Abstract
Objectives
To explore the causal associations between oxidative stress markers and the occurrence of various types of lymphomas.
Methods
This two-sample Mendelian randomization (MR) study employed summary data from genome-wide association studies of oxidative stress markers and various lymphoma types. Analysis was conducted using the inverse variance weighted, with confirmation using the weighted median, weighted mode, and MR-Egger regression methods. Heterogeneity, horizontal pleiotropy, outliers, and robustness were tested using the Cochran Q, MR-Egger regression, MR pleiotropy residual sum and outlier (MR-PRESSO), and leave-one-out methods.
Results
Genetically predicted myeloperoxidase (MPO) was causally associated with follicular lymphoma (odds ratio (OR) = 1.33, P = 0.0173). Uric acid showed a causal link to diffuse large B-cell lymphoma (DLBCL) (OR = 1.003, P = 0.043). Glutathione peroxidase (GPX) was associated with follicular lymphoma (OR = 1.16, P = 0.033). Catalase (CAT) was inversely associated with non-Hodgkin lymphoma (NHL) (OR = 0.8921), non-follicular lymphoma (OR = 0.8755), and follicular lymphoma (OR = 0.8364) (all P < 0.05). Glutathione-S transferase (GST) was inversely associated with NHL (OR = 0.9248) and follicular lymphoma (OR = 0.8313) (all P < 0.05). Heterogeneity was noted for uric acid and DLBCL, but no pleiotropy was detected; after outlier removal, the association with uric acid was non-significant.
Conclusions
The findings suggest causal relationships between oxidative stress markers and lymphoma risk, notably MPO and GPX with follicular lymphoma, and inverse associations for GST and CAT with different lymphoma types. Limitations include heterogeneity for some markers, requiring further validation.
Introduction
Lymphoma is a broad and heterogeneous family of cancers of the lymphatic system arising from lymphocytes, representing approximately 5% of all cancers. 1 The two main types of lymphoma are Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL). NHLs represent the 10th most common cause of cancer, with an estimated 553,010 new cases in 2022, and the 11th cause of cancer-related death, with 250,475 deaths in 2022. 2 HL is the 26th cause of cancer (82,409 new cases in 2022), and the 28th cause of cancer-related death (22,701 deaths in 2022). 2 The prognosis of newly diagnosed HL is generally excellent in patients who have received and responded to therapy.3,4 NHLs include several histological types: FL, diffuse large B-cell lymphoma (DLBCL), chronic lymphocytic leukemia/small lymphocytic lymphoma, mantle cell lymphoma, marginal zone lymphoma, extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue, mature T and natural killer (NK)-cell lymphoma, and adult T-cell leukemia-lymphoma, among others.5,6 The prognosis of NHL is highly variable and depends upon the exact subtype, ranging from indolent (e.g. follicular lymphoma (FL)) to very aggressive diseases (e.g. DLBCL).5,6
HL and NHL can be suspected in patients with painless persistent lymphadenopathy with or without B-symptoms (i.e., weight loss of >10%, night sweats, and/or fever). The diagnosis of HL and NHL is usually established based on blood test results, a biopsy of the lymph nodes and bone marrow, and immunophenotyping by flow cytometry and/or immunohistochemistry of the surgical specimen.3–6 Several blood parameters are disrupted in patients with lymphoma, including complete blood count, erythrocyte sedimentation rate, lactate dehydrogenase levels, liver and kidney function markers, and β2-microglobulin.7–9
Oxidative stress is involved in a wide variety of diseases, including cancer, cardiovascular diseases, diabetes, and neurological, kidney, respiratory, psychiatric, and gastrointestinal diseases. 10 Oxidative stress is involved in lymphoma pathogenesis because it participates in DNA, protein, and lipid damage, and develops somatic oncogenic mutations.11–13 Still, several biomarkers commonly evaluated in clinical practice, including markers of oxidative stress, could be used to screen for patients at risk of lymphoma, but their association with lymphoma is often biased by the confounding and risk of reverse causality often observed in epidemiological studies, 14 and their exact causality in relation to lymphoma remain unknown and probably differ among subtypes of lymphomas. 9 Determining biomarkers that could predict the occurrence of lymphoma could have public health importance for identifying and monitoring patients at risk. Still, lymphomas are uncommon, and a longitudinal study would require hundreds of thousands of patients followed over decades to reach an answer.
Genome-wide association studies (GWASs) revolutionized the field of genetic medicine by providing data on millions of genetic variations and their association with various phenotypes, especially for complex traits and diseases.15,16 Mendelian randomization (MR) is a statistical method based on the random distribution of alleles during gamete generation and embryo conception. MR is based on the associations of common single-nucleotide polymorphisms (SNPs) as instrumental variables (IVs) with different exposures and the association of the identified IVs with the outcomes to explore possible causal associations between exposures and outcomes (or diseases).17,18 Upon specific assumptions, MR studies can help determine causality without bias from confounders and reverse causality. 14 Two-sample MR uses the associations between SNPs and exposures and between SNPs and outcomes from different GWASs to combine them into a causality estimation.
Therefore, this study aimed to explore the causal associations between oxidative stress markers and the occurrence of various types of lymphomas. The results could help provide additional evidence for the screening of lymphoma in the population.
