Abstract
Background:
The limitations of prostate-specific antigen (PSA) in diagnosing prostate cancer (PCa) necessitate the exploration of novel biomarkers. Recent studies suggest a potential link between coagulation markers, particularly fibrinogen and D-dimer, and PCa.
Methods:
A retrospective single-center analysis on 466 biopsy-undergone patients was conducted, categorized into PCa and benign prostatic hyperplasia (BPH) groups. Baseline and coagulation parameter levels were analyzed. Utilizing a Mendelian randomization (MR) approach, we investigated the causative relationship between D-dimer and PCa risk.
Results:
Individuals with PCa, compared with those with BPH, exhibited significantly higher D-dimer levels (P < .001), total PSA (P < .001), and PSA density (P < .001). Fibrinogen levels did not exhibit significant differences (P = .505). The MR analysis suggested a probable causal link between elevated D-dimer levels and an increased risk of PCa (odds ratio: 1.81, 95% confidence interval: 1.48-2.21, P = 7.4 × 10−9).
Conclusions:
This research highlights D-dimer as a potential biomarker for diagnosing PCa, supported by clinical and MR analyses. The study paves the way for future large-scale, multi-center research to corroborate these findings and further explore the relationship between coagulation markers and PCa mechanisms.
Introduction
Prostate cancer (PCa) stands as the predominant cancer within the male genitourinary tract, accounting for approximately 1.4 million new diagnoses and 375 000 fatalities worldwide annually. It is the second most frequent cause of cancer-related mortality in men, after lung cancer.1,2 However, non-clinically significant prostate cancer (non-csPCa) is likely to cause no severe harm and typically requires no intervention. 3 At present, prostate-specific antigen (PSA) functions as a key indicator for identifying PCa. Its role in reducing the disease-specific mortality of PCa has been affirmed by past studies. However, the lack of specificity associated with PSA leads to unnecessary prostate biopsies and overtreatment of patients with non-csPCa. 4 With the advancement in research on molecular pathways related to PCa, numerous biomarkers for PCa have been discovered. According to current studies, the diagnostic efficacy of these biomarkers appears to surpass that of PSA. Examples include the 4K Score, Prostate Health Index (PHI), and SelectMDx. However, compared to the simplistic PSA and its derivatives, they incur higher costs, hindering their widespread application in most medical centers. 5
As research in tumor molecular biology progresses, a growing body of evidence suggests a tight correlation between coagulation function anomalies and the development and advancement of cancer. 6 Cancer cell activity disturbs the equilibrium of the body’s coagulation system, triggering coagulation-related processes within the tumor’s stroma and its adjacent microenvironment. This process is not only related to disease progression and prognosis but could also serve as a signal for undetected malignant tumors. 7 The complex interaction between cancer and the coagulation system results in abnormal expression of coagulation indicators in cancer patients. Differing concentrations of coagulation markers could disclose the potential biological characteristics of tumors, and the identified aberrant expression of these markers has been linked to the clinical pathologic traits of malignancies including gastrointestinal and lung cancers.8,9 As research into the relationship between PCa and the coagulation system advances, more evidence is emerging to support the roles of fibrinogen (Fib) and D-dimer as potential tumor markers for PCa. 10
Fib, a key marker of the coagulation system, is intimately connected with the growth of malignant cells, the formation of new blood vessels, evasion of immune detection, and the spread of cancer. 11 Allin and colleagues observed a notable correlation between Fib levels and both the initiation and advancement of PCa. 12 Recent studies have uncovered that patients with PCa exhibiting hyperfibrinogenemia are more likely to present with elevated levels of PSA, higher Gleason scores, and risk stratification. 13 D-dimer, indicative of coagulation and fibrinolysis system activation, represents a breakdown product of cross-linked fibrin. Recently, McNally and Kalkan et al observed a significant elevation in D-dimer levels among PCa patients.3,14
The potential of peripheral blood coagulation parameters reflecting systemic coagulation status as biomarkers for PCa diagnosis has been suggested by previous observational studies. However, these traditional observational studies are susceptible to confounding bias and causal inference bias. The advent of Mendelian randomization (MR) studies offers a fresh perspective. Utilizing genetic variations that are randomly allocated in meiosis as instrumental variables (IVs), MR investigations explore the causative link between exposure and its outcome, circumventing the confounders and reverse causation often encountered in alternative observational study designs. 15
In this study conducted at a single center, we explored the differences in peripheral blood coagulation parameter levels, namely Fib and D-dimer, between PCa patients and non-cancer individuals. Parameters displaying differences were subsequently selected for MR analysis, with the objective of investigating the potential utility of peripheral blood coagulation parameters as biomarkers for diagnosing PCa.
