Abstract
Epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors represent the standard of care in patients with EGFR mutation-positive (EGFRm+) non-small cell lung cancer (NSCLC). The availability of several EGFR tyrosine kinase inhibitors approved for use in the first-line or later settings in NSCLC warrants an in-depth understanding of the pharmacological properties of, and clinical data supporting, these agents. The second-generation, irreversible ErbB-family blocker, afatinib, has been extensively studied in the context of EGFRm+ NSCLC. Results from the LUX-Lung 3 and 6 studies showed that afatinib was more active and better tolerated than chemotherapy in patients with tumors harboring EGFR mutations. Subanalysis of these trials, along with real-world data, indicates that afatinib is active in patients with certain uncommon EGFR mutations (S768I/G719X/L861Q) as well as common mutations (Del19/L858R), and in patients with active brain metastases. In LUX-Lung 7, a head-to-head phase IIb trial, afatinib improved progression-free survival and time-to-treatment failure versus the first-generation reversible EGFR tyrosine kinase inhibitor, gefitinib, albeit with a higher incidence of serious treatment-related adverse events. Nevertheless, afatinib is generally well tolerated, and adverse events are manageable through supportive care and a well-defined tolerability-guided dose adjustment scheme. In this review, we provide a detailed overview of the pharmacology, efficacy, and safety of afatinib, discuss treatment sequencing strategies following emergence of different resistance mechanisms, and shed light on the economic impact of afatinib. We also provide a comparison of afatinib with the available EGFR tyrosine kinase inhibitors and discuss its position within treatment strategies for patients with EGFRm+ NSCLC.
Introduction
Lung cancer is the most common cancer in the US, with ∼228,000 estimated new cases of lung or bronchus cancer in 2020. 1 It is also the most common cause of cancer-related deaths, with more than 135,000 people estimated to die from the disease in 2020. 1 Non-small cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancer cases. 2 Recent advances in tumor molecular analysis have facilitated testing for possible genetic mutations and aberrations that drive tumor growth and proliferation. Numerous oncogenic drivers have been identified in NSCLC, including mutations in the genes encoding the epidermal growth factor receptor (EGFR), KRAS, and anaplastic lymphoma kinase (ALK), among others.2–4 Identification of abnormalities in these molecular pathways has prompted the development of agents with the aim of targeting specific components of these pathways.
EGFR is a member of the ErbB family of receptor tyrosine kinases, which includes EGFR (ErbB1, HER1), HER2 (ErbB2, Neu), HER3 (ErbB3), and HER4 (ErbB4).5,6 The tyrosine kinase activity of EGFR triggers numerous cellular signaling pathways that regulate growth, proliferation, and survival. 6 EGFR is a well-established molecular target in NSCLC, and mutations have been identified in 10–15% of Caucasian and up to 50% of Asian patients with adenocarcinoma, the most frequent NSCLC subtype.2,6 The most common EGFR mutations are deletions in exon 19 (Del19) and the point mutation L858R in exon 21, which together account for 85–90% of all presenting EGFR mutations. 7 Numerous uncommon mutations within exons 18–21 of the EGFR gene, including S786I, G719X, and L861Q, have also been identified in NSCLC tumors. 8 These activating mutations in EGFR lead to increased signaling downstream of the receptor, resulting in cellular growth and proliferation, and driving tumor development by promoting metastatic spread and resistance to apoptotic signals.6,9 The presence of EGFR mutations that drive tumorigenesis makes EGFR an attractive therapeutic target.
Advances in EGFR mutation testing
Advances in molecular screening of tumors have improved patient outcomes by allowing the development and use of targeted therapeutics. Initial tumor testing for EGFR or other mutations is routinely undertaken using direct biopsy of the tumor10,11; however, around 30% of NSCLC patients may be unable to provide a biopsy sample that is suitable for EGFR mutation analysis at diagnosis or following disease progression.11–13 In addition, tissue biopsies, particularly re-biopsies at the point of progression on initial treatment, are invasive and costly, and therefore not always feasible.10,11,14–16 Given these drawbacks, the less invasive liquid biopsy may be preferred over traditional tumor biopsy, particularly in monitoring for resistance. 11