Materials and methods
Study design
This study used publicly available data from GWASs to investigate the causal association between oxidative stress and lymphoma (Figure 1). Because this study used publicly available anonymized data, no additional ethical approval was necessary. This study assumed that (a) the SNPs used as IVs for oxidative stress are associated with oxidative stress (relevance assumption); (b) there are no common causes between the SNPs and lymphoma (independence assumption); and (c) there are no independent pathways between the SNPs and lymphoma other than through oxidative stress (no horizontal pleiotropy assumption). 14
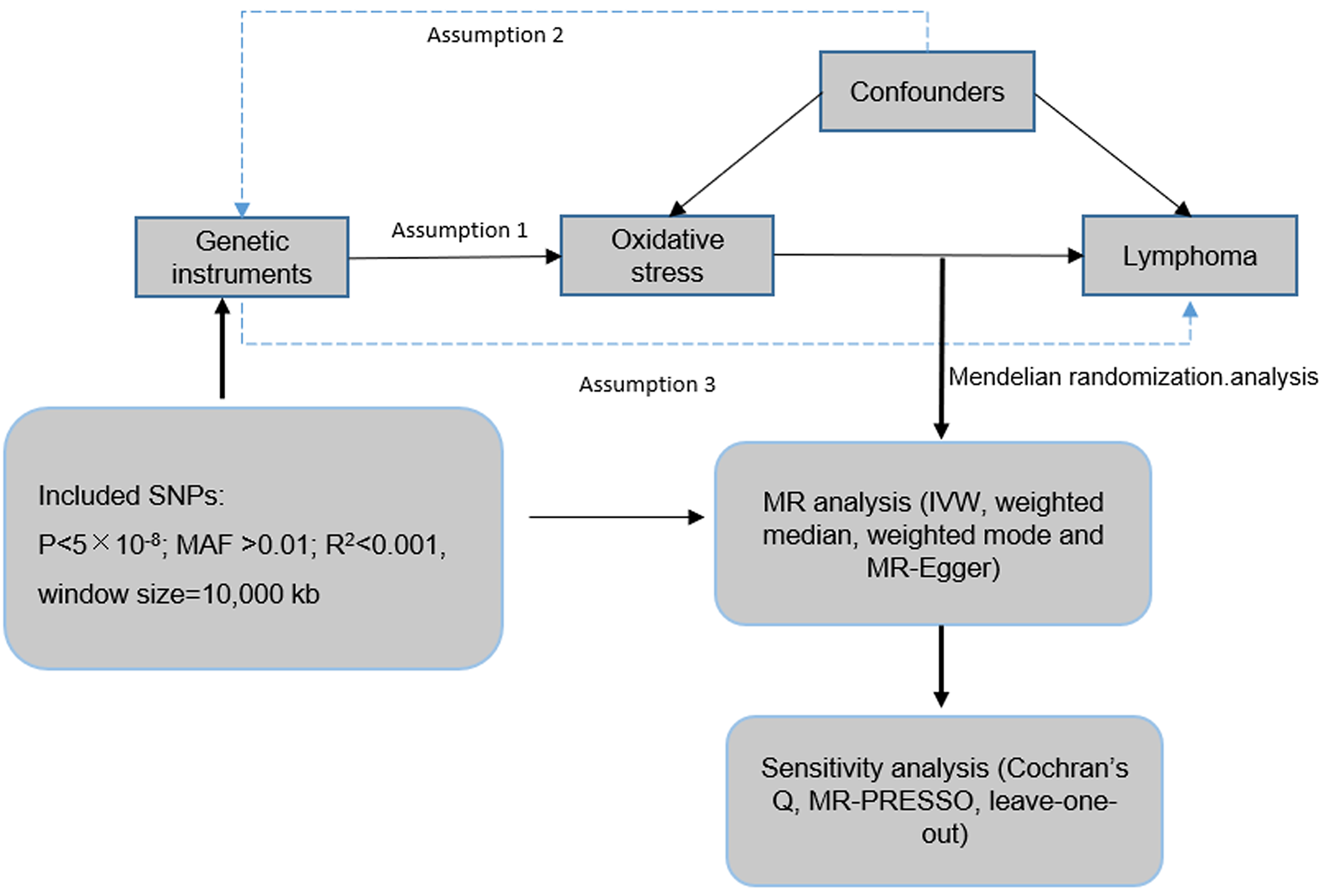
Schematic representation of the Mendelian randomization study.
Data source
The outcome datasets were obtained from the R12 cohort of the FinnGen Project, which is a GWAS of about half a million Finnish residents. The outcomes included HLs, NHLs, DLBCLs, FLs, non-FLs, mature T or NK cell lymphomas, and other unspecified NHLs (Supplementary Table S1). The exposure datasets related to oxidative stress were from published papers19–23 and the UK Biobank, a large GWAS of individuals of European ancestry living in the United Kingdom (Supplementary Table S1).
Instrumental variable selection
The IVs included in this study had to meet the following criteria. SNPs significantly associated with each exposure were initially selected at the conventional genome-wide significance level of P < 5 × 10−8, which is widely used in GWAS to control for multiple testing and to minimize false positive findings. However, for several exposures (cerebral dopamine neurotrophic factor, neurotrophin-3, neurotrophin-4, alpha-linolenic acid measurement, and beta-nerve growth factor levels), very few or no variants met this stringent threshold, which would have resulted in insufficient instrument strength and loss of statistical power in the MR analyses. To retain an adequate number of instruments while still focusing on reasonably strong and biologically plausible signals, a less stringent threshold of P < 5 × 10−6 was therefore applied, consistent with previous MR studies that have relaxed the genome-wide cut-off in settings with limited genome-wide significant variants.24–27 The SNPs with a minimum minor allele frequency of >0.01 were screened out. 28 Linkage disequilibrium (LD) between SNPs was removed based on R2 < 0.001 and a window size of 10,000 kb. 29 When a selected IV was not present in the summary data of the outcome, SNPs with high LD (R2 > 0.8) were searched with the IV as proxy SNPs for replacement. 30 The F-value was calculated for each SNP in the IV to assess the strength of the IV. The calculation formula was F = R2 × (N − 2)/(1 − R2), where R2 is the proportion of exposure variance explained by the SNP in the IV. The requirement for the F-value to exclude potential weak instrumental bias between the IV and the exposure factor was >10. 31
Mendelian randomization analysis
In the present study, the inverse variance weighted (IVW) method was used as the primary MR method to assess the causal association between exposure and the outcome risk by calculating the odds ratio (OR) and 95% confidence interval (CI). 32 IVW is the main method for MR interpretation, and it calculates the inverse variance of each SNP as weights and computes the weighted average of the effect sizes. 32 IVW is a random effect model that can accommodate a certain degree of heterogeneity. 32 In addition, the MR-Egger, 33 weighted median, 34 and weighted mode 35 were used to examine the robustness of IVW results. The MR-Egger method examines the presence of an intercept and provides an accurate causal effect estimate when considering pleiotropic bias. 33 In the weighted median method, half of the IVs are assumed to be valid for analyzing the causal association between the exposure and the outcome. 34 All analyses were performed using the “TwoSampleMR” package in R. 36 Visualization was performed using scatter plots and sensitivity analysis plots.
