Abstract
Background:
Pulmonary toxicities caused by immune checkpoint inhibitors are a prominent concern for clinicians. Clinical Practice Guidelines (CPGs) are critical for managing these toxicities.
Methods:
A systematic search of CPGs on checkpoint-associated pulmonary toxicities (ca-PT) was conducted in October 2022. PubMed, Embase, Cochrane Library, CINAHL, and Web of Science were searched. AGREE II and AGREE-REX were used to appraise CPGs and recommendations quality, respectively. Descriptive statistics, intraclass correlation coefficient, Kruskal-Wallis (H) test, and Spearman’s correlation were used for analyses. P-values < .05 were considered statistically significant. Matrices were used to determine recommendation differences between CPGs. The study’s design was based on the PRISMA 2020 checklist for systematic reviews. Protocol registration number: CRD42022358435.
Results:
Eight CPGs (two high-quality, three moderate-quality, and three low-quality) were identified. All CPGs covered pneumonitis. One CPG covered pleural effusions and pneumonitis/SARs-CoV-2-infection. Three CPGs covered sarcoidosis-like-reactions. CPGs for pulmonary fibrosis, airway disease, bronchiolitis, and diffuse alveolar damage, were unavailable. No CPG recommendation was based on a prospective study, and none were appraised as high-quality. Also, recommendations were not specific to histopathologic subtypes. AGREE II’s “rigor of development,” the domain that evaluates a guideline’s methodological approach and strategies in gathering scientific evidence, correlated strongly with AGREE-REX’s “overall quality” pneumonitis recommendations, r = .952; P < .01. Approximately 73% of recommendations on pneumonitis were similar between high-quality CPGs. About 16% to 74% of low-quality CPGs were similar to those recommended by high-quality CPGs.
Conclusion:
Prospectively designed research projects focusing on all types of ca-PT and their histopathologic subtypes are urgently needed. Due to the lack of high-quality recommendations in available CPGs, the disparities in treatment recommendations between high-quality CPGs, and the similarities in recommendations that exists between high-quality and low-quality CPGs, clinicians should thoroughly assess and responsibly appraise all available CPG recommendations in formulating treatment strategies for ca-PT.
Keywords
Background
Checkpoint-associated pulmonary toxicities (ca-PT) are lung injuries associated with immune checkpoint inhibitor (ICI) therapy. The exact pathophysiologic mechanism of ca-PT has not been fully elucidated. It is postulated to be related to ICI-associated immune dysregulations and increased T-cell activities against cross-antigens expressed in malignant and normal lung tissues, the accumulation of inflammatory cells (predominantly lymphocytes and macrophages) in the lung parenchyma, the upregulation of pro-inflammatory cytokines and chemokines, as well as the reduced anti-inflammatory effects of regulatory T cells. Increased autoantibody levels and enhanced complement-mediated inflammation have also been implicated.1-3 Checkpoint-associated (ca)-pneumonitis is the most commonly occurring type of ca-PT. However, pleural effusions, airway disease, bronchiolitis, pulmonary fibrosis, diffuse alveolar damage, and sarcoidosis-like reactions have also been reported.1,3 Furthermore, ca-pneumonitis can present as interstitial lung disease (ILD), and histopathologic subtypes like cryptogenic organizing pneumonia, non-specific interstitial pneumonia, hypersensitivity pneumonitis, and acute interstitial pneumonia have been described.1-3 The incidence of ca-pneumonitis varies in different analyses. Still, it is primarily affected by drug, class (PD-1/PD-L1 blockade versus CTLA-4), cancer type, disease setting, and ICI regimen complexity (monotherapy versus combination therapy).1,3 Data from clinical trials show that incidences of any-grade pneumonitis are about 4% in anti-PD1 monotherapy, 2% in anti-PDL1 monotherapy, and less than 1% in anti-CTLA-4 monotherapy. The incidence of high-grade pneumonitis associated with any ICI is less than 1%. Pneumonitis associated with anti-PD1/PDL1 plus anti-CTLA-4 combination therapy is higher (about 10%) compared with ICI given as monotherapy (<5%). Preexisting ILD, former or current smokers, and men are at a higher risk of developing ca-PT than patients without any preexisting lung abnormalities, non-smokers, and women.1-4 Ca-PT has become a prominent concern for clinicians treating patients with ICIs.1-3 With no distinctive symptomatic, pathophysiologic, or radiographic features, ca-PT potentially poses significant challenges to clinicians in identifying, diagnosing, and managing patients promptly and effectively. Even though ca-PT can present with symptoms that include dyspnea, cough (new, persistent, or worsening), chest pain, fever, and hypoxia (potentially leading to respiratory failure), there are cases where patients were asymptomatic, and only with more sophisticated diagnostic apparatus like computed tomography scans were detectable inflammations observed. Ca-PT can cause irreversible pulmonary damage, and although fatalities are uncommon, some data show that about 0.4% of fatalities are associated with anti-PD-1/PD-L1 monotherapy and 1.2% with anti-PD1/PDL1 plus anti-CTLA-4 combination therapy. Pneumonitis encompasses the most frequent fatal events (about 35% of ca-PT cases). 1 Also, about 0.2% to 4.0% of patients treated with anti-PD-1/PD-L1 monotherapy discontinued treatment due to pneumonitis.4-6 Managing ca-PT is still based on clinical experience, data from tiny sample sizes that are retrospective or sometimes based on case reports, and algorithms from expert opinions or consensus. These limitations make Clinical Practice Guidelines (CPGs) published on managing ca-PT vital resources for clinicians making treatment decisions.7,8 Questions that therefore come up are whether quality CPGs with quality treatment recommendations on ca-PT are available, whether their recommendations for the treatment of ca-PT are like how non-ICI-associated pulmonary diseases are treated, and whether CPG treatment recommendations for ca-PT are tailored toward specific types of ca-PT, their histopathologic subtypes, and the grade/severity of the toxicity. We addressed these questions by completing a systematic review (SR) of all available CPGs with recommendations on at least one ca-PT. We reviewed all identified CPGs to assess the types of ca-PT and their histopathologic subtypes covered in these CPGs. We used AGREE II (Appraisal of Guidelines, Research and Evaluation II) to appraise the quality of CPGs and AGREE-REX (Appraisal of Guidelines Research and Evaluation–Recommendations Excellence) to determine the quality of CPGs’ treatment recommendations.8-10 Furthermore, we determined the number of high-quality CPGs available for managing ca-PT and investigated the correlation between CPG treatment recommendations and quality. We also assessed the differences in treatment recommendations between high-quality CPGs and low-quality CPGs.
