Abstract
Background:
The Node Reporting and Data System (Node-RADS) is a recently proposed classification system for the categorization of lymph nodes in radiological images. This study was conducted to retrospectively evaluate the diagnostic accuracy of the Node-RADS score for metastatic cervical lymph nodes on magnetic resonance imaging (MRI) of patients with nasopharyngeal carcinoma (NPC).
Methods:
We retrospectively analyzed cervical lymph nodes of NPC cases. Two radiologists independently evaluated each lymph node on the MRI scans using Node-RADS. Interobserver agreement between 2 radiologists for Node-RADS score assessment was evaluated by linear weighted kappa statistics. The correlation between metastasis and the Node-RADS score of each lymph node was analyzed using multivariate regression analysis. To investigate the diagnostic performance of the Node-RADS score, we further conducted receiver operating characteristic curve analysis. Correspondently, the sensitivity, specificity, positive predictive value, and negative predictive value of each different cutoff (>1, >2, >3, and >4) were computed.
Results:
In all, 119 patients with NPC were assessed, including 203 cervical lymph nodes consisting of 140 (69%) of 203 metastatic and 63 (31%) of 203 benign. The kappa agreement between the 2 readers for the Node-RADS score was 0.863 (95% CI = 0.830-0.897, P < .001). Node-RADS score on MRI scan was shown to be an independent predictive factor of lymph node metastasis after multivariate regression analysis (odds ratio [OR] = 6.745, 95% CI = 3.964-11.474, P < .001). Node-RADS achieved an area under the curve (AUC) of 0.950 (95% CI = 0.921-0.979) in diagnosing metastatic lymph nodes. When Node-RADS >2 was identified as the best cutoff based on balanced values, the sensitivity and positive predictive value were 0.92 and 0.94, respectively.
Conclusions:
Our study suggests that the Node-RADS score has high accuracy in predicting NPC cervical lymph node metastasis. Nevertheless, this conclusion requires confirmation in a larger cohort of patients with NPC.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is a highly prevalent cancer of the head and neck in South China and Southeast Asian countries. 1 The early stage of NPC usually has no obvious symptoms, and patients are often not detected in time. At the onset of diagnosis, most patients have cervical lymph node (CLN) metastases. A meta-analysis of CLN metastasis involving 2920 cases of NPC showed that the incidence of involvement of levels II, III, IV, and V, and retropharyngeal lymph nodes are 70%, 45%, 11%, 27%, and 69%, respectively. 2 The metastatic status of the CLNs is an essential indicator in the staging of NPC and has crucial implications for the formulation of treatment plans, such as induction chemotherapy and target volume delineation.3,4
Cervical lymph nodes in NPC are primarily evaluated by imaging, including ultrasound, computed tomography (CT), positron emission tomography (PET), and magnetic resonance imaging (MRI). Clinically, MRI is the most preferred method for head and neck soft tissue evaluation since it has high soft tissue resolution and efficiency in detecting metastatic CLNs.5,6 Current MRI diagnostic criteria for CLN metastasis of NPC include nodal short-axis length, necrosis in the center or rim enhancement, and extra-nodal neoplastic spread.7,8 However, when interpreting an MRI of NPC, borderline-sized lymph nodes without necrosis in the center or extra-nodal neoplastic spread always remain a difficulty in diagnosing CLNs for the radiologist.
Recently, Elsholtz et al 9 described a novel scoring system called Node Reporting and Data System (Node-RADS) based on CT and MRI to diagnose cancer lymph node metastasis. This scoring system includes size and configuration dimensions to perform imaging evaluation of lymph node infiltration with comprehensive scores. This scoring system has no restrictions on tumor type or anatomic location. Node-RADS has shown encouraging diagnostic performance in prostate, lung, and bladder cancer since it launched in 2021.10 -12 However, no previous report has explored its role in NPC. We assumed that higher Node-RADS scores are correlated with a higher risk of CLN metastasis. Therefore, this study reviewed MRI of patients with NPC undergoing core-needle biopsy or lymphadenectomy and examined the comprehensive diagnostic accuracy of Node-RADS in NPC.