Sensitivity analysis
Sensitivity analyses were performed to detect potential heterogeneity and pleiotropy. Cochran's Q-test was used to assess heterogeneity among IVs. A Q-test P-value of >0.05 indicated low heterogeneity and that the estimates among IVs varied randomly and had a minimal impact on the IVW results. 37 Because of the risk of an influence of genetic variation heterogeneity on the estimation of association effects and to meet the third MR assumption, the MR-Egger regression method was used to determine the presence of horizontal pleiotropy. 37 The absence of pleiotropy was considered when the intercept term of the MR-Egger regression tended toward zero or lacked statistical significance. In addition, an MR pleiotropy residual sum and outlier (MR-PRESSO) analysis was used to detect potential outliers (i.e. SNPs with P < 0.05).37,38 The outlier SNPs were removed to recalibrate the causal associations and correct for horizontal pleiotropy. A leave-one-out analysis was used to assess the robustness and consistency of the results. 39
Results
Instrument variable selection
A total of 619 instrumental variables related to oxidative stress were extracted. The number of IVs ranged from 5 to 239, with all F statistics being >10, varying from 20.91 to 142,042.3 (Supplementary Table S2), indicating the reliable strength of the instrumental variables. The R2 values for all exposures varied between 0.00329 and 0.3390 (with the lowest value for retinol and the highest for total bilirubin) (Supplementary Table S2), suggesting that the genetic explanatory power differs across various exposures. Supplementary Table S3 shows the results of SNPs after exposure and outcome matching, including details of the SNPs that were not matched in the outcomes and the proxy SNPs (48 SNPs were not matched in the summary data, of which 18 proxy SNPs were found).
Mendelian randomization analysis results (significant analyses).
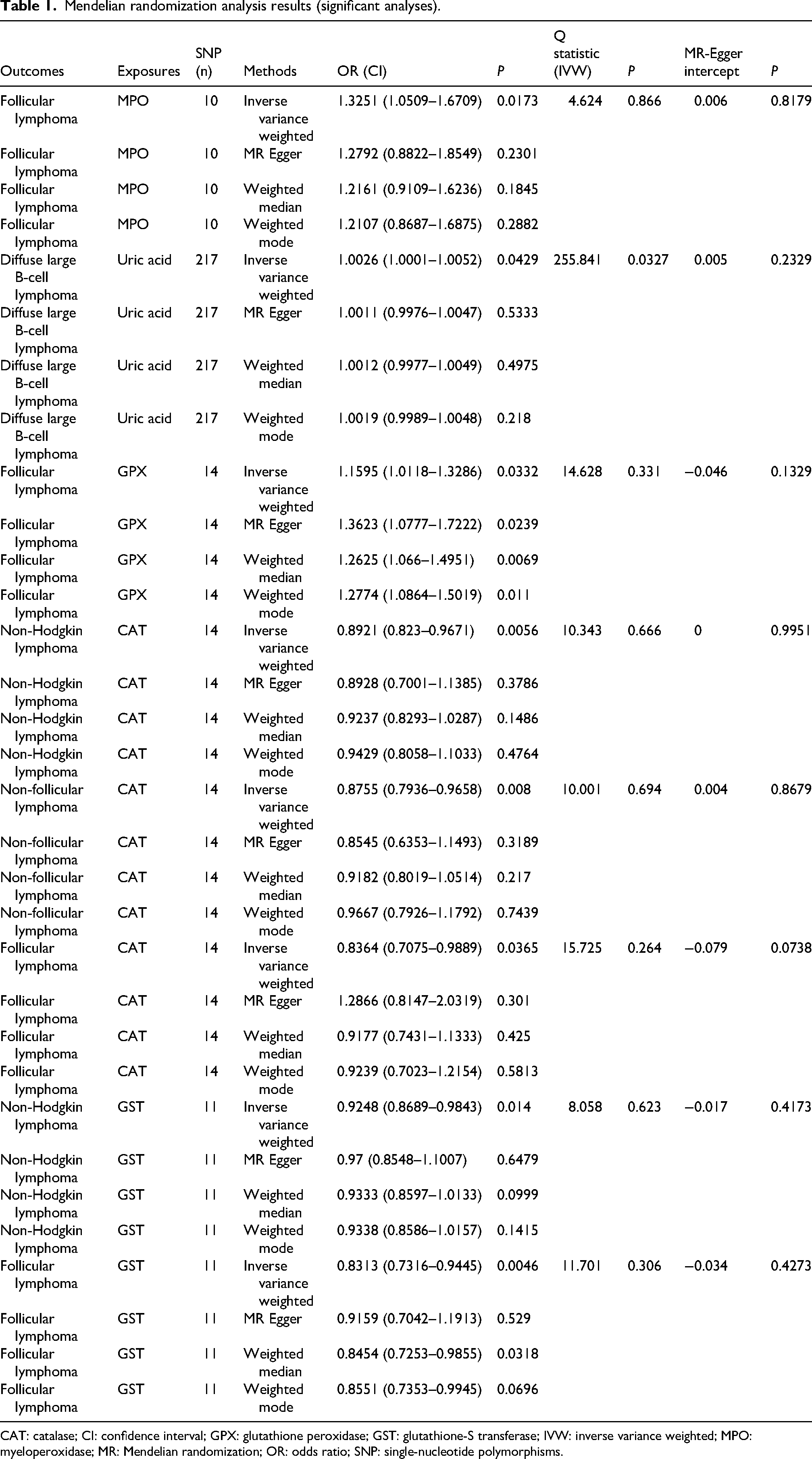
CAT: catalase; CI: confidence interval; GPX: glutathione peroxidase; GST: glutathione-S transferase; IVW: inverse variance weighted; MPO: myeloperoxidase; MR: Mendelian randomization; OR: odds ratio; SNP: single-nucleotide polymorphisms.