Methods
Study design
This SR was designed using the PRISMA 2020 checklist for SRs (Supplemental Appendices 1 and 2). 11 The study protocol was registered with the international prospective register of SRs (CRD42022358435) and published online in the Open Science Framework repository before the study began. 12 We completed an SR of all CPGs with treatment recommendations on ca-PT (Table 1).
Eligibility criteria pertaining to PICAR statement.

Abbreviations: AGREE II, Appraisal of Guidelines for Research and Evaluation version 2; AGREE-REX, Appraisal of Guidelines Research and Evaluation–Recommendations Excellence; ca-PT, checkpoint-associated pulmonary toxicities; CPG, Clinical Practice Guideline; ICI, Immune-Checkpoint Inhibitor; irAEs, Immune-related adverse events; PICAR, Population-Intervention-Comparator-Attributes-Recommendation.
DistillerSR online software (DistillerSR Version 2.38.0 Evidence Partners, Ottawa, Canada, 2022) was used to facilitate duplicate removals, record screening, data entry, AGREE II/AGREE-REX quality appraisals, and to ensure the independence and blinding of all reviewers. The same three independent reviewers participated in all levels of the study. A standardized evidence quality evaluation grading rubric template for AGREE II and AGREE-REX was created before project initiation (see Supplemental Appendix 3).
Data sources and searches
PubMed, Embase, Cochrane Library, CINAHL, and Web of Science were searched. All searches were done in October 2022. The search strategies used a combination of controlled vocabulary that included the terms “immune-related adverse events, checkpoint-associated adverse events, immunotherapy-related adverse events, pulmonary toxicity, pneumonitis, lung toxicity, interstitial lung disease, sarcoidosis, pleural effusion, immune-checkpoint inhibitors, immunotherapy, and clinical practice guidelines” (see Supplemental Appendix 4). Google Scholar (https://scholar.google.com), well-known guideline repositories, and websites of major national and international organizations were searched for additional CPGs.
Selection of CPGs
Three reviewers (KD, HJ, and RS) independently screened records returned from the literature search. A first-level screen of titles and abstracts and a second-final-level screen of the full text of potentially eligible records were done using the eligibility criteria outlined in our PICAR (Population & clinical areas, Interventions, Comparators, Attributes of CPGs, and Recommendation characteristics) statement. CPGs proceeded to the next level if all three reviewers agreed that a CPG meets the study’s criteria for inclusion. Discrepancies in the selection process were resolved by consensus if at least one of the three reviewers’ opinions differed. CPGs were excluded in all cases where all three reviewers agreed on their exclusion. We selected CPGs based on the National Academy of Medicine standards for practice guidelines. We limited our choices to those commissioned by a national, international, professional association, or health ministry. 13 No Limitations were placed on region or year of publication. Only the latest CPG version, available in English, was selected.
CPG quality assessments
AGREE II is a tool that is used in evaluating CPG quality. The tool’s broader scope primarily targets the CPG’s developmental processes and quality.8,9 KD, HJ, and RS, independently and blinded from each other’s assessments, used AGREE II to appraise all selected CPGs. The reviewers rated twenty-three key items organized within the following six domains: (1) Scope and Purpose; (2) Stakeholder Involvement; (3) Rigor of Development; (4) Clarity of Presentation; (5) Applicability; and (6) Editorial Independence. With the aid of a grading rubric (Supplemental Appendix 4), each reviewer allotted domain points independently; each domain captures a unique dimension of guideline quality. Each AGREE II item was rated on a 7-point scale (1 = strongly disagree to 7 = strongly agree). A quality score was calculated for the six AGREE II domains (see Supplemental Appendix 3). Domains assessed with scores of seventy percent, or more were considered effectively addressed. A CPG was considered high-quality if it scored ⩾ 70% in at least three of the six AGREE II domains, including the “Rigor of Development” domain. CPG was moderate-quality if three or more domains were adequately addressed (standardized score ⩾ 70%), except for the “Rigor of Development” domain, or if the CPG adequately addressed at least two AGREE II domains (standardized score ⩾ 70%), except the “Rigor of Development” domain, but still, the “Rigor of Development” domain must have a standardized score of at least 50%. CPGs not meeting the high-quality or moderate-quality criteria were classified as low-quality.8,9,14
AGREE-REX assesses a CPG’s recommendations quality. The tool addresses three factors that must be considered to determine the quality of CPG recommendations. The domains are (1) Clinical Applicability, (2) Values and Preferences, and (3) Implementability. The AGREE-REX “overall quality score” describes a guideline’s recommendations’ overall quality. The tool evaluated the treatment recommendations for each type of ca-PT discussed in each selected CPG. Reviewers discussed and determined each domain score by consensus using the AGREE-REX grading rubric. Treatment recommendations appraised with an AGREE-REX overall score of at least 70% were considered high-quality. Those with less than 50% overall quality scores were considered low-quality treatment recommendations. Treatment recommendations with overall quality scores between 50% and 70% were considered moderate quality (see Supplemental Appendix 3).8,10
Treatment recommendation matrices and data extracted
Treatment recommendation matrices were created to assist in sorting out similarities and differences in ca-PT management strategies and to estimate the number of recommendations that were similar or closely matched between high-quality CPGs and between high-quality/low-quality CPGs. Data collected into the matrices included ICI-associated recommendations, corticosteroid-related recommendations, experts’ consultation recommendations, inpatient care/hospitalization recommendations, antimicrobial recommendations, recommended interventions to prevent complications for long-term corticosteroid use, and monitoring (laboratory/assessments/diagnostic) recommendations. One reviewer (KD) independently reviewed and entered all relevant data from each CPG into the matrix. Two reviewers (HJ and RS) reviewed every data entry for accuracy. Each reviewer independently compared recommendations between CPGs and noted those that were similar or closely matched between CPGs. Discrepancies in the recommendation comparison process were resolved by consensus if at least one of the three reviewer’s opinions differed. A recommendation between two CPGs on any particular subject was noted as different (not similar or closely related) in all cases where all three reviewers agreed they were different. The percentage of similar or closely matched recommendations on any topic between two CPGs was estimated by dividing the total number of similar or closely related recommendations between CPGs by the total number of recommendations made by both CPGs, multiplied by 100.
