Abstract
Despite the major advancements in the management of chronic phase (CP) chronic myeloid leukemia (CML), blast crisis (BC) remains a major therapeutic challenge. BC can be myeloid, lymphoid, or mixed lineage with myeloid BC being the most common type. BC in CML is mediated by aberrant tyrosine kinase activity of the BCR::ABL fusion protein. The introduction of BCR::ABL tyrosine kinase inhibitor (TKI) has been a gamechanger in the treatment of CML and there has been a significant reduction in the incidence of BC. The main treatment goal in BC is to achieve a second CP and consolidate that with an allogeneic stem cell transplantation (SCT) in eligible patients. The outcomes in BC remain dismal even in the current era. In this review, we provide an overview of the biology and current therapeutic approach in myeloid BC.
Keywords
Introduction
Chronic myeloid leukemia (CML) is a triphasic myeloproliferative neoplasm (MPN) characterized by reciprocal translocation between chromosome 9q34 and chromosome 22q11.2 resulting in the formation of a BCR::ABL1 oncogene. Most CML patients (85%-90%), are diagnosed in the chronic phase (CP), however, in the absence of treatment, will inevitably advance to BC. 1 BC is the most dreaded complication of CML, and it is notoriously difficult to treat. Approximately, 20% to 25% of patients may develop de novo blast crisis (BC) without the intervening accelerated phase (AP). Although BC mimics de novo acute leukemia, it is a separate clinical entity altogether characterized by resistance to standard therapy. Fortunately, the risk of transformation to BC has diminished markedly (1%-1.5% per year) in the tyrosine kinase inhibitor (TKI) era. 2 Most patients (70%-80%) with CML-BC present with myeloid phenotype, whereas up to one third can transform to lymphoid BC. 3
The World Health Organization (WHO) defines BC as ⩾ 20% blasts in the bone marrow or peripheral blood or evidence of extramedullary disease. Clinical and laboratory features provide important clues regarding the cell lineage. Similarly, cytogenetic profile can provide additional information to differentiate the two types of BC. Mutations in TP53 and isochromosome i(17q) are more commonly seen in myeloid BC while hypodiploidy, monosomy 7, and CDKN2A mutations are frequently associated with lymphoid BC. 4 Flow cytometry (FC) and immunohistochemistry (IHC) can help confirm the cell lineage of the BC.
Mechanism of Progression to CML Blast Crisis
The precise mechanisms involved in transformation to blast crisis (BC) remain unclear. BC is believed to be mediated by increased BCR::ABL tyrosine kinase activity that drives secondary molecular and cytogenetic changes leading to clonal evolution and differentiation arrest.5,6 The fact that the risk of blast transformation is markedly reduced in the TKI era, further supports this theory. Mutations in TET2, ASXL1, CBL family, and IDH family are often seen in advanced phase CML with myeloid phenotype, and they highlight the aggressive nature of the disease. 7 Also, mutations in the polycomb repressive complex have been implicated in CML progression. 8
Management of Myeloid BC
Despite significant advancements in the management of CP-CML, BC remains a therapeutic challenge with dismal outcomes. It is critical to make the distinction between myeloid and lymphoid BC as it has important therapeutic and prognostic implications. All CML patients with newly diagnosed BC should have a detailed work up that includes complete blood counts, peripheral blood smear, immunophenotyping by FC, cytogenetics, BCR::ABL quantitative reverse transcription polymerase chain reaction (qRT-PCR), BCR::ABL kinase domain mutation and next-generation sequencing (NGS) myeloid panel. All patients who are candidates for an allogenic stem cell transplantation (SCT) should undergo a donor search and appropriate evaluation. The primary objective of treatment in patients with CML-BC is to achieve a second CP and proceed to allogenic SCT in eligible patients. In patients with CML-BC, a deep molecular response correlates with better outcomes. CML-BC patients who achieve a major molecular response (MMR) or complete cytogenetic response (CCyR) have dismal outcomes, and hence, the goal should be to achieve molecularly undetectable leukemia. 9 Several important factors, such as age, comorbidities, performance status, de novo versus progression on TKI, BCR::ABL kinase mutations have to be considered prior to selecting therapy for myeloid BC. Historically, single-agent cytarabine and combinations with idarubicin have been used with some degree of success, but remissions were often short lived.10,11
Single-Agent TKI in CML Myeloid BC
In CP-CML, BCR::ABL is the sole driver mutation, whereas, most patients (50%-80%) with BC tend to have additional cytogenetic alterations (ACAs) making them genetically complex. Unlike in CP-CML, single-agent TKI therapy (including third generation) are overall less effective in BC with responses being transient and the median survival is less than 1 year. The TKI response in BC depends on the cell lineage and ACA. The TKI selection depends on the prior therapy and the mutational profile.12,13 Patients with myeloid BC and ACA have a worse outcome when compared with those with lymphoid BC. 14 The most commonly used TKIs in BC are listed below. Clinical trial data supporting use of all the below-listed TKIs in BC are provided in Table 1.
