Abstract
Background:
Breast cancer (BC) is the most common neoplasm in women worldwide. Liquid biopsy (LB) is a non-invasive diagnostic technique that allows the analysis of biomarkers in different body fluids, particularly in peripheral blood and also in urine, saliva, nipple discharge, volatile respiratory fluids, nasal secretions, breast milk, and tears. The objective was to analyze the available evidence related to the use of biomarkers obtained by LB for the early diagnosis of BC.
Methods:
Articles related to the use of biomarkers for the early diagnosis of BC due to LB, published between 2010 and 2022, from the databases (WoS, EMBASE, PubMed, and SCOPUS) were included. The MInCir diagnostic scale was applied in the articles to determine their methodological quality (MQ). Descriptive statistics were used, as well as determination of weighted averages of each variable, to analyze the extracted data. Sensitivity, specificity, and area under the curve values for specific biomarkers (individual or in panels) are described.
Results:
In this systematic review (SR), 136 articles met the selection criteria, representing 17 709 patients with BC. However, 95.6% were case-control studies. In 96.3% of cases, LB was performed in peripheral blood samples. Most of the articles were based on microRNA (miRNA) analysis. The mean MQ score was 25/45 points. Sensitivity, specificity, and area under the curve values for specific biomarkers (individual or in panels) have been found.
Conclusions:
The determination of biomarkers through LB is a useful mechanism for the diagnosis of BC. The analysis of miRNA in peripheral blood is the most studied methodology. Our results indicate that LB has a high sensitivity and specificity for the diagnosis of BC, especially in early stages.
Keywords
Introduction
Breast cancer (BC) is the most frequently diagnosed malignancy and the leading cause of cancer-related deaths in women worldwide. In 2020, more than 2.2 million new cases were diagnosed and 684 996 deaths were reported globally. 1 This neoplasm originates in the epithelial cells that line the mammary ducts responsible for transporting milk to the nipple or in the lobules, which are the glandular structures that produce milk.
Despite recent developments for early detection of this disease, additional innovative and effective diagnostic methods in the early stages are needed to obtain the best possible outcomes during treatment. To date, progress in this area has been slow and continues to be an important challenge.2,3
Although ultrasound and mammography are the most widely used methods, both procedures depend on the radiologist’s expertise, as well as the quality and technology of the equipment used during these procedures. Furthermore, as mammography applies ionizing radiation, the ability to use in patients younger than 30 years of age is limited.3-5 During the last decade, nuclear magnetic resonance of the mammary glands has been used as a complementary method, with high sensitivity in the detection of small lesions. This approach, however, is an expensive procedure with a significant rate of false-positives. 6
Nevertheless, to confirm the diagnosis of BC, all of the above methods require a tissue biopsy as an adjunct, which is an invasive procedure. The development of non-invasive techniques and methods that allow early diagnosis of BC is highly relevant, and several methods are being studied and researched worldwide. An example of the above would be the use of serum markers such as carcinoembryonic antigen (CEA) and Ca153, which may be interesting strategies, but show low sensitivity and specificity. 7
Liquid biopsy (LB) is an approach that has also recently emerged. It identifies circulating biomarkers that can serve as a valuable and promising tool for early diagnosis of BC. This procedure, which is non-invasive, can be performed on blood and other body fluids such as urine, saliva, nipple discharge, volatile respiratory fluids, nasal secretions, breast milk, and tears. Cellular components, such as circulating tumor nucleic acids (ctDNA), circulating tumor cells (CTC), vesicle-encapsulated extracellular RNA (EV-mRNA), and circulating microRNA (miRNA) molecules, are among the major components identified. 8
The molecular classification of the disease based on the expression of estrogenic hormone receptors (ER), progesterone receptors (PR), human epidermal growth factor 2 (HER2), and Ki-67 proliferative index allows the following BC subtypes to be identified: luminal A (ER and/or PR+, Her2−, Ki-67 low), luminal B (ER and/or PR+, Her2−, Ki-67 high) or (ER and/or PR+ Her2+), Her2-enriched (ER and PR− Her2+), and triple-negative (ER− PR− Her2−), each of which is related to a specific gene expression and useful in the diagnosis of neoplasia. 9 In addition to molecular classification, the histological grade and stage of the disease are being investigated to determine their benefit in the early diagnosis of BC.
The aim of this study was to analyze the available evidence on the use of biomarkers obtained by LB in the early diagnosis of BC.