Statistical analyses
Statistical analyses were conducted using IBM Corp. Released in 2021. IBM Statistics for Windows, version 28.0.0.0 (190). Armonk, NY: IBM Corp. Descriptive analyses included mean (±standard deviation, SD). The average intraclass correlation coefficient (ICC), including the 95% confidence interval, was applied to estimate inter-rater reliability (IRR). Subgroup differences were explored with the Kruskal–Wallis (H) test. Spearman’s correlation was used in examining the relationship between the distinct domains. P-values < 0.05 were considered statistically significant.
Results
Literature search
Screening and guideline selection results have been summarized in a PRISMA flow diagram (Figure 1, Supplemental Appendix 4).

PRISMA 2020 flow diagram of included CPGs on checkpoint-associated pulmonary toxicities.
Four thousand three hundred and fifty records were identified. After removing duplicates and records deemed ineligible by the reviewers, the titles and abstracts of 4331 records were screened, excluding 4290 records. The full text of the resulting 41 records was screened, excluding 33. Eight CPGs with recommendations on managing ca-PT resulted in the SR.
General characteristics of identified CPGs
The organizations affiliated with the eight identified CPGs were Alberta Health Services (AHS), the American Society of Clinical Oncology (ASCO), Cancer Care Ontario (CCO), the European Society for Medical Oncology (ESMO), the Multinational Association of Supportive Care in Cancer (MASCC), National Comprehensive Cancer Network (NCCN), Spanish Society of Medical Oncology (SEOM), and Society for Immunotherapy of Cancer (SITC).15-22 AHS, ASCO, CCO, ESMO, NCCN, SEOM, and SITC CPGs covered other ICI-related toxicities besides ca-PT. MASCC was the only CPG with recommendations solely on ca-PT (ca-pneumonitis, ca-pleural effusions, ca-sarcoidosis-like reactions, and ca-pneumonitis with SARs-CoV-2-related pneumonia). 19 Besides MASCC, ESMO, and SITC had recommendations or comments on ca-sarcoidosis-like reactions.18,19,22 Three CPGs (CCO, MASCC, and SITC) had their definition/characterization for what is ca-pneumonitis slightly different from how it is defined in the Common Terminology Criteria for Adverse Events (CTCAE).17,19,22,23 Three CPGs (ASCO, ESMO, and NCCN) definition of ca-pneumonitis was closely matched to how it is defined in the CTCAE.16,18,20,23 Two CPGs (AHS and SEOM) did not define ca-pneumonitis.15,21 Apart from NCCN, which defined its terminology to grade ca-pneumonitis severity, CTCAE was utilized by all other CPGs. 23 Recommendations for managing ca-pneumonitis from all eight CPGs were generalized for any ca-pneumonitis regardless of the histopathologic subtype. Also, all recommendations from all eight CPGs were expert opinion or consensus-based (consensus methodology differed between guidelines). No recommendations from any of the eight CPGs were based on results from a prospective clinical trial. Per AHS and ASCO, the grade of recommendations (GOR)/level of evidence (LOE) for all recommendations made in the guideline was moderate strength, based on the best available evidence, with benefits outweighing harms. CCO and MASCC did not assign LOE/GOR to all recommendations made in the guideline. ESMO and SEOM adapted their grading system from the Infectious Diseases Society of America (IDSA)-United States Public Health Service (USPHS) grading system. 24 All ca-pneumonitis recommendations made by ESMO were either level IV (retrospective cohort studies or case-control studies) or level V (studies without control groups or based on case reports or merely by expert opinions). GOR of ESMO’s ca-pneumonitis recommendations was ranked as grade A (strong evidence for efficacy with a substantial clinical benefit, strongly recommended). ESMO’s recommendations for managing ca-sarcoidosis-like reactions had no GOR/LOE linked. SEOM’s LOE for all ca-pneumonitis recommendations was ranked as level V (studies without a control group, case reports, or merely by expert opinions). GOR for SEOM’s ca-pneumonitis was ranked as grade B (strong or moderate evidence for efficacy but with a limited clinical benefit, generally recommended). All recommendations on ca-pneumonitis by NCCN were ranked as category 2A (based upon low-level evidence, there is uniform NCCN consensus that the intervention is appropriate). 20 SITC relied on the Oxford Center for Evidence-Based Medicine (OCEBM) grading system for recommendations. SITC’s LOE for all ca-pneumonitis recommendations were either ranked as LE3 (non-randomized, controlled cohort, or follow-up study) or LE4 (case series, case-control, or historically controlled study). SITC’s recommendations on ca-sarcoidosis-like reactions had no LOE/GOR linked. 25 AHS, CCO, and MASCC originated in Canada. ASCO, NCCN, and SITC’s CPGs originated in the United States, ESMO’s headquarters was based in Switzerland, and SEOM’s in Spain (see Table 2 for details).
General characteristics of CPGs on checkpoint-associated pulmonary toxicities.

Abbreviations: AHS, Alberta Health Services; ASCO, American Society of Clinical Oncology; ca, checkpoint-associated; CCO, Cancer Care Ontario; CPGs, Clinical Practice Guidelines; CT, computed tomography; CTCAEv, Common Terminology Criteria for Adverse Events version; ESMO, European Society for Medical Oncology; EU, European Union; HQ, Headquarters; IDSA-USPHS grading system, Infectious Diseases Society of America-United States Public Health Service Grading System; ILD, Interstitial Lung Disease, recs, recommendations; MASCC, Multinational Association of Supportive Care in Cancer; N, No; N/A, Not applicable; NCCN category Category 2A, Based upon low-level evidence, there is uniform NCCN consensus that the intervention is appropriate; NCCN, National Comprehensive Cancer Network; NR, Not reported; OCEBM, Oxford Centre for Evidence-Based Medicine; Org, Organization; SEOM, Spanish Society of Medical Oncology; SITC, Society for Immunotherapy of Cancer; USA, United States of America; Y, Yes.
Newer CPG version/edition has been published with at least one recommendation notably different from the older version/edition.
Volume/version/edition was updated at least once in the same year.
CPG defines its grade/severity of ICI-associated pulmonary toxicities.
CPG quality appraisals via AGREE II
Figure 2 shows a heatmap of AGREE II standardized domain scores of CPGs with recommendations for at least one ca-PT (see Supplemental Appendix 5 for AGREE II individual appraiser scores).

Heat map of AGREE II CPG appraisal scores of CPGs on checkpoint-associated pulmonary toxicities.