Outcomes with single-agent TKIs in CML myeloid BC.

Abbreviations: BC, blast crisis; CCyR, complete cytogenetic response; CHR, complete hematological response; CML, chronic myeloid leukemia; CyR, cytogenetic response; HR, hematological response; MCyR, major cytogenetic response; NA, not applicable; TKI, tyrosine kinase inhibitor.
Imatinib is a first-generation TKI and clinical outcomes of imatinib monotherapy in BC remain unsatisfactory with most responses being transient. Pivotal trials that have investigated imatinib in BC are listed in Table 1. The recommended dose of imatinib in BC is 800 mg daily.
Dasatinib is a second-generation oral multitargeted TKI that is 325-fold more potent than imatinib. In a phase 1 study, imatinib-resistant or -intolerant patients treated with single-agent dasatinib showed impressive hematological and cytogenetic responses in advanced phase CML. 15 In CML-BC, dasatinib at a dose of 140 mg once daily and 70 mg twice daily are equally efficacious, but the former is better tolerated. 16
Nilotinib is a second-generation TKI that is more potent than imatinib. Most studies of nilotinib in CML-BC were done in patients intolerant or resistant to imatinib at a dose of 400 mg twice daily. Major adverse events to monitor while on nilotinib include hyperglycemia, pancreatitis, and vascular events. Nilotinib is approved in CP and AP-CML, but not in BC.
Bosutinib is a small molecule BCR::ABL and src TKI that is approved for all three phases of CML after prior treatment with a TKI. Patients treated with bosutinib in the second-line setting after failure of imatinib tend to have a much better response rate compared with those treated with bosutinib in the third line and beyond. The recommended dose in CML-BC is 500 mg once daily. Bosutinib does not have activity against T315I mutation. 17
Ponatinib is an oral multitargeted third-generation pan BCR::ABL TKI that has impressive clinical activity in BC. Ponatinib is unique as it is the only TKI that can overcome the T315I mutation. It is recommended that patients who progress to BC on a second-generation TKI should be started on ponatinib rather than switching to a different second-generation TKI. 18 The phase 2 PACE (Ponatinib Ph- positive acute lymphoblastic leukemia [ALL] and CML Evaluation) study evaluated ponatinib 45 mg daily in patients with all three phases of CML, including blast crisis (N = 62; myeloid blast crisis = 52). The median progression-free survival in patients with BC was 3.7 months and overall survival at 3 years was 9% 19
Currently, data supporting use of asciminib in CML-BC are not available.
In summary, outcomes of single-agent TKIs in CML-BC remain unsatisfactory, and it should ideally be used as a “bridge” to get patients to a second CP and eventually to an allogenic SCT. Except nilotinib, all other TKIs are currently approved in CML-BC.
Combination Therapy: Chemotherapy plus TKI in Myeloid BC
Clinical outcomes tend to be better when TKIs are combined with chemotherapy rather than either treatment modalities used alone. Myeloid BC is usually treated with chemotherapy regimens for acute myeloid leukemia (AML) in combination with a second- or third-generation TKI, and this recommendation is endorsed by the European Leukemia Net.31-33 In young fit patients, intensive chemotherapy (IC) plus TKI is an excellent induction regimen, whereas in elderly patients, hypomethylating agents (HMAs) in combination with a TKI is better tolerated. IC or HMA combined with TKI have a better response rate, lower relapse rate, and improved overall survival (OS) when compared with TKI alone. 34
(A) IC plus TKI:
Young patients with CML myeloid BC and good performance status can be treated with at combinations of TKIs and dose-intense chemotherapy. An overview of the studies is provided below and in Table 2.