MR-PRESSO (significant analyses).
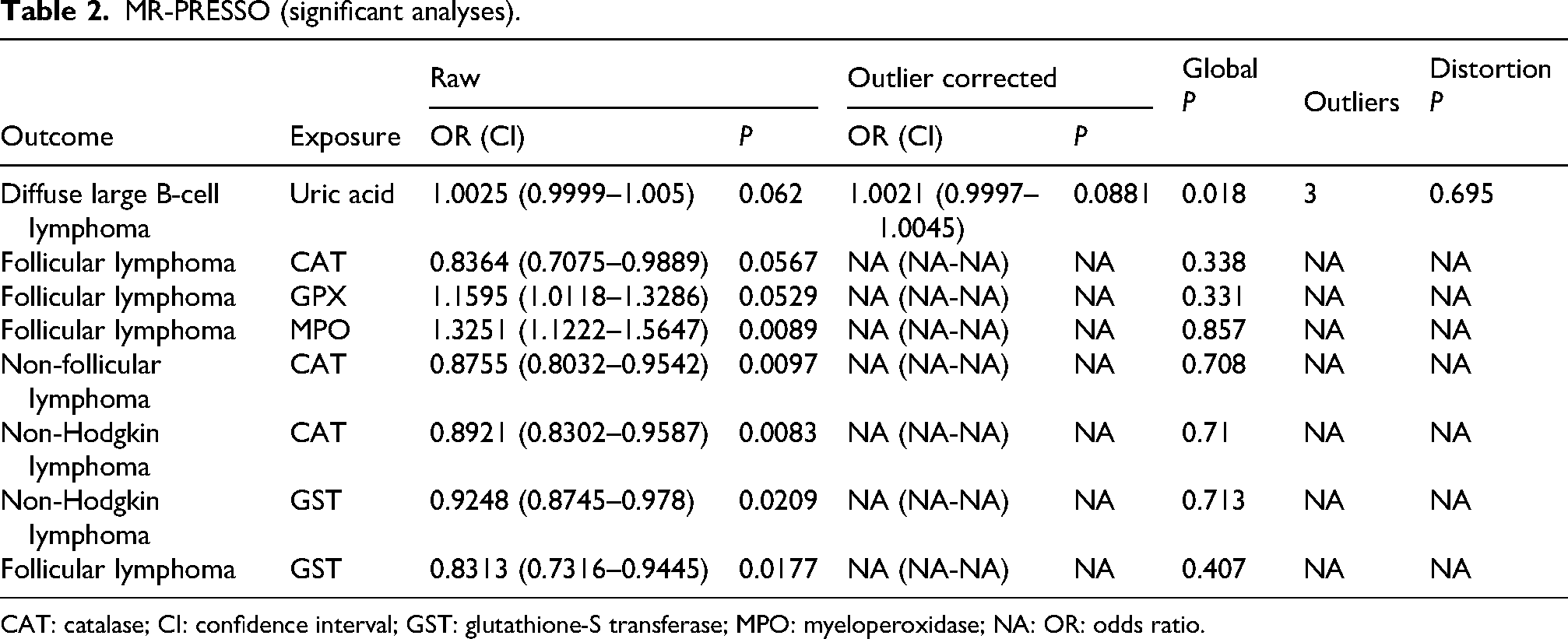
CAT: catalase; CI: confidence interval; GST: glutathione-S transferase; MPO: myeloperoxidase; NA: OR: odds ratio.
