Abstract
Leptomeningeal metastasis (LM) is a serious complication of advanced non–small cell lung cancer (NSCLC), and the incidence of LM has been increasing yearly in recent times. There is no consensus on the best treatment modality for LM, which underscores a difficult problem in the management of advanced NSCLC patients. The existing treatments include molecular targeted therapy, systemic chemotherapy, local radiotherapy, antivascular tumor therapy, intrathecal chemotherapy, and immunotherapy, but their efficacy is not satisfactory. In this article, we briefly describe the clinical manifestations, diagnosis, and treatment of NSCLC-LM and discuss progress regarding evaluation of the efficacy of LM treatment to better provide a necessary reference for clinical practice and clinical trial evaluation.
Keywords
Introduction
“The riddle of the Sphinx” symbolizes elusiveness and often a complex, mysterious, and incomprehensible problem. When malignant tumor cells metastasize to the meninges, subarachnoid space, or cerebrospinal fluid (CSF), patients will exhibit serious clinical symptoms due to the resulting condition known as leptomeningeal metastasis (LM). Leptomeningeal metastasis is one of the serious complications of advanced malignant tumors and often occurs in the late stage of non–small cell lung cancer (NSCLC), breast cancer, melanoma, and other malignant tumors. 1 Leptomeningeal metastasis can occur in approximately 3% to 5% of advanced NSCLC cases. 2 In recent years, the incidence of LM has increased year by year, which is obviously correlated with the prolongation of overall survival (OS) due to the improvement of treatment and the improvement of related diagnostic techniques.3,4 Once NSCLC-LM develops, the prognosis is extremely poor, severely affecting the OS and quality of life (QOL) of patients. Without aggressive treatment, the median OS is only 4 to 6 weeks. With the development of innovative drugs and therapeutic techniques, the penetration of antitumor drugs in the brain and CSF has greatly improved, and the OS of patients has increased from 1 to 3 months in the past to 3 to 11 months, 5 especially with the advent of the era of molecular targeted therapy. Driver gene-positive NSCLC mainly involves epidermal growth factor receptor (EGFR) positivity and anaplastic lymphoma kinase (ALK) or ROS1 rearrangement, which are more likely to be associated with LM. MET and RET gene mutations are far less common than EGFR, and preclinical and clinical data (1,2)indicate that compared to traditional chemotherapy the use of MET and RET TKIs is more likely to penetrate the BBB, resulting in better drug treatment outcomes. Yin et al 6 found that driver gene-positive NSCLC is more prone to LM than wild-type NSCLC, and given the advantages of high efficiency, low toxicity, and portability, molecular targeted drugs have become the preferred treatment option for patients with driver gene-positive NSCLC-LM. These drugs largely improve patients’ QOL and notably prolong OS; therefore, molecular targeted therapy is an important research direction and treatment focus for NSCLC-LM.
Leptomeningeal metastasis can occur in malignancy through a variety of pathways, the mechanisms of which are still unclear; the spread may involve hematogenous means, infiltration of metastases in the brain parenchyma, or progression along neural pathways into the CSF and the meninges of the brain or spinal cord. Owing to the presence of special structures, such as the choroid in the blood‒brain barrier (BBB) (Figure 1), 7 malignant tumor cells evade the surveillance of the immune system after the occurrence of LM, 8 and tumor cells transferred to the central nervous system (CNS) escape the attack of multiple immune cells and survive. In addition, due to the “protection” of the BBB, most intravenous chemotherapeutic drugs cannot enter the CNS, and this results in the concentration of CSF drugs being too low, which affects the therapeutic effect in LM. Non–small cell lung cancer–LM represents one of the bottlenecks and most challenging problems in the treatment of lung cancer metastases, and poor control of LM will seriously affect the treatment of the primary lung lesion, indirectly or directly affect the QOL of patients and substantially reduce the life expectancy of NSCLC-LM patients.