Based on our predefined AGREE II criterion, appraisal of the eight CPGs with ca-PT management recommendations showed that ASCO and NCCN were high-quality CPGs; AHS, ESMO, and SITC were ranked moderate-quality; and CCO, MASCC, and SEOM were low-quality CPGs. An almost perfect agreement was present among all appraisers in this study, with an average ICC of 0.963 (95% CI 0.952, 0.971) (see Supplemental Appendix 6 for statistical analysis details).
The “Scope & Purpose” domain evaluates the main objective of a CPG and its target population.8,9 AHS, ASCO, ESMO, NCCN, SEOM, and SITC adequately addressed this domain with scores greater than 70%. CCO and MASCC poorly fulfilled this criterion with scores less than 50%. Significant differences existed between the MASCC/NCCN and MASCC/ASCO CPGs, P < .05. The “Stakeholder Involvement” domain evaluates whether relevant stakeholders were involved in developing the CPG and whether the views of the intended guideline users were represented.8,9 ASCO and NCCN adequately addressed this domain, scoring over 70% of the domain’s criteria. AHS, CCO, ESMO, and SITC moderately addressed the domain’s requirements with scores between 50% and 70%. There were no significant differences in appraisal scores between guidelines. The “Rigor of Development” domain evaluates a guideline’s methodological approach, strategies, and transparency in searching and gathering scientific evidence.8,9 ASCO and NCCN adequately addressed this domain, fulfilling the required criteria with scores of 87.5% and 72.2%, respectively. AHS, ESMO, and SITC moderately addressed this domain with scores between 50% and 70%. CCO, MASCC, and SEOM, with scores less than 50%, poorly addressed this domain. There were significant differences between MASCC/NCCN, MASCC/ASCO, CCO/NCCN, CCO/ASCO, and SEOM/ASCO, P < .05. The “Clarity of Presentation” domain evaluates the CPG’s structural design, formatting, and presentation. The domain also assesses whether the main recommendations can easily be identified and described precisely and unambiguously.8,9 AHS, ASCO, ESMO, NCCN, and SITC adequately addressed this domain with scores greater than 70%. SEOM moderately addressed this domain with a score of 57.4%. CCO and MASCC poorly addressed this domain criterion with less than 50% scores. Significant differences existed between the MASCC/ASCO and NCCN/ASCO CPGs, P < .05. The “Applicability” domain assesses the pertinent processes for successful guideline implementation. These processes include facilitators, barriers, and additional resources needed for implementation. The domain also evaluates whether the guideline provided any provisions for monitoring and auditing.8,9 ASCO, with a score of 77.8%, was the only CPG that adequately addressed this domain criterion. The other seven guidelines poorly addressed this domain criterion, with scores of less than 50%. There were significant differences between CCO and ASCO, P < .05. The “Editorial Independence” domain evaluates the funding, competing, and other interests of the CPG authors and experts involved in the guideline development.8,9 AHS, ASCO, NCCN, and SITC adequately addressed this domain criterion with scores greater than 70%. ESMO and MASCC moderately addressed this domain with scores of 61.1% and 50%, respectively. CCO had the lowest rating, fulfilling less than 10% of the domain’s criteria. There were no significant differences in appraisal scores between guidelines in this domain.
Checkpoint-associated pneumonitis
AGREE-REX quality appraisal rankings of ca-pneumonitis CPGs
Figure 3 is a heatmap of AGREE-REX quality appraisal rankings of treatment recommendations for ca-pneumonitis (see Supplemental Appendix 5 for AGREE-REX consensus scores).

Heatmap of AGREE-REX quality appraisal rankings of ca-pneumonitis.
There were no high-quality treatment recommendations from any of the CPGs for ca-pneumonitis; none of the CPGs scored over 70%. The two high-quality CPGs (ASCO and NCCN) had their treatment recommendations for ca-pneumonitis ranked as moderate-quality, with overall quality scores of 59.3% and 61.1%, respectively. Two moderate-quality CPGs (ESMO and SITC) were also appraised with moderate-quality treatment recommendations for ca-pneumonitis, with scores between 50% and 70%. AHS, another moderate-quality CPG, and three low-quality CPGs (CCO, MASCC, SEOM) had treatment recommendations for ca-pneumonitis rated low-quality with less than 50% AGREE-REX overall scores.
Recommendation differences between ca-pneumonitis CPGs
Table 3 shows ICI-related recommendation differences between CPGs per grade of ca-pneumonitis (see Supplemental Appendix 7 for full comparison details).
ICI-related recommendations for ca-pneumonitis.

Abbreviations: AHS, Alberta Health Services; ASC0, American Society of Clinical Oncology; ca, checkpoint-associated; CCO, Cancer Care Ontario; CPG, Clinical Practice Guideline; d/c, discontinue; ESMO, European Society for Medical Oncology; ICI, Immune Checkpoint Inhibitor; MASCC, Multinational Association of Supportive Care in Cancer; NCCN, National Comprehensive Cancer Network; SEOM, Spanish Society of Medical Oncology; SITC, Society for Immunotherapy.
[a] If there is evidence of radiographic improvement or resolution, can consider resuming ICI with close monitoring; [b] Proceed with close monitoring for worsening symptoms; [c] Consider holding ICI until resolution if the patient is on steroids; [d] Consider delaying ICI therapy with clinical observation; [e] If infiltrates have resolved, ICI therapy may be cautiously resumed with close follow-up; [f] Permanent withdrawal is recommended if the progression of toxicity despite drug cessation; [g] Progressive grade 1 pneumonitis requires a hold: consider resuming on radiographic evidence of improvement; [h] If there is a resolution of symptoms and radiographic improvement to grade 1 or less, can consider re-challenging with ICI; [i] Hold ICI till clinical improvement to grade < 1; [j] Withhold therapy until resolution to grade 0 to 1 without complications & prednisone dose tapered to less than 10 mg/day; [k] Rechallenge with ICI therapy upon complete resolution of symptoms can be considered on an individual basis with close monitoring; [l] Drug re-challenge following resolution of infiltrates in carefully selected patients with grade 2 pneumonitis is also reasonable, with follow-up; [m] Resume once pneumonitis has resolved to ⩽grade 1 and patient is off steroids. Resume once pneumonitis has resolved to ⩽grade 1 and the patient is on a steroid dose of ⩽ 10 mg/day of prednisone; [n] Patients may be re-challenged with ICIs upon complete resolution of symptoms.