Mendelian randomization results
Genetically predicted myeloperoxidase (MPO) was causally associated with the risk of FL (OR = 1.3251, 95% CI: 1.0509–1.6709, P = 0.0173; other MR analyses P > 0.05) (Figures 2(a) and 3(a)). Uric acid was causally associated with DLBCL (OR = 1.0026, 95%CI: 1.0001–1.0052, P = 0.0429; other MR analyses P > 0.05) (Figures 2(b) and 3(b)). Glutathione peroxidase (GPX) was causally associated with FL (OR = 1.1595, 95% CI: 1.0118–1.3286, P = 0.0332; MR-Egger P = 0.0239; weighted median P = 0.0069; weighted mode P = 0.0110) (Figures 2(c) and 3(c)). CAT was causally associated with NHL (OR = 0.8921, 95% CI: 0.8230–0.9671, P = 0.0056; other MR analyses P > 0.05) (Figures 2(d) and 3(d)), non-FL (OR = 0.8755, 95% CI: 0.7936–0.9658, P = 0.0080; other MR analyses P > 0.05) (Figures 2(e) and 3(e)), and FL (OR = 0.8364, 95% CI: 0.7075–0.9889, P = 0.0365; other MR analyses P > 0.05) (Figures 2(f) and 3(f)). GST was causally associated with NHL (OR = 0.9248, 95% CI: 0.8689–0.9843, P = 0.014; other MR analyses P > 0.05) (Figures 2(g) and 3(g)) and FL (OR = 0.8313, 95% CI: 0.7316–0.9445, P = 0.0046; MR-Egger P = 0.5290; weighted median P = 0.0318; weighted mode P = 0.0696) (Figures 2(h) and 3(h)) (Table 1). Among the statistically significant IVW results, Cochran's Q-test revealed heterogeneity for only the analysis of uric acid and DLBCL (P = 0.033). No horizontal pleiotropy was observed using the MR-Egger regression analysis (Table 1 and Figure S1(a) to (g)). The leave-one-out analyses did not reveal SNPs driving the results (Figure S2(a) to (g)). The MR-PRESSO analysis revealed three outliers for the analysis of uric acid and DLBCL. After removing the three outliers, the causal association was no longer statistically significant (Table 2).
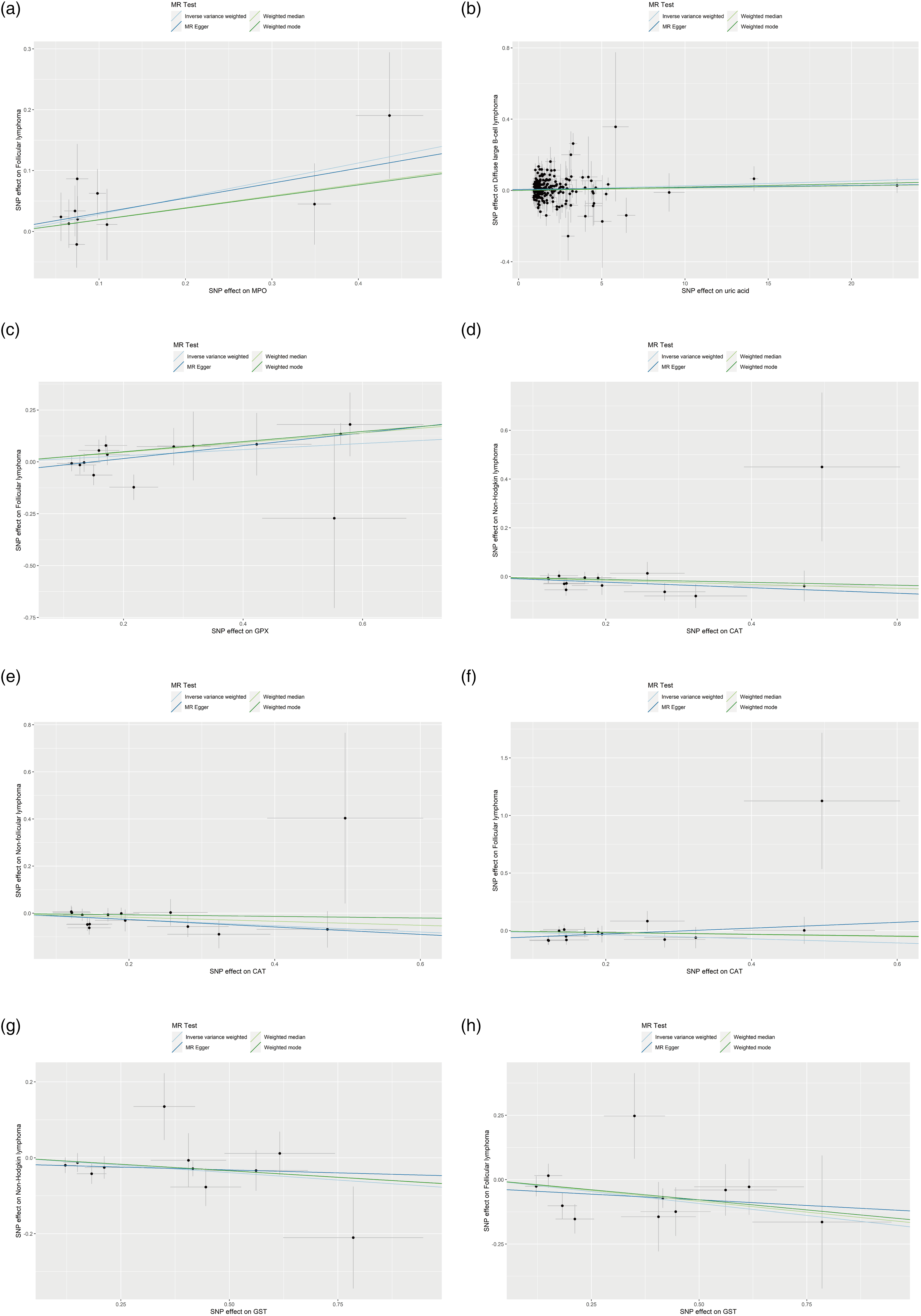
Scatter plots for the Mendelian randomization analyses of (a) myeloperoxidase (MPO) and follicular lymphoma, (b) uric acid and diffuse large B-cell lymphoma (DLBCL), (c) glutathione peroxidase (GPX) and follicular lymphoma, (d) catalase (CAT) and non-Hodgkin lymphoma (NHL), (e) CAT and non-follicular lymphoma, (f) CAT and follicular lymphoma, (g) glutathione-S transferase (GST) and NHL, and (h) GST and follicular lymphoma.