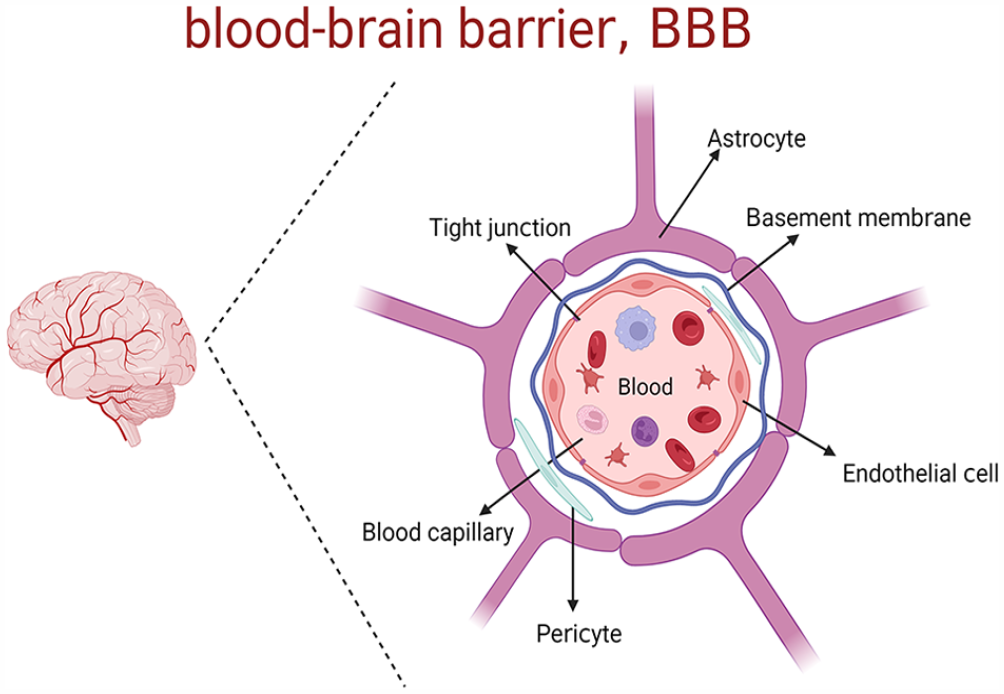
The structure of the BBB mainly includes endothelial cell, pericyte, tight junction, astrocyte, and basement membrane. This special structure not only prevents external substances from entering the CNS but also prevents drugs from entering the brain, leading to a decrease in drug utilization. BBB indicates blood‒brain barrier; CNS, central nervous system.
Clinical Manifestations and Diagnosis of NSCLC-LM
The clinical symptoms of LM are complex and difficult to distinguish from those of parenchymal brain metastases (Figure 2).9,10 The clinical diagnosis of LM is mainly based on the combination of CSF exfoliation cytology, imaging examination, and corresponding clinical symptoms or signs (Figure 3). 11 Patients with LM often present with elevated intracranial pressure (dizziness, headache, vomiting, and optic papillary edema), meningeal irritation (cervical ankylosis, Kernig’s sign, and Brudzinski’s sign), and cerebral neuropathy. 12 The results of enhanced magnetic resonance imaging (MRI) of the head often suggest meningeal enhancement or meningeal thickening. For patients with NSCLC with positive driver genes who are treated with molecularly targeted drugs, the appearance of symptoms, such as dizziness, headache, nausea, and vomiting, and other symptoms of increased intracranial pressure warrant a high degree of suspicion of LM. When this occurs, clinicians can usually choose to perform lumbar puncture and send CSF for exfoliative cytology to determine the presence of LM. Cerebrospinal fluid cytology is the gold standard for the diagnosis of LM, but its sensitivity is low, resulting in a low CSF positivity rate of 40% to 50%. 13 The positivity rate of repeated lumbar puncture is 80% to 90%. Therefore, there is considerable randomness in finding tumor cells by CSF cytology. On one hand, it is difficult to detect tumor cells in CSF in the early stage of LM, and on the other hand, factors such as the limitation of CSF sampling volume and delay in detection can markedly affect the positivity rate. 14

The clinical manifestations of NSCLC-LM are diverse and can be accompanied by different neurological symptoms, mainly consisting of 3 aspects. Patients with LM often present with elevated intracranial pressure (dizziness, headache, vomiting, and optic papillary edema), meningeal irritation sign (cervical ankylosis, Kernig’s sign, and Brudzinski’s sign), and cerebral neuropathy. NSCLC-LM indicates non–small cell lung cancer-leptomeningeal metastasis.
