Abstract
Background:
Patients with gastric cancer (GC) who underwent radical surgery require long-term follow-up (usually 5 years). The purpose of this study was to explore individualized follow-up strategies for patients with GC.
Methods:
This is a retrospective cohort study that established a clinicopathologic database of patients who underwent gastrectomy from January 2010 to December 2020 at Ningbo No. 2 Hospital. Follow-up was performed until March 2023. The rate of new-onset recurrence of patients with GC was explored annually according to different pTNM stages, defining a recurrence rate of less than 1% as adequate follow-up time.
Results:
Of the 1606 patients who were eligible, the total number of patients who completed the 5- and 10-year follow-up was 1107 and 586, respectively. A total of 444 cases were diagnosed with recurrence. The recurrence rate for stage IA patients was consistently less than 1% during the follow-up time. The adequate follow-up time (the rate of new-onset recurrence less than 1%) was 5 years for stage IB and IIA patients, and 8 years for stage IIB and IIIA patients, respectively. In contrast, stage IIIB patients were always at risk of recurrence during the follow-up time (>1%). Time to a new recurrence rate for stage IIIC patients was 6 years.
Conclusion:
Among patients who underwent radical gastrectomy, the rate of new-onset recurrence varied among patients with different pTNM stages. This study suggests that the follow-up of GC can be individualized and refer to pTNM stage.
Introduction
Gastric cancer (GC) is the fifth most common malignant tumor and the fourth leading cause of cancer death worldwide. 1 The main incidence area of GC is Southeast Asian countries, 2 among which China accounts for 42% of the world’s patients with GC. 3 In China, the proportion of patients with advanced GC accounts for more than 40% of all patients. 4 The overall 5-year survival rate of patients with advanced GC is only 20% to 30%5,6 with a poor prognosis. Currently, the conventional treatment options for GC include surgery, radiotherapy and chemotherapy, targeted therapy, and immunotherapy, 7 with regular follow-up visits. The purpose of follow-up is to observe the effect of treatment and to detect disease recurrence at an early stage, thus contributing to further treatment.
According to the National Comprehensive Cancer Network (NCCN) and the Japanese guidelines,8,9 the current recommendation for the frequency of follow-up after radical surgery for early GC is every 6 months for the first 3 years postoperatively, then every 1 year until 5 years postoperatively. The frequency of follow-up after radical surgery and unresectable palliative treatment for advanced GC is every 3 to 6 months for the first 2 years, then 6 to 12 months until 5 years, and annually for more than 5 years. It is widely recognized internationally that the follow-up endpoint for GC is usually considered to be 5 years after surgery due to the likelihood of tumor recurrence after 5 years, 10 but there is no high-level medical evidence to support which strategy is optimal. Besides, some studies have suggested that patients require individualized follow-up due to differences in their tumor prognosis. 11
A study by Yago et al 12 concluded that the duration of adequate surveillance of patients with GC who had undergone radical gastrectomy should vary at each stage. Another study conducted by Qiu et al 13 suggested that patients in different age groups should be followed up with different strategies due to different peaks of recurrence and types of recurrence. To date, only a few studies have mentioned individualized follow-up for GC. On the other hand, pTNM stage is the most widely used tool to predict tumor prognosis. 14 Therefore, the aim of this study is to investigate the relationship between pTNM stage and time to recurrence and try to identify appropriate follow-up strategies. Our hypothesis is that patients with GC with different pTNM stages require individualized follow-up.
Methods
Patients
This is a retrospective cohort study using prospectively collected data. A total of 1606 patients who underwent radical gastric surgery at Ningbo No. 2 Hospital from January 2010 to December 2020 were eventually enrolled. The inclusion criteria were as follows: (1) histologically confirmed primary gastric adenocarcinoma; (2) underwent radical surgery; (3) no previous history of gastrectomy or other malignancies; (4) did not receive neoadjuvant chemoradiotherapy; and (5) complete follow-up data. The study was approved by the Human Research Ethics Committee, Ningbo No. 2 Hospital (PJ-NBEY-KY-2019-153-01). All the patients provided written informed consent.
