Abstract
Background:
Intravenous vitamin C (IVC, ascorbate [Asc]) and alpha-lipoic acid (ALA) are frequently coadministered in integrative oncology clinics, with limited understanding of combination effects or drug-drug interactions. As high-dose IVC has anticancer activity through peroxide (H2O2), it is hypothesized that IV ALA, a thiol antioxidant, might have untoward effects when combined with IVC.
Methods:
In vitro combination index (CI) was investigated in 6 types of human cancer cells, using clinically relevant concentrations of Asc (0.625-20 mM) and ALA (0.25, 0.5, and 1 mM) evaluated by nonconstant ratio metrics. Cellular H2O2 was measured using HeLa cells expressing a fluorescent probe HyPer. Mouse xenografts of the metastatic breast cancer MDA-MB-231 were treated with intraperitoneal injections of ALA (10, 20, and 50 mg/kg) and Asc (0.2, 0.5, and 4 g/kg) at various dose levels.
Results:
Cancer cell lines were sensitive to Asc treatment but not to ALA. There is no evidence ALA becomes a prooxidant at higher doses. The CIs showed a mixture of synergistic and antagonistic effects with different ALA and Asc combination ratios, with a “U” shape response to Asc concentrations. The ALA concentrations did not influence the CIs or cellular H2O2 formation. Adding ALA to Asc dampened the increase of H2O2. Toxicity was observed in mice receiving prolonged treatment of ALA at all doses. The Asc at all doses was nontoxic. The combination of ALA and Asc increased toxicity. The ALA at all doses did not inhibit tumor growth. The Asc at 4 g/kg inhibited tumor growth. Adding ALA 50 mg/kg to Asc 4 g/kg did not enhance the effect, but lower doses of ALA (10 or 20 mg/kg) dampened the inhibitory effect of Asc.
Conclusions:
These data do not support the concurrent or relative concurrent use of high-dose intravenous ALA with prooxidative high-dose IVC in clinical oncology care with potentially increased toxicity.
Keywords
Introduction
Integrative medicine physicians who practice oncology employ a wide array of therapeutics, including conventional therapies and natural products.1-5 Among these therapeutic choices, intravenous therapies frequently administered in integrative oncology clinics are ascorbic acid (vitamin C), glutathione (GSH), and alpha-lipoic acid (ALA).
Intravenous ascorbate (Asc) or vitamin C (IVC) has been studied extensively for decades.6-14 It is considered a pharmacologic agent when intravenously administering ascorbic acid.15-18 Blood levels of orally administered vitamin C are tightly controlled by gastrointestinal tract absorption, whereas IVC bypasses this tight control, resulting in markedly higher systemic concentrations. 15 Compared with oral vitamin C, IVC is a prooxidative pharmacologic agent.13,17,18 It is only high-dose IVC that generates hydrogen peroxide.
A recent pharmacokinetic study confirms safety even at doses of up to 100 gr/infusion in healthy and oncology participants. 19 These higher concentrations produce well-documented effects not seen with oral administration, specifically forming ascorbyl radical and hydrogen peroxide (H2O2), making IVC a prooxidative therapy.13,18,20 In preclinical cancer models and oncology clinical trials, IVC results in cancer cell inhibition, death, and prolonged patient survival.7,9,11-14,20,21
Alpha-lipoic acid was first isolated from liver specimens in 1951 by biochemist Lester Reed, PhD. 22 The ALA is a medium-chain fatty acid containing 2 sulfhydryl groups. It is produced endogenously, with additional systemic levels dependent on dietary intake. The ALA exists in a redox pair, with the reduced form as dihydrolipoic acid and the oxidized form as ALA. Isomer forms include R-ALA and S-ALA, with the R form representing the natural form that benefits human systems.23,24
The ALA is a well-known antioxidant that is fat-soluble and water-soluble.25-29 It is recognized as a critical antioxidant required to recycle other antioxidants systemically through its reducing capacity, including vitamin C and, indirectly, vitamin E. The ALA is crucial for enriching the thiol pool and regenerating intracellular GSH and all other thiol-containing compounds.26,28,30,31 The concern is that adding a thiol compound like ALA regenerates GSH and other thiol-containing antioxidants that have the potential to react with any oxidizing agent like IVC in the system.26,31,32
As an essential antioxidant, ALA is critical to mitochondrial respiration. 28 The ALA is also an important cofactor for increasing the enzyme pyruvate dehydrogenase, which reduces pyruvate dehydrogenase kinase. 33 These are important for ALA’s reported antineoplastic actions. 26
The literature describes many important oral and intravenous ALA health benefits in chronic disorders. 34 Here, we focus only on its antineoplastic properties. Preclinical studies have demonstrated apoptosis induction35-37 and multiple other mechanisms for ALA’s antineoplastic properties, including regulating mitochondria respiration,35,38 inducing endoplasmic reticulum (ER) stress, 39 inhibiting nuclear factor-kappa B (NF-kB), 40 inhibiting transforming growth factor beta (TGF-β) signaling and epithelial to mesenchymal transition, 35 and epigenetic modification. 41 It is hypothesized that ALA induces tumor reduction and dormancy in the clinical setting rather than a cure for the disease. 38
When ALA is administered intravenously as an antineoplastic agent, it is usually approximately 600 mg of ALA in 100 mL carrier fluid, generally normal saline. The solution is infused more than 45 minutes and given 2 to 3 times per week. 38 The Food and Drug Administration (FDA) reports no significant adverse effects associated with its use, but pharmaceutically prepared products are unavailable. 42 In the United States, at the time of this report, injectable ALA is obtained through compounding pharmacies.
A limited pharmacokinetic study in healthy adults using intravenous ALA was reported in 1998. 43 The ALA 200 mg was mixed in 150 mL of normal saline and infused more than 20 minutes. No untoward adverse events were identified. The T ½ was 25 minutes and not statistically different compared with oral dosing. Most pharmacokinetic studies have been completed using oral ALA.29,43 There is a concern about toxicity with high-dose ALA, but only when the dosing is many-fold over those used in clinical practice. 44 A primate study used 90 to 100 mg/kg, far above the usual 10 to 20 mg/kg human doses. There was acute liver necrosis and death in half of the primates, making the LD50 of injected ALA at 90 to 100 mg/kg. 45 On histology, the mitochondria were significantly edematous with marked architecture disruption. Large muscles, liver, heart, and kidneys showed necrotic foci.