Summary of studies showing outcomes of patients with myeloid BC treated with dose-intense chemotherapy plus TKI.
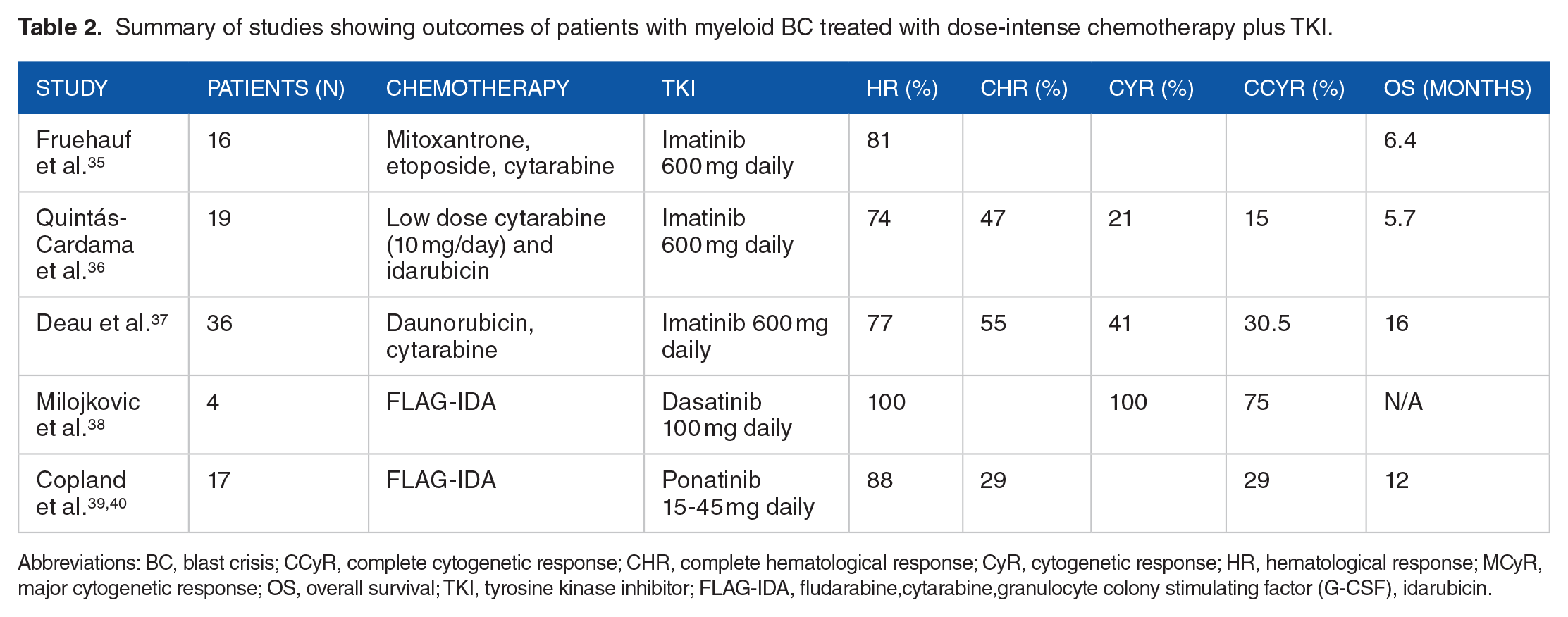
Abbreviations: BC, blast crisis; CCyR, complete cytogenetic response; CHR, complete hematological response; CyR, cytogenetic response; HR, hematological response; MCyR, major cytogenetic response; OS, overall survival; TKI, tyrosine kinase inhibitor; FLAG-IDA, fludarabine,cytarabine,granulocyte colony stimulating factor (G-CSF), idarubicin.