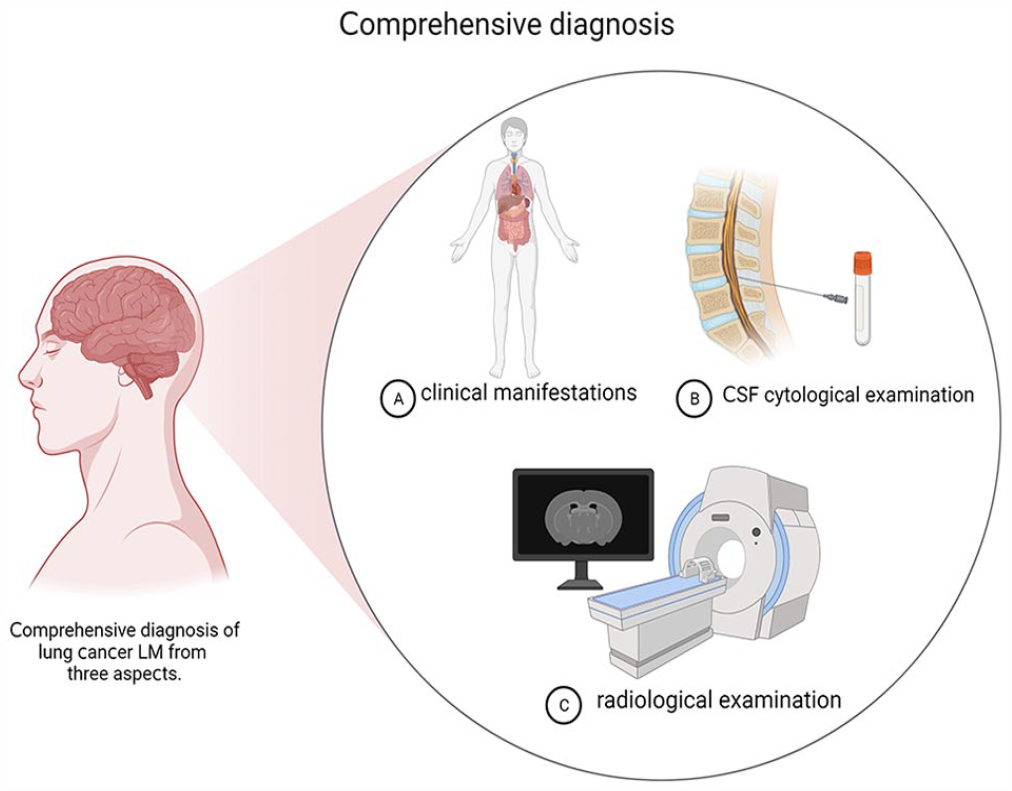
The comprehensive diagnosis of NSCLC-LM is mainly based on the combination of CSF cytological examination, radiological examination, and corresponding clinical symptoms or signs. CSF cytology is the gold standard for the diagnosis of LM. However, to overcome the problem of low sensitivity of CSF cytology, liquid biopsy of CSF is necessary. CSF indicates cerebrospinal fluid; NSCLC-LM, non–small cell lung cancer–leptomeningeal metastasis.
Cerebrospinal fluid cytology is the cornerstone of diagnosing LM, but its diagnostic sensitivity is low, especially in the early stages of LM, which leads to delayed treatment timing.2,15 Sasaki et al 16 and Verheul et al 17 have confirmed that the detection of tumor markers (TMs) in CSF can be used for early diagnosis of LM. Under physiological conditions, the generation of TMs in the CNS is minimal. Under pathological conditions, the BBB is damaged, which can promote the movement of TMs into the CSF from the blood; additionally, TMs can be synthesized in the sheath of cancer cells secreted by the CNS, which can increase TMs in the CSF. The increase in the TM concentration in the CSF depends on the secretion and release of local cancer cells or TMs in serum infiltrate through the BBB, with the former being more likely. 18 Therefore, the increase in the TM concentration in the CSF is often used for auxiliary diagnosis of LM. 19 Wang and colleagues20,21 suggest that the CSF CEACAM6 or CEA level is useful for diagnosing LM. However, the diagnostic sensitivity of TMs in the CSF for LM is low, especially in the early stages of LM disease, and the clinical application of TMs in the CSF is limited. Thus, it is crucial to find indicators with high diagnostic sensitivity for LM in the CSF.
To cope with the shortcomings of CSF cytology, liquid biopsy of CSF is necessary. 22 The sensitivity of liquid biopsy techniques can be as high as 93%. 14 The detection of CSF circulating tumor cells (CTCs) can not only improve the early diagnosis of LM but also have a substantial impact on tumor prognosis. 23 The presence of CSF CTCs is greatly correlated with the prognosis of tumors. 24 At present, the detection of CSF CTCs is not popular because of its high technical requirements, but it is believed that in the future, it will shine in the treatment of NSCLC-LM.
Treatment of NSCLC-LM
Treatment strategies for oncogene-addicted NSCLC are therefore distinct from those for non-oncogene-addicted NSCLC and depend on the specific genetic mutation present. The optimal treatment options and modalities for NSCLC-LM are still immature 25 and in the exploratory stage, particularly for non-oncogene-addicted NSCLC. 26 Owing to the existence of the BBB and its special function in the human body, intravenous administration of chemotherapeutic drugs commonly used in antitumor therapy is not accompanied by high permeability in the CNS, resulting in greatly reduced CSF concentrations of drugs.9,27 If appropriate levels of chemotherapeutic drugs cannot be achieved in the CNS to control and alleviate metastatic CNS lesions, the treatment effect in LM patients is substantially affected. For those with “oncogene addiction,” there are currently molecularly targeted drugs, namely, EGFR, ALK, and ROS1 tyrosine kinase inhibitors (TKIs) that strongly penetrate the BBB and prevent the spread of CNS cancer.
