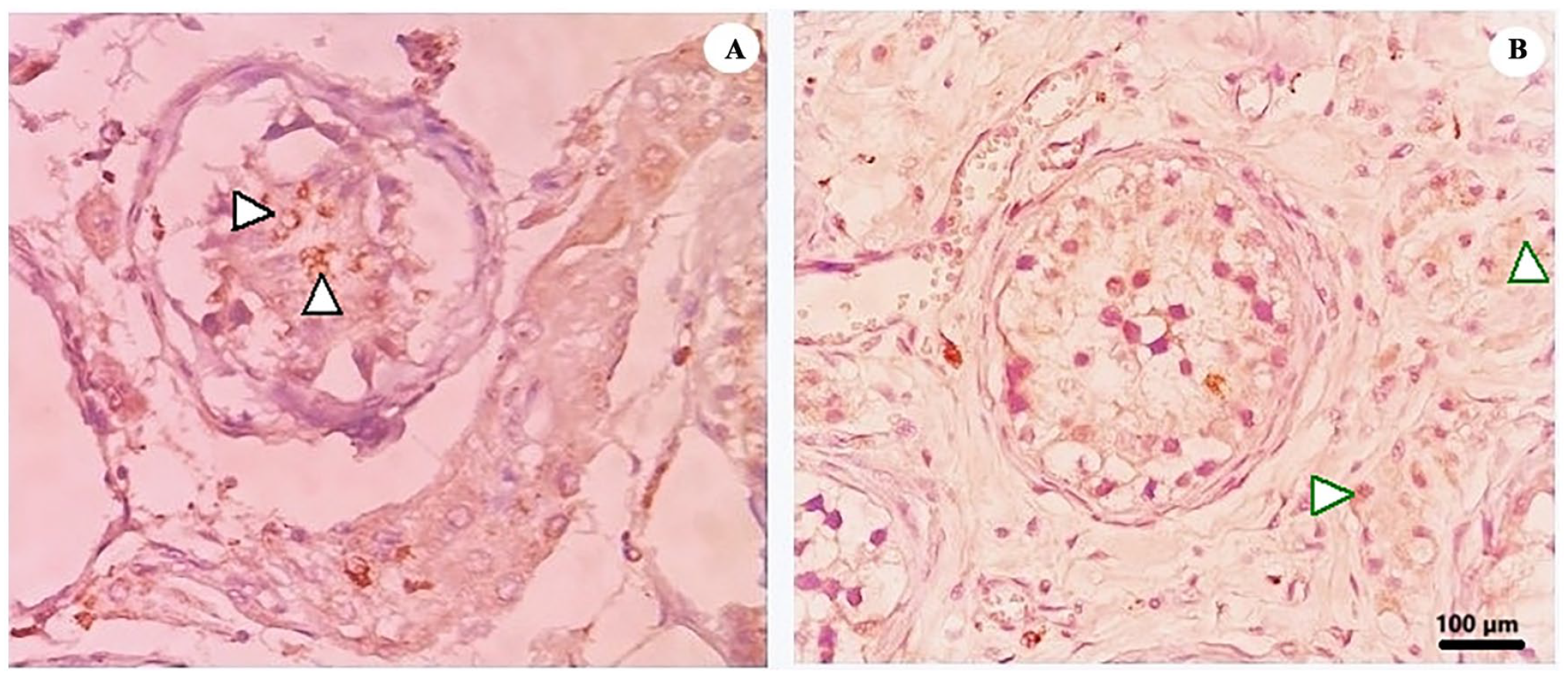
Cases showing less widespread and weak-to-moderate staining for SARS-CoV-2. (A) Weak-to-moderate basolateral staining in Sertoli cells (×20) (black arrows). (B) Granular staining observed in Leydig cells and occasional stromal cells (×20) (green arrows).
Immunohistochemical analysis of the ACE2 antigen revealed strong expression in all cases, predominantly in Leydig and Sertoli cells within the tumor microenvironment. However, this finding did not reach statistical significance for further analysis (Figure 6).

Cases showing ACE2 staining across all groups. (A) Strong and diffuse staining in Leydig cells (×20) (green arrows). (B) Granular strong cytoplasmic staining in a seminoma case (×20). (C) Strong and diffuse cytoplasmic staining in a mixed germ cell tumor case (×20).