Methods
Research design
This study had the approval of the Research Ethics Committee on Biomedical Research (RECBR), West China Hospital of Sichuan University (no. 2023-912, 2023.06.02). In this study, patients with NPC who underwent core-needle puncture biopsy or lymph node dissection in our hospital from 2012 to 2022 were retrospectively analyzed. Those with a pathological diagnosis of NPC CLN metastasis were enrolled as the positive group. We included lymph node biopsy or dissection with pathologic confirmation of benign nodes as the negative group. All of these patients underwent pretreatment MRI. Patients without available MRI scans and those with unavailable data were excluded.
Magnetic resonance imaging scan examination and Node-RADS assessment
Head and neck T1- and T2-weighted MRI images (T1WI and T2WI) were acquired for all patients enrolled in this study using a 3.0T scanner (Philips, Eindhoven, the Netherlands) with a slice thickness of 3 mm (T1WI echo time (TE)/repetition time (TR)/inversion time (TI) = 2.950/6.600/0.000 ms; T2WI TE/TR/TI= 95.360/4078.000/0.000 ms). The manufacturer’s recommended acquisition parameters were used.
Magnetic resonance imaging was retrospectively assessed by a reader with 10-year MRI experience. The Node-RADS scoring system was used to evaluate suspicious CLNs. 9 Scoring was performed on T2-weighted axial images by 1 reader with 8 years and 1 reader with 5 years of experience in MRI analysis. The Node-RADS scores were recorded separately by 2 notetakers to ensure that the raters rated the same nodes. Size and configuration were the 2 main imaging findings evaluated. Cervical lymph node size was considered enlarged if the short axis exceeded 10 mm. The texture was classified as homogeneous, heterogeneous, focal, or gross necrosis for configuration purposes. The border was described as either smooth or irregular. The shape was classified as kidney bean with fat hilus or spherical without fat hilus. Briefly, classification categories range from 1 to 5, reflecting how likely a malignant tumor is as follows: very low, low, equivocal, high, and very high, respectively. The final lymph node category resulted from these characteristics (Figure 1). The Node-RADS results were compared with the pathologic results.

Explanation of the Node-RADS scoring system, an example. Node-RADS indicates Node Reporting and Data System.
Statistical analysis
All statistical analysis in this study was done on a lymph nodal basis. We evaluated the interobserver consistency of the Node-RADS score. The senior radiologist’s assessment was used for subsequent statistical analyses. The evaluated score of each node was matched to the corresponding pathologic results. We analyzed the relationship between CLN Node-RADS score and CLN metastasis by establishing univariate and multivariate logistic regression models. The diagnostic capabilities of Node-RADS for CLN metastasis were then described by area under the curve (AUC) of receiver operating characteristic (ROC) analysis. Correspondently, sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of different Node-RADS score cutoff values (>1, >2, >3, >4) were estimated. All tests were 2-sided with a P < .05 considered to be statistical significance. All statistical and graphics analyses were carried out using R 4.2.3.
Results
Patient population
Overall, 119 patients with NPC who had confirmed diagnosis via pathological examination between July 2012 and September 2022 were included. All cases had MRI of the head and neck before induction chemotherapy followed by concurrent chemoradiotherapy. The demographic and disease characteristics of the patients with NPC included are outlined in Table 1.
Demographic and clinical characteristics of patients with NPC.