The use of liquid biopsies, which may be obtained from blood, saliva, or urine, provides a minimally invasive method for collecting cell-free circulating tumor DNA and circulating tumor cells.17,18 This approach allows for regular testing of patients at various stages of treatment, for efficacy monitoring and real-time identification of potential new mutations.15,18,19 Liquid biopsies have been tested in the context of EGFR mutation-positive (EGFRm+) NSCLC and can be used for detection of Del19 and L858R.10,14,15,20,21 EGFR mutations detected in plasma and urine have been shown to be concordant with those identified by tumor biopsy, supporting the use of liquid biopsies as a screening tool.10,14,22 In addition, use of liquid biopsies from plasma to monitor tumor progression and identify EGFR mutations associated with resistance to therapy (e.g., acquired T790M) has been documented, with such biopsies showing 60–80% sensitivity and specificity approaching 100% in detecting specific resistance mutations.15,16,21,23–25
EGFR tyrosine kinase inhibitors
EGFR-driven tumors become dependent on EGFR signaling for growth and survival, a phenomenon known as oncogene addiction.5,26 Oncogene addiction postulates that some tumors become dependent on a single oncogene for growth and survival, such that inhibition of this oncogene is sufficient to block tumor growth and induce tumor regression. 26 Consequently, EGFRm+ tumors are particularly susceptible to treatments that target the EGFR pathway. 9
Three generations of EGFR tyrosine kinase inhibitors (TKIs) have been developed and are available for clinical use. The first-generation TKIs, gefitinib and erlotinib, are reversible inhibitors of both wild-type and common EGFR mutations,27,28 and have shown improved efficacy and tolerability compared with chemotherapy in numerous phase III trials (gefitinib: IPASS, WJTOG3405, NEJ002; erlotinib: EURTAC, OPTIMAL, ENSURE).29–34 In two head-to-head trials of gefitinib versus erlotinib (CTONG0901 and WJOG5108L), there were no significant differences between the two first-generation agents with regard to duration of progression-free survival (PFS), objective response rate (ORR), or overall survival (OS) in any line of treatment.35,36 The second-generation EGFR TKIs include afatinib and dacomitinib, which are irreversible inhibitors of wild-type and mutant EGFR, ErbB2, and ErbB4, thereby inhibiting signaling from all possible homo- or hetero-dimers of ErbB family receptors (Figure 1).27,37,38 This includes ErbB3-containing heterodimers, as the second-generation agents prevent the trans-phosphorylation of the ErbB3 receptor and subsequent signal transduction.27,37,38 When compared with chemotherapy, first-line afatinib has demonstrated significant improvements in PFS for patients with EGFRm+ NSCLC, and in OS for patients with Del19-positive disease.39,40 Compared in head-to-head trials, both afatinib and dacomitinib have shown significantly improved PFS versus gefitinib.41,42 With regard to OS in the same trials, there was no statistically significant difference with afatinib versus gefitinib, and exploratory analysis demonstrated prolonged OS with dacomitinib versus gefitinib.43,44 The third-generation TKI, osimertinib, selectively inhibits both EGFR-TKI-sensitizing and EGFR T790M resistance mutations, with lower activity against wild-type EGFR. 13 The “gatekeeper” T790M mutation is a well-characterized mechanism of acquired resistance to first- and second-generation TKIs, which has been reported to occur in >60% of cases.45–47 Osimertinib has shown robust efficacy in patients who have progressed on first- or second-generation TKIs.47,48 In addition, in the recent FLAURA head-to-head trial, first-line treatment with osimertinib led to superior PFS and OS compared with a first-generation TKI (gefitinib or erlotinib).13,49
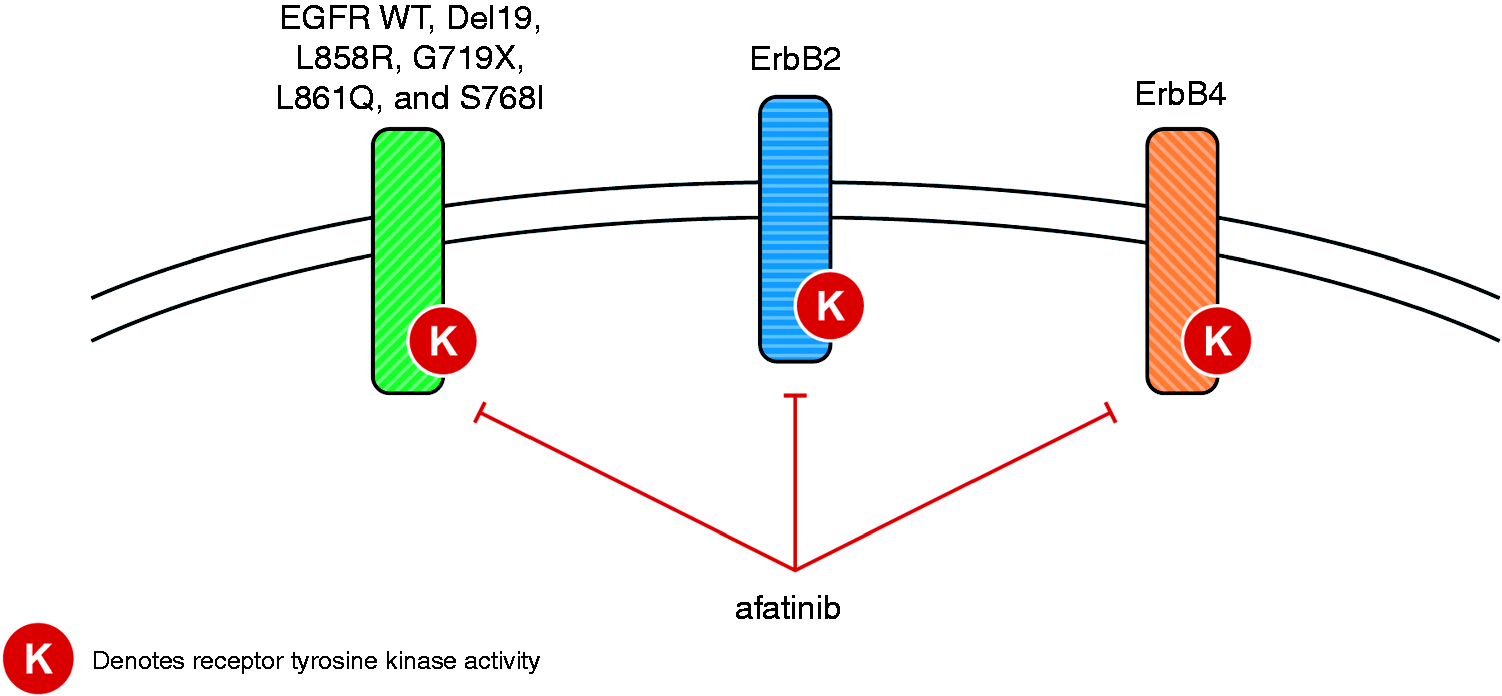
Inhibition of ErbB family members, including WT and mutant EGFR, by afatinib. EGFR: epidermal growth factor receptor; WT: wild-type.
EGFR TKIs represent the standard of care for first-line treatment of EGFRm+ NSCLC; gefitinib, erlotinib, afatinib, dacomitinib, and osimertinib have all been approved by the Food and Drug Administration (FDA) in this setting.50–54 The availability of multiple EGFR TKIs with distinct pharmacological, efficacy, and safety profiles raises the question of which one should be used at which stage in the treatment sequence, and also highlights the need to identify an optimal sequencing strategy for each of the TKIs, in order to achieve long-term treatment benefit and optimal survival. In this review, we will focus on the second-generation TKI, afatinib, providing an in-depth overview of its pharmacology, efficacy, and safety. We will discuss the use of afatinib in clinical settings, particularly in patients with NSCLC harboring common and uncommon EGFR mutations, patients with central nervous system (CNS) metastases, and in real-world settings. Furthermore, we will provide insight into the use of sequential first-line afatinib and second-line osimertinib.