To address the problem of a low drug entry rate into the brain, clinicians can choose intrathecal (IT) chemotherapy to directly reach the foci of LM and give local CNS treatment; this approach is associated with fewer adverse effects than systemic chemotherapy and allows rapid and uniform distribution of drugs in the CSF and rapid achievement of effective drug concentrations.
Intrathecal chemotherapy involvers lumbar puncture to inject chemotherapeutic drugs directly into the subarachnoid space, allowing drugs to reach the site of meningeal metastasis via CSF circulation. When LM occurs in lung cancer, commonly used IT chemotherapeutic agents include methotrexate (MTX), but MTX is mainly used to treat hematologic tumors and is effective against lung cancer. A small clinical study of intrathecal pemetrexed (IP) has been conducted with significant results. Sun et al 28 established a mouse model to evaluate the safety and pharmacokinetics of IP, and the results confirmed that the efficacy of IP at a dose of 1 mg/kg was significant. The drug could be maintained at a higher concentration in the CSF for a longer period of time, with significant improvement in neurological symptoms and QOL, and there was a longer progression-free survival (PFS) and no serious adverse events (AEs). On the basis of a mouse model, Pan et al’s phase I clinical trial of IP in 13 patients with NSCLC-LM 29 showed that IP at a dose of 10 mg and in combination with vitamin supplements once or twice a week provided controlled toxicity and good efficacy. The results of several studies30,31 likewise confirmed the significant efficacy and manageable safety of IP in patients with LM. The results of a phase I and II clinical study of NSCLC-LM with EGFR mutations that failed to respond to treatment with a TKI 32 showed that an IP dose of 50 mg could be the recommended dose with a high response rate and manageable adverse effects. Zheng et al 33 also recommend IP as a potentially effective treatment, and IP can be combined with TKI molecular targeted therapy or radiation therapy with considerable efficacy. However, the efficacy and safety of IP in NSCLC-LM still need to be verified by clinical data from a large sample, and the optimal dose, frequency, and treatment period of IP still need to be investigated in more clinical trials.
Studies have shown that NSCLC with EGFR gene mutations has the highest incidence of LM. 34 The incidence of LM can be as high as 50% for EGFR and approximately 5% for ALK fusions. The usage of molecular targeted therapy in patients with positive driver genes has become a key research direction in studies of NSCLC-LM. Molecularly targeted therapies have been gradually developed in a transition from drugs with poor initial brain entry rates to drugs with high brain entry rates; such progress can lead to significantly improved OS and QOL in NSCLC-LM patients.35,36
The CNS penetration rate of first-/second-generation EGFR-TKIs is low, and their standard dose is not effective in patients with CNS metastases. 37 Third-generation EGFR-TKIs are mainly used for patients with CNS metastases, and the representative drugs are osimertinib, almonertinib, and furmonertinib. The results of several studies38-40 have shown that osimertinib has a greater ability to penetrate the BBB than first-/second-generation EGFR-TKIs. The AURA study 41 showed that osimertinib achieved an objective response rate (ORR) of 55% in T790M-positive asymptomatic CNS metastatic NSCLC patients after EGFR-TKI treatment, with median PFS and median OS of 11.1 and 18.1 months, respectively. The results of the AURA study showed a significant improvement in CNS remission rates. The BLOOM study 42 is an important study in NSCLC-LM. Osimertinib (160 mg QD) was administered after positive diagnosis by CSF cytology for EGFR-mutated NSCLC-LM with an ORR of 62% and a median OS of 11.0 months. The results of one study35,43 showed that osimertinib improved OS in EGFR-mutant NSCLC patients who developed LM regardless of T790M mutation status. Both third-generation almonertinib and furmonertinib are innovative drugs developed in China with independent intellectual property rights, and both have also shown excellent BBB penetration. 44 The preliminary results of several ongoing clinical studies show that their efficacy in the population with CNS metastases is not inferior to that of osimertinib in the FLAURA study. 45 We need to wait for final results expected in the future.