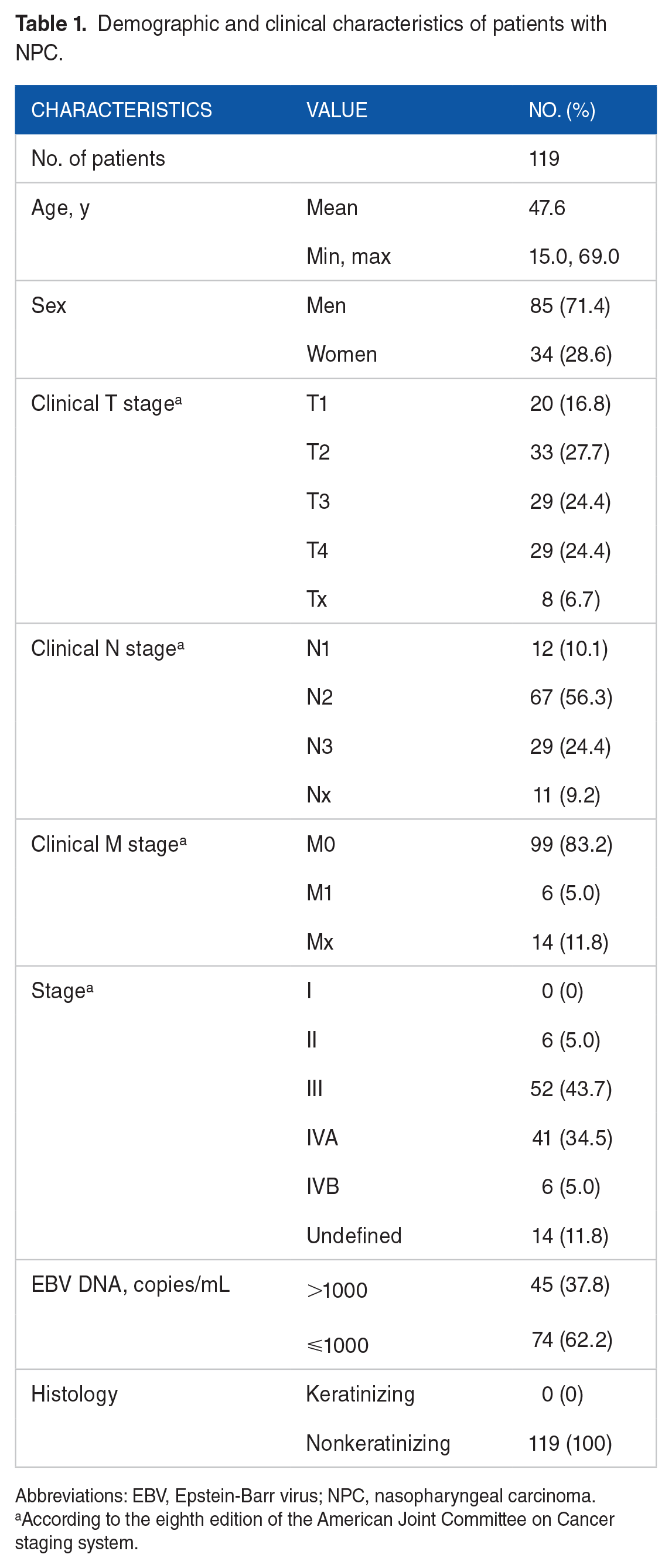
Abbreviations: EBV, Epstein-Barr virus; NPC, nasopharyngeal carcinoma.
According to the eighth edition of the American Joint Committee on Cancer staging system.
Inter-reader agreement
Interobserver agreement between 2 physicians for Node-RADS score assessment was evaluated by linear weighted kappa statistics. As shown in Table 2, in a total of 300 CLNs evaluated, the 2 physicians agreed on the RADS score for 238 lymph nodes. The percentage of agreement was 79.3%. Overall, the kappa agreement between the 2 readers for the Node-RADS score was almost perfect, with κ = 0.863 (95% CI = 0.830-0.897, P < .001).
Interobserver agreement for Node Reporting and Data System score for cervical lymph nodes of nasopharyngeal carcinoma.

Abbreviations: CI, confidence interval; Node-RADS, Node Reporting and Data System; NPC, nasopharyngeal carcinoma.
Lymph node metastasis rates and correlation with the Node-RADS score
Among all CLNs evaluated, a total of 203 CLNs had performed core-needle biopsy or lymph node dissection to confirm the presence of NPC metastasis. In total, 140 CLNs were indicated cancer-positive and 63 were indicated cancer-negative. Readers assigned 203 lymph nodes to 5 scores while scores 1, 2, 3, 4, and 5 had 47 (23.1%), 19 (9.4%), 26 (12.8%), 47 (23.2%), and 64 (31.5%) lymph nodes, respectively. The correspondence between the Node-RADS score assessed by readers and the results of core-needle biopsy or lymph node dissection is demonstrated in Figure 2. The Mantel-Haenszel test indicated a linear correlation between Node-RADS score and CLN metastasis (P < .001) as shown in Figure 2.

Lymph node metastasis rates according to Node-RADS score and Sankey diagram. Node-RADS indicates Node Reporting and Data System.