Afatinib pharmacokinetics and interactions
Afatinib (BIBW 2992; N-[4-[(3-chloro-4-fluorophenyl)amino]-7-[[(3S)-tetrahydro-3-furanyl]oxy]-6-quinazolinyl]-4-(dimethylamino)-2-butenamide) is an adenosine triphosphate (ATP)-competitive anilinoquinazoline derivative containing a reactive acrylamide group. 27 Afatinib is administered orally, at a recommended dose of 40 mg once daily, with peak plasma concentrations achieved 2–5 h post-dose. 50 Administration after a high-fat meal results in an approximately 40% reduction in the area under the time–concentration curve from time 0 to infinity (414 ng · h/ml fed; 676 ng · h/ml fasted) 55 ; consequently, afatinib should be taken on an empty stomach, at least 1 h before or 2 h after eating. 50 The elimination half-life of afatinib is 37 h following repeat dosing,50,56 and steady-state plasma concentrations are achieved within 8 days of multiple dosing.50,55–57
Excretion occurs primarily via the feces (85%), with the parent compound representing 89% of the recovered dose. 58 Moderate-to-severe renal impairment has been shown to have a minor influence on afatinib pharmacokinetics. 59 Afatinib treatment can be considered without the need for starting dose modifications in patients with mild or moderate renal impairment, although a starting dose of 30 mg/day is recommended in patients with severe renal impairment. 50 Close monitoring of patients with severe renal impairment is advised, and dose adjustment is recommended if tolerability issues arise. 59 No afatinib starting dose adjustments are needed for patients with mild or moderate hepatic impairment. 60 However, afatinib has not been studied in patients with severe hepatic impairment, and these patients should be closely monitored and the dose adjusted, if needed. 50
Afatinib is highly soluble across the physiological pH range of 1–7.5 and is not expected to interact with acid-reducing agents.61,62 This is in contrast to gefitinib, erlotinib, and dacomitinib, which exhibit pH-dependent solubility, such that absorption and bioavailability may be decreased when co-administered with treatments that increase gastric pH (e.g., histamine H2-receptor antagonists and proton pump inhibitors). 61 In vitro studies indicate that plasma protein–binding of afatinib is high (∼94%). 60 However, enzyme-catalyzed metabolism of afatinib plays a negligible role in its metabolism in vivo. 61 Unlike other EGFR TKIs, which undergo metabolism via cytochrome P450 (CYP) enzymes, CYP enzymes play a negligible role in the metabolism of afatinib. 61 Smoking status does not impact the pharmacokinetics of afatinib, 62 whereas for erlotinib, smoking reduces exposure.61,63 Moreover, afatinib does not relevantly inhibit or induce CYP enzymes, including those involved in drug metabolism. 62 Therefore, the risk of interactions between afatinib and co-administered drugs that undergo CYP enzyme metabolism is thought to be minimal. 62 In contrast, other EGFR TKIs interact with different CYP enzymes to varying extents, potentially impacting the metabolism of concomitant drugs.51,61
Afatinib is a substrate and inhibitor of P-glycoprotein in vitro, and concomitant use of strong P-glycoprotein inhibitors can increase exposure to afatinib.62,64 Interestingly however, concomitant administration of ritonavir, a well-known, potent inhibitor of P-glycoprotein and BCRP (another ATP binding cassette drug transporter inhibitor) has shown no relevant impact on afatinib pharmacokinetics in healthy male adults.62,64 P-glycoprotein inducers such as rifampicin can reduce afatinib exposure.50,62,64 In vitro, afatinib is both a substrate and inhibitor of BCRP, 50 and gefitinib, erlotinib, and osimertinib also function as inhibitors and/or substrates of P-glycoprotein and/or BCRP to varying degrees.51,53,61,65
Afatinib efficacy and safety data from clinical trials
Afatinib versus chemotherapy: LUX-Lung 3 and LUX-Lung 6
Afatinib was approved for the first-line treatment of metastatic NSCLC harboring non-resistant EGFR mutations based primarily on the outcomes of the global phase III LUX-Lung 3 trial that compared first-line afatinib with cisplatin/pemetrexed in 345 patients with advanced EGFRm+ adenocarcinoma. 39 LUX-Lung 6 was another phase III trial that compared first-line afatinib with gemcitabine/cisplatin in 364 Asian patients with EGFRm+ NSCLC. 40
In both trials, which included patients with NSCLC harboring common and/or uncommon EGFR mutations, afatinib treatment resulted in improved median PFS versus chemotherapy: LUX-Lung 3 (afatinib vs cisplatin/pemetrexed): 11.1 versus 6.9 months; hazard ratio (HR), 0.58 (95% confidence interval (CI), 0.43–0.78), P = 0.001; LUX-Lung 6 (afatinib vs gemcitabine/cisplatin): 11.0 versus 5.6 months; HR, 0.28 (95% CI, 0.20–0.39), P < 0.0001.39,40 While OS analysis of the two studies, as well as both studies combined, indicated no significant OS improvement with afatinib versus chemotherapy (LUX-Lung 3/6 combined: 25.8 vs 24.5 months; HR, 0.91 (95% CI, 0.75–1.11), P = 0.37), a pre-specified subgroup analysis demonstrated that afatinib prolonged OS versus chemotherapy in patients with Del19-positive tumors, in both LUX-Lung 3 (33.3 vs 21.1 months; HR, 0.54 (95% CI, 0.36–0.79), P = 0.0015) and LUX-Lung 6 (31.4 vs 18.4 months; HR, 0.64 (95% CI, 0.44–0.94), P = 0.023). 66 It appears that not all EGFR TKIs confer the same OS benefits; in a meta-analysis comparing patients with Del19-positive NSCLC who were treated with erlotinib, gefitinib, or afatinib, only afatinib was associated with a statistically significant OS benefit versus chemotherapy. 67
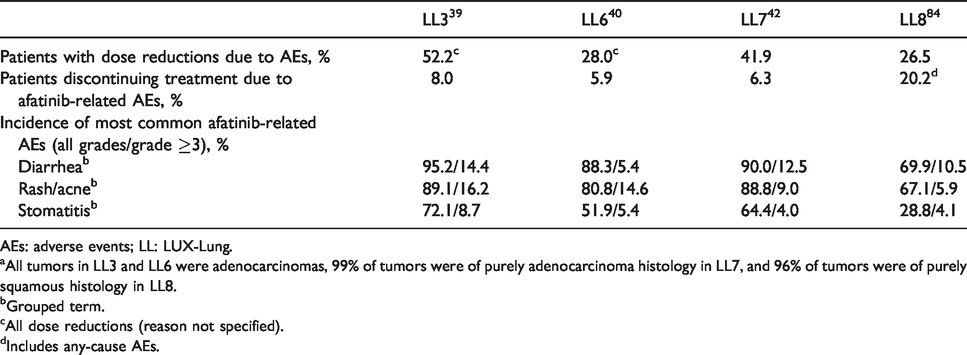
Consistent with the results observed with other EGFR TKIs,31,32 afatinib was better tolerated than chemotherapy in both studies.39,40 The most common adverse events (AEs) were class-related: gastrointestinal (diarrhea, stomatitis) and cutaneous (rash/acne).39,40,68 The discontinuation rate for afatinib due to treatment-related AEs was low, despite patients being on treatment longer with afatinib than with chemotherapy (LUX-Lung 3: 8% versus 12%; LUX-Lung 6: 6% versus 40%, with afatinib and chemotherapy, respectively; Table 1).39,40
Incidence of AEs leading to dose reduction and treatment discontinuation, and the most common afatinib-related AEs in the major afatinib clinical trials. a
AEs: adverse events; LL: LUX-Lung.