AZD3759 is a new generation of EGFR-TKI and specially designed to penetrate the BBB.46,47 In preclinical studies, AZD3759 showed excellent penetration of the BBB. 48 The Kpuu, CSF (ratio of free drug concentration in CSF to plasma) and Kpuu, brain (ratio of free drug concentration in brain to plasma) of AZD3759 are approximately 1, indicating that almost all AZD3759 can reach the brain. 49 The BLOOM study 50 is a phase I clinical study exploring the efficacy and safety of AZD3759 in patients with EGFR-sensitive mutant NSCLC with brain metastasis. Among the enrolled patients, the Kpuu, CSF values of AZD3759 can be as high as 1.11, indicating that AZD3759 can freely penetrate the BBB, so it can effectively treat patients with CNS metastasis, especially LM patients with high lethality. A total of 67 patients were included in this study. Among 21 patients with evaluable brain/meningeal metastases, 52% (11/21) achieved tumor reduction, with 14% (3/21) showing clear local remission. Among the patients initially treated with EGFR-TKI, 18 patients had evaluable CNS target lesions, with an intracranial ORR of 83% (15/18). Among LM patients who have previously received EGFR-TKI treatment, the ORR can reach 28% (5/18) and the disease control rate (DCR) can reach 78% (14/18). Thus, AZD3759 is a choice for EGFR-mutant NSCLC patients with CNS metastasis, especially LM patients.51,52
Most ALK driver-positive patients who receive the first-generation ALK-TKI crizotinib develop CNS metastasis,53,54 mostly due to poor crizotinib penetration in the CNS or disease progression. 55 In response to the shortcomings of poor CNS penetration of the first-generation drugs, the second-generation ALK-TKI alectinib is a drug with efficient CNS activity, mainly because alectinib is not effluxed by the P-glycoprotein transporter protein, 56 which is the key to its increased drug concentration in the brain and CSF. In an ALEX study 57 of the efficacy of alectinib versus crizotinib in CNS metastasis, alectinib demonstrated superior CNS activity and significantly delayed CNS progression compared with crizotinib. The third-generation ALK-TKI, lorlatinib, is specifically designed to penetrate the BBB, and animal models and preclinical studies in human trials have yielded results demonstrating lorlatinib’s potent antitumor activity and high BBB permeability.58-60 The rapid uptake of lorlatinib by the CNS was confirmed in animal models by imaging techniques. 61 Lorlatinib treatment is the preferred regimen for ALK driver-positive patients with CNS metastases. 62
A retrospective study 63 included 97 patients with NSCLC-LM molecular characteristics between 2015 and 2021, most of them had EGFR mutations and other mutations, including ALK, ROS1, KRAS, and TP53 mutations and MET amplification. Currently, there are relatively few reports of rare oncogenes, such as RET rearrangement or HER2 mutation causing LM. There is a case report 64 of lung adenocarcinoma accompanied by rare oncogene HER2 exon 20 insertion mutations that causes LM. After 3 days of administration of poziotinib, the symptoms improved significantly and the PFS was nearly 2 months. Therefore, we speculate that the CSF concentration and permeability of poziotinib may be significantly higher than those of other TKIs.
For NSCLC patients with positive driver genes, TKIs are still the first choice for treatment, but unresolved drug resistance issues are common.65,66 Therefore, it is particularly important to clarify the gene mutation status of the metastatic lesion after the occurrence of TKI resistance to guide subsequent treatment. For intracranial metastases, tissue biopsy is limited in clinical application due to difficulty in obtaining specimens and invasiveness. Liquid biopsy technology based on next-generation gene sequencing (NGS) provides a safer and more effective approach for precise treatment of lung cancer. Liquid biopsy can determine the tumor burden, drug efficacy, and drug resistance of NSCLC patients by detecting circulating tumor DNA (ctDNA) in peripheral blood, CSF, and other body fluids.67,68 In addition, few previous studies69-72 have confirmed that ctDNA in CSF compared with peripheral blood can more accurately reflect gene mutations in intracranial metastases. Using NGS liquid biopsy technology, detecting ctDNA changes in the CSF compared with peripheral blood of LM patients can more accurately and comprehensively reflect the true gene mutation status of LM. This strategy has broad application prospects in guiding medication decisions, monitoring drug resistance, efficacy evaluation, and prognosis judgment.4,73
The optimal treatment of NSCLC-LM is still in the phase of clinical exploration. The main goals of current treatment are to improve symptoms, improve QOL, and prolong survival time. Therefore, symptomatic supportive therapy is also necessary for LM patients and can rapidly relieve severe clinical symptoms in the short term. 74 However, the therapeutic effect cannot be maintained for a long period of time, and patients are prone to recurrent attacks and complete remission of the disease. For some patients, surgical treatment, mainly ventriculoperitoneal (VP) or Ommaya reservoirs, is available to rapidly relieve the clinical symptoms caused by high intracranial pressure.