Materials and Methods
Study population and data source
Between January 2018 and December 2022, a total of 702 patients were diagnosed with either PCa or benign prostatic hyperplasia (BPH) following prostate biopsy at the Department of Urology, Shandong Provincial Hospital. Following a strict adherence to exclusion criteria, 466 patients were included in this study. Exclusion criteria encompassed (Ⅰ) past or present malignancies in other organ sites; (Ⅱ) diseases like coagulation-related disorders, collagen diseases, and abnormal liver or kidney function that could potentially alter peripheral blood coagulation parameter levels; (Ⅲ) significant cardiovascular diseases or any anticoagulant treatment; and (Ⅳ) incomplete clinical data. Demographic and clinical data of the patients included in the study were collected, such as age, total prostate-specific antigen (TPSA), prostate volume (PV), prostate-specific antigen density (PSAD), Fib, D-dimer, and histopathological findings. Within this study, csPCa was defined as a Gleason score of 7 (3 + 4) or higher. This retrospective study was approved by the Biomedical Research Ethic Committee of Shandong Provincial Hospital (approval no. SWYX2023-503).
The principal genetic instrument for D-dimer originated from a recent Genome-Wide Association Study (GWAS). This research examined the genetic structure of host proteins implicated in SARS-CoV-2 infection across a cohort of 10 708 subjects, employing an aptamer-based method. 16 The study identified 220 host DNA sequence variants explaining the variance of 97 proteins, with D-dimer being one of the proteins mentioned. To encompass more single-nucleotide polymorphisms (SNPs) of D-dimer, a more lenient threshold (P < 5 × 10−5) was employed; this method has also been used in MR studies for exploring new SNPs.17,18 Two SNPs demonstrated a significant correlation with D-dimer levels (P < 5 × 10−5, linkage disequilibrium [LD] r 2 < 0.001, kb = 10 000). The F-statistic was 17, surpassing the standard threshold of 10, indicating that the instrument possesses strong potential for predicting D-dimer levels. The GWAS summary statistics for PCa originated from meta-analysis research encompassing 79 148 PCa instances and 61 106 controls. 19 We sourced summary statistics from this investigation, isolating two SNPs linked to D-dimer levels, which included the influence of these SNPs on PCa; we additionally extracted the effect sizes and standard errors.
Statistical analyses
Data were analyzed statistically using SPSS® software, version 26.0, provided by IBM in Chicago, IL, United States. For quantitative variables adhering to a normal distribution, the presentation was in the form of mean ± standard deviation, with comparisons between groups executed via the t-test. Data not conforming to a normal distribution were depicted as the median (interquartile range), and the analysis of two independent samples was conducted using the Mann–Whitney U test for comparison. The area under the curve (AUC) of the receiver operating characteristic (ROC) served as a quantitative metric of diagnostic precision. All analytical results were deemed statistically significant with a P-value threshold of less than .05.
Analytical procedures were carried out utilizing the TwoSampleMR package (version 0.5.7) within the R computing environment (version 4.3.1). A heterogeneity test prefaced the evaluation of genetic IVs for MR estimates. With only two significant SNPs linked to D-dimer levels, multiple MR methods were employed, including inverse variance weighted (IVW), weighted mode-based, MR-Lasso, MR-Robust, and MR-Robust Adjusted Profile Score (MR-RAPS). The IVW method integrates all IVs, weighted by their variance, for a comprehensive effect size estimate. The weighted mode–based method, focusing on the most frequent causal estimate, assumes it reflects the true causal effect. MR-Lasso, addressing pleiotropy, applies penalization to reduce bias from invalid instruments. MR-Robust adjusts IV weights to account for heterogeneity and pleiotropic variants, providing a more accurate causal effect. MR-RAPS, managing genetic heterogeneity and invalid IVs, enhances robustness using a profile score for pleiotropy and instrument variability. A Leave-One-Out sensitivity analysis assessed the influence of individual SNPs on the overall estimate, enhancing the stability and reliability of the causal inference by identifying outliers and pleiotropic SNPs.