As described above, ALA’s neoplastic effects are related to mitochondrial respiration and metabolic stress pathways. This contrasts with IVC, which appears to function as a prooxidative therapy by forming hydrogen peroxide and specifically targets neoplastic cells with abnormal catalase metabolism. The IVC leaves normal cells with normal metabolic systems intact. 17
Oral and intravenous therapies are often given simultaneously or in tandem with a limited understanding of combination effects or drug-drug interactions. It has been shown previously in preclinical studies that administering IVC and intravenous glutathione (IVGSH) in tandem to mimic the real-world application has untoward effects on the antineoplastic actions of IVC by reducing IVC prooxidative effects. 32
Based on prior findings where IVGSH inhibited the prooxidative effects of IVC, we hypothesized that intravenous alpha-lipoic acid (IVALA), a similar thiol antioxidant, might also show similar untoward effects when combined with IVC. Thus, it was proposed that the combination effects of parenteral Asc and ALA be investigated using cellular and mouse cancer models. It was also proposed to determine whether increasing doses of ALA becomes a prooxidant similar to IVC chemistry, which has not yet been described.
Materials and Methods
Cell culture, in vitro combination treatments, and cell viability assays
Human cancer cell lines HCT116 (colon cancer), MDA-MB231 (metastatic breast cancer), Ovcar5 (ovarian cancer), PANC-1 (pancreatic cancer), PC3 (prostate cancer), and SK-Mel2 (metastatic melanoma) were obtained from American Type Culture Collection (ATCC) and maintained in the laboratory. Cells were cultured in recommended media with 10% fetal bovine serum. Cells were seeded into 96-well culture plates at 5000 cells/well and cultured overnight, and then exposed to serial concentrations of Asc (0.625-20 mM), ALA (0.25, 0.5, and 1 mM), and the combinations of Asc and ALA. Control cells were treated with the vehicle (1% dimethyl sulfoxide [DMSO]). The combination treatments used a non-constant ratio design in a 96-well-plate matrix.46,47 Each horizontal row of the 96-well plate contains serially diluted ALA concentrations (0. 0.25, 0.5, and 1 mM) with 3 repeats for each concentration, and the vertical lines of the plate were serially diluted Asc concentrations (0, 0.625, 1.25, 2.5, 5, 10 and 20 mM). The layout of the plate is shown in Supplementary Figure 1. The MTT assays were used to determine cell viability after 24 hours of treatment. Based on the Chou-Talalay method, the dose-response curve, IC50 values, and combination indices (CIs) were calculated using CompuSyn software. The CIs were used to assess drug synergism, additive effect, or antagonism. 48
Assessment of H2O2 in HeLa-HyPer-Cyto cells
HyPer is a genetically encoded fluorescent sensor capable of specifically detecting intracellular hydrogen peroxide (H2O2).49,50 HeLa-HyPer-Cyto cells express the HyPer protein in the cell cytoplasm. Cells were seeded into a black-wall 96-well culture plate at 5000 cells/well and cultured to 80% confluence and then were exposed to Asc (0.625-20 mM), ALA (0.25-1 mM), or the combination of Asc and ALA. Fluorescence was detected at 0, 2, 4, 8, 21, 22, 23, and 24 hours of treatment and was normalized to the untreated cells. Imaging for green fluorescence (500/528 nm) was performed with an Olympus IX71 fluorescence microscope and DP71 camera (Olympus, Center Valley, Pennsylvania). Fluorescence intensity was detected with a Synergy 2 fluorescence and luminescence plate reader (BioTek, Winooski, Vermont) at 488 ± 10 nm excitation and 525 ± 10 nm emission. The changes in HyPer fluorescence intensity were normalized to that of untreated control cells at time 0.
Mouse tumor model, dose-finding, and treatment procedures
All animal experiments followed a protocol (#2021-2064) approved by the Institutional Animal Care and Use Committee at the University of Kansas Medical Center, USA. Female athymic nu/nu mice were purchased from the Jackson Laboratory (Bar Harbor, Maine). The MDA-MB-231 cells were injected subcutaneously into athymic nu/nu mice at 106 cells/injection. Tumors were allowed to form for 10 days to be palpable. Then, mice were grouped to have an average equal tumor burden, and treatment commenced. All treatments were administered by intraperitoneal (IP) injections. The ALA was administered in the combination groups approximately 30 minutes before the Asc injection.
Pilot dose-finding experiments were performed in either tumor-bearing or tumor-free nu/nu mice using ALA alone at 50, 30, or 20 mg/kg or Asc alone at 0.2, 0.5, or Asc 4 g/kg. The combination treatments used ALA 50 mg/kg plus Asc 0.2 g/kg, Asc 0.5 gm/kg, or Asc 4 gm/kg, as well as ALA 30 mg/kg plus Asc 4 g/kg, and ALA 20 mg/kg plus Asc 4 g/kg.
The ALA doses were hypothesized from extrapolation of the PK ALA data 43 that parenteral dosing with 50 mg/kg should yield ~0.80 mM plasma Cmax, a 30 mg/kg dose should yield ~0.50 mM, a 20 mg/kg dose should yield ~0.30 mM, and 10 mg/kg dose should yield ~0.10 mM plasma Cmax. Research-grade (Sigma-Aldrich St. Louis, Missouri; Cat#T1395) and pharmaceutical-grade ALA (Vertisis Custom Pharmacy, Scottsdale, Arizona) were used. Research-grade ALA was prepared in 45% polyethylene glycol (Sigma-Aldrich, Cat#202398) in 55% sterile water. The control group was injected with the vehicle.
The Asc doses were based on published PK data18,20,32 that a 0.2 g/kg IP dose will give a Cmax of ~1.2 mM, a 0.5 g/kg dose will give a Cmax of ~3 mM, and high dose of 4 g/kg for Cmax of ~ 24 mM. Asc (Sigma-Aldrich, Cat#A92902) was dissolved in water as a 1 M stock solution with pH adjusted to 7 using NaOH. The Asc was stored at −80°C. A single vial was thawed for each use.