It was found that vascular endothelial growth factor (VEGF) mediates tumor neovascularization, causing increased vascular permeability. 75 Vascular endothelial growth factor causes extravasation of plasma and proteins, leading to brain edema. 76 This suggests that VEGF may play an important role in the development of LM. The antiangiogenic drug bevacizumab, a recombinant humanized IgG1 monoclonal antibody against VEGF, specifically binds to VEGF. 77 On one hand, using bevacizumab can reduce neovascularization, inhibit tumor growth, reduce brain edema, 78 and improve patients’ QOL and overall treatment efficiency. On the other hand, bevacizumab can inhibit immature angiogenesis and induce normalization of blood vessels, thus increasing perfusion within the tumor and increasing the drug delivery rate. This suggests that combining bevacizumab with other chemotherapeutic drugs or targeted drugs in treatment can significantly increase the CNS penetration rate of other drugs, increasing the concentration of these drugs in the CSF and, thereby, giving full play to drug efficacy. Bevacizumab can also be combined with radiotherapy to induce normalization of tumor blood vessels, improve oxygenation, and reduce the proportion of oxygen-depleted cells in the tumor, thus enhancing radiotherapy sensitivity. 79 It can significantly improve the tumor control rate in patients with CNS metastases.
Systemic and IT chemotherapy with radiotherapy is usually administered for non-oncogene-addicted NSCLC. The main mode of head radiotherapy for NSCLC-LM is whole-brain radiotherapy (WBRT). 80 It can rapidly relieve acute neurological symptoms and clinical manifestations in a short period of time. However, due to the dose limitation of normal brain tissue, some patients still have difficulty in local tumor control. If the dose of radiotherapy is increased, high-dose WBRT can cause radioactive brain damage and irreversible damage to the brain parenchyma, and it is still controversial whether radiotherapy can benefit patient survival.9,81,82 Therefore, the purpose of radiotherapy is mostly palliative treatment.
Immunotherapy has gained popularity in recent years and has shown remarkable efficacy in the treatment of lung cancer. Immune checkpoint inhibitors (ICIs) have become a common first-line regimen in lung cancer treatment. However, due to the poor prognosis of LM patients, they have been excluded as participant in most clinical trials of ICIs. A single-center, retrospective study of 32 NSCLC patients treated with ICIs after LM 83 showed that patients with good physical status scores were more likely to benefit from immunotherapy. A single-arm phase II clinical trial 84 enrolling 18 patients with solid tumors with LM showed that immunotherapy has a better efficacy and safety profile in LM. There is no definitive evidence of the efficacy of immunotherapy in LM, and future clinical studies with large samples are needed to verify this.
Antibody drug conjugate (ADC) is a new type of antitumor drug that combines the advantages of both targeted therapy and chemotherapy while conferring a considerable reduction of toxic side effects compared with chemotherapy. It is a novel therapeutic tool that delivers a potent cytotoxic load through cytocytosis and ligand cleavage by monoclonal antibodies that specifically recognize tumor cell target antigens.85,86 Antibody drug conjugates are more commonly used in hematologic malignancies, but for solid tumors, only 7 ADCs have been approved worldwide, mainly breast cancer and uroepithelial cancer, involving a small number of phase I/II clinical studies.87-90 The biggest breakthrough in ADC application in solid tumors with CNS metastases has come from a phase II clinical trial on brain metastases from breast cancer. 91 A total of 15 patients were enrolled, all of whom had HER2-positive breast cancer with brain metastases. After treatment with ADC targeting the HER2 locus, 11 of the 15 patients (73.3%) had shrunken brain metastases, including 2 patients (13.3%) who had complete disappearance of brain metastases; the drug was well tolerated, with no deterioration in brain function or QOL during the treatment period. HER2 gene abnormalities have been shown to be associated with the development and invasion of many malignant tumors, such as breast, gastric, lung, and colorectal cancers. 92 HER2 mutations have a low incidence, are exhibited in only a few cases, and are associated with poor treatment outcomes in the real world of NSCLC.93,94 This clinical study provides the basis and ideas for exploring novel treatment options in NSCLC with CNS metastasis and new ADC-related strategies for NSCLC-LM patients.
Efficacy Evaluation of NSCLC-LM Treatment
The evaluation of efficacy in LM treatment has long been a complex issue in neuro-oncology. On one hand, it is mainly difficult to distinguish whether neurological symptoms are directly related to LM or due to disease progression, and on the other hand, it is difficult to accurately determine the lesion size on imaging, which will affect the determination of efficacy after drug administration. There is still a lack of a quantifiable evaluation system regarding treatment effects in LM, and efficacy evaluation is of great importance for clinical practice and clinical trials. Owing to the lack of evaluation criteria, most clinical trials will exclude patients with LM; therefore, energy, time, and multidisciplinary participation need to be directed at development of a treatment evaluation system for LM in the future.
Zhang and Yang 95 acknowledged that the efficacy of NSCLC-LM was evaluated by the Response Evaluation Criteria in Solid Tumors (RECIST 1.1) 96 with Brain Metastases, but it is controversial whether this criterion is applicable to the evaluation of the efficacy in patients with LM. The reason is that the size of the lesion cannot be accurately measured on cranial scan and enhanced MRI or X-ray computed tomography (CT) after LM for changes in cranial LM to be evaluated and treatment effect determined. Alternatively, the neurological system has not been evaluated in a specialized manner to allow determination of treatment effect. The neurological system was not evaluated, so the impact of the treatment plan on neurological function could not be assessed, and the improvement in neurological function could not be judged. This is one of the more problematic issues in LM efficacy evaluation criteria.