Results
Baseline characteristics
The mean age of the 466 patients was 68.7 ± 8.6 years. The median TPSA concentration of all patients included in the study was 16.0 ng/ml (8.8, 43.5); the median PV was 45.5 ml (32.7, 67.0); and the median PSAD was 0.38 ng/ml2 (0.18, 0.94). The median Fib level was 3.07 g/L (2.72, 3.60), while the median D-dimer level was 0.32 ug/ml (0.22, 0.56; Table 1)
Clinical characteristics of patients according to all cohort.

BPH, benign prostate hyperplasia; Fib, fibrinogen; PCa, prostate cancer; PSAD, prostate-specific antigen density; TPSA, total prostate-specific antigen.
Overall results
In this study comprising 466 patients, they were categorized into PCa and BPH groups based on biopsy pathology results, with baseline characteristics detailed in Table 1. Compared to the BPH group, patients diagnosed with PCa were older (P < .001), had smaller PV (P < .001), and exhibited higher levels of TPSA (P < .001), PSAD (P < .001), and D-dimer (P < .001). Nevertheless, the comparison of Fib levels between the two cohorts did not yield a statistically significant difference (P = .505).
Based on the Gleason score, patients were divided into csPCa and non-csPCa groups for an inter-group comparison. There were no significant variances noted in age, PV, and Fib levels across the two cohorts. However, the csPCa group exhibited higher levels of TPSA (P < .001), PSAD (P < .001), and D-dimer (P = .019; see Table 2 for details)
Subgroup analysis according to pathologic Gleason score.
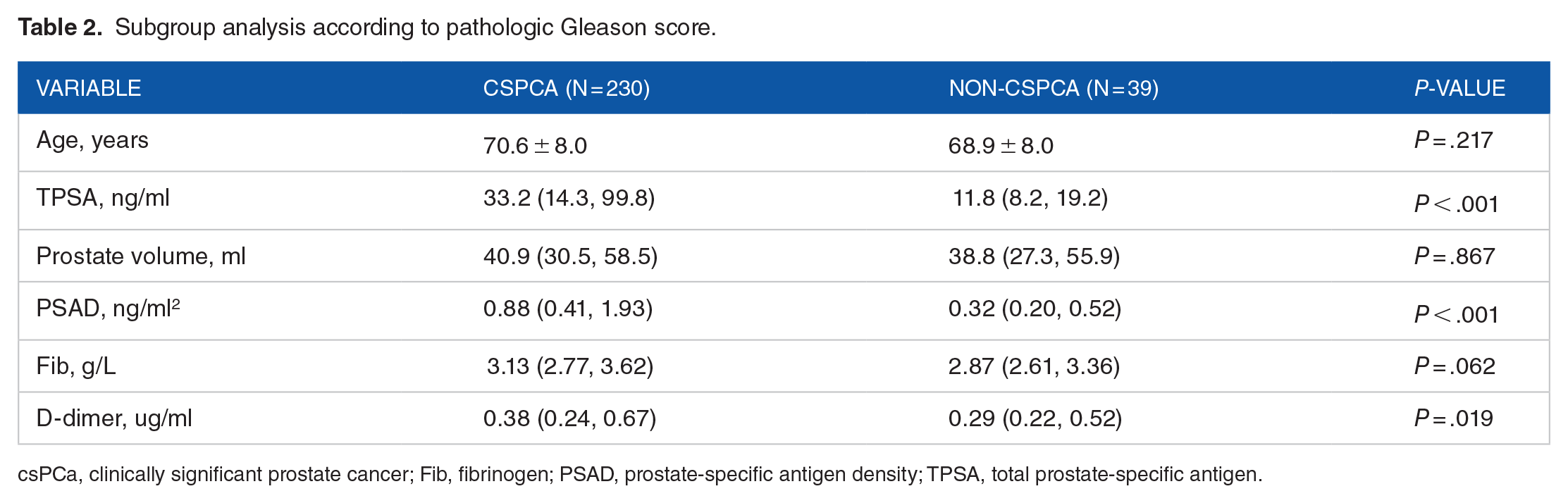
csPCa, clinically significant prostate cancer; Fib, fibrinogen; PSAD, prostate-specific antigen density; TPSA, total prostate-specific antigen.
Based on the analysis results, we selected TPSA, PSAD, and D-dimer for further ROC-AUC curve analysis to clarify the diagnostic value of each variable for csPCa. As shown in Figure 1, all variables demonstrated a predictive value for csPCa (all AUCs > 0.6). Among these, PSAD had the highest diagnostic value with an AUC of 0.772, followed by TPSA (AUC of 0.756) and D-dimer (AUC of 0.617).