Fruehauf S et al reported a phase 1/2 trial of 16 patients with CML myeloid BC-treated imatinib in combination with mitoxantrone, etoposide, and cytarabine (MEC). Hematological response (HR) rate was 81% and median OS was 6.4 months. 35
Quintas Cardama et al reported a pilot study of 19 patients with CML myeloid BC who were treated with imatinib, low-dose cytarabine, and idarubicin. Fourteen patients (74%) achieved a HR and nine (47%) had a complete HR. Median survival was 5 months. 36
Deau et al conducted a dose escalation study of 36 patients with CML myeloid BC treated with standard 7 + 3 regimen consisting of daunorubicin combined with imatinib mesylate (600 mg/day) and cytarabine (200 mg/day for 7 days). A HR was observed in 28 patients (77.7%) with 20 (55.5%) achieving a complete HR. Median OS was 16 months and for those with a HR, it was 35.4 months 37
Milojkovic et al reported a small study of four patients who developed BC (myeloid-2, biphenotypic -1, lymphoid-1) on imatinib, and they were treated with a combination of dasatinib and FLAG-IDA (fludarabine, cytarabine, granulocyte colony-stimulating factor [G-CSF], and idarubicin). All patients achieved morphological remission and three of four had CCyR and MMR. 38
The MATCHPOINT phase 1/2 trial combined ponatinib with FLAG-IDA for patients in CML-BC. Eleven of 16 (69%) patients achieved a second CP after one cycle. Twelve of 17 (71%) patients proceeded to allogenic SCT. Three patients (18%) died from treatment-related toxicity. The 1-year OS was 45.8%.39,40
A combination of cladribine, idarubicin, and cytarabine (CLIA) is highly effective in newly diagnosed AML patients aged ⩽ 65. 41 Extrapolating data from the MATCHPOINT study, CLIA in combination with ponatinib could be a safe and effective regimen in CML myeloid blast crisis. A phase 2 study (NCT02115295) is currently investigating the role of CLIA regimen in combination with venetoclax in various myeloid neoplasms including CML myeloid BC.
(B) Older patients unfit for standard induction chemotherapy:
Older patients or those with comorbidities that preclude the use of dose-intense chemotherapy, can be treated with HMA-based regimens.
(i) HMA plus TKI
Abnormal DNA methylation of genes plays a crucial role in CML disease progression and resistance to TKIs. 42 Hence, logically HMA in combination with TKIs could prove to be an effective therapeutic option in myeloid BC. In a retrospective study of 162 patients with CML myeloid BC treated with different chemotherapy regimens, HMA had a similar objective response rate (ORR) compared with IC. In elderly patients, HMA-based regimens achieved a better survival presumably due to better tolerance and lower induction mortality.43,44 Some important trials that have shown encouraging activity of HMA and TKI in patients with CML myeloid BC are listed in Table 3.
Outcomes with HMA-based therapy with/without TKI.

Abbreviations: CCyR, complete cytogenetic response; CHR, complete hematological response; CyR, cytogenetic response; HR, hematological response; HMA, hypomethylating agent; MCyR, major cytogenetic response; NA, not applicable; TKI, tyrosine kinase inhibitor.
(ii) Venetoclax plus TKI
Maiti et al investigated outcomes of patients with advanced Philadelphia-positive myeloid neoplasms (N = 9, myeloid BC) treated with venetoclax and various TKIs. The ORR in patients with myeloid BC was 75% (6/8), three patients achieved CCyR and four had minimal residual disease negative status. The median relapse-free survival was 3.9 months. 50 The synergistic activity of BCL-2 and BCR::ABL inhibition needs to be investigated further in larger studies.
Homoharringtonine-Based Regimens
Homoharringtonine (HHT), is a natural plant alkaloid with clinical activity in all phases of CML. Listed below are some clinical trial data looking at combinations of HHT and TKI in CML-BC.
In a phase 2 trial, 15 CML patients (BC, N = 9) in various phases of the disease (myeloid BC -3) were treated with HHT and Imatinib. The ORR was 40% at 4 months. Two patients with myeloid BC achieved CHR and one had CCyR. The median OS in the entire cohort was 4.6 months. 51
Standard-dose imatinib combined with G-CSF and low-dose HHT was evaluated in a small study of 11 patients with CML myeloid BC who had failed imatinib therapy. The HR rate was 100% and seven (64%) achieved a CHR. MCyR and CCyR were 100% and 27%, respectively. 52
A phase 1/2 study evaluated subcutaneous HHT in patients with CML (myeloid BC, N = 8) who have failed imatinib therapy. HHT was found to be safe with all five evaluable patients achieving HR. 53 Response data were not available for patients with myeloid BC.