Forty percent of ICI-related recommendations by ASCO and NCCN (high-quality CPGs) were similar or closely matched. A comparison of ASCO’s recommendations with low-quality CPGs showed that 75% of ICI-related recommendations made by ASCO were similar or almost similar to recommendations made by AHS, SITC, and SEOM. Sixty percent of ASCO’s ICI-related recommendations were similar or closely matched to recommendations made by CCO and MASCC, and forty percent of ASCO’s ICI-related recommendations were similar or almost similar to ESMO’s. Our evaluation of NCCN and low-quality CPGs showed that almost all ICI-related recommendations made in NCCN were similar or closely matched to ESMO’s recommendations. Seventy-five percent of recommendations made by NCCN were similar or closely matched to recommendations made in CCO. Fifty percent of NCCN’s recommendations were similar or closely matched with AHS, SEOM, and SITC. Thirty-three percent of NCCN’s ICI-related recommendations closely matched MASCC’s treatment recommendations.
Table 4 summarizes the corticosteroid-related recommendation differences between CPGs per grade of ca-pneumonitis (see Supplemental Appendix 7 for full comparison details).
Corticosteroid-related recommendations for ca-pneumonitis.

Abbreviations: AHS, Alberta Health Services; ASC0, American Society of Clinical Oncology; CCO, Cancer Care Ontario; CPG, Clinical Practice Guideline; d, day; ESMO, European Society for Medical Oncology; IVIG, intravenous immunoglobulin; kg, kilogram; MASCC, Multinational Association of Supportive Care in Cancer; MMF, mycophenolate mofetil; NCCN, National Comprehensive Cancer Network; NR, not reported; SEOM, Spanish Society of Medical Oncology; SITC, Society for Immunotherapy.
[a] Consider Prednisone PO or methylprednisolone IV 1 mg/kg/d; [b] Monitor symptoms every 2 to 3 days. If symptoms worsen treat as grades 2 or 3/4; [c] Repeat radiographic imaging in 3-4 weeks. If no improvement, should be treated as grade 2; [d] Dosed at 1 mg/kg of ideal body weight/day; [e] Corticosteroids 2 mg/kg of ideal body weight/day; [f] Subset of patients may develop chronic pneumonitis and may require longer taper; [g] Consultation with or referral to an expert should be arranged and therapeutic escalation should occur; [h] No improvement in pneumonitis symptoms within 72 hours (or if symptoms are life-threatening); [i] There is no standard agent in this setting; [j] the clinical impact of augmented immunosuppressive therapies and optimal dose, duration, and timing of these agents have not been studied. Evidence favoring the use of IVIG has also emerged; [k] Taper in consultation with pulmonary service.
ASCO and NCCN made similar or almost similar corticosteroid-related recommendations about 57% of the time. Sixty percent of corticosteroid-related recommendations made by ASCO were similar or closely matched to recommendations made by AHS and SITC, 30% were similar to ESMO and MASCC, 40% were similar to CCO, and 15% were similar to SEOM. With NCCN, about 47% of corticosteroid-related recommendations were similar to that of AHS, 30% were similar or almost similar to CCO and ESMO, 25% were similar or closely matched to MASCC and SITC, and 15% were similar to SEOM.
Table 5 summarizes specialist/expert consultations and inpatient care/hospitalization recommendation differences between CPGs per grade of ca-pneumonitis (see Supplemental Appendix 7 for full comparison details).
Experts/specialists recommendations and inpatient/hospitalization recommendations for ca-pneumonitis.

Abbreviations: AHS, Alberta Health Services; ASC0, American Society of Clinical Oncology; CCO, Cancer Care Ontario; CPG, Clinical Practice Guideline; ESMO, European Society for Medical Oncology; MASCC, Multinational Association of Supportive Care in Cancer; NCCN, National Comprehensive Cancer Network; SEOM, Spanish Society of Medical Oncology; SITC, Society for Immunotherapy.
[a] Consider pulmonary and infectious diseases consult; [b] Pulmonary and infectious disease consults if necessary; [c] Consider pulmonary consultation; [d] If there is no improvement within 72 h of corticosteroid use, consultation with or referral to an expert should be arranged, and therapeutic escalation should occur; [e] Patients should be referred to a specialist when they experience toxicities of grade ⩾ 3, if toxicities of any grade do not respond to steroid treatment, or if toxicities require hospitalization.
Eighty percent of expert consultations and hospitalization recommendations made in ASCO and NCCN were similar or almost similar. All recommendations made by SITC differed notably from those made by NCCN or ASCO. A little over twenty percent of recommendations made in AHS, ESMO, MASCC, and SEOM were similar to those made in NCCN and ASCO. A little over 50% of CCO’s expert consultation and hospitalization recommendations were similar to those in NCCN and ASCO.
Table 6 summarizes laboratory and other diagnostics recommendation differences between CPGs per grade of ca-pneumonitis (see Supplemental Appendix 7 for full comparison details).
Laboratory/assessments/diagnostics/other recommendations for ca-pneumonitis.

Abbreviations: AHS, Alberta Health Services; ASC0, American Society of Clinical Oncology; BAL, Bronchoalveolar lavage; CCO, Cancer Care Ontario; CPG, Clinical Practice Guideline; CT, Computed tomography; ESMO, European Society for Medical Oncology; H&P, History and physicals; MASCC, Multinational Association of Supportive Care in Cancer; NCCN, National Comprehensive Cancer Network; SEOM, Spanish Society of Medical Oncology; SITC, Society for Immunotherapy.
Over 80% of laboratory/assessment/diagnostic/and antimicrobial recommendations made in CPGs were similar or almost similar in AHS, ASCO, NCCN, and ESMO. Only 11% of SEOM’s recommendations were similar or closely related to those made in ASCO or NCCN. A little over 50% of recommendations made in CCO and MASCC’s recommendations were similar or almost similar to those made in ASCO and NCCN. About 35% of laboratory/assessment/diagnostic/and antimicrobial recommendations made in SITC were similar to those made by both ASCO and NCCN.