aAll tumors in LL3 and LL6 were adenocarcinomas, 99% of tumors were of purely adenocarcinoma histology in LL7, and 96% of tumors were of purely squamous histology in LL8.
bGrouped term.
cAll dose reductions (reason not specified).
dIncludes any-cause AEs.
Afatinib versus gefitinib: LUX-Lung 7
The phase IIb LUX-Lung 7 study was the first global head-to-head comparison of a first-generation EGFR TKI (gefitinib) and a second-generation EGFR TKI (afatinib) in patients with EGFRm+ NSCLC. 42 The three co-primary endpoints of the study were PFS, OS, and time-to-treatment failure (TTF). As an endpoint, TTF captures patients who are continued on treatment beyond trial-defined radiological progression in the absence of clinical deterioration. Afatinib significantly improved PFS (median 11.0 vs 10.9 months; HR, 0.73 (95% CI, 0.57‒0.95), P = 0.017) and TTF (median 13.7 vs 11.5 months; HR, 0.73 (95% CI, 0.58‒0.92), P = 0.007) versus gefitinib. PFS was also numerically longer with afatinib across most patient subgroups investigated, including EGFR mutation type (Del19 or L858R). 42 Afatinib also significantly improved ORR versus gefitinib (70% vs 56%; P = 0.0083), with a longer median duration of response (10.1 months (interquartile range (IQR), 5.6–16.8) vs 8.4 months (IQR, 6.2–13.1)), and a trend towards improved OS with afatinib vs gefitinib observed (median 27.9 vs 24.5 months; HR, 0.86 (95% CI, 0.66‒1.12), P = 0.258). 43
The overall frequency of grade ≥3 treatment-related AEs was higher with afatinib than with gefitinib (31.3% vs 17.6%). Grade ≥3 diarrhea, rash/acne, and stomatitis were more frequent with afatinib, while elevated alanine aminotransferase/aspartate aminotransferase was more common with gefitinib. 42 Serious treatment-related AEs were reported in 11% of afatinib-treated and 4% of gefitinib-treated patients; however, discontinuations due to treatment-related AEs were similar between treatment arms (6% in each arm). 42
Afatinib in patients with uncommon EGFR mutations
Uncommon mutations within exons 18–21 are present in up to 23% of EGFRm+ NSCLC tumors.69–75 While most phase III studies of EGFR TKIs in EGFRm+ NSCLC included only patients with tumors harboring common (Del19/L858R) EGFR mutations,29–34 the LUX-Lung 2, 3, and 6 studies included patients with uncommon mutations. 75 In a pooled analysis of patients with uncommon mutations (n = 75 across the three studies), afatinib showed efficacy in a subgroup of patients with tumors harboring the G719X, L861Q, and S768I EGFR mutations, as well as other, rarer mutations, but not against tumors harboring T790M or exon 20 insertions. 75 Median PFS (95% CI) was 13.8 months (6.8–not evaluable) for patients with tumors harboring G719X, 8.2 months (4.5–16.6) for L861Q, and 14.7 months (2.6–not evaluable) for S768I. Median OS (95% CI) was 26.9 months (16.4–not evaluable), 17.1 months (15.3–21.6), and not evaluable (3.4–not evaluable), and ORR was 78%, 56%, and 100%, for patients with NSCLC harboring these three uncommon mutations, respectively. The European Medicines Agency indicated afatinib for the first-line treatment of NSCLC with any activating EGFR mutation on first approval in 2013. 76 These results from LUX-Lung 2, 3, and 6 led to the expansion of the FDA label to include patients with metastatic NSCLC harboring non-resistant mutations, including G719X, L861Q, and S768I. 50
Afatinib in patients with baseline brain metastases
Brain metastases are common in patients with NSCLC, occurring in around 25% of patients during the course of their disease. 77 The incidence of brain metastases is higher among patients with EGFRm+ NSCLC than in patients whose tumors have wild-type EGFR (31.4 vs 19.7% in one report, P < 0.001). 77 However, patients with brain metastases have been excluded from clinical trials. 41 This is of particular significance as it limits the ability to generalize the outcomes of such clinical trials to patients in the clinic. Although little is known about the brain penetrance of afatinib in humans, preclinical pharmacology studies in rats suggest low distribution to the brain. 78 Nevertheless, several clinical studies with afatinib and osimertinib, including head-to-head trials with first-generation TKIs, have included patients with EGFRm+ NSCLC with CNS involvement, which has shed light on the effect of these TKIs on patients with CNS metastases.