HHT in combination with cytarabine (HA regimen) is also an effective treatment option in myeloid BC. In a small study of 34 patients treated with HA regimen, the overall HR rate was 60% and 21% had a cytogenetic response. 54
HHT-based therapies appear promising and merit further investigation in combination with third-generation TKIs.
Novel and Emerging Therapies in CML Myeloid BC
Venetoclax-based regimens. BCL-2 is expressed on CML stem cells, and it has been shown in mice models that a combination of BCL-2 inhibitor and BCR::ABL TKI has synergistic properties and can eliminate the stem cells that are responsible for relapse. 55 A phase 2 clinical trial (NCT04188405) will investigate decitabine, venetoclax, and ponatinib in patients with Philadelphia-positive myeloid leukemias including CML myeloid BC.
Azacitidine (75 mg/m2 daily for 7 days) is currently being investigated in combination with ponatinib (45 mg daily) in an open-label phase 2 study (Ponaza trial) (NCT03895671).
Janus kinase inhibitor (JAK). Preclinical studies have showed that HMA when combined with JAK1/JAK2 inhibitor (ruxolitinib) has synergistic properties. 56 A phase 2 study (N = 25, BC = 15) of decitabine plus ruxolitinib showed an ORR of 44% and median OS in the entire cohort was 9.5 months in patients with advanced MPN. 57
A phase 1b study is investigating the triplet combination of fludarabine, cytarabine and pegcrisantaspase in patients with relapsed/refractory leukemia including those with CML myeloid BC (NCT04526795).
Hu8F4, a monoclonal antibody targeting PR1/HLA-A2, is currently being investigated in patients with various advanced hematological malignancies including CML myeloid BC (NCT02530034)
A phase 1 study is investigating the role of HA-1T cell receptor T cell immunotherapy in patients with relapsed/refractory leukemia after allogenic SCT (NCT03326921).
Isocitrate dehydrogenase (IDH) inhibitor: Somatic mutations in IDH1 and IDH2 can be detected in patients with CML-BC and patients harboring these mutations could benefit from a targeted IDH1/IDH2 inhibitor added to TKI and chemotherapy. 58 Also, IDH2 inhibitor in combination with ruxolitinib is being investigated in a phase 2 clinical trial of accelerated and blast phase MPN (NCT04281498).
Gemtuzumab ozogamicin (GO), a CD33-targeted monoclonal antibody has been used successfully either as a single agent or combined with chemotherapy to achieve CHR and CCyR in patients with CD33-positive CML myeloid BC. 59 Jabbour et al reported that in heavily pretreated patients with myeloid neoplasms, including those with CML myeloid BC, cytarabine combined with twice daily fludarabine and GO could be an effective regimen achieving an ORR of 26% with a 21% CR. 60
Asciminib is a novel, first-in-class allosteric ABL myristoyl pocket inhibitor approved in patients with CP-CML that has failed two or more TKIs. The phase 3 ASCEMBL trial included 233 patients with CP-CML and MMR was 25.5% with asciminib and 13.2% with bosutinib. CCyR with asciminib and bosutinib was 40.8% and 24.2%, respectively. 61 In ponatinib-naïve patients with T315I mutation, asciminib was able to achieve an MMR of 66.7% and in patients pretreated with ponatinib, the MMR dropped to 32.1%. 62 Overall, asciminib is an exciting new drug with a unique mechanism of action and is more potent than most of the existing TKIs. A phase 1 study is investigating asciminib in combination with either imatinib or dasatinib or nilotinib in Philadelphia chromosome-positive myeloid neoplasm including myeloid BC (NCT02081378).
Vodobatinib, a novel third-generation BCR::ABL TKI is being investigated in a phase 1/2 study in treatment refractory CML (NCT02629692).
Olverembatinib (HQP1351) is a third-generation BCR::ABL TKI that is currently being investigated in a phase 2 study in combination with decitabine in patients with advanced CML (NCT05376852).