Furthermore, univariate and multivariate logistic regression models were established to analyze the correlation of the Node-RADS scores with the CLN metastasis. A univariate logistic regression indicated that the Node-RADS scores are correlated with CLN metastasis (odds ratio [OR] = 6.213, 95% CI = 3.887-9.931, P < .001). A multivariate logistic regression model adjusted for clinical data showed that Node-RADS scores (OR = 6.745, 95% CI = 3.964-11.474, P < .001; C-index = 0.9590) were independent risk factors for CLN metastasis. Considering a Node-RADS 3 to 5 positive (OR = 149.607, 95% CI = 42.897-521.761, P < .001) or a Node-RADS 4 to 5 (OR = 116.256, 95% CI = 25.559-528.808, P < .001) positive are also predictors for NPC CLN metastasis (Supplemental Table 1).
Diagnostic performance
When applying ROC analysis for the diagnostic detection of CLNs, the AUC of the Node-RADS score achieved 0.950 (95% CI = 0.921-0.979) (Figure 3).

Result of the ROC curve analysis for discriminating lymph node metastasis with Node-RADS score with an AUC of 0.950. AUC indicates area under the curve; Node-RADS, Node Reporting and Data System; ROC, receiver operating characteristic.
We set different cutoff values (from 5 to 1) of Node-RADS score, with sensitivity and NPV rising from 45.7% to 96.4% and from 45.3% to 89.4%, respectively. Correspondingly, specificity and PPV declined from 100.0% to 66.7% and from 100.0% to 86.5%, respectively, as shown in Table 3. When using the cutoff value of Node-RADS score 3, the sensitivity, specificity, PPV, and NPV were 92.1%, 87.3%, 94.2%, and 83.3%, with the highest accuracy of 90.6% (Table 3).
The test characteristics at different cutoff points.
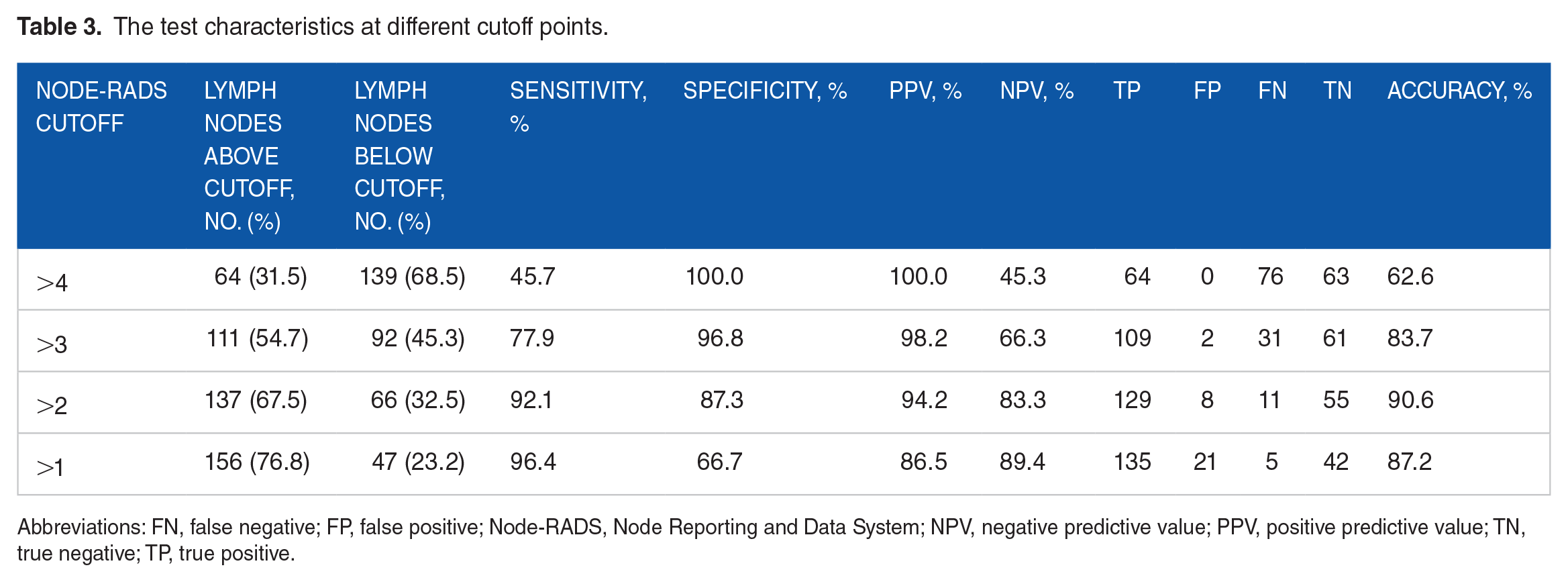
Abbreviations: FN, false negative; FP, false positive; Node-RADS, Node Reporting and Data System; NPV, negative predictive value; PPV, positive predictive value; TN, true negative; TP, true positive.