Histological examinations
The medical records and pathologic reports were reviewed for each patient. Clinicopathologic factors included age, sex, tumor location, type of surgical resection, tumor size, histologic type, lymphovascular invasion, pTNM stage, presence of adjuvant chemotherapy, and number of lymph node dissection. Staging was defined according to the American Joint Committee on Cancer Staging Manual, 8th edition. 15 The severity of GC depended largely on the depth of local infiltration, the degree of lymph node metastasis, and the presence of distant metastases.
Adjuvant therapy
In principle, 5-fluorouracil (5-FU) or platinum-based adjuvant chemotherapy is recommended for all stage II-III patients.16,17 Of note, some patients with early GC (stage I) have received adjuvant chemotherapy, including adjuvant S-1 monotherapy. These patients usually have a number of risk factors including 1 to 2 positive lymph nodes (T1N1M0), muscle invasion (T2N0M0), poorly differentiated lymphovascular invasion, tumor deposition, or age less than 50 years. In addition, some stage II-III patients refuse chemotherapy because of age, financial costs, or personal preference.
Follow-up
Follow-up evaluation was based on medical history, clinical findings, blood test results inclusive of tumor markers, imaging, and endoscopy.8,9 Blood tests were performed every 3 months in the first year after surgery and every 3 or 6 months thereafter. Computed tomography (CT) scans were performed every 6 months after surgery. Endoscopy was performed annually to screen for GC remnants and esophageal cancer. In addition to this routine follow-up regimen, patients would come forward for review if they develop suspicious clinical symptoms and suspect disease recurrence. For suspected metastases to the bones, brain, lungs, and/or other sites, bone imaging, CT, magnetic resonance imaging (MRI) of the chest or brain, positron emission tomography (PET)-CT, and puncture biopsy or surgical exploration were performed. 9
Disease-free survival (DFS) was defined as the time from surgery to death, local recurrence, or distant recurrence.
18
Patients who did not record these events were recorded on the last known date of contact. The median follow-up for the entire cohort was 53 months. The follow-up of all patients included in this study ended in March 2023. The 5 internationally recognized patterns of GC recurrence include local recurrence, lymph node metastasis, peritoneal metastasis, hematogenous metastasis, and mixed recurrence.
19
Recurrence here refers to primary GC recurrence only. In this study, the rate of recurrence after a specified time point was defined as the rate of new-onset recurrence.
12
This rate is the ratio of the number of recurrences in the current year minus the number of recurrences that have occurred to number of patients still in follow-up in the current year minus those who have relapsed (
Statistical analysis
Univariate analyses were applied to identify all potential clinicopathologic factors associated with prognosis. These factors were finally subjected to multivariate regression using Cox regression. Hazard ratios (HRs) and 95% CIs were used as indicators of correlation. All statistical tests were performed using 2-sided analysis and P < .05 was considered statistically significant. Kaplan-Meier analysis was used to show DFS in each pathological stage of GC. The above analyses were performed using the SPSS software program (version 25.0; Chicago, Illinois).
Results
Patient characteristics
From January 2010 to December 2020, a total of 2183 patients underwent gastrectomy at Ningbo No. 2 Hospital, as shown in Figure 1. A total of 577 patients were excluded according to the inclusion criteria. Finally, 1606 patients were eligible for analysis. The proportion of stage I, II, and III patients in these cases was 38.7%, 19.0%, and 42.3%, respectively. The median follow-up time for patients in each stage was 62 months for stage I, 58 months for stage II, and 40 months for stage III, respectively. The total number of patients completing 5-year follow-up (including normal closure or death) was 1107, and the total number of patients completing 10-year follow-up (including normal closure or death) was 586.

A total of 1606 patients underwent gastrectomy for gastric cancer from 2010 to 2020. The number at each stage is shown.