Doses used in tumor treatment were determined based on the toxicities observed. Tumor-bearing mice were divided into 8 to 10 mice per group with equal tumor burden and treated with the pharmaceutical grade of ALA at decided doses, Asc at the decided doses, and the combinations of ALA and Asc. Mice were monitored daily for signs of toxicity, such as recumbency, abnormal appearance (hunched, etc), restlessness, reluctance to move and mutilation, labored breathing, hemorrhage, flaccid or spastic paralysis, circling, convulsion or blindness, or inability to ambulate. Body weight was recorded during the treatment. At the end of the study, mice were euthanized by CO2 asphyxiation followed by immediate bilateral thoracotomy, and tumor weight was recorded.
Statistical Analysis
Cellular fluorescence and tumor weight among groups were compared by 1-way analysis of variance (ANOVA) with post-hoc Tukey honestly significant difference (HSD) tests. Pairing t-tests were used for single-point comparisons between groups. All statistical analysis was 2-sided and assessed for significance at the 5% level.
Results
Sensitivity of cancer cells to alpha-lipoic acid, ascorbate, and the combination of alpha-lipoic acid + ascorbate
The in vitro effects were evaluated using a panel of 6 different types of human cancer cell lines, including HCT116: colon cancer, MDA-MB231: metastatic breast cancer, Ovcar5: ovarian cancer, PANC-1: pancreatic cancer, PC3: prostate cancer, and SK-Mel2: metastatic melanoma. Clinically relevant concentrations of Asc (0-20 mM) were used. High concentrations of ALA (0-1 mM) were also used, which covered the concentration range or were higher than the estimated Cmax to be achieved by likely high doses of IV in humans.
All 6 tested cancer cell lines were sensitive to Asc treatment at concentrations below 5 mM (Figure 1A), a clinically relevant concentration achievable through intravenous administration. These data are consistent with published studies. All 6 tested cancer cell lines were insensitive to ALA treatment or only marginally affected, as 1 mM ALA did not reach 50% inhibition of cell viability in all tested cell lines (Figure 1B). The estimated IC50 values for each single-drug treatment are listed in Table 1.
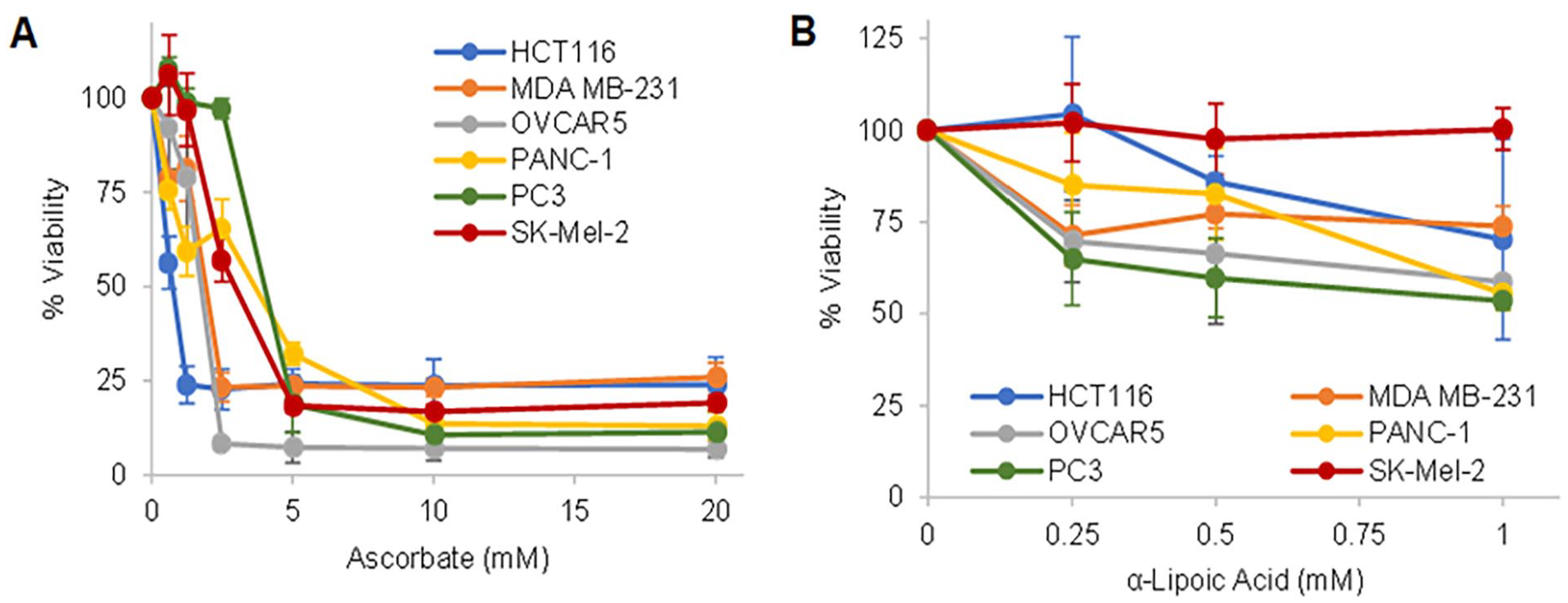
Dose-response curves of the 6 tested cancer cell lines to ascorbate (A) or alpha-lipoic acid (B) treatment. Cells were treated for 24 hours, and cell viability was assessed by MTT assay and compared with untreated. Data represent mean ± SD of 2 to 3 independent experiments, each done in triplicates.
IC50 values of ascorbate (Asc) and alpha-lipoic acid (ALA) in cancer cell lines.