Receiver operating characteristic (ROC) curves for the prediction of csPCa with various variables.
Subsequently, we combined these three variables to predict csPCa, yielding an AUC of 0.791 (95% confidence interval [CI]: 0.723-0.858), indicating improved predictive performance compared to the use of each variable alone, as shown in Figure 2.

Receiver operating characteristic (ROC) curves for combined prediction of csPCa using multiple variables.
Mendelian randomization
Based on the analysis of single-center clinical data, it was found that there is a significant difference in D-dimer levels between cancer and non-cancer patients, as well as between csPCa and non-csPCa patients. However, no significant differences were observed in Fib levels across these groups. Consequently, we have opted to conduct an MR study to explore the relationship between D-dimer and PCa, as illustrated in Figure 3.
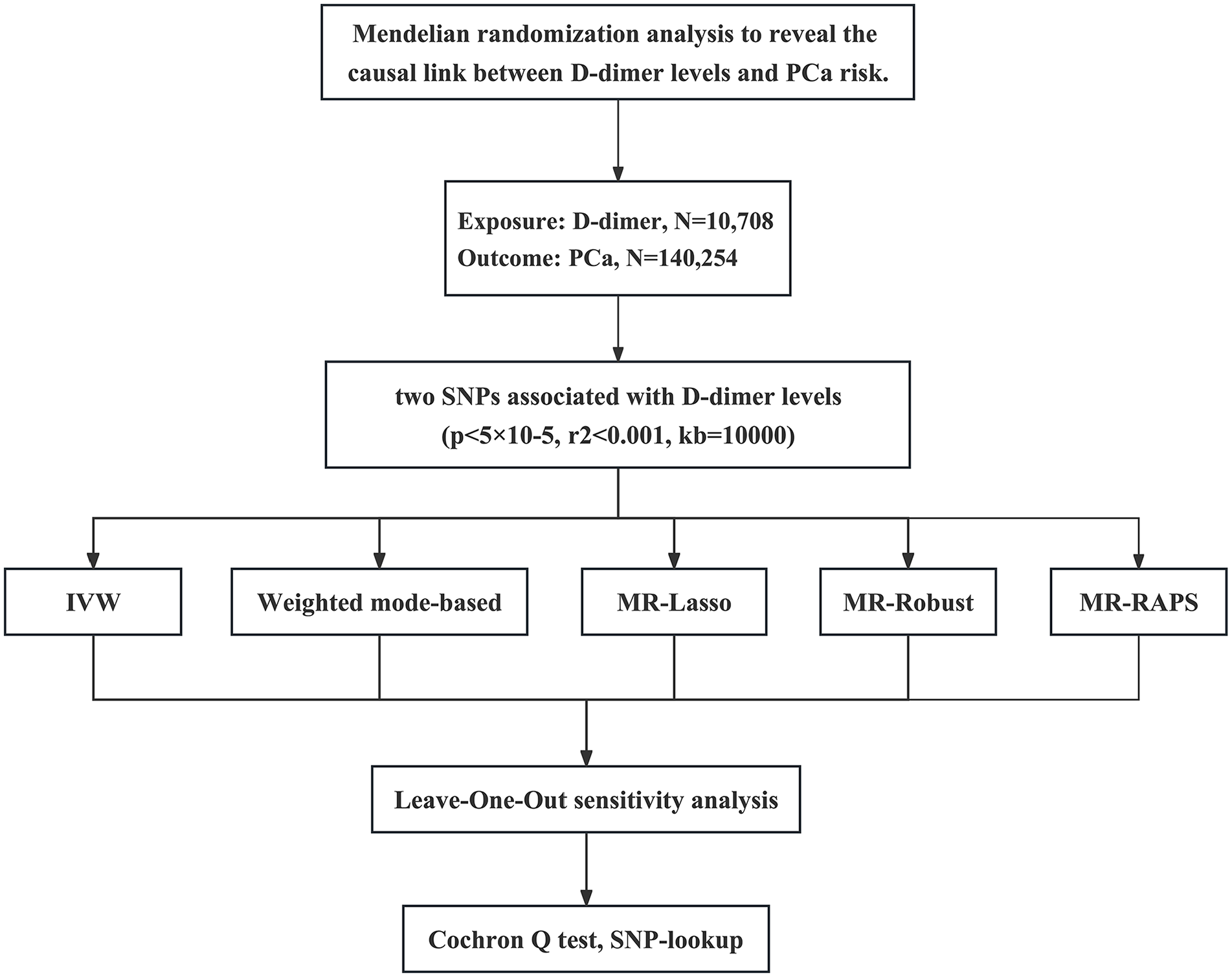
Workflow of Mendelian randomization study revealing causality from D-dimer on prostate cancer.