Correlations among the distinct AGREE II domains and AGREE-REX overall treatment recommendation quality of ca-pneumonitis CPGs
Significant correlations existed between AGREE II domains and the AGREE-REX overall quality of ca-pneumonitis treatment recommendations (Figure 4). The AGREE II “Scope and Purpose” domain showed positive and significant (P < .05) correlations with the AGREE II domains “stakeholder Involvement” r = .970; “Rigor of Development” r = .922; “Applicability” r = .747; and “Editorial Independence” r = .838. The domain also showed positive and significant (P < .05) correlations with AGREE-REX “Overall Quality” estimation, r = .826. The “Stakeholder Involvement” domain showed positive and significant (P < .05) correlations with the AGREE II domains, “Rigor of Development” r = .881, “Applicability” r = .755, and “Editorial Independence” r = .786. The domain also showed a positive and significant (P < .05) correlation with the AGREE-REX “Overall Quality” estimation, r = .738. The “Rigor of Development” domain showed positive and significant (P < .05) correlations with the AGREE II domain “editorial independence” r = .762. The domain also showed strong and significant (P < .01) correlations with the AGREE-REX “Overall Quality” estimation, r = .952. The “Applicability” domain correlated significantly (P < .05) with AGREE-REX’s “Overall Quality” estimation, r = .731 (see Supplemental Appendix 6 for statistical analysis details).
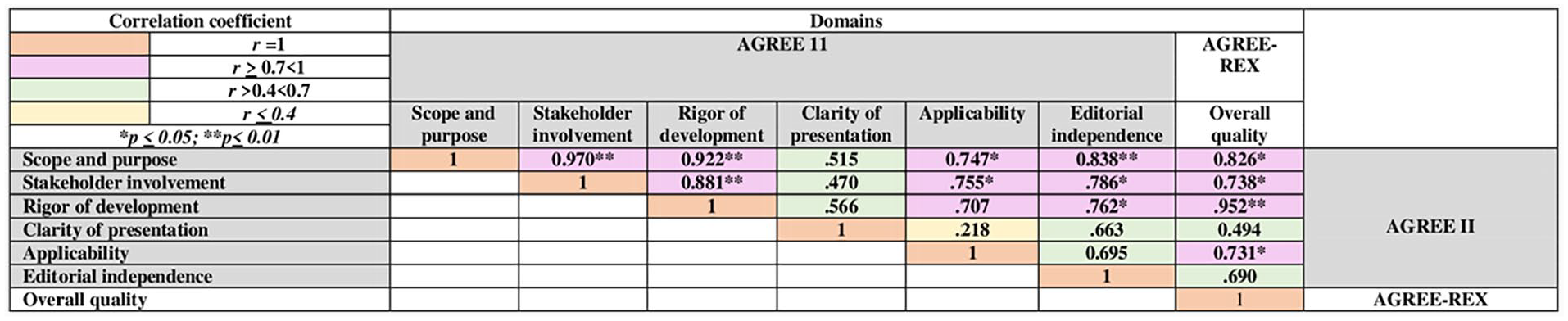
Correlations among the distinct AGREE II domains and AGREE-REX overall treatment recommendations quality of ca-pneumonitis CPGs.
Checkpoint-associated sarcoidosis-like reactions
AGREE-REX quality appraisal rankings of ca-sarcoidosis-like reactions CPG treatment recommendations
No high-quality or moderate-quality treatment recommendations from the three CPGs addressed ca-sarcoidosis-like reactions. None of the CPGs scored over 50% (Figure 5). The two moderate-quality CPGs (ESMO and SITC) and one low-quality CPG (MASSC) were appraised by consensus with the following AGREE-REX overall quality scores: ESMO (14.8%), MASCC (11.1%), and SITC (5.6%) (see Supplemental Appendix 5 for AGREE-REX consensus scores).

Heatmap of AGREE-REX quality appraisal rankings of ca-sarcoid-like reactions treatment recommendations.
Recommendation differences between ca-sarcoidosis-like reactions CPGs
Recommendations made by all three CPGs for ca-sarcoidosis-like reactions were not graded by severity (Table 7). In addition, there were no LOE/GOR-linked recommendations. ESMO gave the most elaborate recommendations among the three CPGs, recommending that in cases where ICI was benefiting patients and if the patient is asymptomatic to ca-sarcoidosis-like reactions, ICI therapy can be continued. However, if a patient is symptomatic, lower corticosteroid doses (
Recommendation differences between CPGs on ca-sarcoidosis-like reactions.

Abbreviations: CPG, Clinical Practice Guideline; ESMO, European Society for Medical Oncology; ICI, Immune Checkpoint Inhibitor; irAE, Immune-related adverse events; MASCC, Multinational Association of Supportive Care in Cancer; SITC, Society for Immunotherapy.
Checkpoint-associated pleural effusion & checkpoint-associated pneumonitis w/ SARs-CoV-2-related pneumonia
MASCC’s AGREE-REX overall quality score was 9.3% for ca-associated pleural effusion and 14.8% for ca-pneumonitis w/ SARs-CoV-2-related pneumonia (Figure 6).

MASCC AGREE-REX quality appraisal rankings of ca-pleural effusion and ca-pneumonitis w/ SARs-CoV-2-related pneumonia.
Results summary
Table 8 summarizes the main findings of the SR, quality appraisals, and contrasts between the eight CPGs available for managing ca-PT.
Summary of study results.

Abbreviations: AGREE II, Appraisal of Guidelines for Research and Evaluation version 2; AGREE-REX, Appraisal of Guidelines Research and Evaluation–Recommendations Excellence; AHS, Alberta Health Services; ASCO, American Society of Clinical Oncology; CCO, Cancer Care Ontario; CPGs, Clinical Practice Guidelines; ESMO, European Society for Medical Oncology; ILD, Interstitial Lung Disease; IVIG, intravenous immunoglobulin; MASCC, Multinational Association of Supportive Care in Cancer; MMF, mycophenolate mofetil; NCCN, National Comprehensive Cancer Network; Recs, Recommendations; SEOM, Spanish Society of Medical Oncology; SITC, Society for Immunotherapy of Cancer.
—Not applicable.
Results have been noted in another column.
Discussion
Main findings
This SR was done to determine the number, quality, and treatment recommendation differences of ca-PT CPGs. Eight CPGs (AHS, ASCO, CCO, ESMO, MASCC, NCCN, SEOM, and SITC) on managing ca-pneumonitis are available. Apart from ca-sarcoidosis-like reactions that were briefly covered by three CPGs (ESMO, MASCC, SITC), ca-pleural effusion and ca-pneumonitis w/ SARs-CoV-2-related pneumonia (briefly covered in MASCC), CPGs for less commonly presenting ca-PT (airway disease, bronchiolitis, pulmonary fibrosis, diffuse alveolar damage, and others), are not available. In addition, diagnostic and management strategies presented in CPGs on ca-pneumonitis were largely generalized and not tailored to specific histopathologic subtypes (cryptogenic organizing pneumonia, non-specific interstitial pneumonia, hypersensitivity pneumonitis, and acute interstitial pneumonia). For ca-sarcoidosis-like reactions and ca-pleural effusion, management strategies recommended by CPGs were very brief, with little regard to the severity of the toxicity.