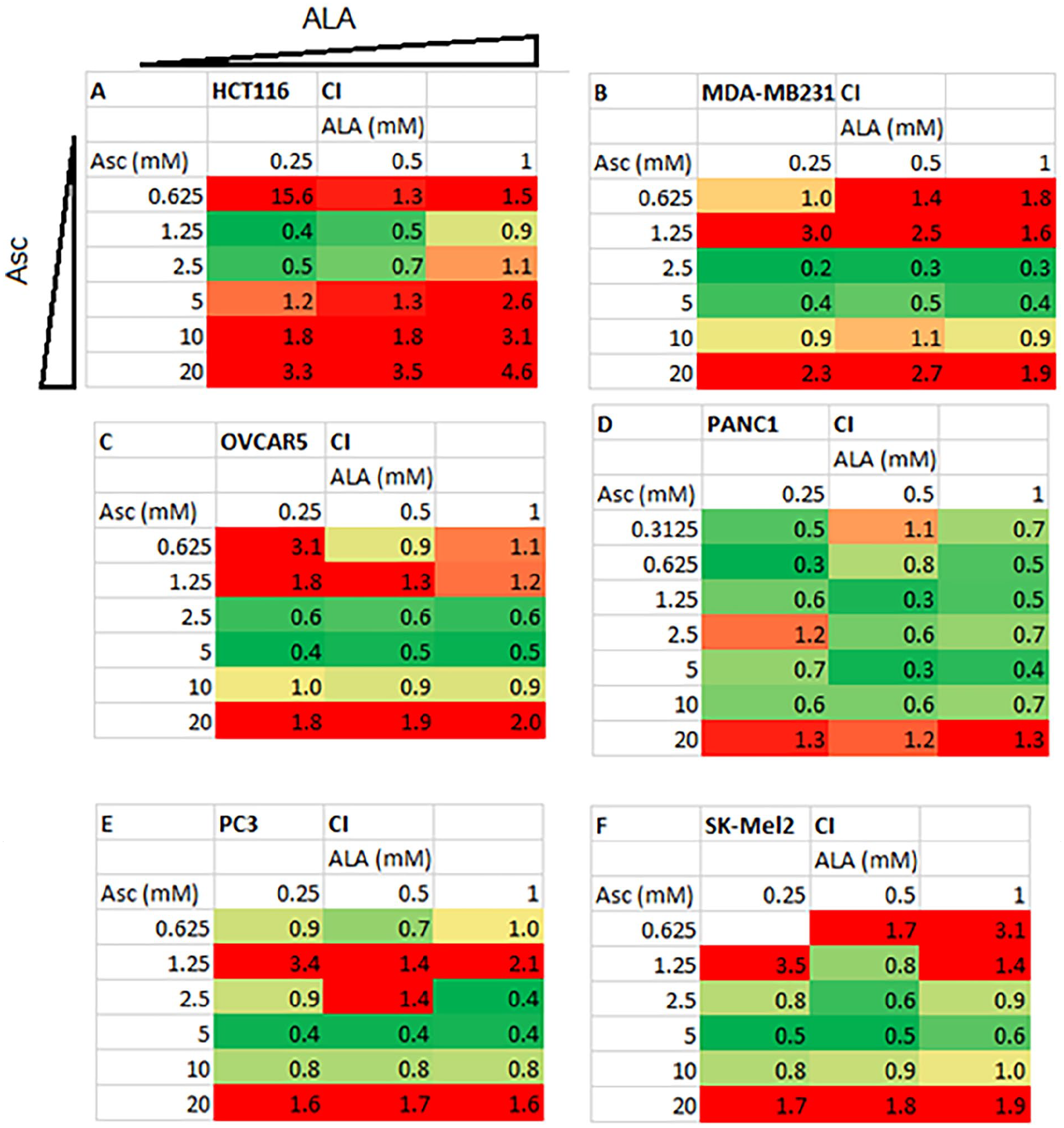
The combination treatment used a non-constant ratio design in a 96-well-plate matrix, with 6 Asc concentrations from 0.625 to 20 mM and 3 ALA concentrations from 0.25 to 1 mM. The combination indexes (CIs) were determined using the Chuo-Talalay method (Table 2A-F). 48 CI < 1 indicates the synergistic effect, CI = 1 indicates the additive effects, and CI > 1 indicates the antagonistic effects.
Combination indexes (CI) of ALA and Asc in inhibiting cancer cell viability.

The tested cells were treated with combinations of ALA and Asc in a 96-well plate for 24 hours. The combinations used 6 concentrations of Asc (0.625-20 mM) and 3 concentrations of ALA (0.25-1 mM). The layout of the drug concentrations in the combination treatment is shown in Supplementary Figure 1. Cell viability was detected using MTT assays, and percentages of viability vs untreated control were used to calculate CIs using CompuSyn (see section “Materials and Methods”). A-F each demonstrates the CIs in the 6 tested cancer cell lines. The CIs are color-coded with CI < 1 indicates the synergistic effect (green), CI = 1 indicates the additive effects (yellow), and CI > 1 indicates the antagonistic effects (red).
Because the CI values showed a mixture of synergistic to antagonistic effects with different combination ratios of ALA and Asc, we then studied the CI values related to Asc concentration, considering all the ALA concentrations used with that specific Asc concentration for all tested cell lines. The results are shown in Figure 2A. Combined with ALA, the results suggested a “U” shape response in the CIs to Asc concentration. Synergistic or additive effects were mainly achieved within the range of Asc concentrations from 2.5 to 10 mM, which are clinically relevant levels. However, beyond this range, either higher or lower concentrations of Asc showed antagonistic effects with ALA. On the contrary, the CIs were not affected by different concentrations of ALA used (Figure 2B).