In our comprehensive MR study, we investigated the link between D-dimer concentrations and the likelihood of a PCa diagnosis, employing two IVs, SNPs rs75156689 and rs189262247. Due to the limitation of having only two SNPs, the present study could not employ the MR-Egger method. However, we utilized the online tool PhenoScanner V2 (www.phenoscanner.medschl.cam.ac.uk) designed for exploring human genotype-phenotype associations, to assess the potential pleiotropic effects for the underlying confounding factors. 20 The principal analysis utilized the IVW approach, which revealed an odds ratio (OR) of 1.81, indicating a significant association (95% CI: 1.48-2.21; P-value: 7.4 × 10−9). The robustness of this association was further substantiated through alternative MR approaches. The weighted mode-based method yielded an OR of 1.73 (95% CI: 1.27-2.36; P-value: 5.1 × 10−4), while MR-Lasso produced an OR of 1.81 (95% CI: 1.47-2.22; P-value: 1.8 × 10−8), paralleling the IVW findings. Furthermore, the MR-Robust method corroborated these results with an OR of 1.81 (95% CI: 1.57-2.08; P-value: 2.5 × 10−16), and the MR-RAPS method suggested an OR of 1.86 (95% CI: 1.33-2.60; P-value: 2.6×10-4), reinforcing the potential causal link. These findings are visually summarized in Figure 4, which presents a forest plot of the results across the different MR methodologies.
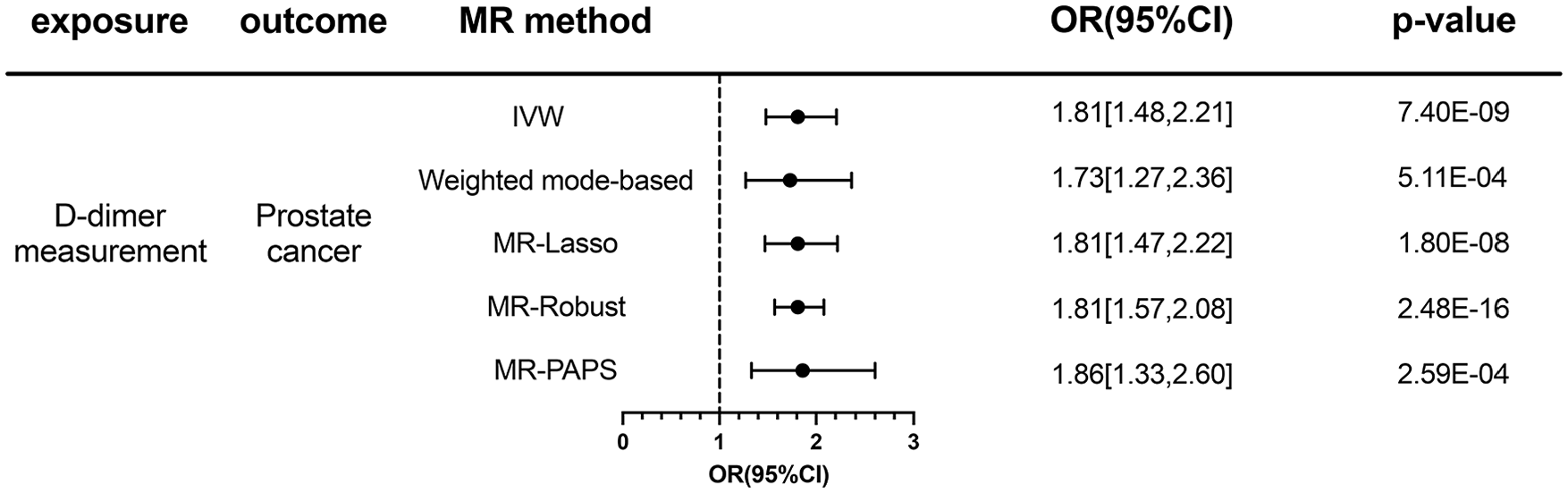
Association between D-dimer count and risk of prostate cancer using multiple MR methods.