In the LUX-Lung 3, 6, and 7 trials, the inclusion criteria allowed recruitment of patients with asymptomatic and stable brain metastases.39,40,42 Baseline brain metastases were present in 12%, 13%, and 16% of patients in LUX-Lung 3, 6, and 7, respectively.42,79 In a combined analysis of the LUX-Lung 3 and 6 studies, 81 patients with brain metastases at baseline (afatinib: n = 48; chemotherapy: n = 33) showed improved PFS (8.2 vs 5.4 months; HR, 0.50 (95% CI, 0.27–0.95), P = 0.0297) and ORR (combined analysis: 73% vs 24%; LUX-Lung 3: 70% vs 20%, p = 0.0058; LUX-Lung 6: 75% vs 28%, P = 0.0027) with afatinib compared with chemotherapy, while there was no improvement in OS (22.4 vs 25.0 months; HR, 1.14 (95% CI, 0.66–1.94), P = 0.6412). 79 Improvements in PFS in patients with brain metastases were similar to those observed among patients without brain metastases. In LUX-Lung 7, 50 patients with brain metastases (afatinib: n = 26; gefitinib: n = 24) showed similar PFS improvement between the two treatments (afatinib vs gefitinib: 7.2 vs 7.4 months; HR, 0.76 (95% CI, 0.41–1.44)) and also compared with patients without brain metastases. 42
Competing risk analyses of LUX-Lung 3 and 6 showed that in patients with baseline brain metastases the cumulative incidence of CNS progression (31.3%) was lower than that of non-CNS progression (52.1%; Figure 2). 28 A recent single-center, real-world study of 306 EGFRm+ NSCLC patients from Taiwan with or without baseline brain metastases assessed the impact of first-line erlotinib, gefitinib, or afatinib in treating and preventing brain metastases. 80 PFS was significantly longer and OS numerically longer with afatinib (compared with erlotinib or gefitinib) in the overall patient population, although it must be noted that higher proportions of patients treated with afatinib had better Eastern Cooperative Oncology Group (ECOG) performance status and Del19 mutations. PFS and OS were similar across the three EGFR TKIs in a subgroup analysis comparing patients with an ECOG performance status of 0–1 and Del19 mutations. Another subgroup analysis, involving patients without baseline brain metastases, found that after adjusting for possible confounding factors, those in the afatinib group had a significantly lower HR for the development of subsequent brain metastases than did those in the gefitinib group. However, the three treatments showed similar efficacy in patients with baseline brain metastases.
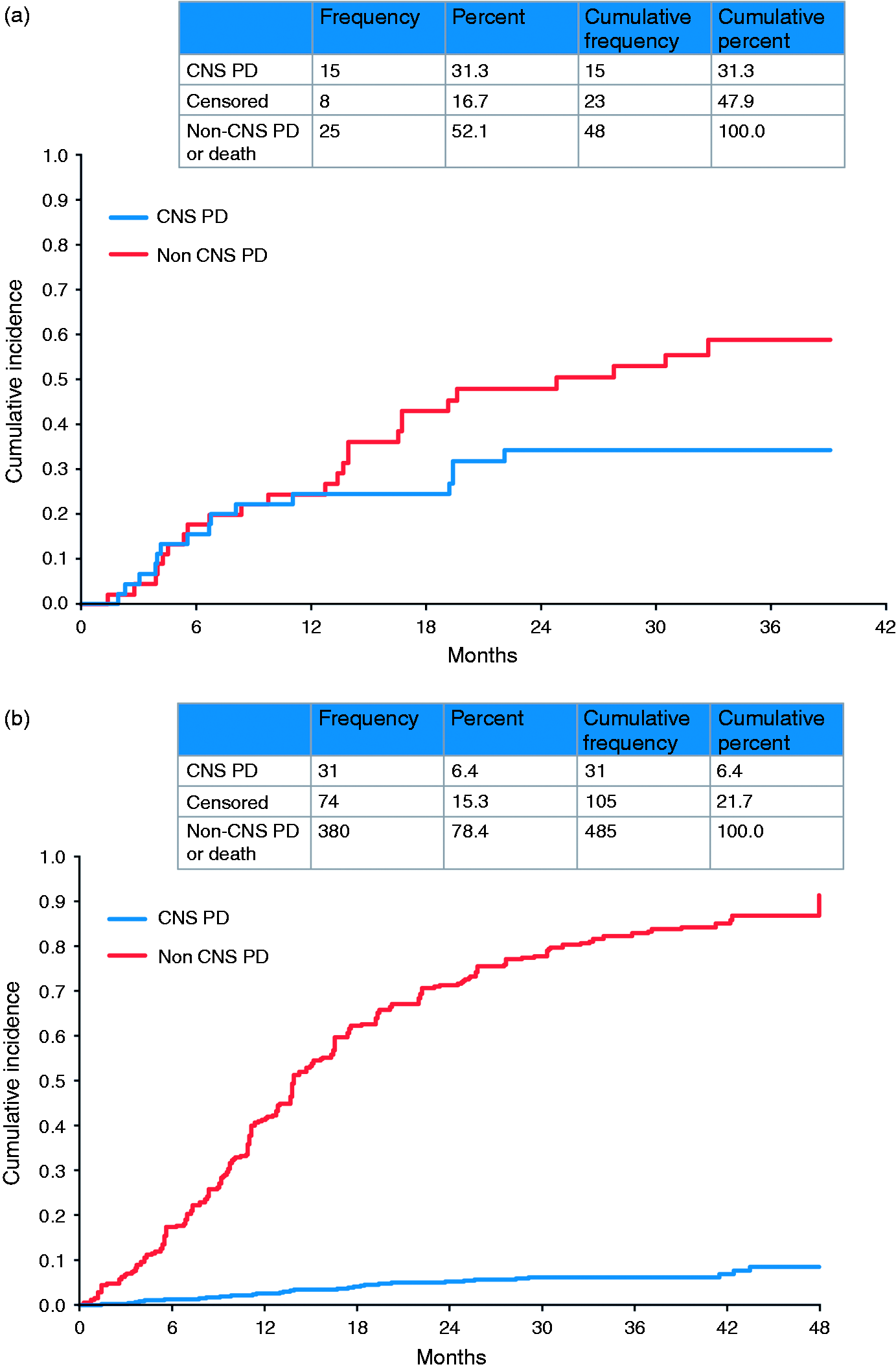
Competing risk analysis for progression in afatinib-treated patients with baseline brain metastases ((a) LUX-Lung 3 and 6) and without baseline brain metastases ((b) LUX-Lung 3, 6, and 7). CNS: central nervous system; NSCLC: non-small cell lung cancer; PD: progressive disease. Reproduced under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International Public License (Girard et al.). 30
It is also worth noting that osimertinib has also demonstrated efficacy in patients with brain metastases.13,81 Based on analysis of CNS activity in the FLAURA trial, osimertinib appears superior to first-generation TKIs in treating CNS metastases and in reducing the incidence of de novo CNS lesions. 81
Afatinib in squamous cell carcinoma
While EGFR mutations are rare in lung squamous cell carcinoma (SCC) tumors, they are often associated with increased EGFR protein expression and occasionally, altered EGFR copy number. 82 Certain clinical features, such as an absence of tobacco exposure have indicated a higher likelihood of a targetable mutation (including EGFR and ALK) and it is therefore reasonable to recommend testing for such alterations in non-smoker patients with lung SCC. 83 Identification of increased EGFR copy number and high levels of EGFR protein expression in SCC have served as rationale to assess the impact of EGFR-targeted agents in the treatment of SCC. 82 The LUX-Lung 8 study compared afatinib with erlotinib in patients with stage 3 b/4 SCC of the lung who had progressed after at least four cycles of platinum-based chemotherapy. 