Discussion
The consensus on the radiologic evaluation of tumor metastatic lymph nodes is still controversial. Node Reporting and Data System is a recently proposed criterion that showed great potential in diagnosing lymph node metastasis on CT or MRI. To our best knowledge, this is the first study to validate the performance of Node-RADS in diagnosing CLN metastasis in NPC.
Nodal metastasis has been considered an independent predictive factor for poor prognosis of patients with NPC. Meanwhile, the level of lymph nodes is considered to be the most crucial lymph node variable in the clinical N (cN) stage evaluation. The metastatic spread of CLN chains in patients with NPC follows a typical pattern, a predisposition to lymph node metastasis in level II lymph nodes. It sequentially involves level III, IV, and supraclavicular fossa lymph nodes or extends posteriorly to involve level V lymph nodes. 13 Nasopharyngeal carcinoma has a high radiosensitivity, and radiotherapy is its main treatment modality. In clinical practice, only a minority of patients are diagnosed with early stage (I-IIa) NPC, while most are diagnosed with locally advanced (III-IVa) disease. 5 Tang et al 14 suggested that in low-risk patients with NPC, intensity-modulated radiotherapy alone caused 3-year failure-free survival not inferior to concurrent radiotherapy. Induction chemotherapy and concurrent radiotherapy for locally advanced NPC significantly improved overall and disease-free survival for all histologic types. 15 The metastatic status of the CLNs is an essential indicator in the cN staging of NPC and has crucial implications for the target volume delineation and planning radiotherapy.3,4
In clinical practice, MRI is the most ideal approach for head and neck soft tissue evaluation due to its high soft tissue resolution. The current MRI diagnostic criteria for CLN metastasis include nodal short-axis length, necrosis in the center or rim enhancement, and extra-nodal neoplastic spread.7,8 However, lymph node assessment remains a diagnostic challenge for radiologists when interpreting borderline-sized lymph nodes on MRI images. Under these circumstances, optimal treatment and counseling require accurate, objective, and normalized evaluation of NPC CLN metastasis.
Recently, the Node-RADS was proposed for malignant lymph node assessment. It provides a normalized, thorough assessment of the status of the lymph nodes, including criteria for both size and configuration. 9 In our study, we assumed that a higher Node-RADS score was independently associated with a higher risk of CLN metastasis in NPC and further examined the diagnostic performance of the Node-RADS score. Our study cohort provides an initial indication of the potentially useful role of Node-RADS in patients with NPC. Besides, lymph node metastasis rates based on Node-RADS scores showed a positive trend. Node-RADS scores were shown to be an independent predictive factor of status using multivariable logistic regression adjusted for clinical data (OR = 6.717, P < .001). Node-RADS scores showed high accuracy with an AUC of 0.95 when analyzed using the ROC curve. These encouraging results suggest the feasibility of using Node-RADS in diagnosing NPC CLN metastasis. In contrast, previous studies showed heterogeneous diagnostic performance of Node-RADS in different categories of cancer. Lucciola et al 10 indicated that the AUC of Node-RADS in diagnosing prostate cancer lymph nodes metastasis was merely 0.583. The application of Node-RADS in prostate cancer may be constrained. However, Node-RADS achieved a high AUC of 0.94 in diagnosing lung cancer lymph node metastasis 11 and a high AUC of 0.91 in bladder cancer. 12 Our research showed more outstanding diagnostic performance in NPC, providing new possibilities for the extensive application of Node-RADS.