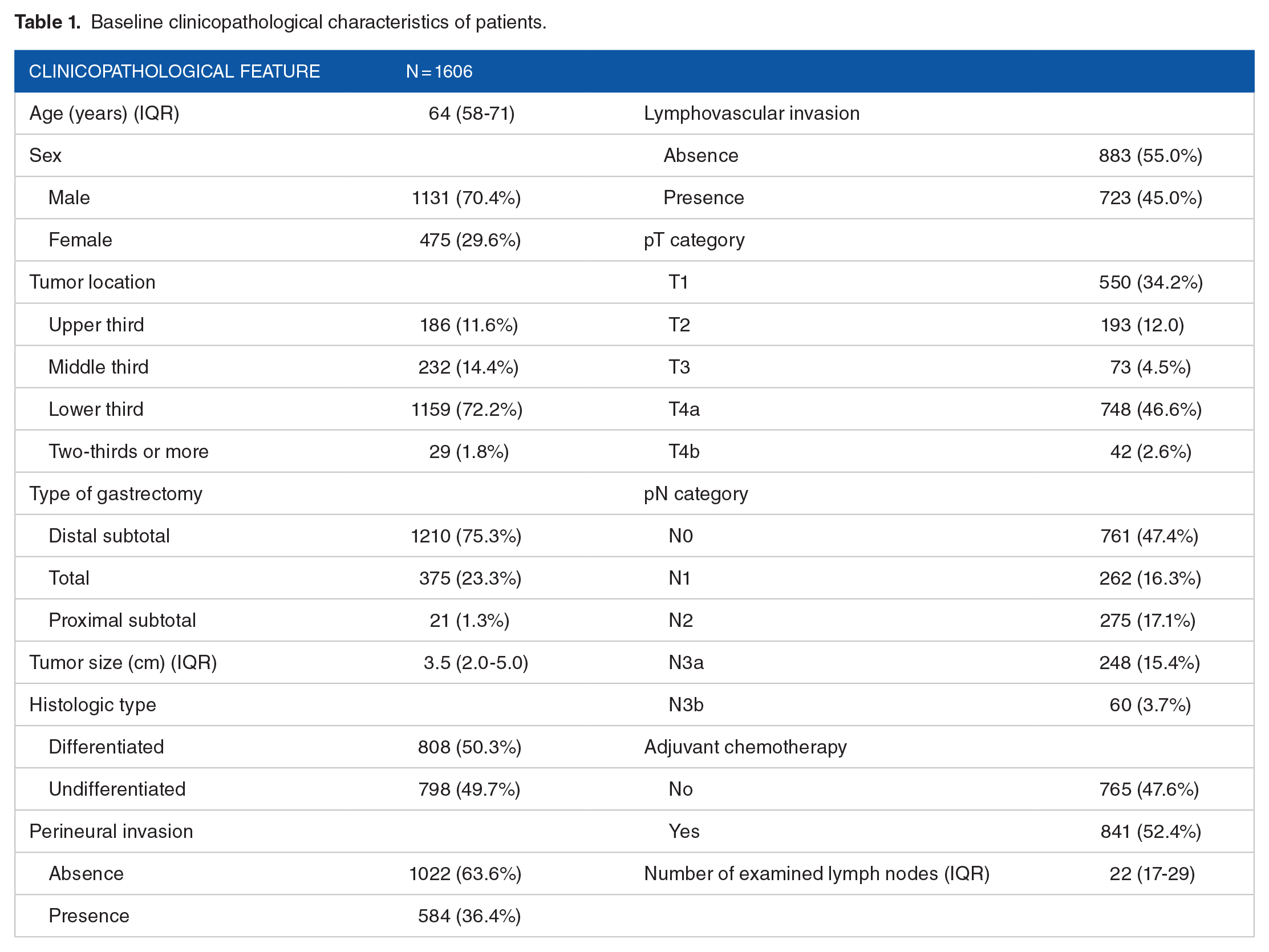
The demographic and tumor characteristics of the 1606 patients who underwent therapeutic gastrectomy are listed in Table 1. Of the total population, the median age was 64 years, 70.4% of the patients were men and 29.6% women, 72.2% of patients have tumors located in the lower third of the stomach, 75.3% of the patients underwent distal subtotal resection, and the proportion of differentiated GC was close to that of undifferentiated GC. In terms of invasion of peripheral tissues, perineural invasion was found in 36.4% while lymphovascular invasion in 45.0%. Lymph node metastases were present in 52.6% of patients, and the median number of lymph nodes examined was 22. Among all patients, 52.4% received adjuvant chemotherapy.