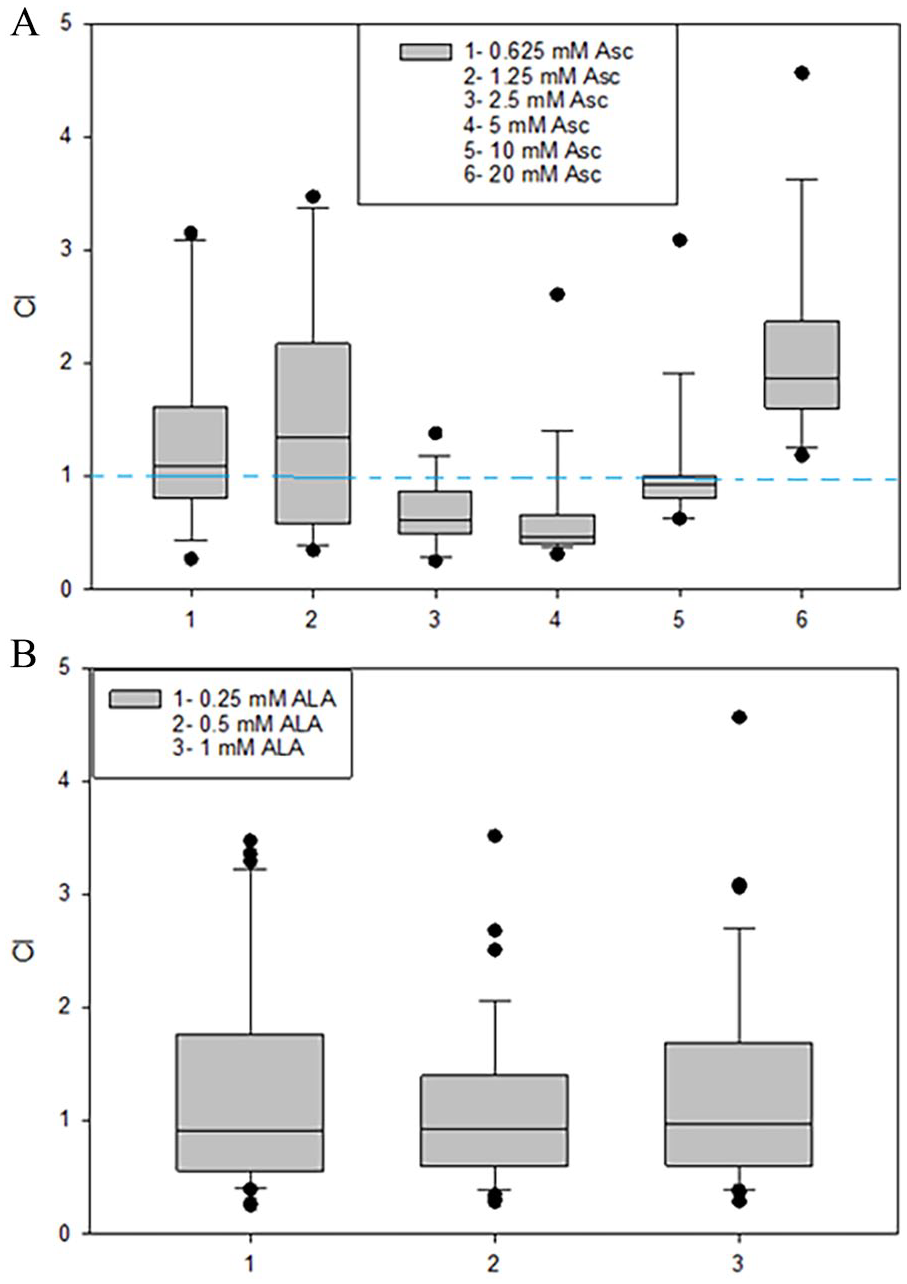
CI values according to Asc concentrations (A) or ALA concentrations (B). (A) The CI values related to Asc concentration. The x-axis shows the 6 groups of Asc concentrations, and the y-axis shows the CI values. The CI values were averaged for all cell lines, and all 3 ALA concentrations were combined with that specific Asc concentration. The CIs showed a U-shape change as Asc concentration increased. (B) The CIs related to ALA concentration. The x-axis shows the 3 ALA concentration groups. No change in CIs was seen as ALA concentration increased.
H2O2 formation by alpha-lipoic acid, ascorbate, and the combination of alpha-lipoic acid + ascorbate
Because H2O2 formation is the central mechanism by which Asc exhibits cancer cell inhibition, and because ALA is an antioxidant with the potential to affect Asc-induced H2O2 formation, we measured H2O2 when ALA and Asc were concurrently used on cancer cells. Hela-HyPer-Cyto cells express an H2O2 fluorescent sensor protein HyPer.49,50 The fluorescence from Asc or ALA treatment alone is shown in Figure 3A and B. From 0 to 24 hours, ALA treatment up to 1 mM alone did not increase fluorescence in Hela-HyPer-Cyto cells, indicating no H2O2 formation. A slight increase in fluorescence was found at 2 hours but was not significant compared to untreated controls and did not persist (Figure 3A). Ascorbate increased fluorescence in a dose-dependent manner. At each concentration, the fluorescence increased over time, indicating the accumulation of H2O2 within 24 hours.
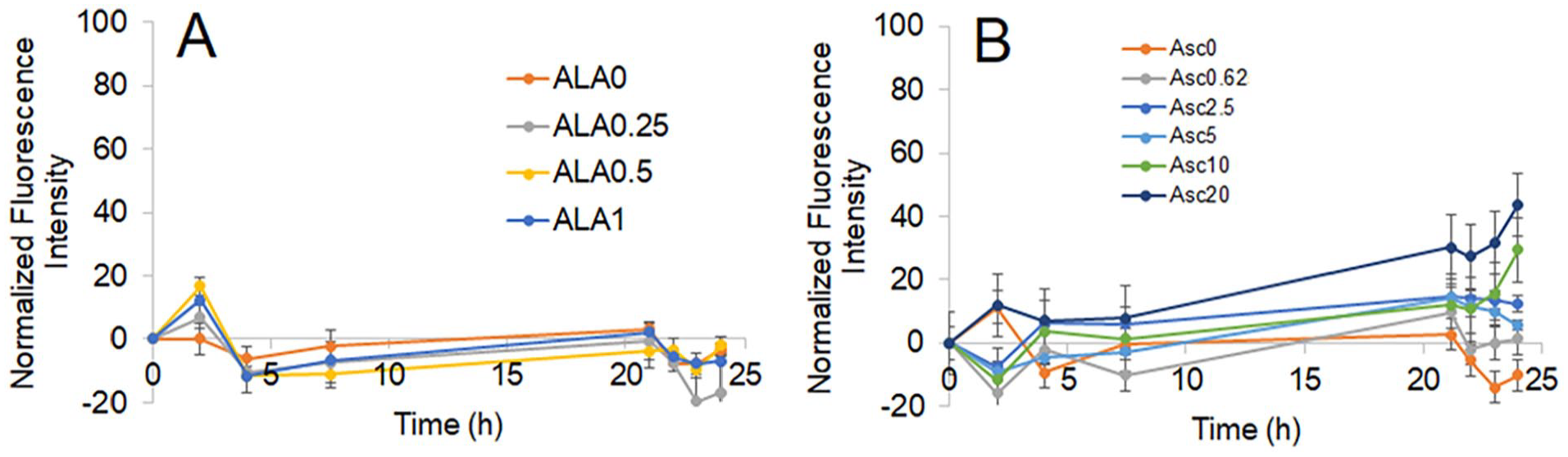
Normalized fluorescence of Hela-HyPer-Cyto cells under ALA or Asc treatment. (A) Cells were treated with ALA at 0, 0.25, 0.5, or 1 mM. (B) Cells were treated with Asc at 0.625, 1.25, 2.5, 5, 10 or 20 mM. Fluorescence was detected at 488/525 nm excitation/emission and then normalized to time 0 of the same treatment. The changes in fluorescence reflect concentrations of H2O2 in the cells.