In the face of challenges in determining the efficacy of new treatments for malignant glioma, such as antiangiogenic drugs, the 2010 Response Assessment in Neuro-Oncology (RANO) working group proposed a new response assessment standard for high-grade glioma based on the Macdonald criteria established in the 1990s 97 to address its limitations. This effort has been extended to other RANO working groups related to CNS tumors, including the Response Assessment in Neuro-Oncology Brain Metastases (RANO-BM) system. 98 In 2015, the RANO working group proposed the RANO-BM efficacy assessment system, which, in contrast to the more widely used RECIST system for solid tumors, changed the evaluation of target lesions from a 1-dimensional measurement of the longest diameter to a vertical cross-2-diameter assessment of nodule size. It is important to note that the RANO-BM emphasizes the valid assessment of both target and nontarget lesions in brain metastases while including both in the criteria for defining efficacy. Progression of clear nontarget lesions should likewise be used as a criterion for progression and interruption of treatment. A more comprehensive systematic evaluation of the nervous system aims to develop standard response and progression criteria to be used in clinical trials for the treatment of brain metastases. This evaluation system proposes a comprehensive evaluation of neurological signs and symptoms and other aspects to comprehensively and systematically determine the improvement in neurological function; this is a milestone in the LM evaluation system development.
In 2018, the Chinese Society of Clinical Oncology, Chinese Association of Anticancer, proposed the Diagnosis and Treatment Consensus of Brain and Leptomeningeal Metastasis from Lung Cancer at the 15th China Lung Cancer Summit Forum. 99 The expert group recommended the use of the RECIST evaluation system for solid tumors in combination with the RANO-BM evaluation criteria for efficacy evaluation of LM treatment. For some researchers, 100 the specific evaluation work can be explained as follows: on the basis of the RECIST evaluation system, combined with RANO-BM evaluation criteria as a supplement to the evaluation reference, the importance of independent intracranial and extracranial evaluation and comprehensive evaluation is emphasized. In the evaluation of efficacy, intracranial lesions should be evaluated according to the RANO-BM criteria, and extracranial lesions should be evaluated according to the RECIST 1.1 criteria. It should be noted that the evaluations of both are independent of each other and do not interfere with each other. By consensus, CNS PFS and non-CNS PFS were recommended as clinical endpoints, and the concept of bicompartmental PFS was innovatively developed. 71 Compared with the management and assessment of lesions that occur in intracranial or extracranial unilateral progression, bicompartmental PFS offers better guidance and clinical significance, with emphasis on the specificity of intracranial lesion assessment and the overall disease characteristics and disease progression. Disease progression in 1 of the 2 compartments should be judged as CNS progression.
The RANO-BM standard mainly corresponds to brain metastases, and for most brain metastases, the distance between the 2 vertical cross paths can be measured, and brain metastases are very different from meningeal metastases in imaging performance.101,102 Brain metastases are easy to diagnose in imaging, but LM lesions are not easily determined in imaging, which is currently a major challenge for imaging.
The 2017 RANO working group 103 developed a consensus recommendation for LM, namely, the RANO-LM evaluation system, which proposes a comprehensive evaluation of LM after treatment: standardized neurological examination, CSF analysis, and imaging assessment. This evaluation system mainly involves a more comprehensive systematic evaluation of patients from 3 perspectives, and it has been widely used in clinical practice and clinical trials in recent years. This represents the latest research progress on the evaluation of LM treatment efficacy. The RANO-LM evaluation system contains 3 aspects. The first aspect, the neurological examination, is divided into 10 main sections, namely, gait, strength, sensation, vision, eye movements, facial strength, hearing, swallowing, level of consciousness, and behavior, and the examiner assigns a graded score to each aspect. As most neurological impairment due to LM is irreversible, the best response to treatment is usually stabilization of neurological function. 104 The RANO-LM recommends but does not include neurological symptoms in the RANO-LM evaluation system because there is currently no way to capture LM-related symptoms, such as headache, nausea, vomiting, or seizures. The second aspect, CSF exfoliative cytology, usually involves a qualitative analysis with results reported as negative, atypical, suspicious, or positive. Response Assessment in Neuro-Oncology-LM supports a binary outcome measure of positive or negative, so that atypical is considered negative and suspicious is considered positive. Response Assessment in Neuro-Oncology–LM reflects the idea that quantitative CSF exfoliative cytology may detect a more accurate efficacy-related response in theory, but feasibility is not high, and CSF exfoliative cytology is rarely performed in practice. It is possible that the poor sensitivity of CSF cytology may lead to false negatives, which may be as high as 50%, ie, tumor cells are still present in the CSF but not detected, leading possibly to “conversion.” 