84 Treatment with afatinib resulted in significantly longer PFS (2.6 vs 1.9 months; HR, 0.81 (95% CI, 0.69, 0.96), P = 0.0103) and improved OS (7.9 vs 6.8 months; HR, 0.81 (95% CI, 0.69, 0.95), P = 0.0077) compared with erlotinib. Biomarker analysis indicated that, among patients treated with afatinib, PFS and OS were numerically longer in patients with ErbB mutation-positive tumors than in those without mutations; this was not observed for erlotinib. Interestingly, the benefit of afatinib over erlotinib among patients with ErbB-positive tumors appeared to be driven by mutations in HER3 (ErbB3), HER4 (ErbB4), and in particular, HER2 (ErbB2). 85 Based on the overall LUX-Lung 8 results, afatinib has been approved by the FDA for the treatment of metastatic, squamous NSCLC progressing after platinum-based chemotherapy. 50
Management of afatinib-related AEs
The most common afatinib-related AEs across the LUX-Lung 3, 6, 7, and 8 studies were diarrhea, rash/acne, and stomatitis (Table 1).39,40,42,84 A supportive care strategy combining patient education, frequent communication, routine monitoring, early recognition, proactive management, and adherence to the recommended tolerability-guided dose adjustment schema is important to help maximize clinical benefits, optimize symptom management, and reinforce adherence, thereby limiting treatment discontinuation. 86 The management of afatinib-related AEs has been well characterized, and guidelines are available that describe recommended strategies for prevention and management.68,87 For example, patients who experience diarrhea should receive medication, such as loperamide, immediately.
In addition, the availability of several different doses (20, 30, or 40 mg) of afatinib facilitates the implementation of a tolerability-guided dose adjustment strategy (described below) that can help patients to remain on afatinib long term. 50 Of note, analysis of data from LUX-Lung 3 indicated that higher exposure to afatinib increased the risk of experiencing grade ≥3 toxicity or grade ≥2 diarrhea. 78 Afatinib should be withheld in patients who experience any adverse reactions of grade ≥3, diarrhea of grade 2 persisting for ≥2 consecutive days while taking anti-diarrheal medication, or cutaneous reactions of grade 2 that last >7 days or are intolerable. Treatment should be resumed at a reduced dose when the adverse reaction has fully resolved, improved to grade 1, or returned to baseline. Dosing should be reduced by 10 mg decrements, to a minimum of 20 mg/day. 50 Importantly, afatinib dose reductions in the LUX-Lung 3 and 6 trials (in line with the criteria described above) resulted in a major reduction in treatment-related grade ≥3 AEs (from 73.0% to 20.5% in LUX-Lung 3 and from 80.6% to 11.9% in LUX-Lung 6) without affecting therapeutic efficacy, as assessed by PFS. 88 Furthermore, in a noninterventional, observational study conducted in a real-world setting, dose reductions with afatinib were shown to reduce the frequency and intensity of adverse drug reactions, without compromising treatment effectiveness. 89
It has been suggested that the severity of afatinib-related skin reactions may be prophylactically reduced using a general skin care regimen combined with oral antibiotics, such as tetracyclines. 90 Others recommend the regular use of emollients and protection from excessive sun exposure. 68 For patients who receive prophylactic treatment but still develop skin reactions in response to EGFR TKIs, topical antibiotics, corticosteroids, and potentially antihistamines are recommended. If the patient’s skin condition does not improve, dose reduction or treatment discontinuation is recommended, along with referral to a dermatologist.68,90
Acquired resistance and treatment sequencing strategies
Despite the efficacy of EGFR TKIs in the first-line treatment of EGFRm+ NSCLC, acquired resistance to therapy occurs in the majority of patients. 45 Identifying the molecular mechanisms of acquired resistance is essential in order to determine the subsequent treatment that would yield greatest benefit. The most common mechanism of resistance to first-generation EGFR TKIs is the development of the gatekeeper T790M mutation, which occurs in approximately 50–70% of patients.45,46,91 Likewise, T790M is detected in around 50–70% of patients with resistance to afatinib91,92 and is also likely to be associated with clinical resistance to dacomitinib. 93 A variety of other mechanisms of resistance to the first- and second-generation EGFR TKIs have been identified, including other EGFR alterations, Erb2 amplification, MET amplification, and transformation to squamous or small cell morphology.45,94,95