The combination effects of Asc and ALA were then detected at 21 to 24 hours, and the average fluorescence intensity is shown in Figure 4. Data showed that adding ALA decreased the fluorescence when Asc concentrations were low, ie, 0.625-2.5 mM. At higher Asc concentrations of 5 to 20 mM, ALA did not influence the fluorescence produced by Asc.

Normalized fluorescence of Hela-HyPer-Cyto cells under combination treatment of ALA and Asc. Cells were treated with Asc (0-20 mM) and ALA (0-1 mM). Fluorescence was detected at 24 hours of treatment at 488/525 nm excitation/emission and normalized to untreated control cells. The relative fluorescence intensity reflects H2O2 inside the cells. At lower Asc concentrations (0.625-2.5 mM), adding ALA inhibited H2O2 formation from Asc. At higher concentrations of Asc, ALA did not influence H2O2 formation.
Tumor effects and toxicities of the combination treatment of alpha-lipoic acid + ascorbate
In a pilot dose-finding experiment, tumor-free Athymic nu/nu mice were given IP ALA at 50, 30, or 20 mg/kg to evaluate the ALA dosing regimen (Table 3). Research-grade ALA was used, and mice were treated for 2 weeks. The Asc 4 g/kg was combined with the ALA treatment. Signs of toxicity were observed with ALA 50 mg/kg in 2 of 3 mice on day 10, indicated by reduced activities and an insignificant body weight reduction. When Asc 4 g/kg was added to ALA 50 mg/kg on day 11, 2 deaths were observed on day 13. The lower ALA doses of 20 and 30 mg/kg did not show toxicity alone or in combination with Asc.
Adverse events in the non-tumor-bearing mice.

Mice (N = 3 per group) were given daily intraperitoneal injections of ALA at 50 mg/kg (group 1), 30 mg/kg (group 2), or 20 mg/kg (group 3), or the combination of ALA 30 mg/kg + Asc 4 g/kg (group 4), and ALA 20 mg/kg + Asc 4 g/kg (group 5). The Asc was given 30 minutes after ALA administration. For group 1, Asc at 4 g/kg was added on day 11. Research-grade ALA was used.
Tumor-bearing mice were also treated with ALA at 50 mg/kg using the research-grade compound. The Asc at 0.2, 0.5, and 4 g/kg were combined with the ALA (Table 4). Significant toxicities were observed. In groups 7 and 8, where 0.5 or 4 g/kg of Asc was combined with 50 mg/kg of ALA, 2/10 in group 7 and 6/10 mice in group 8 had lowered body temperatures after 2 doses of the combination treatment. Even after the treatment was halted in these 2 groups—group 7 had 3 of 10 deaths, and group 8 had 5 of 10 deaths. The ALA 50 mg/kg + Asc 0.2 g/kg treatment (group 6) also had 3 of 10 deaths after 6 to 7 days of treatment. The ALA single-drug treatment at 50 mg/kg (group 5) caused 2 of 10 deaths after 6 to 7 days of treatment. No adverse events were observed with vehicle treatment (control) or Asc single-drug treatment at all 3 doses (groups 2, 3, and 4). The results suggested that a high dose of ALA at 50 mg/kg had toxicities to mice, and adding Asc increased the toxicity.
Adverse events in tumor-bearing mice with ALA 50 mg/kg combined with Asc at 3 doses.

Research-grade ALA was used. Mice were treated with daily IP injections, and Asc was given 30 minutes after ALA.
A pharmaceutical-grade ALA was also evaluated at 50 mg/kg and in combination with Asc at 0.2, 0.5, and 4 g/kg (n = 10 per group). Adverse effects were seen in the Asc 4 g/kg + ALA 50 mg/kg group. There was an insignificant drop in average body weight on day 5, contributed by 3 of 10 mice; all had less than 10% body weight loss. Two of the mice had body weight recovery as the treatment continued. However, the third mouse died on day 17. All the other groups had no adverse events or changes in body weight (Figure 5). These data suggested better tolerance to the treatment when pharmaceutical-grade ALA was used.
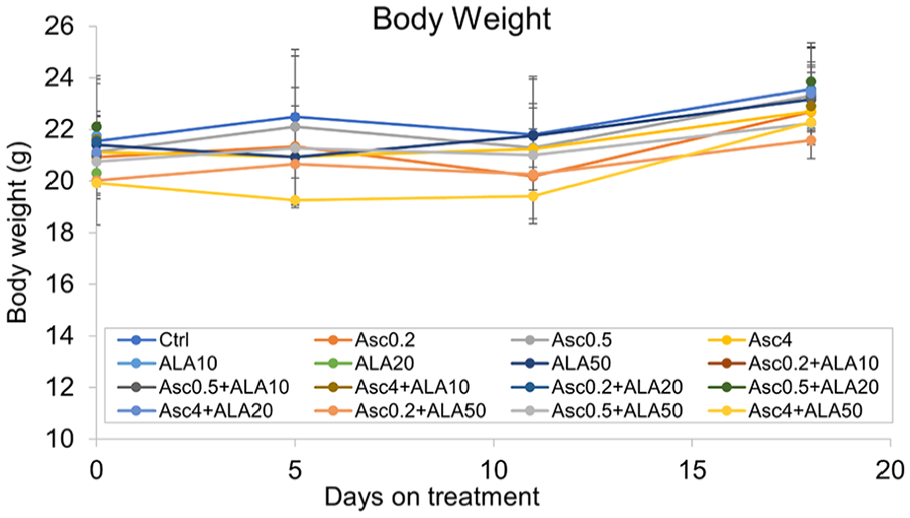
Average body weight of each group of mice under combination treatment of ALA and Asc. N = 9 to 20 per group.