Baseline clinicopathological characteristics of patients.
Risk factor analysis
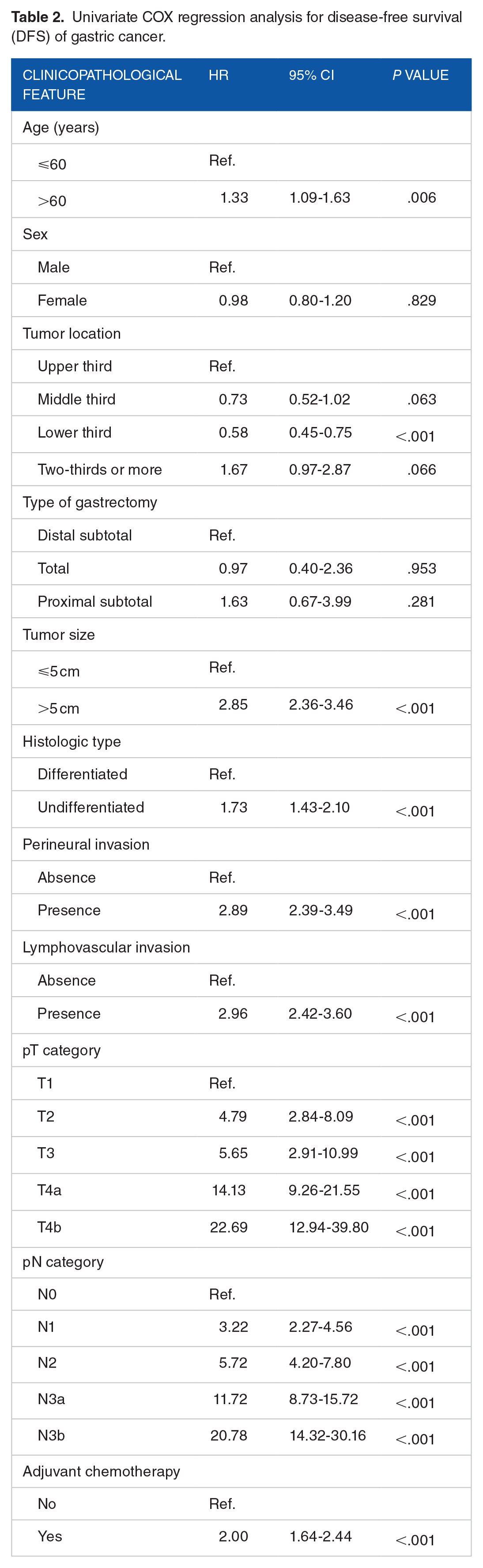
To clarify the risk factors of GC, univariate and multivariate regression analyses were performed. Univariate regression analysis showed that the risk factors of GC included age, tumor location, type of gastrectomy, tumor size, type of differentiation, perineural invasion, lymphovascular invasion, T stage, N stage, adjuvant chemotherapy, and so on, as shown in Table 2. After multivariate analysis, it was found that age, T stage, and N stage were the independent factors affecting the prognosis of GC, as detailed in Table 3. Compared with patients younger than or equal to 60 years of age, patients older than 60 years of age had a higher risk of recurrence (HR = 1.42, P = .001).
Univariate COX regression analysis for disease-free survival (DFS) of gastric cancer.
Multivariate COX regression analysis for disease-free survival (DFS) of gastric cancer.

Disease-free survival rate
In this study, T stage and N stage were reaffirmed as the most important prognostic factors affecting GC. Kaplan-Meier curves of DFS were plotted according to the pTNM stages, as shown in Figure 2. A significant difference was found in DFS by stage (P < .001).