To assess the consistency of the results and potential influence of individual SNPs, a leave-one-out sensitivity analysis was conducted. The outcomes, illustrated in Figure 5, show that the relationship between D-dimer concentrations and the risk for PCa is consistent throughout the analyses, with none of the individual SNPs predominating the findings.

Leave-one-out analysis for the causal effect of D-dimer on prostate cancer.
Considering the results of the heterogeneity test, a Cochran’s Q-value was determined to be 1.05, accompanied by a Q-test P-value of .304, indicating an absence of notable heterogeneity among the employed SNPs. This enhances the credibility of our findings, indicating a consistent effect size across the genetic instruments and strengthening the argument for a potential causal relationship.
Together, the findings from our comprehensive MR study indicate a significant causal link between higher D-dimer levels and a heightened probability of being diagnosed with PCa. This assertion is supported by the convergence of results from diverse MR methodologies and reinforced by rigorous sensitivity analyses.
Discussion
This study is a pioneering exploration into the relationship between peripheral blood coagulation parameters, specifically D-dimer levels, and PCa, using a combined approach of clinical data analysis and MR. In a retrospective analysis of 466 patients undergoing prostate biopsies, those with PCa demonstrated significantly elevated D-dimer levels compared to patients without cancer. A subgroup analysis after Gleason scoring revealed similar discrepancies in D-dimer levels. No significant variations in Fib levels were observed. These results suggest a potential link between increased D-dimer levels and the development and progression of PCa. A subsequent two-sample MR analysis provided support for a causal relationship between elevated D-dimer levels and increased PCa risk.
D-dimer is a degradation product of fibrin induced by plasmin, serving as a biomarker indicative of fibrinolysis activation. In cancer patients, the structural activation of the coagulation cascade due to pro-coagulant factors leads to the generation of thrombin, subsequently forming fibrin. Fibrin not only forms a protective barrier around malignant tumor cells, helping them evade endogenous defense mechanisms, but also fosters tumor angiogenesis, invasion, and metastasis. 21 The elevation of D-dimer levels in plasma may reflect the continuous metabolism of fibrin in the actively remodeling tumor stroma during the onset and progression of the tumor, generating more D-dimer as a result of degradation. 22 However, the precise mechanism underlying the association between D-dimer and PCa remains inadequately elucidated. Previous research has unveiled that elevated levels of D-dimer serve as prognostic markers for increased mortality risk across a variety of malignant tumors, including PCa. 23 Lei and colleagues’ study found a significant increase in plasma D-dimer concentrations among individuals with advanced PCa. Similarly, Alevizopoulos et al 10 found an association between D-dimer and higher tumor staging and grading. 11 Kohli et al identified a significant elevation of D-dimer across various stages of PCa and posited that this subclinical activation appears to be unrelated to other pre-thrombotic risk factors such as advancing age. 24 Furthermore, research conducted by investigators in Korea and Turkey also indicated a marked rise in plasma D-dimer concentrations in patients with PCa relative to those without cancer.22,25 This aligns with our research findings, and through further MR investigation, we surmise that an elevation in D-dimer may augment the risk of PCa.
Fib, a pivotal protein in human coagulation processes with a molecular weight of 340 kDa, has been reported to be associated with the onset and progression of various cancers. 26 However, the underlying biological mechanism of this association remains not fully elucidated. Drawing upon various preceding studies, Fib is thought to influence the initiation and advancement of PCa via a range of mechanisms. First, Fib deposition around solid tumors provides a stable structural framework for the extracellular matrix of tumor cells, potentially offering protection against immune system recognition; it can also serve as a scaffold for tumor cell growth factors, ultimately promoting tumor angiogenesis and proliferation. 27 Second, tumor cells express Fib receptors, facilitating the binding of Fib to tumor cells. This increases the adhesive capability of tumor cells within the vascular system of target organs, thereby promoting immune evasion and propelling tumor metastasis. 28 In PCa patients, tumor cells have the capacity to stimulate the liver for increased Fib production, owing to their ability to secrete pertinent cytokines. Concurrently, an endogenous synthesis of Fib takes place within the tumor cells. These dual pathways collectively contribute to the elevated levels of Fib observed in the plasma of PCa patients. 29 The study by Caine et al revealed a notable decline in Fib levels post-radical prostatectomy, which may indirectly substantiate the aforementioned viewpoints. 30 In addition, Thurner and colleagues observed that patients with PCa exhibiting raised plasma Fib levels tend to have a less favorable prognosis. 29 Conversely, Hong et al reported no significant difference in Fib levels when comparing PCa patients to non-cancer individuals, with no notable correlation between Fib and tumor staging or grading. 22 Simultaneously, a large-sample population study revealed no significant association between Fib and the risk of PCa. 12 Our findings are in concordance with those reported by Hong and Allin; however, our data emanate solely from a single center with a limited sample size. To elucidate the relationship between Fib and PCa further, multi-center studies with larger sample sizes are warranted.