A pharmaceutical-grade ALA was then used to treat MDA-MB-231 tumor-bearing mice alone or in combination with Asc. Table 5 summarizes the treatment groups conducted in 2 independent experiments, with one experiment using ALA 50 mg/kg and combined with Asc 0.2, 0.5, and 4 g/kg, and the other experiment testing ALA 20 and 10 mg/kg in combination with Asc 0.2, 0.5 and 4 g/kg. Mice were euthanized after 18 days of treatment, and total tumor weight was measured. The results are shown in Figure 6.
Adverse events in tumor-bearing mice treated with ALA, Asc, or the combination of ALA and Asc.

Pharmacologic grade ALA was used. Mice were treated with daily IP injections, and Asc was 30 minutes after ALA.
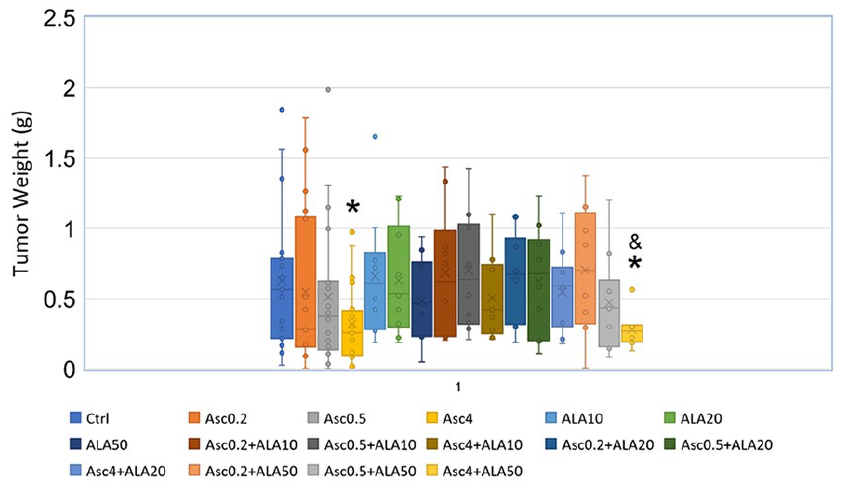
Final tumor weight at necropsy. Box-Whisker graph showing the distribution of tumor weight in each group. At the end of the treatment, mice were euthanized, and tumors were weighed. Data were combined from 2 independent experiments, each with 9 to 10 mice per group. The X mark in the box represents the average value, and the short line in the box represents the median. *P < .05 compared with the control group; &P < .05 compared with the ALA 50 mg/kg group. Asc 4 g/kg significantly inhibited tumor growth compared with control. The combination of ALA 50 mg/kg + Asc 4 g/kg treatment resulted in significantly smaller tumors than the control or ALA 50 mg/kg treatment. However, had no difference than Asc 4 g/kg. The combination of ALA 20 mg/kg or 10 mg/kg + Asc 4 g/kg had larger tumors than Asc 4 g/kg treatment alone but no difference than control.
Consistent with previous reports, Asc 4 g/kg significantly reduced the average tumor weight compared with the control group (P = .01). As expected, the lower doses of Asc did not inhibit tumor growth. The ALA alone did not inhibit tumor growth, and adding ALA to Asc did not provide benefit in tumor inhibition. In the combination groups, only the highest dose (ALA 50 mg/kg + Asc 4 g/kg) showed significant tumor inhibition compared with the control.
This combination treatment of ALA 50 mg/kg + Asc 4 g/kg is significantly better than the ALA 50 mg/kg alone. However, there is no difference between the ALA 50 mg/kg + Asc 4 g/kg and the Asc 4 g/kg alone groups. Adding ALA 20 or ALA 10 mg/kg to Asc 4 g/kg dampened the tumor-inhibitory effects of Asc, rendering the tumor weight of these combination treatment groups no different than the control.
One death was observed in the ALA 50 mg/kg + Asc 4 g/kg group on day 17. One mouse in each of the groups of ALA 20 mg/kg, Asc 4 g/kg + ALA 10 mg/kg, Asc 4 g/kg + ALA 50 mg/kg, and Asc 0.5 g/kg + ALA 20 mg/kg was euthanized on day 17 because of significant drop of Body Score Index (BSI, < 2), indicating significant toxicities with prolonged treatment with the combination (Table 5). After a week, a daily dose of ALA caused jaundice in 10% to 30% of mice receiving ALA at all the tested doses or the combination treatment.
Dehydration was observed in 5% to 10% of mice receiving Asc single-drug treatments and about 30% to 40% receiving combination treatments (Table 5). Most cases of dehydration resolved without additional care, but some needed treatment with subcutaneous ½ saline. The Asc alone did not cause any other adverse events besides dehydration.
Data herein suggest that adding high-dose ALA to high-dose Asc did not improve the tumor-inhibitory effects. Instead, it added toxicity.
Discussion
Medicine has significantly escalated the use of prescribed pharmaceutical drugs, resulting in polypharmacy and drug-drug interactions with increased adverse events.51,52 It is recognized that drug regimens are increasingly complex and, therefore, without research on the composite, can be potentially harmful. Polypharmacy and adverse drug reactions are also a concern in oncology populations.53-55 More research and understanding of drug-drug interactions must be available to guide practitioners.
Integrative oncology is no different. There can be little comfort in the belief that products are safe because they are natural if there is limited pharmacokinetic information, cytochrome P450 information, or evaluation of drug-drug interactions. Integrative oncology clinics employ many products from conventional oncology practice and integrative pharmacopeia. These include various natural products given orally or intravenously and many in combination. There needs to be more understanding of the impact of these products concerning their interactions with one another.