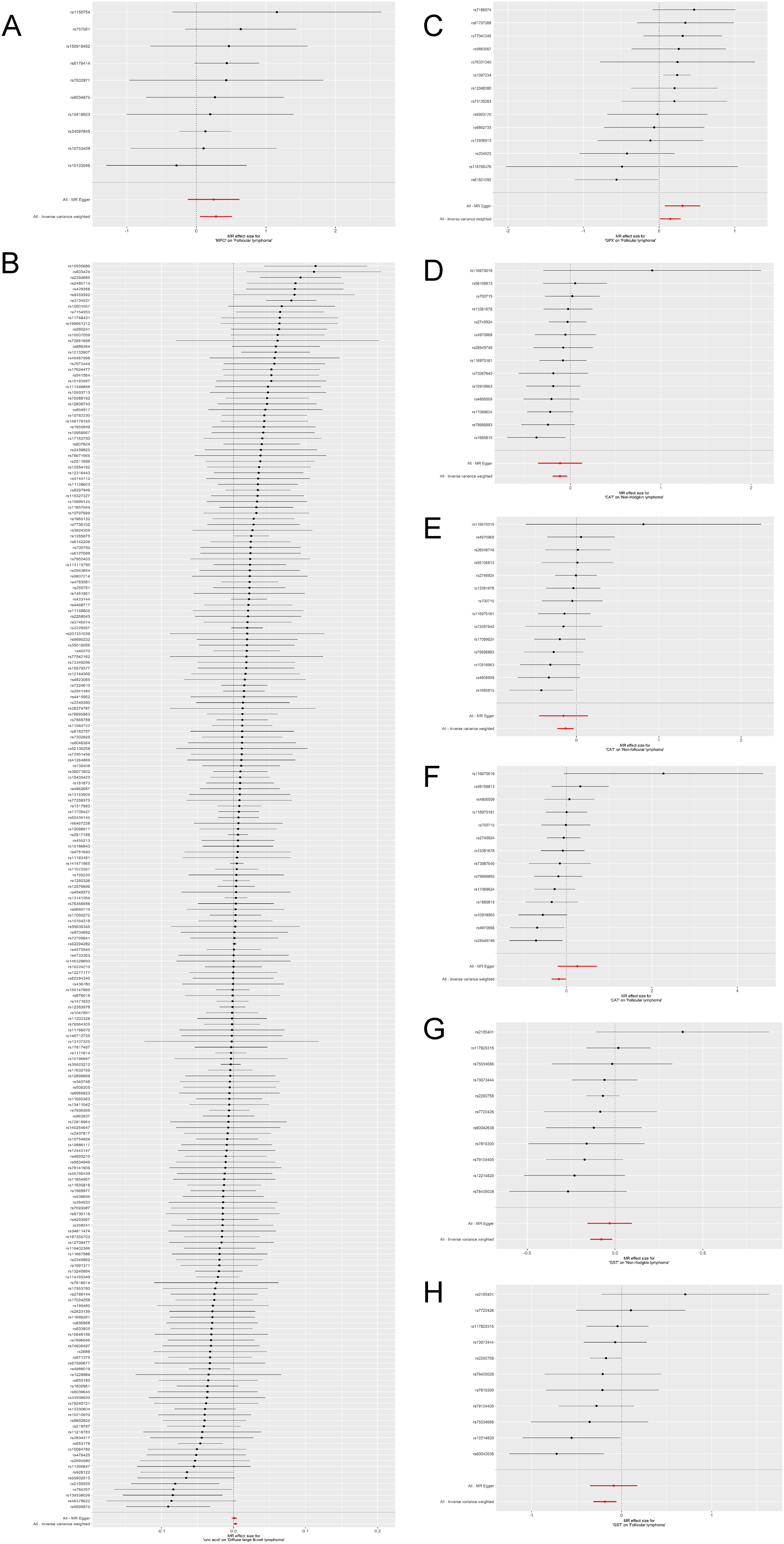
Forest plots for the Mendelian randomization analyses of (a) myeloperoxidase (MPO) and follicular lymphoma, (b) uric acid and diffuse large B-cell lymphoma (DLBCL), (c) glutathione peroxidase (GPX), and follicular lymphoma, (d) catalase (CAT) and non-Hodgkin lymphoma (NHL), (e) CAT and non-follicular lymphoma, (f) CAT and follicular lymphoma, (g) glutathione-S transferase (GST) and NHL, and (h) GST and follicular lymphoma.
All other analyses had negative IVW results (Supplementary Table S4). The Cochran Q test showed heterogeneity in the analysis of 18 negative IVW results. No horizontal pleiotropy was observed using the MR-Egger regression analysis (Supplementary Table S5). The MR-PRESSO analysis revealed that removing the outliers did not change the negative results (Supplementary Table S6).
Discussion
This two-sample MR study explored the causal associations between oxidative stress markers and the occurrence of various types of lymphomas. The results suggested direct causal associations between MPO and FL and GPX and FL, and inverse causal associations between GST and NHL and FL, and between CAT and NHL, non-FL, and FL. These results suggest that oxidative stress is involved in lymphoma pathogenesis. Monitoring the blood levels of those markers could eventually assist in screening patients and risk assessment, but additional studies are necessary before clinical application.