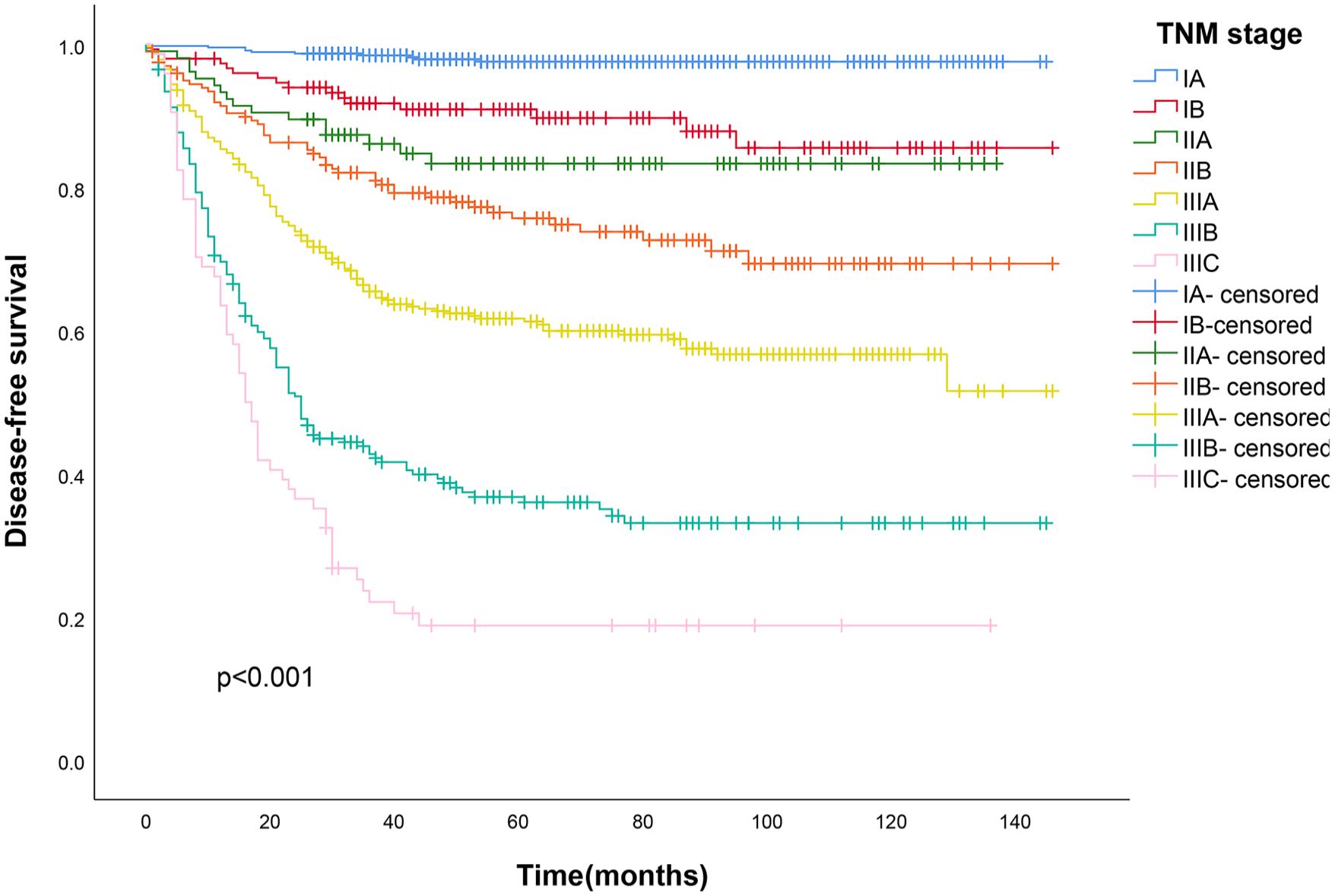
Disease-free survival curves with Kaplan-Meier analysis at each stage.