Discussion
In our study, a significant increase in the prevalence of TGCTs was observed during the 36-month period starting from December 2019, the onset of the COVID-19 pandemic, compared with the total tumor cases archived in our hospital. Our findings strongly indicate a potential link between SARS-CoV-2 and the increased incidence of TGCTs, suggesting that the virus may influence tumorigenesis beyond its well-documented effects on testicular damage and infertility.
The detection of viral nucleoprotein positivity in cases dating back to 2018 raises the possibility of a subclinical presence of SARS-CoV-2 or a related coronavirus strain before the pandemic was officially declared. Interestingly, strong immunohistochemical expression was also noted in a single archival case from 2015. While the presence of strong immunoreactivity in this case is intriguing, it remains unclear whether this reflects cross-reactivity with other viral antigens or an early, undocumented exposure to a coronavirus-related strain. This observation is particularly notable given that 2015 coincided with an avian influenza outbreak (H5N1) in India, which was reported in our region. Previous studies, such as those by Bai et al, have shown that infections with avian influenza viruses, particularly H7N9, can induce orchitis and reduce testosterone levels, emphasizing the broader impact of viral pathogens on male reproductive health. 29
In addition, recent research by Lucio Carrasco et al 25 has indicated that the SARS-CoV-2 Nucleocapsid (N) protein may contribute to testicular hormonal imbalance by disrupting Leydig cell function, leading to a significant reduction in testosterone levels. Given our observation of strong immunoreactivity in both Leydig and Sertoli cells, which suggests a direct impact of SARS-CoV-2 on testicular endocrine function, we hypothesize that this viral infection could contribute not only to testicular hormonal imbalance but also to the development of intratesticular germ cell neoplasia (ITGCN). In contrast to some findings in the literature, no immunoreactivity was observed in gonocytes, suggesting that the impact of SARS-CoV-2 may be specific to the endocrine (Leydig) and supporting (Sertoli) cells of the testis, rather than directly affecting the germ cells. This is consistent with the findings of a recent study, which also identified SARS-CoV-2 infection in ACE2-positive Leydig and Sertoli cells in an ex vivo human testis model. They reported a decrease in steroidogenic enzymes over time, suggesting a potential subtle disruption of Leydig cell function. 29 Furthermore, the findings from the autopsy study by Duarte-Neto et al support our observations, as they also detected viral particles in Sertoli and Leydig cells. In addition, they reported histopathological changes such as thickened basal membranes and reduced spermatogenesis in the testicular tissue of deceased COVID-19 patients. This study highlights the direct damage caused by SARS-CoV-2 to testicular cells, with electron microscopy revealing viral particles within the cytoplasm of fibroblasts, endothelial cells, Sertoli cells, Leydig cells, spermatids, and epithelial cells of the rete testis. 30
However, we did not include histopathological findings in our study because our tumor-bearing testis samples already exhibited histopathological features consistent with hypogonadism, such as thickened tubular basal membranes and only Sertoli-cell-like changes. These findings are characteristic of tumor predisposition and are not unique to SARS-CoV-2 infection, but we might consider that SARS-CoV-2 could contribute to the hypogonadal environment required for tumor development. This impaired testicular development means that some early PGCs or gonocytes are blocked in their process of differentiation, and as such, these germ cells retain their early (embryonic) marker profile. The reason for this development block is not yet entirely clear, but it is likely a combination of microenvironmental factors and epigenetic defects. Sertoli cells, through their interactions with the KIT/KITLG pathway, are key regulators of gonocyte survival, migration, and differentiation.6,7 Alterations in this pathway, potentially influenced by viral infection, could further compromise proper germ cell maturation. Since low testosterone levels are known to affect normal germ cell differentiation, SARS-CoV-2-induced dysfunction of Leydig cells may create an environment conducive to the initiation or progression of intratubular germ cell neoplasia (ITGCN), a condition characterized by abnormal germ cells residing within the seminiferous tubules, with the potential to develop into invasive TGCTs. 31