In T790M-positive tumors, following progression after first- or second-generation EGFR TKIs, second-line osimertinib has demonstrated significant efficacy compared with chemotherapy (median PFS: 10.1 vs 4.4 months; HR, 0.30 (95% CI, 0.23‒0.40), P < 0.001). 48 In the LUX-Lung 7 trial, the 3-year OS rate was up to 90% in patients who received a third-generation EGFR TKI (osimertinib (10.3% of patients) or olmutinib (3.4%)) after discontinuing afatinib. 43 However, given the recent positive results of the FLAURA study showing improved efficacy with first-line osimertinib compared with a first-generation TKI in patients with EGFRm+ NSCLC, 13 and the subsequent approval of osimertinib in this setting, 51 the most appropriate sequencing of the EGFR TKIs for optimal benefit is unclear. The second- and third-generation TKIs have shown significantly longer PFS compared with first-generation TKIs, and the third-generation TKI, osimertinib, has shown superior OS compared with first-generation TKIs.41,42,49 While the major mechanism of resistance to first- and second-generation TKIs is well defined, mechanisms of acquired resistance to osimertinib are more heterogeneous, and include EGFR C797S, acquired KRAS, and targetable gene fusions.96–98 These data suggest that the net survival benefit could potentially be optimized by reserving osimertinib for second-line use in the >60% of patients who develop T790M-mediated resistance (Figure 3). This is supported by recent data from a global, noninterventional study of patients with EGFRm+ NSCLC receiving sequential afatinib and osimertinib in a real-world setting, which showed median time to treatment failure of 28.1 months (90% CI, 26.8–30.3) and median OS of 41.3 months (90% CI, 36.8–46.3). 99 In addition, MET amplification has also been shown to co-exist with the T790M mutation, 95 suggesting that the combination of osimertinib with a MET inhibitor may be beneficial for such patients. 100 Detractors of this sequencing strategy will note that first, many patients experience disease progression on first-line EGFR TKIs and are not able to receive second-line therapy, due to death or deterioration of performance status, among other reasons. Indeed, discontinuation of erlotinib or gefitinib in patients with acquired resistance has been reported to cause rapid progression (disease flare). 101 In the FLAURA trial, 12% of patients treated with osimertinib and 17% treated with first-generation EGFR TKIs died without commencing a second-line therapy. 13 Second, it is impossible to prospectively predict which patients will develop T790M and thus benefit from this sequencing strategy. One study identified de novo T790M mutations more frequently within L858R tumors than within Del19 tumors among 20 EGFR TKI-naïve patients, 102 while mutation analysis of EGFR TKI-pretreated patients in the osimertinib AURA extension and AURA2 trials detected T790M mutations in a higher proportion of patients with Del19 mutations versus those with L858R. 91 Patients who do not develop T790M upon progression on first- or second-generation EGFR TKIs may have been better treated with osimertinib as first-line therapy, which has shown the longest median PFS of all EGFR TKIs, at 18.9 months (assessed by investigator review) in the FLAURA trial. 13
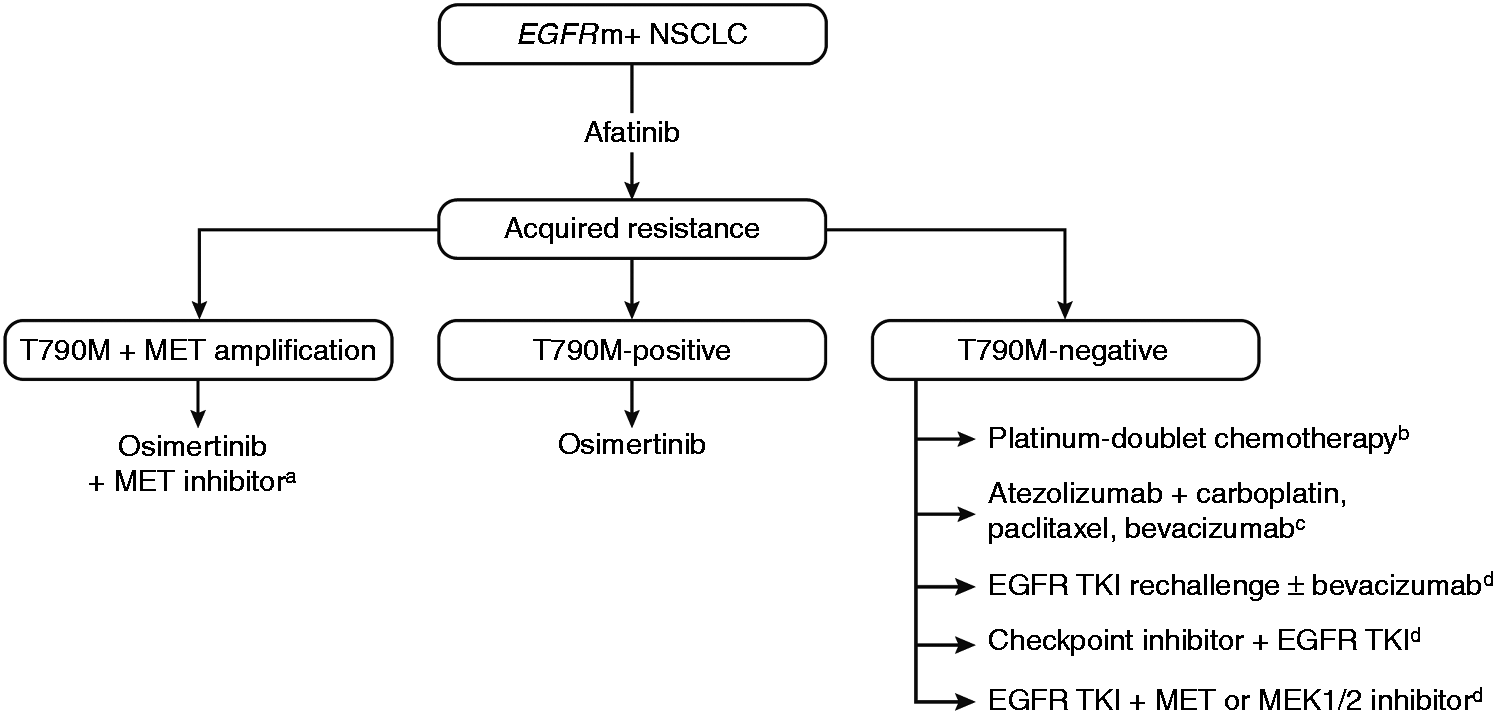
Potential treatment strategies following acquired resistance to afatinib. (a) Investigational therapy 100 ; (b) platinum-doublet chemotherapy represents the principal treatment option outside of the clinical trial setting 103 ; (c) this combination showed considerable efficacy in EGFRm+ NSCLC but has not yet been investigated specifically in patients with EGFRm+ T790M-negative tumors 109 ; and (d) Investigational treatment options with some activity in EGFRm+ NSCLC in early-phase studies.103,105–108EGFR: epidermal growth factor receptor; EGFRm+: EGFR mutation-positive; NSCLC: non-small cell lung cancer; TKI: tyrosine kinase inhibitor.