Recurrence rate
Annual recurrence rates were calculated based on the recurrence in each postoperative year of follow-up. The recurrence rates and follow-up endpoints for each postoperative year are shown in Table 4, which shows that early GC cases, such as stage IA, have a lower recurrence rate throughout the period. On the contrary, the recurrence rate was higher in early stage of advanced GC. During the follow-up period, the recurrence rate for patients with stage IA GC was consistently less than 1%. The recurrence rate for patients with stage IB GC decreased to 0.95% in year 4, and was 1% in year 5, and has been less than 1% since then. The recurrence rate for patients with stage IIA GC was 1.82% in year 5 and was 0 from year 6 until the end of the current follow-up period. The recurrence rate for stage IIB patients decreased to 1.25% in year 8 and remained at 0 thereafter. The recurrence rate for stage IIIA patients decreased to 1.17% at year 8 and remained at 0 from year 9 to the end of the current follow-up. The recurrence rate for stage IIIB patients was consistently higher than 1% during the follow-up time, and the risk of recurrence was always present. The recurrence rate for stage IIIC patients was 9.09% at year 5 and remained at 0 from year 6 to the end of the follow-up period.
Recurrence risk among patients with recurrence free at the specified time point and follow-up endpoints.

Recurrence pattern
The recurrence pattern and incidence of each postoperative cycle are shown in Table 5. According to the statistics of the recurrence, postoperative recurrence of GC was mainly dominated by peritoneal metastasis and hematogenous metastasis. The proportion of peritoneal metastasis was the highest in the first 2 years after surgery, accounting for 51.9% and 39.1%, respectively. With prolonged follow-up, the proportion of hematogenous metastases gradually increased, accounting for 38.0% in the third postoperative year after the resection surgery. In contrast, the proportion of peritoneal metastasis showed a decreasing trend, and the proportion of local recurrence was low during the follow-up time, always less than 10%. In addition, the total number of recurrences of various recurrence patterns in the first 3 years was 380, accounting for 85.6% of the total number of recurrences, and the total number of recurrences in the first 5 years was 423, representing 95.3% of the total number of recurrences, whereas the rate of recurrence after 5 years was 4.7%.
Pattern and rate of recurrence according to follow-up time.

Discussion
The high incidence of advanced GC is mainly in Asian countries represented by China, and patients in these areas are more in need of follow-up. 3 In our study, TNM stage proved to be the most important independent prognostic factor, so we categorized patients with GC according to TNM stage and further aimed to discover the adequate follow-up time and the optimal follow-up methods required for different types of GC.
In the present study, GC was found to be quite prevalent among male patients, up to more than 70%, which may be closely related to the epidemiological characteristics of Helicobacter pylori and the characteristics of alcohol consumption. In China, the male-to-female H pylori infection rate is roughly 4:1 to 6:1, 20 and alcohol exposure rate of men is much higher than that of women. 21 The incidence of distal GC is also significantly higher than that of proximal GC, which is different from countries such as Europe and the United States where gastric reflux is the main cause of GC. 22 In contrast, Chinese people are more susceptible to H pylori infection, 23 which is closely related to the development of GC in the sinus section and the lower part of the stomach.
In this study, age was found to be an independent factor affecting the prognosis of GC. Elderly patients are in poorer health, less tolerant of adjuvant therapy, and have a progressive decline in their immune function. 24 Moreover, a higher percentage of patients undergo D2 lymph node dissection in younger patients compared with older patients with more comorbidities. 25 Therefore, the prognosis of elderly patients is poorer, which is similar to the findings of Hemminki et al. 26 However, it has also been found that GC has a poorer prognosis in younger adults. 25 This may be due to the higher degree of malignancy of such tumors. Lauren’s diffuse and poorly differentiated histological types of tumors also occurred more frequently. Besides, some studies have found that tumor size, degree of differentiation, and adjuvant chemotherapy are prognostic factors for tumors,14,27 but these factors were not statistically different in our study. The pTNM stage is another important factor affecting the prognosis of GC. In this study, the percentage of patients with advanced GC (stage II-III) was as high as 61.3%, which is significantly different from Japan, Korea, and other countries.