A similar trend has been observed in prostate cancer (PCa) during the pandemic, with an increased detection rate of PCa and a higher prevalence of cases with elevated Gleason scores (GS 7 and 9-10). 32 The expression of SARS-CoV-2 entry molecules, such as TMPRSS2, in the prostate suggests a potential viral tropism for prostatic glandular cells. The SARS-CoV-2 infection induces chronic inflammation, which has been associated with DNA methylation changes and cancer progression—factors implicated in around 60% of PCa cases. Furthermore, SARS-CoV-2 exacerbates the upregulation of TMPRSS2 expression, potentially enhancing TMPRSS2 fusions and contributing to PCa development. Although the virus does not directly encode known oncoproteins, its role in modulating TMPRSS2 expression and maintaining chronic inflammation may influence prostate carcinogenesis. 33
In light of our findings, and considering the disturbances in testicular homeostasis, as well as the potential role of SARS-CoV-2 in fostering a hypogonadal milieu, an important question arises: does this viral infection directly induce de novo tumor formation, or does it simply act as a catalyst that accelerates the progression of pre-existing lesions? Recent studies have highlighted a bidirectional relationship between cancer and COVID-19. Cancer patients are more vulnerable to severe COVID-19 outcomes due to their immunocompromised state, older age, and increased expression of key viral entry molecules such as ACE2 and TMPRSS2. 34 This susceptibility is particularly evident in patients with lung cancer (including non–small-cell lung cancer, NSCLC), GI cancers (pancreatic, colorectal, and upper GI), and gynecological cancers.35 -37 Tumors in these patients often exhibit more aggressive behavior, potentially driven by heightened inflammatory responses, cytokine storms, and virus-induced epigenetic alterations. In addition, the pro-inflammatory state triggered by SARS-CoV-2 may serve as a trophic stimulus, accelerating tumor progression or reactivating dormant cancer cells.
Our findings suggest that ACE2 expression in testicular tissue is restricted to Sertoli and Leydig cells, with no detectable expression in spermatogonia. This observation led us to conclude that early-stage germ cells are not primary targets for SARS-CoV-2. Consistent with our observation, recent studies have highlighted that while SARS-CoV-2 is rarely found in seminal fluid, viral particles have been detected within mature spermatozoa up to 90 days post-infection.38,39 Given that spermatozoa are transcriptionally inactive, this raises the possibility that viral persistence is mediated through alternative mechanisms. One potential explanation is the involvement of DNA-based extracellular traps (ETs), which have been implicated in systemic inflammatory responses to COVID-19. These traps may serve as a defense mechanism by capturing viral particles within the spermatozoa, rather than facilitating active replication. 39 This phenomenon could explain why SARS-CoV-2 is absent in spermatogonia yet detected in mature sperm, reinforcing the idea that testicular infection is not driven by direct viral invasion of germ cells but rather by immune-mediated mechanisms within the reproductive tract.
However, some studies have shown that ACE2 is expressed not only in the myoid, Sertoli, and Leydig cells, which are involved in nursing and steroidogenesis, respectively, but also in spermatogonia, suggesting that these cell types could be highly susceptible to SARS-CoV-2. 20 In addition, TMPRSS2 is shown to be expressed in Sertoli cells, spermatogonia, spermatocytes, and spermatid cells. 40 In contrast, our study observed ACE2 expression in cells other than germ cells using immunohistochemical methods. Notably, ACE2 was upregulated in both seminoma and MGCTs, independent of whether these tumors occurred during the pandemic period.
This overexpression aligns with bioinformatics data indicating aberrant ACE2 mRNA expression in various malignancies, including colon, kidney, pancreatic, rectal, stomach, and lung adenocarcinomas, likely due to epigenetic modifications such as hypo-DNA methylation. 41
The upregulation of ACE2 in germ cell tumors may represent an adaptive response to the tumor microenvironment. Common features of malignancies, such as hypoxia, inflammation, and oxidative stress, could drive ACE2 expression, potentially aiding tumor cell survival through its immunomodulatory functions. Emerging evidence indicates that SARS-CoV-2 and tumor cells exploit overlapping molecular pathways, including immune suppression, oxidative stress, disruption of DNA damage response (DDR) signaling, activation of stemness pathways, and suppression of tumor suppressor proteins. 42
The SARS-CoV-2 infection of Sertoli cells may play a pivotal role in the development of germ cell tumors by disrupting the testicular microenvironment. Sertoli cells, essential for supporting and nurturing germ cells, could be directly impacted by viral invasion. This disruption might impair the normal maturation process of germ cells, preventing them from properly differentiating. In addition, Leydig cells, responsible for testosterone production, could also be infected, leading to a hypogonadal environment. The resulting testosterone deficiency could disrupt spermatogenesis, further promoting tumorigenesis.