In contrast to T790M-positive tumors, subsequent treatment options for patients with T790M-negative acquired resistance are less well defined, reflecting the large variety of possible resistance mechanisms identified. 103 Currently, the standard treatment for these tumors is platinum-doublet chemotherapy. However, several other targeted treatments and combinations are under investigation in this setting (Figure 3). In tumors with MET amplification, the use of MET inhibitors together with EGFR TKIs has shown preliminary efficacy.95,104 Other approaches include the combination of afatinib with EGFR antibodies or bevacizumab, which have shown promise but some may be associated with considerable treatment-related toxicities.105–108 In addition, novel combinations incorporating checkpoint inhibitors may be effective for patients with T790M-negative acquired resistance. Recent results from the IMPOWER 150 trial indicate that the combination of atezolizumab, bevacizumab, and chemotherapy is effective in patients with NSCLC, regardless of PD-L1 expression and EGFR or ALK genetic alteration status. 109 This combination was assessed in patients with EGFRm+ NSCLC who had received prior EGFR TKI treatment, and it is possible that these findings may translate into the more specific setting of EGFRm+ NSCLC with T790M-negative acquired resistance. 109
Finally, rather than switching to second-line therapy, continuation of EGFR TKI treatment beyond radiological progression may help maintain clinical benefit for some patients with acquired resistance to first-line EGFR TKIs. 110 This approach may be beneficial to help avoid rapid disease progression with worsening of disease-related symptoms following treatment discontinuation (rebound tumor flare), which, as previously mentioned, can occur in some patients after discontinuation of EGFR TKIs. 110 Indeed, one analysis found that 14/61 patients (23%) who discontinued erlotinib/gefitinib treatment experienced a disease flare (defined as hospitalization or death due to disease progression), with a median time to disease flare following EGFR TKI discontinuation of 8 days. 101
Cost-effectiveness of afatinib
Economic assessments are essential to the healthcare decision-making process. In EGFRm+ NSCLC, EGFR TKIs have a clear benefit over platinum-based chemotherapy in terms of cost-effectiveness. 111 For example, one US study showed that erlotinib and afatinib were more cost-effective than cisplatin-pemetrexed, with erlotinib holding a cost-effectiveness advantage over afatinib. 112 Importantly, a budget impact analysis of first-line afatinib use in patients with EGFRm+ NSCLC in the US estimated that increasing the treatment share of afatinib in a health plan would lead to an increase in the proportion of treated patients who remained progression-free after 5 years, while having only a small impact on the health plan budget. 113 In contrast, a recent analysis of the cost-effectiveness of first-line EGFR TKIs in patients with EGFRm+ NSCLC suggested that osimertinib is not a cost-effective first-line therapy compared with first- or second-generation TKIs. 114 Further cost-benefit analyses of afatinib and other EGFR TKIs in patients with EGFRm+ NSCLC and SCC are needed to adequately assess the economic impact of the different treatment options and to identify the optimal sequencing strategy.
Summary and conclusions
In a little over a decade, developments in the molecular analysis of lung tumors have revealed that alterations in EGFR are major oncologic drivers in certain subtypes of lung cancer, with EGFR TKIs quickly becoming the “weapon of choice” for the treatment of patients with EGFRm+ NSCLC or SCC. Afatinib has been shown to provide significant improvements over chemotherapy and gefitinib in the first-line setting in patients with EGFRm+ NSCLC,39,40,42,43 and over erlotinib in the second-line setting in patients with SCC following chemotherapy. 84 Afatinib also has proven efficacy in patients with tumors harboring certain uncommon EGFR mutations, 75 and in patients with brain metastases. 79 The clinical effectiveness of afatinib has been confirmed by recent results of real-world studies.89,99 In addition, afatinib-related AEs are manageable by means of tolerability-guided dose adjustments, along with the use of AE-specific treatment administered either prophylactically or as needed.68,87,89,90 The optimal EGFR TKI sequencing strategy is still under debate. While some data show that a sequential treatment strategy consisting of afatinib followed by osimertinib can provide a viable, long-term treatment option for patients who develop T790M-mediated resistance, 99 this strategy would not be beneficial for the 30–50% of afatinib-treated patients who do not develop the T790M mutation.91,92 Head-to-head studies, including those of afatinib and osimertinib, are needed to adequately assess the optimal sequence of EGFR TKI therapies to achieve long-term treatment benefit. Additional studies are required to define optimal management approaches for patients with T790M-negative resistance, as are further pharmacoeconomic analyses to confirm preliminary findings that afatinib is cost-effective in the treatment of EGFRm+ NSCLC.112,113
Footnotes
Acknowledgments
The authors meet criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICMJE). The authors received no direct compensation related to the development of the manuscript. Writing and editorial support was provided by Hashem Dbouk, PhD, of GeoMed, an Ashfield Company, part of UDG Healthcare plc, which was contracted and compensated by Boehringer Ingelheim Pharmaceuticals Inc. (BIPI). BIPI was given the opportunity to review the manuscript for medical and scientific accuracy, as well as intellectual property considerations.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RDH declares research funding to his institution from Pfizer, and from AstraZeneca that supports his salary; VRA and PM declare no conflicts of interest. TB declares advisory board participation outside of the submitted work for AstraZeneca.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