According to pTNM stage, we found that there were significant differences in survival curves between different stages, further validating that the 8th edition pTNM stage as a powerful tool for GC staging. For patients with stage IA, the probability of recurrence was always <1%, and the 5-year DFS can reach 97.6%, which is a good prognosis. According to previous studies, 28 however, it is recommended that the follow-up period for stage IA patients be shortened to 2 years. For stage IB and IIA patients, the prognosis is slightly worse than that of stage IA patients, with 5-year DFS of 90.9% and 83.3%, respectively. Due to the low recurrence rate, a 5-year follow-up is sufficient for these patients, and the study by Lauricella et al 29 is also in line with our findings. For stage IIB and IIIA patients, the 5-year DFS was 74.8% and 61.0%, respectively, with a significant decrease in the recurrence rate at year 8, and the recurrence rate remained 0 from year 9 to the end of the current follow-up. Therefore, we recommended that the adequate follow-up for stage IIB and IIIA patients should preferably be prolonged to 8 years. For stage IIIB patients, the risk of recurrence always existed until the closure of this follow-up. The results of Liu et al 30 in this regard were similar to ours. Their research also suggests that the survival rate of late stage recurrence within 2 years is only 23.4%, indicating a high risk. Therefore, it can be assumed that stage IIIB patients should be followed for a longer period of time according to their own situation. Overall, for patients with stage IA to IIIB GC, the required follow-up is longer and the likelihood of GC recurrence is higher as the tumor stage becomes more advanced.
For stage IIIC patients, the results showed that although the recurrence rate was always 0 at the beginning of year 6, it was still 9.09% at year 5. This was due to the fact that the number of stage IIIC patients decreased during the long-term follow-up for various factors such as death and loss of visits. Therefore, it is recommended that stage IIIC patients should still be followed up for as long as possible. In the present study, the number of patients followed up at year 5 was only 11, and the results can somewhat have fortuitous. However, the results of a study by Yago et al 12 also showed that the recurrence rate of stage IIIC patients plummeted at year 5, with a similar overall trend. The fact that these 2 studies coincidentally yielded the same results suggests that there is a specificity in stage IIIC. Patients with stage IIIC tumors are more malignant and had more severe tumor progression, lymphovascular infiltration, and vascular infiltration, all of which lead to higher recurrence rates. These factors make stage IIIC patients more likely to develop abdominal metastases. 31 Recurrence occurs at an early stage in stage IIIC patients with low recurrence potential, so there is a significant decrease in the recurrence rate at year 5.
In general, the recurrence pattern of GC is divided into 5 categories. Local recurrence accounted for a low percentage of the follow-up time because local recurrence is predominantly in gastric stump cancer. Current surgical inventions are usually accompanied by pathologic examination to ensure negative margins, and it usually takes 15 years for gastric stump cancer to develop. 32 In this study, patients with advanced stage and advanced age were the majority, and therefore, local recurrence may not be have been observed during the follow-up period. Lymph node metastasis as a recurrence pattern accounted for no more than 20% per year in this study. Lymph node metastasis was previously considered to be a major feature of GC. 33 However, advances in surgical procedures have led to a significant reduction in the number of patients with lymphatic metastases compared with the past, and D2 lymph node dissection, which has a high rate of clearance (the mean number of lymph nodes cleared in this study was 22), is now mostly used in China. 34 In this study, the proportion of hematogenous metastasis gradually increased as follow-up proceeded. With the prolongation of time, the patient’s immunity declines and the underlying cancer cells enter the portal vein or body circulation and then spread to other parts of the body. 35 They are commonly found in the lungs, liver, and pancreas, with hepatic metastasis being the most common, followed by peritoneal, adrenal glands, kidneys, brain, as well as the ovaries, bone marrow, and skin. At present, the main recurrence pattern of GC is abdominal metastasis. Gastric cancer cells are highly invasive crossing the stomach wall and invading the peritoneum and other organs in the abdominal cavity. In general, patients with abdominal metastasis are usually in advanced stages with poor treatment outcomes. 36 They are more prone to organ failure and death. Therefore, the mortality rate of this recurrence pattern is extremely high, which is similar to the findings of Kurokawa et al. 37 In addition, we found that the proportion of abdominal metastasis was highest in the first 2 years after surgery, followed by a decreasing trend. The reason considered is that patients with abdominal metastasis recurred within a short period of time, and after 2 years, the number of recurrences gradually decreased and the percentage declined. 38