Besides, diagnostic performance at different cutoffs was examined with the sensitivity of 45.7% to 96.4% and the specificity of 66.7% to 100% in this study. Finally, a Node-RADS score of 3 was identified as the best cutoff value based on balanced sensitivity and specificity values. Using a Nodes-RADS score of 3 as the cutoff, 66 (32.5%) of 203 nodes would be considered “negative” at the cost of missing 11 positive nodes within those 66 (16.7%) below the Nodes-RADS cutoff. As a result, implementing this cutoff would lead to the inclusion of approximately one-third of patients with a low risk of lymph node metastasis with an NPV of 83.3%. In particular, higher specificity with lower sensitivity or vice versa would result from a higher or lower cutoff. Nevertheless, the choice of the cutoff should be made on the basis of clinical situations. Therefore, further studies should evaluate Node-RADS cutoff based on specific clinical situations, such as the decision to administer neoadjuvant chemotherapy, the assessment of chemotherapy response, and the development of radiotherapy plans.
Interestingly, in the original classification standard introduced by Elsholtz et al, 9 only a score above 3 is regarded as “high probability” or “very high probability” metastatic. A Node-RADS score of 3 can be merely considered “equivocal.” However, our study demonstrated that a score of 3 is supposed to be suspected of metastatic lymph nodes. This also indicates the heterogeneity of Node-RADS judging criteria in different cancer categories. In the future, different judging criteria may be required according to different cancer types. Overall, the Node-RADS score may present a more standardized and accurate tool for identifying lymph node involvement in patients with NPC. These conclusions need to be further validated in larger cohorts and directly compared between Node-RADS and other established cN status modalities.
Limitations
Despite the encouraging results, our study also has several limitations. First, our study is a single-center retrospective study with limited sample size. The performance of Node-RADS in NPC needs to be further validated in multicenter retrospective/prospective studies with larger cohorts. Second, the image resolution and the slice thickness of MRI vary in different scanners and centers. This may potentially influence the measurement of the short-axis length of lymph nodes. In addition, some NPC frequently metastatic retropharyngeal nodes were excluded from the study cohort due to a lack of pathological results. Nevertheless, we still laid a solid foundation for the radiologic diagnosis of Node-RADS in NPC CLN metastasis.
Conclusions
The results of our study indicate that Node-RADS has good diagnostic value in differentiating benign from malignant CLNs in patients with NPC. In conclusion, this study lays the groundwork for implementing the Node-RADS scoring system for CLNs in patients with NPC.
Supplemental Material
sj-docx-1-onc-10.1177_11795549241231564 – Supplemental material for Diagnostic Performance of Node Reporting and Data System Magnetic Resonance Imaging Score in Detecting Metastatic Cervical Lymph Nodes of Nasopharyngeal Carcinoma
Supplemental material, sj-docx-1-onc-10.1177_11795549241231564 for Diagnostic Performance of Node Reporting and Data System Magnetic Resonance Imaging Score in Detecting Metastatic Cervical Lymph Nodes of Nasopharyngeal Carcinoma by Xinggang Yang, Jiaqing Yang, Jia Li, Junyan Leng, Yu Qiu and Xuelei Ma in Clinical Medicine Insights: Oncology
Footnotes
Author Contributions
XY, JY, JL, and XM contributed to conceptualization. XY, JY, JL, JYL, and YQ contributed to data curation. XY, JY, and XM contributed to formal analysis. XY, JY, JYL, and YQ contributed to investigation. XY, JY, JL, and XM contributed to methodology. XM, XY, and JY contributed to project administration. XY, JY, JL, JYL, YQ, and XM contributed to resources. XY and JY contributed to software. XM, JY, and XY contributed to supervision. XM, JY, JYL, and YQ contributed to validation. JY and XY contributed to visualization. XY and JY contributed to writing—original draft. XM, JY, and XY contributed to writing—review and editing. XY, JY, JL, JYL, YQ, and XM contributed to final approval of the version to be published.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the 1·3·5 project for disciplines of excellence-Clinical Research Incubation Project, West China Hospital, Sichuan University (grant no. 20HXFH052), and National Key Research and Development Program of China (grant nos. 2023YFC3605902 and 2023YFC3605900).
Consent for Publication
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to restrictions.
Ethics Approval and Consent to Participate
This retrospective study was approved by the Research Ethics Committee on Biomedical Research (RECBR), West China Hospital of Sichuan University (no. 2023-912, 2023.06.02). Written informed consent was waived by the RECBR for this retrospective study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