42 More importantly, CSF cytology results are influenced by a number of objective factors, such as whether a sufficient volume of CSF is obtained, the choice of site for CSF acquisition, and the means of rapid processing after CSF acquisition, all of which can markedly affect the results of CSF analysis. 105 Disease progression in CSF cytology is defined as the conversion of negative CSF cytology to positive or the unsuccessful conversion of positive to negative after induction therapy. The third and most challenging of the 3 aspects is neurological imaging assessment. Abnormal MRI often suggests meningeal enhancement of the brain or spinal cord, which can be classified as nodular, linear, and curvilinear or may be focal or diffuse. The RANO-LM working group recommended that nodules ⩾5 × 10 mm in diameter be defined as measurable lesions and evaluated serially. As the imaging features of most LM lesions are usually small in size and irregular in geometry, current MRI techniques do not allow for quantitative assessment, so the group adopted a scorecard model for imaging assessment. The RANO-LM expert group concluded that T2-FLAIR enhancement sequences of MRI are more useful for the assessment of LM lesions and that T2-FLAIR sequences could be added for further analysis of future meningeal lesions. Ideally, neurological examination, MRI, and CSF evaluation should be performed at the same time frame, but MRI should precede lumbar puncture to minimize the occurrence of false-positive meningeal enhancement on MRI after lumbar puncture.
However, this evaluation is still controversial; for example, neurological examination cannot accurately distinguish whether the symptoms associated with LM are due to the progression of systemic disease or the effects of parenchymal metastases or are related to adverse effects after treatment, which cannot be accurately determined according to the RANO-LM evaluation system. In addition, due to the low cytologic sensitivity of CSF, there is a high risk of false negatives, and it is inconclusive whether a negative CSF is an important part of the evaluation; its use as an important indicator of overall improvement is still controversial. Many experts106,107 have proposed NSCLC-LM treatment effect evaluation criteria that are currently available. However, the evaluation criteria are not widely used in clinical practice because they have not been further validated or are controversial.
We are also actively engaged in the joint multicenter development and practical validation of the evaluation criteria regarding the efficacy of treatment of meningeal metastases. In the context of referring to other evaluation systems, we initially attempted to propose refinable evaluation criteria, conduct statistical analysis, quantify the score according to each evaluation component, and assign different weights to each component based on the results of statistical analysis. On the basis of the RANO-LM evaluation system, the relevant evaluation contents are added or deleted, and the main points and basis for addition or deletion are as follows. (1) Delete the evaluation of “CSF cytology turning negative”: Due to the low sensitivity of CSF cytology examination, there are many clinical patients with obvious improvement in clinical symptoms after treatment in whom CSF cytology is still positive after treatment, which means stable according to the RANO-LM evaluation system. Moreover, since the clinical importance of CSF cytology is not clear, whether CSF cytology can be used as an important part of the evaluation to represent the improvement of the overall condition has not been established conclusively. 3 (2) Increase in “ QOL” or “physical activity status” scores: the presence of CNS metastases is associated with poor prognosis. 82 The expert consensus on NSCLC-LM treatment and the main goal of treatment is to improve symptoms, improve QOL, and prolong survival, so a separate evaluation of QOL should be added. (3) Characteristics of the measurement, observation of the level of increased intracranial pressure and the occurrence of LM, the degree of association of clinical symptoms, and exclusion of the factors of the occurrence/extent of intracranial pressure change after treatment with bevacizumab: for patients with IT chemotherapy, we can dynamically monitor the changes in intracranial pressure and adjust the drug dose and frequency in time with clinical symptoms.
The above views arise from a synthesis of information from various evaluation systems and the results of clinical practice. The work is still in the data processing and analysis stage, and more studies with large samples will be needed to verify the views.
Summary
Non–small cell lung cancer–LM treatment remains a challenge for clinical work. Affected by many factors, such as driving gene status, treatment process, and physical status score, individualized treatment is needed in the future, and multidisciplinary participation is also needed. The assessment of efficacy of NSCLC-LM treatment is subject to multiple uncertainties, and there is a long way to go in the future.
Footnotes
Author Contributions
All authors contributed to the article conception and design. The first draft of the article was written by YZ, LY, and LW, and all authors commented on previous versions of the article. YW and HC generated the images for this article. QW and YuW made a substantial contribution to the concept of the work. All authors read and approved the final article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The “XINRUI” Project of Cancer Supportive Care and Treatment Research (grant no. cphcf-2022-139) and WU JIEPING MEDICAL FOUNDATION (grant no. 320.6750.2022-17-24).
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