It has been reported that the recurrence rate of stage I GC after radical surgical resection is 2.7%. 39 Among them, distant metastasis and gastric stump cancer accounted for 51.9% and 16.5%, respectively. The time of recurrence was mostly within 2 years after surgery. A study found that endoscopic mucosal resection combined with chemotherapy can effectively reduce early recurrence. 40 The 3 major recurrence patterns of advanced GC are distant metastasis, abdominal metastasis, and local recurrence, each accounting for about one-third of the total. 41 Patients with GC tend to recur within 1 to 3 years after undergoing surgery. It is now internationally recognized that stage II and III patients can reduce the recurrence rate after receiving adjuvant chemotherapy.
In this study, the recurrence rate was 95.3% in the first 5 years, which proves that the 5-year follow-up time is still of great reference value for the general GC population. For stage IA patients, the recurrence rate is lower, and the peak of recurrence is earlier. Hence, the follow-up time can be appropriately shortened. For patients with stage IIB and above, the risk of recurrence is higher and the duration of recurrence risk is longer, so it is recommended to appropriately lengthen the years of follow-up. Individualized follow-up improves the accuracy of patient follow-up, reduces patients’ anxiety, and lowers health care costs. Meanwhile, some studies have also pointed out that a reasonable follow-up strategy should not only emphasize the effectiveness of testing but also consider the cost-effectiveness. 42 A study by Wu et al 11 noted that an individualized follow-up strategy could lead to optimal cost-effectiveness for stage II patients, while stage III patients were more advantageous to be followed according to the NCCN guideline of the most intensive follow-up. This also reflects that the concept of individualized follow-up is receiving increasing attention.
With a sample size of up to 1600 cases and a follow-up period of up to 10 years, our study is one of the more comprehensive analyses of individualized follow-up of GC in the Chinese field. The study intended to explore the recurrence regularity of GC according to the stage of the tumor. This study can provide a reference for follow-up strategies in areas with a high prevalence of advanced GC. However, this study still has limitations. The annual recurrence rates among patients with various stages were highly fluctuating, which may be due to the limited sample size of patients with advanced GC and the need for a larger sample size for validation.
Conclusions
The ideal follow-up time required after radical GC surgery may vary depending on the pTNM stage. For patients with stage IA, it is recommended that the follow-up time be shortened to less than 2 years, whereas for patients with stage IIB and above, if the risk of recurrence is more than 5 years, a longer follow-up period is clinically warranted.
Footnotes
Author Contributions
CZ, TH, and LG contributed to drafting, conception, and design. MQ contributed to manuscript writing. XL, XZ, and XC contributed to data collection. YS, PC, and FW performed procedures and data analysis. TH polished this article. All authors contributed to manuscript revision, read, and approved the submitted version.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Zhu Xiu Shan Talent Project of Ningbo No. 2 Hospital (Project Number: 2023HMYQ09), and Ningbo Clinical Research Center for Digestive System Tumors (Grant No. 2019A21003) and Ningbo Medical Key Discipline (Grant No. 2022-B09).
Date Availability Statement
The datasets supporting this article’s conclusions are included within the article and its additional files.
Ethical Approval
This study was a retrospective study. Approval of the research protocol: All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the Human Research Ethics Committee, Ningbo No. 2 Hospital (PJ-NBEY-KY-2019-153-01). All the patients provided written informed consent.