Oxidative stress is involved in cancer pathogenesis, 10 including lymphoma, due to DNA, protein, and lipid damage and the occurrence of somatic oncogenic mutations.11–13 Unfortunately, the available data are mainly from epidemiological studies, which are often biased by confounders and reverse causality. 14 In the present study, the exposures MPO, GPX, GST, and CAT showed causal associations with various types of lymphoma. MPO is an enzyme that contributes to oxidative stress and inflammation. MPO is found in neutrophils and monocytes and is a key part of the innate immune system. MPO produces reactive oxygen species (ROS) and reactive nitrogen species, which can damage cells and tissues. 40 MPO plays a role in oxidative stress and cancer through DNA damage and promotion of tumor growth and progression. 41 GPX is an antioxidant enzyme that protects cells from oxidative stress by reducing hydrogen peroxide and other hydroperoxides to water. 42 Despite its antioxidant role, GPX appears to have dual roles in cancers and can act either as a suppressor or promoter, and is upregulated in lymphoma. 43 Those known properties of MPO and GPX support the direct causal associations of MPO and GPX with FL observed in the present study. On the other hand, GSTs are a group of enzymes that protect cells from oxidative stress by binding glutathione to toxic molecules. This process makes the molecules less toxic and easier for the cell to remove.44–46 GST protects DNA from oxidative damage that can lead to DNA mutations and carcinogenesis. 45 On the other hand, once cancer has developed, GST appears to promote cancer cell survival, proliferation, and drug resistance. 47 CAT is an antioxidant enzyme that helps to neutralize oxidative stress by breaking down hydrogen peroxide into water and oxygen and is a key part of the body's defense against oxidative stress.48,49 CAT also possesses a dual role in cancer, being protective before cancer development and being harmful once cancer develops.50,51 Nevertheless, the present study examined the genetically predicted risk of lymphoma development, and both GST and CAT appeared protective against lymphoma development.
Still, lymphoma is a very heterogeneous group of diseases. 1 Although a wide variety of oxidative stress markers were tested in the present study, only a few showed associations with a small number of lymphoma types. Disease pathogenesis is often influenced by a combination of genetic and environmental factors.52,53 Therefore, in the presence of negative results in MR studies, environmental factors should be investigated. Negative MR results do not mean that there is no link between exposure and outcome. The absence of association can be because the IVs are insufficient to model the effect of exposure on the outcome, which is a known limitation of MR.
In this study, many oxidative stress-related markers showed no evidence of a causal association with specific lymphoma subtypes. On the one hand, if the genetic instruments are sufficiently strong and the MR assumptions are reasonably satisfied, such results may indicate that lifelong differences in these biomarkers do not materially influence lymphoma risk, at least within the detectable effect size range of our design.54,55 On the other hand, several methodological limitations of MR could also contribute to the absence of detectable effects, including limited power for modest or subtype-specific effects, weak or sparse instruments, measurement error in the underlying GWAS, and residual horizontal pleiotropy, all of which tend to bias estimates toward the null.27,33,56 Moreover, oxidative stress biology is strongly shaped by environmental and lifestyle exposures (such as chemical solvents, air pollution, and other pro-oxidant agents), and accumulating evidence links these exposures—via oxidative damage and chronic inflammation—to lymphomagenesis.57,58 Because conventional MR using germline variants cannot fully capture context-specific, time-varying, or exposure-intensity–dependent effects, the absence of an MR signal should be interpreted as “no association detected in this genetic framework” rather than definitive evidence of no association, and future work integrating detailed environmental exposure data with genetic instruments is needed to clarify these complex relationships.54,59
In the analysis of genetically predicted uric acid and DLBCL, we observed substantial heterogeneity and detected several outlier variants using the MR-PRESSO framework. Biologically, this heterogeneity is plausible because uric acid metabolism is tightly linked to a broad spectrum of cardiometabolic disorders and chronic inflammatory pathways, reflecting a complex and pleiotropic genetic architecture.60–62 At the same time, DLBCL is a highly heterogeneous lymphoma with distinct metabolic subtypes and diverse immune microenvironmental states, and multiple studies have highlighted extensive metabolic and immunologic reprogramming in DLBCL cells.63–65 Consequently, some MR-PRESSO outlier SNPs may represent true horizontal pleiotropy; for example, variants that influence inflammatory, metabolic, or obesity-related traits in addition to uric acid, whereas others could reflect technical artifacts such as batch effects or subtle differences in genotyping, imputation, or sample structure between the exposure and outcome GWAS.38,66 Notably, the association between uric acid and DLBCL was no longer statistically significant after removal of MR-PRESSO outliers, and the causal effect estimates were attenuated, indicating that the signal is either weak or sensitive to pleiotropic instruments and should therefore be interpreted with particular caution.