In this altered microenvironment, infected plasma cells, along with other immune cells, would likely exacerbate the inflammatory response. This inflammatory and hypoxic condition within the testicular tissue could create a pro-tumorigenic milieu that promotes tumor initiation or progression. Chronic inflammation and hypoxia are known to foster a cellular environment conducive to malignancy, where abnormal cells can thrive and proliferate. Furthermore, in the presence of pre-existing genetic predispositions or mutations, this inflammatory state may facilitate the growth of already abnormal cells, thus accelerating tumor progression. Therefore, SARS-CoV-2 infection may not only contribute to the initiation of germ cell tumors but also facilitate their growth and expansion by modulating both the hormonal and immune landscapes of the testicular environment.
While the retrospective design of this study and the absence of long-term follow-up limit our ability to establish definitive causal relationships between SARS-CoV-2 and tumor development, the findings offer a strong foundation for future research. The fluctuating severity of the pandemic, intermittent lockdowns, and disruptions to routine healthcare services further complicated consistent data collection and systematic follow-up.
To substantiate the immunohistochemical observations presented here, upcoming studies should consider incorporating cell isolation techniques and PCR-based viral RNA detection to differentiate infected from uninfected cell populations within the tumor microenvironment. These confirmatory approaches could help identify virus-induced oncogenic signaling pathways, advancing our understanding of how viruses like SARS-CoV-2, through specific cellular targets such as Sertoli and Leydig cells, may contribute to tumorigenesis. Ultimately, the insights gained from this study, along with future research, could help guide the development of targeted interventions not only for SARS-CoV-2–associated TGCT but also for other viruses with similar tropism.
Conclusions
This study reveals a possible association between SARS-CoV-2 and testicular tumorigenesis, as suggested by an increased frequency of germ cell tumors diagnosed during the pandemic period. Although this link is not yet as well-established as that between HPV and cervical cancer, the known tropism of SARS-CoV-2 for the male genital tract highlights the need for further scientific investigation.
While SARS-CoV-2 does not appear to directly transform germ cells, it may contribute to tumorigenesis by altering the testicular immune environment and inducing a hypogonadal state. This effect likely results from viral infection of both Sertoli and Leydig cells, disrupting their physiological roles and impairing germ cell maturation, thereby fostering a tumor-permissive microenvironment.
The ACE2 expression was absent in spermatogonia but observed in tumor germ cells from both pandemic and pre-pandemic periods, as well as in Sertoli and Leydig cells. This distribution suggests a potential role for ACE2 in facilitating viral entry and modulating inflammatory and proliferative responses within the testicular tumor microenvironment.
Taken together, these findings underscore the importance of exploring the molecular and cellular mechanisms of virus-associated tumorigenesis, which may inform future strategies for early detection and targeted treatment of virus-related testicular pathology.
Footnotes
Acknowledgements
Not applicable.
Author contributions
Mahi Balcı: Made a substantial contribution to the concept or design of the work; Acquisition; Analysis or interpretation of data; Paper design; Immunohistochemical evaluation; Writing paper.
Merva Aydemir Akkaya: Drafted the article or revised it critically for important intellectual content; Immunohistochemical evaluation; Writing paper.
Each author approved the version to be published and has participated sufficiently in the work to take public responsibility for appropriate portions of the content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Kırıkkale University Research Project Unit (The Project Number: 2023/073).
Ethics statement
The study was approved by the Kırıkkale University, Non-Interventional Research Ethics Committee (Protocol #2023-073) and has been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments. The requirement for written informed consent was waived as the study was retrospective, all data was deidentified, and the research contained no more than minimal risk to the participants or their privacy.
Consent for publication
Not applicable.
Availability of data and materials
The data supporting the findings of the study are available from the corresponding author upon reasonable request.