Two CPGs (ASCO and NCCN) were high-quality CPGs. Three CPGs (AHS, ESMO, and SITC) were moderate-quality, and three CPGs (CCO, MASCC, SEOM) were low-quality. None of the eight CPGs had high-quality treatment recommendations for ca-pneumonitis. The two high-quality CPGs (ASCO, NCCN) and two moderate-quality CPGs (ESMO, SITC) were appraised with moderate-quality recommendations for managing ca-pneumonitis. AHS (a moderate-quality CPG) and all three low-quality CPGs (CCO, MASCC, SEOM) were appraised with low-quality recommendations on managing ca-pneumonitis. MASCC’s recommendations on sarcoidosis-like reactions, ca-pleural effusion, and ca-pneumonitis w/ SARs-CoV-2-related pneumonia, and ESMO/SITC’s recommendations on sarcoidosis-like reactions were all low-quality treatment recommendations. No recommendations in any of the CPGs are attributed to a prospective study. Retrospective cohort studies, case-control studies, studies without control groups, case reports, and expert opinions supported recommendations in all CPGs. About 73% of all recommendations made in ASCO and NCCN on managing ca-pneumonitis were similar or closely related. We estimated that about 27% of ca-pneumonitis recommendations differed between NCCN and ASCO. Another remarkable observation was that many ca-pneumonitis treatment recommendations in low-quality CPGs (CCO, MASCC, and SEOM) were similar to those made in ASCO and NCCN. Approximately 50% of management recommendations made by CCO for treating ca-pneumonitis were either similar or almost similar to recommendations made by ASCO and NCCN. A little over 50% of recommendations made in MASCC for treating ca-pneumonitis were either similar or almost similar to recommendations made by ASCO, and 43% were similar or almost similar to recommendations made in NCCN. About 16% of recommendations made by SEOM were similar or almost similar to those made by ASCO and NCCN. What is noteworthy from these results is that ASCO and NCCN (appraised as high-quality CPGs) had their treatment recommendations for ca-pneumonitis ranked as moderate-quality. Even though some of the same treatment recommendations were made in low-quality CPGs (CCO, MASCC, and SEOM), the treatment recommendations were appraised as low-quality. These findings are consistent with other studies and underscore the importance and clear benefits of developing high-quality CPGs.8,26 CPGs that strictly adhere to an evidenced-based developmental process and clearly explain methodological approaches, strategies, and transparency in searching and gathering scientific evidence; even in the absence of available prospective clinical trials, the clinical applicability of the recommendation, ease of Implementing the recommendation, and therefore the overall quality of the treatment recommendations are positively influenced.8,26 Correlation analyses among the distinct AGREE II domains and AGREE-REX “Overall Quality” estimation of ca-pneumonitis CPGs validated our findings. “Rigor of Development” (the domain that evaluates a guideline’s methodological approach, strategies, and transparency in searching and gathering scientific evidence) significantly (P < .01) correlated strongly and positively with the overall quality of ca-pneumonitis CPG treatment recommendations, r = .952.
Checkpoint-associated pulmonary toxicities versus non-ICI-related pulmonary diseases
Notwithstanding that, it is beyond the scope of this study to review the appropriateness of therapeutic strategies for managing non-ICI-related pneumonitis and other pulmonary diseases; certain basic information can be looked at to see if fundamental differences or similarities in management strategies exist. 1-4 Although ca-pneumonitis management strategies do not necessarily mimic how non-ICI-related pneumonitis is treated, it is clear that corticosteroids and immunosuppressive therapies are vital in both disease states.1-3,27-30 For non-ICI-related pneumonitis, most of the references recommend that clinicians put in a considerable amount of effort in making a confident diagnosis of the specific type of pneumonitis to be treated, and formulate a histopathologic-specific, patient-centered, personalized management strategy.28-30 Therefore, pharmacologic treatments of non-ICI-related pneumonitis/ILD vary depending on the type and histology of the disease. Steroids and other immunosuppressants benefit inflammatory conditions, such as hypersensitivity pneumonitis, but must be avoided in idiopathic pulmonary fibrosis. Patients with acute or subacute inflammation, demonstrated by consolidation or ground glass opacity; steroids or steroid-sparing immunosuppressants (eg, cyclophosphamide, cyclosporine, and azathioprine) have been used either as monotherapy or in combination with other immunosuppressant agents. For patients with fibrosing ILDs, tyrosine kinase inhibitors (TKIs) like nintedanib have been used either in addition to immunosuppressants or as monotherapy.27-30 This contrasts with the findings in our study regarding ca-PT. Although most CPGs acknowledge that ca-pneumonitis has histopathologic subtypes, we found that the general approach to treatment is primarily dictated by the severity of the toxicity regardless of the histopathologic subtype. Also, the general pharmacotherapeutic strategy for treating ca-pneumonitis (particularly in grades 2-4 cases) is to use corticosteroids (doses/durations/frequencies varied depending on CPG) as first-line therapy. Recommendations for using other immunosuppressant agents (types of agents/doses/frequencies/duration varied depending on CPG) were only added to corticosteroids in cases where corticosteroids were determined to be refractory. None of the CPGs had recommendations for ca-pulmonary fibrosis, and TKIs like nintedanib are currently not recommended in any ca-PT CPG.15-22,27-30
Implications for practice
The results of our research have significant implications for clinicians in practice. Because no prospective trials are available for treating ca-pneumonitis and other ca-PT, clinicians must continue to rely on CPGs in their clinical decision-making when treating patients. However, based on our findings, high-quality CPGs do not necessarily give clinicians high-quality treatment recommendations, and low-quality CPGs do not necessarily mean their treatment recommendations are useless. This study showed differences between ca-PT CPGs and some clear overlaps in management strategies, regardless of CPG quality. Clinicians should, therefore, thoroughly assess and responsibly appraise treatment recommendations of all available CPGs when treating patients with ca-PT to develop the best management options. Because current CPG recommendations are generalized for all ca-pneumonitis and not tailored toward histopathologic subtypes, it is necessary to use a multidisciplinary team of experts, particularly pulmonologists and infectious diseases specialists, when treating patients. This is because they are well versed in determining histopathologic subtypes of pulmonary diseases (in the case of pulmonologists), ruling out infections (in the case of infectious diseases experts), and therefore must be included in clinical decisions when treating patients with ca-PT.