FL being the only subtype associated with MPO and GPX can be framed as biologically plausible because FL is a germinal center (GC)-derived lymphoma that depends heavily on redox-regulated survival and selection signals within a ROS-rich microenvironment, whereas other B-cell lymphoma subtypes are more strongly driven by alternative oncogenic pathways and may be less dependent on MPO/GPX-mediated oxidative balance.11,67 FL arises from GC B cells (centrocytes/centroblasts), which undergo intense proliferation, somatic hypermutation, and class-switch recombination in a structurally and functionally specialized GC microenvironment. These processes generate high levels of ROS, necessitating tight control of oxidative stress and redox homeostasis to allow survival of GC B cells that carry extensive DNA damage yet avoid catastrophic apoptosis.11,67,68 MPO, predominantly expressed in myeloid cells within the GC niche, contributes to local generation and modulation of ROS and hypochlorous species, shaping B-cell selection, antigen presentation, and inflammatory signaling that can influence GC B-cell survival and clonal evolution. GPX family members, particularly GPX1/GPX4, detoxify hydrogen peroxide and lipid peroxides, buffering oxidative damage and supporting anti-apoptotic and drug-resistance programs in B-cell malignancies; polymorphisms reducing GPX1 activity have been linked to increased mature B-cell lymphoma risk, with signals enriched in FL, and GPX overexpression has been implicated in therapy resistance and ferroptosis suppression in aggressive lymphomas.11,68–71 Because FL cells remain GC-like and continue to rely on GC-style redox regulation and microenvironmental support, genetically determined variation in MPO/GPX activity may more directly perturb the homeostasis, selection thresholds, and survival of GC B cells that give rise to FL. In contrast, subtypes such as DLBCL, mantle cell lymphoma, and marginal zone lymphoma are frequently driven by alternative pathogenic programs (e.g. NF-κB activation, BCR signaling dependence, chronic antigenic stimulation, or immune evasion), and although oxidative stress pathways are still relevant for their biology and prognosis, their initiation and maintenance may be less specifically governed by MPO/GPX-mediated redox balance, attenuating subtype-specific genetic associations for these enzymes.11,67–69
This study used a two-sample MR design and GWAS data from hundreds of thousands of patients. On the other hand, the study also had limitations. The present study was based on GWAS summary statistics derived from populations of European ancestry, which substantially constrains the generalizability of the findings to non-European groups. Because allele frequencies, linkage disequilibrium structure, environmental exposures, and gene-environment interactions can differ across ancestral backgrounds, the effect estimates and inferred genetic architecture observed here may not accurately reflect risk patterns in other populations, and any extrapolation should be made with caution. Future studies are needed to replicate and extend these results in large, ancestrally diverse cohorts or in cohorts of different ancestry (e.g. Asians) to validate the robustness of the conclusions and to enhance their relevance for global populations. Second, several SNPs could be selected as IVs, affecting the causal associations. Third, for some exposures, only a small number of genome-wide significant variants were available, so a more relaxed significance threshold had to be adopted to select IVs to retain sufficient statistical power. This strategy inevitably increases the risk of including weaker or potentially invalid instruments, which could bias effect estimates toward the null or introduce pleiotropy, although it was mitigated by LD clumping and applying robust sensitivity methods such as MR-Egger, weighted median, and related approaches. Alternative approaches specifically developed for settings with many weak instruments (e.g. MR-RAPS, debiased IVW or Egger-type estimators, and methods that explicitly model pleiotropy) exist but were not implemented here, and future work should evaluate whether these techniques materially change the observed associations.
In conclusion, this study suggests direct causal associations between MPO and FL, and GPX and FL, and inverse causal associations between GST and NHL and FL, and between CAT and NHL, non-FL, and FL. These results suggest that oxidative stress is involved in lymphoma pathogenesis. Monitoring such biomarkers could eventually help lymphoma risk stratification. Future studies could support the public health importance of such biomarkers.
Supplemental Material
sj-tif-1-jbm-10.1177_03936155261428137 - Supplemental material for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study
Supplemental material, sj-tif-1-jbm-10.1177_03936155261428137 for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study by Bin Zhu and Wenli Zhao in The International Journal of Biological Markers
Supplemental Material
sj-tif-2-jbm-10.1177_03936155261428137 - Supplemental material for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study
Supplemental material, sj-tif-2-jbm-10.1177_03936155261428137 for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study by Bin Zhu and Wenli Zhao in The International Journal of Biological Markers
Supplemental Material
sj-docx-3-jbm-10.1177_03936155261428137 - Supplemental material for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study
Supplemental material, sj-docx-3-jbm-10.1177_03936155261428137 for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study by Bin Zhu and Wenli Zhao in The International Journal of Biological Markers
Supplemental Material
sj-docx-4-jbm-10.1177_03936155261428137 - Supplemental material for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study
Supplemental material, sj-docx-4-jbm-10.1177_03936155261428137 for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study by Bin Zhu and Wenli Zhao in The International Journal of Biological Markers
Supplemental Material
sj-xlsx-5-jbm-10.1177_03936155261428137 - Supplemental material for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study
Supplemental material, sj-xlsx-5-jbm-10.1177_03936155261428137 for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study by Bin Zhu and Wenli Zhao in The International Journal of Biological Markers
Supplemental Material
sj-docx-6-jbm-10.1177_03936155261428137 - Supplemental material for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study
Supplemental material, sj-docx-6-jbm-10.1177_03936155261428137 for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study by Bin Zhu and Wenli Zhao in The International Journal of Biological Markers
Supplemental Material
sj-docx-7-jbm-10.1177_03936155261428137 - Supplemental material for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study
Supplemental material, sj-docx-7-jbm-10.1177_03936155261428137 for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study by Bin Zhu and Wenli Zhao in The International Journal of Biological Markers
Supplemental Material
sj-docx-8-jbm-10.1177_03936155261428137 - Supplemental material for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study
Supplemental material, sj-docx-8-jbm-10.1177_03936155261428137 for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study by Bin Zhu and Wenli Zhao in The International Journal of Biological Markers
Supplemental Material
sj-docx-9-jbm-10.1177_03936155261428137 - Supplemental material for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study
Supplemental material, sj-docx-9-jbm-10.1177_03936155261428137 for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study by Bin Zhu and Wenli Zhao in The International Journal of Biological Markers
Supplemental Material
sj-docx-10-jbm-10.1177_03936155261428137 - Supplemental material for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study
Supplemental material, sj-docx-10-jbm-10.1177_03936155261428137 for Causal association between oxidative stress and lymphomas: A two-sample Mendelian randomization study by Bin Zhu and Wenli Zhao in The International Journal of Biological Markers
Footnotes
Acknowledgments
Not applicable.
Ethical considerations
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Funding
The authors disclosed the receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by Shanghai Fengxian District Second Batch of Clinical Treatment Center Construction Project under Grant [number fxlczlzx-a-202105].
Authors’ contributions
Bin Zhu carried out the studies, participated in collecting data, and drafted the manuscript. Bin Zhu and Wenli Zhao performed the statistical analysis and participated in its design. Bin Zhu participated in the acquisition, analysis, or interpretation of data and drafted the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
All data generated or analyzed during this study are included in this published article.
Supplemental material
Supplemental material for this article is available online.