A mainstay of integrative oncology is IVC or Asc, which has a robust and growing translational research underpinning.6-21 When given intravenously at high doses, IVC is well-described as a prooxidative therapy. It has been shown to have synergistic or additive antineoplastic effects when given with chemotherapy and/or radiation. Its prooxidative properties are responsible for the antineoplastic effects. A recent pharmacokinetic study has shown it to be safe in doses of up to 100 g administered intravenously. 19
It has been shown previously that adding parenteral GSH to IVC reduces the prooxidative effects and antineoplastic properties in an animal cancer model. 32 The GSH is a powerful antioxidant that is helpful in treating chemotherapy-related neuropathy and has its place in treatment. However, as an antioxidant, the research cautions and informs us to avoid infusions of Asc and GSH on the same day.
Another integrative oncology therapeutic tool is IVALA. It is an antioxidant important in redox chemistry and mitochondrial respiration. The ALA is given intravenously with an established safety profile and is available through compounding pharmacies.34,38,42,44 It is administered intravenously as an antineoplastic agent in doses from 600 to 1000 mg several times per week. 38
The essential fact regarding administering IVALA with IVC is that adding a thiol compound like ALA replenishes the thiol pool; for example, ALA and regenerated glutathione can react with any oxidizing agent like IVC in the system.26,31,32 The antineoplastic effects of IVC depend on ascorbyl radical formation and hydrogen peroxide production through Fenton chemistry. We hypothesized that the coadministration of IVALA and IVC would reduce the antineoplastic effects of IVC. Indeed, the cell tissue and animal studies described above confirmed a reduction in the tumor-inhibitory effects of prooxidative IVC when adding the antioxidant ALA. These data also show that ALA does not become a prooxidant at higher doses, as seen with high-dose IVC.
Many in vitro and preclinical studies used artificially high concentrations of ALA close to or more than 1 mM that are not feasibly given to humans.35,37,39,56-58 Clinically, it has been reported that ~0.04 mM peak plasma concentration was achieved following 200 mg of intravenous (IV) administration of ALA. 43 Studies in rats noted that 5 mg/kg IV (equivalent to 350 mg for a 70 kg human) had a Cmax of ~0.05 mM. 59 In contrast, in dogs, oral doses of 25 mg/kg had a Cmax of ~0.06 mM, 60 and in sea lions, subcutaneous doses of 20 mg/kg had ~0.05 mM Cmax. 61 A more recent pharmacokinetic study in multiple sclerosis patients reported that oral doses of 1200 mg only had 0.01 mM Cmax. 62 It is unknown in oncology patients what level can be achieved. Higher plasma concentrations might be obtained using higher IV doses, as tolerability allows. As high as 1200 mg IV 63 or 2400 mg oral doses 64 were used in clinical trials. However, blood concentrations were not reported. Extrapolation of the human PK data 43 gives 0.24 mM when the IV dose is 1200 mg, and extrapolation of the rat PK data 64 provides a Cmax of 0.17 mM for a 1200 mg IV dose. It is noted that 1 mM ALA has not been reached by humans. Here, we used relatively high concentrations of ALA at 0.25, 0.5, and 1 mM, and data show that the cancer cells were only marginally sensitive to these ALA concentrations.
A surprising finding was the increased toxicity when coadministering parenteral ALA and Asc in the cancer animal model. The initial experiment dosing ALA at 50 mg/kg and combining it with the 3 doses of Asc showed significantly increased toxicity, leading to death. Because of the toxicity, this experiment was terminated early. As a result of concerns for toxicity with 45% PEG and other potential impurities in the research-grade ALA, we repeated the experiments with pharmaceutical-grade ALA. Pharmaceutical-grade ALA was tested and still showed significant toxicities when used with a high dose of Asc.
The ALA did not exhibit significant tumor reduction compared with the control in our breast cancer model—Asc only at 4 g/kg reduced tumor growth. Adding ALA 50 mg/kg did not further improve the tumor-inhibitory effects, but of concern, adding ALA 10 and 20 mg/kg to Asc 4 g/kg dampened the tumor-inhibitory effects.
A limitation of this study is that the influences of sequence and time intervals between ALA and Asc administration on drug-drug interaction and antitumor effects were not investigated. There has not been an established sequence or interval for ALA and ascorbate clinically, nor have we found literature reports in animals investigating this question. Based on published PK studies, the half-life of ALA was ~25 minutes, and the half-life of Asc was ~ 2 hours. In this study, Asc was injected 30 minutes after ALA, which was approximately 1 ALA half-life interval. More investigations need to be done to study whether sequence and/or administration intervals could influence toxicity or therapeutic effects. The mechanisms for the unexpected toxicity in mice must also be further investigated.
Conclusions
These data do not support the concurrent or relative concurrent use of high-dose intravenous ALA with prooxidative high-dose IVC in clinical oncology care. The combination of ALA and ascorbic acid did not show benefit in tumor inhibition and could be potentially dangerous due to its increased toxicity.
Supplemental Material
sj-docx-1-onc-10.1177_11795549241283421 – Supplemental material for Combination of High-Dose Parenteral Ascorbate (Vitamin C) and Alpha-Lipoic Acid Failed to Enhance Tumor-Inhibitory Effect But Increased Toxicity in Preclinical Cancer Models
Supplemental material, sj-docx-1-onc-10.1177_11795549241283421 for Combination of High-Dose Parenteral Ascorbate (Vitamin C) and Alpha-Lipoic Acid Failed to Enhance Tumor-Inhibitory Effect But Increased Toxicity in Preclinical Cancer Models by Ping Chen, Davis Lamson, Paul Anderson, Jeanne Drisko and Qi Chen in Clinical Medicine Insights: Oncology
Footnotes
Author Contributions
PC, DL, PA, JD, and QC contributed to the study design. PC and QC were responsible for managing the cell tissue and animal experiments. The results of the cell tissue and animal studies were shared with all the authors. QC and JD were responsible for the table and figure design. All authors contributed to the manuscript’s drafting, revision, and final approval.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors disclose receipt of the following financial support from The Clementine Fund (AWD-0002601) and The Peachtree Orthomolecular Medicine Research Fund. The funding agencies had no input in the manuscript’s study design, drafting, or approval.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.