Allogenic SCT in CML-BC
Allogeneic SCT remains the only curative treatment option in patients with CML-BC. Pretransplant remission status determines post-allogenic SCT outcomes. 63 Niederwieser et al reported long-term outcomes of patients who received allogenic SCT for CML-BC (n = 96) and AP (n = 51). The OS at 15 years was 34% and the non-relapse mortality was 28%. Although the cumulative incidence of relapse post-SCT was 43%, there were no relapses beyond 5 years and this clearly establishes the role of SCT in achieving long-term leukemia-free survival. Patients with active BC at the time of SCT did poorly when compared with non-BC patients. Hence, it is critical to achieve a second CP prior to allogenic SCT. A summary of studies looking at outcomes of CML -BC patients who underwent allogenic SCT is provided in Table 4. 64
Summary of studies showing outcomes of patients who received allogenic SCT for CML-BC.

Abbreviations: BC, blast crisis; CML, chronic myeloid leukemia; OS, overall survival; SCT, stem cell transplantation.
In summary, the long-term outcomes in CML-BC with allogenic SCT remain poor. Treatment-related mortality and disease relapse remain the major barriers for improving outcomes. However, the outcomes are still better when compared with non-transplant options. There is an unmet need for novel therapies to reduce the relapse risk.
TKI maintenance post-allogenic SCT
Although endorsed by NCCN, the data supporting the use of post-allogenic SCT, TKI maintenance therapy is fairly limited. 67 Neiderweiser et al showed a significant survival benefit with TKI maintenance post-allogenic SCT in CML-BC. 64 BCR::ABL quantitative polymerase chain reaction (qPCR) should be monitored every 3 months for 2 years post-allogenic SCT. 68
Preventing BC in CML
As clinical outcomes of CML-BC remain suboptimal even in the current era, the emphasis should be to prevent progression to BC. A tailored treatment approach taking into account the disease risk, toxicity profile, comorbidities, and cost of TKIs is key to improving outcomes. 69 Figure 1 shows the algorithm for TKI selection in CP-CML and Figure 2 shows optimal response milestones. The treatment response can be monitored by serial measurements of BCR::ABL1 by qPCR in the peripheral blood. Patients who fail to achieve the milestones should be evaluated for drug resistance, compliance, and appropriate changes should be made.

Algorithm for TKI selection in CP-CML. CML indicates chronic myeloid leukemia; CP, chronic phase; TKI, tyrosine kinase inhibitor.

Optimal milestones in CML-CP therapy. CML indicates chronic myeloid leukemia; CP, chronic phase.
CML-CP patients with major-route ACAs have a poor response to TKIs and are at higher risk for disease progression to BC. 70 Also, patients with mutations in the ABL kinase domain of the Philadelphia chromosome have TKI resistance and they remain at high risk for blast transformation. 71 Hence, patients with high-risk disease would benefit from early use of second- or third-generation TKI like ponatinib (known for pan BCR::ABL1 inhibition) and early allogenic SCT. Close monitoring for emerging mutations is required while on a TKI and appropriate therapeutic changes are essential to prevent progression to BC.
Choosing a TKI Based on the Mutational Profile
T315I mutation—ponatinib.
F317L/V/I/C, T315A mutations, V299L, nilotinib is preferred over dasatinib.72-74
F359V/C/I, Y253H, E255K/V mutations, dasatinib is preferred over nilotinib.75-77
The efficacy of dasatinib and nilotinib is likely similar in most other mutations.
Ponatinib is an option in all the above categories for patients previously treated with a TKI.
Conclusion
CML-BC is rare disease associated with poor outcomes. Figure 3 summarizes the currently treatment approach in CML myeloid BC. There is an unmet need for novel therapies that can achieve a second CP with low-induction mortality that will allow patients to proceed to an allogeneic SCT. Given the limited curative potential of existing therapies, the emphasis should be on prevention of BC.

Current management in CML myeloid BC. BC indicates blast crisis; CML, chronic myeloid leukemia; CP, chronic phase; HMA, hypomethylating agent; SCT, stem cell transplantation; TKI, tyrosine kinase inhibitor; FLAG-IDA, fludarabine,cytarabine,granulocyte colony stimulating factor (G-CSF), idarubicin.
Footnotes
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