Implications for research and CPG development
The results of this study have implications for researchers and CPG developers. None of the recommendations from all eight CPGs were based on a prospective study. This proves that there is an urgent need for prospective studies on the best management strategies for ca-pneumonitis and other ca-PT. Results from a prospective clinical trial will greatly impact the quality of treatment recommendations in CPGs in a positive way. CPG developers will therefore be less reliant on expert opinions. Also, because of the generalized way ca-pneumonitis and other ca-PT are currently treated, research that focuses on treatments based on histopathology, as well as the severity of toxicity, will undoubtedly reveal whether it is more effective treating patients based on histology and severity of toxicity as compared with severity alone. We should all be interested in and curious about this potential research area. There are no guidelines for ca-pulmonary fibrosis and barely any for ca-sarcoidosis-like reactions, ca-pleural effusion, and other less commonly presenting ca-PT. There is, therefore, an urgent need for more research and the development of CPGs for these rarer checkpoint-associated lung toxicities.
Strengths and limitations
This study has several notable strengths. Our methodology included the use of a very comprehensive and robust search strategy that involved multiple databases. We also used a standardized and globally recognized guideline appraisal tool, AGREE II, in identifying guideline methodological quality; AGREE-REX, in determining the quality of CPG treatment recommendations. To ensure uniformity of guideline scores and minimize bias in the appraisal process, we created a grading rubric for AGREE II and AGREE-REX. DistillerSR was used throughout the study for all data meta-synthesis, ensuring reviewers’ independence and complete blinding. Furthermore, the created treatment recommendations matrices facilitated easy comparisons of treatment recommendations between guidelines.
There were limitations to this study. CPGs included were those published or available in English and commissioned by a national, international, professional association, or health ministry. Therefore, other potentially useful guidelines published by individual experts or non-English sources were excluded. Also, our strategy of searching for CPGs by combining adverse events, checkpoint inhibitors, and practice guidelines might have limited or restricted the discovery of potentially useful resources with treatment recommendations on checkpoint-associated pneumonitis and other lung toxicities. We did not limit or restrict CPG publication dates even though we only chose the most current CPG version if more than one version had been published. Furthermore, since we heavily depended on AGREE II in appraising CPGs, if the CPG and their accompanying resources lacked information regarding specific AGREE II domain criteria, we did not try to contact the appropriate organizations to verify these criteria, and that potentially affected the appraisal process. Another limitation is that the AGREE II and AGREE-REX appraisal tools do not have a standardized cut-off to determine quality. An arbitrary cut-off score, determined by researchers and loosely based on other studies, was used to distinguish between guideline scores. In our study, CPG quality was assessed domain-specific, with limits of
Conclusion
Due to the lack of high-quality ca-PT recommendations in CPGs, it is imperative that clinicians involve a multidisciplinary team of experts that includes pulmonologists and infectious diseases specialists and carefully assess and appraise all available CPGs, in formulating best management strategies. Future research should be prospective in design, focused on all types of ca-PT, with management strategies that account for histopathologic subtypes. CPG developers should make every effort to develop CPGs for all known types of ca-PT and not just for the most common types. Also, CPG developers must take an evidenced-based developmental approach, clearly explaining their methodological processes and strategies and being as transparent as possible when making treatment recommendations. Even in the absence of available prospective clinical trials, the clinical applicability of recommendations, ease of implementing recommendations, and, therefore, the overall quality of treatment recommendations are influenced by how the CPG was developed.
Supplemental Material
sj-docx-1-onc-10.1177_11795549231203153 – Supplemental material for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines
Supplemental material, sj-docx-1-onc-10.1177_11795549231203153 for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines by Kofi N Donkor, Hyeree Jang and Reena Sail in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-10-onc-10.1177_11795549231203153 – Supplemental material for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines
Supplemental material, sj-docx-10-onc-10.1177_11795549231203153 for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines by Kofi N Donkor, Hyeree Jang and Reena Sail in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-2-onc-10.1177_11795549231203153 – Supplemental material for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines
Supplemental material, sj-docx-2-onc-10.1177_11795549231203153 for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines by Kofi N Donkor, Hyeree Jang and Reena Sail in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-3-onc-10.1177_11795549231203153 – Supplemental material for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines
Supplemental material, sj-docx-3-onc-10.1177_11795549231203153 for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines by Kofi N Donkor, Hyeree Jang and Reena Sail in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-4-onc-10.1177_11795549231203153 – Supplemental material for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines
Supplemental material, sj-docx-4-onc-10.1177_11795549231203153 for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines by Kofi N Donkor, Hyeree Jang and Reena Sail in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-5-onc-10.1177_11795549231203153 – Supplemental material for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines
Supplemental material, sj-docx-5-onc-10.1177_11795549231203153 for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines by Kofi N Donkor, Hyeree Jang and Reena Sail in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-6-onc-10.1177_11795549231203153 – Supplemental material for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines
Supplemental material, sj-docx-6-onc-10.1177_11795549231203153 for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines by Kofi N Donkor, Hyeree Jang and Reena Sail in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-7-onc-10.1177_11795549231203153 – Supplemental material for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines
Supplemental material, sj-docx-7-onc-10.1177_11795549231203153 for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines by Kofi N Donkor, Hyeree Jang and Reena Sail in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-8-onc-10.1177_11795549231203153 – Supplemental material for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines
Supplemental material, sj-docx-8-onc-10.1177_11795549231203153 for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines by Kofi N Donkor, Hyeree Jang and Reena Sail in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-9-onc-10.1177_11795549231203153 – Supplemental material for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines
Supplemental material, sj-docx-9-onc-10.1177_11795549231203153 for A Systematic Review of Clinical Practice Guidelines for Managing Pulmonary Toxicities Caused by Immune Checkpoint Inhibitors: Quality of Treatment Recommendations and Differences in Management Strategies Between Guidelines by Kofi N Donkor, Hyeree Jang and Reena Sail in Clinical Medicine Insights: Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