The study’s limitations include potential selection bias from single-center, retrospective data and the one-time assessment of blood coagulation parameters before the biopsy. Another limitation is the difference in average age between the BPH group and the PCa group, along with an overall higher average age. Despite evidence suggesting that levels of D-dimer increase with advancing age, possibly due to age-related comorbidities, 10 we implemented stringent exclusion criteria to mitigate the inclusion of elderly patients with comorbidities that might affect D-dimer levels. In the MR analysis, a lenient threshold (P < 5 × 10−5) due to limited D-dimer GWAS data increased SNP inclusion but risked additional bias. Nonetheless, robust F-statistics (rs189262247, F = 16.5; rs75156689, F = 17.3) indicate the absence of weak instrument bias. In addition, according to the ALFA project, 1000Genomes project, Allele Frequency Aggregator, and Han Chinese Genomes Database (PGG. Han 2.0), the two selected SNPs are very rare in the Han population, making it difficult to confirm their potential causal link with PCa. This limitation could impact the generalizability of our findings, and further studies with larger and more diverse populations are necessary to validate these results. The limited SNP availability restricted certain MR techniques, such as MR-Egger, necessitating cautious result interpretation. Comprehensive sensitivity analyses and thorough consistency checks were conducted to mitigate these issues.
Despite the aforementioned limitations, we believe this investigation serves as a pioneering exploration, marking the first attempt to meld clinical data with MR to probe the relationship between peripheral coagulation indices and PCa. By advancing MR analysis on D-dimer, we have substantially mitigated the impact of confounding and reverse causality inherent in other types of observational studies. However, in this study, no significant difference in Fib was observed between the PCa and non-PCa cohorts. Future research endeavors should contemplate employing larger sample sizes and more stringent thresholds to validate our findings. The underlying mechanisms driving the association between PCa and peripheral coagulation parameters warrant further investigation.
Conclusion
In summary, this study demonstrates a significant increase in D-dimer levels in PCa patients compared to non-cancer individuals, with MR analysis supporting the role of elevated D-dimer in increasing PCa risk. However, Fib was not confirmed as a biomarker for PCa. Although preliminary, these findings suggest D-dimer could be a potential biomarker for PCa diagnosis. The exact mechanism linking D-dimer and PCa remains unclear, yet this research provides a solid foundation for further investigation.
Footnotes
Acknowledgements
We sincerely thank the genetics consortiums for sharing the original GWAS datasets.
Author Contributions
Conceptualization: Hao Ning, Jiaju Lyu, and Fei Wu; Methodology: Fei Wu and Feifan Liu; Formal analysis and investigation: Feifan Liu, Jianyu Wang, Delin Wang, Zhenlin Zhao, and Haihu Wu; Writing—original draft preparation: Feifan Liu and Yufeng Song; Writing—review and editing: Hao Ning and Fei Wu; Supervision: Jiaju Lyu. All authors have read and agreed to the published version of the manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shandong Provincial Nature Science Foundation (ZR2020QH240), the National Nature Science Foundation of China (NSFC82002719), the Clinical Medicine Innovation Program of Jinan City (202019125), the China Postdoctoral Science Foundation (2022M711977), the Clinical Medicine Innovation Program of Jinan City (202328070).
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
The datasets generated during the current study are available from the corresponding author on reasonable request.
Ethics Approval
The present study protocol was reviewed and approved by the Biomedical Research Ethic Committee of Shandong Provincial Hospital (NO. SWYX2023-503). Informed consent was waived by the Biomedical Research Ethic Committee of Shandong Provincial Hospital. The research was undertaken by the ethical standards of the 1964 Helsinki Declaration and its later amendments.