Materials and Methods
This study was written following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA 2020) statement 10 and is registered as a protocol in the PROSPERO database (ID: CRD42021255596).
Design
Systematic Review (SR).
Eligibility criteria
Articles related to LB and BC early detection in humans were included, without language restriction; the articles were published between January 2010 and June 2022. Review articles, letters to the editor, case reports, conference abstracts, and duplicate articles were excluded.
Information sources
A systematic search of related literature was conducted from the following sources: WoS, EMBASE, PubMed, SCOPUS. In addition, a manual cross-reference search was performed.
Search strategy
MeSH terms and free words were used: “circulating cell-free DNA,” or “plasma cell-free DNA,” or “serum cell-free DNA,” or “liquid biopsy” or “biomarkers,” or “circulating tumor cells,” or circulating tumor DNA,” or “detection of cancer DNA,” or serum microRNA” and “breast cancer” or “early breast cancer detection” or “screening of breast cancer.” In addition, Boolean operators “AND” and “OR” were used. The searches were adapted to each source of information and the corresponding language.
Selection process
The eligibility assessment of the primary articles was performed by 2 groups of 2 reviewers each (G.D.-C.A. and B.G.-J.P.H.), who worked independently and blinded. Disagreements between review groups were resolved by consensus. Item recruitment closed on May 30, 2022.
Data collection process
For data extraction, an Excel sheet (PC Excel, version 15.24; 2016 Microsoft Corporation) was created. Five authors extracted data from the included studies (G.D., C.A., D.P., J.P.H., and B.G.) and 2 additional authors checked the extracted data (M.M. and L.A.). Disagreements between the reviewers were resolved by consensus.
Variables studied
The variables considered were year of publication, country, number of cases, type of design, body fluid used for LB (peripheral blood, saliva, fluid aspirated from the nipple, sweat, urine, tears, and volatile compounds in the breath), type of biomarkers in the blood (CTC, ctDNA, circulating free DNA [cfDNA], circulating miRNA, circulating extracellular RNA vesicles [EV-RNA], and others), type of biomarkers in other body fluids (CTC, EV-RNA, miRNA, ctDNA, cfDNA, and others), determined biomarkers, sensitivity, specificity, and methodological quality (MQ) of the primary studies.
Study risk of bias assessment
The internal validity (MQ) of the primary studies was assessed using the MInCir-Dg scale 11 (MQ assessment scale for diagnostic studies), composed of 9 items grouped into 3 domains, with a minimum and maximum score of 9 and 45 points, and a cut-off point of 20 points, which defines the dichotomy of the MQ construct for diagnostic studies.
Effect measures
Descriptive statistics (percentages, frequencies) and determination of variable weighted means (weighting of the result of each variable by the MQ of the primary study from which it originated) were used to analyze the extracted data.
Synthesis methods
The identified documents were filtered by duplication between databases. Titles and abstracts were screened using selection criteria. Finally, an in-depth analysis of each of the selected primary articles was performed; critical reading guides were applied, thus organizing the synthesis of the information.
Assessment of reporting bias
Potentially missing studies were identified by cross-reference searches.
Certainty assessment
Not considered.
Ethics
The authors and centers of the primary studies used were masked.
Results
Study selection
In total, 16 142 articles were identified in the aforementioned databases (Table 1). However, 234 articles were retained for full reading, following the elimination of duplicates and articles whose title and abstract did not meet the eligibility criteria; of these, only 13612-147 met the inclusion criteria and are the basis of the qualitative and quantitative analysis of this SR (Figure 1).
Search strategy: databases used and primary articles found.


Flow chart of primary articles used in this SR.
Study characteristics
Of the 136 primary articles, 130 (95.6%) were case-control studies and 6 (4.4%) were cohort studies. The population represented in these articles comprised 34 376 patients, of which 17 709 are BC carriers and 16 667 correspond to controls (defined as healthy subjects with benign breast disease or other types of cancer). In all, 75.7% of the articles were published between 2016 and 2022 (Table 2).
Characterization of the primary articles on the diagnosis of BC by means of LB (n = 136).

Abbreviations: BC, breast cancer; HER2, human epidermal growth factor 2; LB, liquid biopsy.
Results of individual studies
Evidence on the early diagnosis of BC through LB came from 31 countries. China (n = 45) and Egypt (n = 18) were the countries that contributed the most articles (Figure 2).
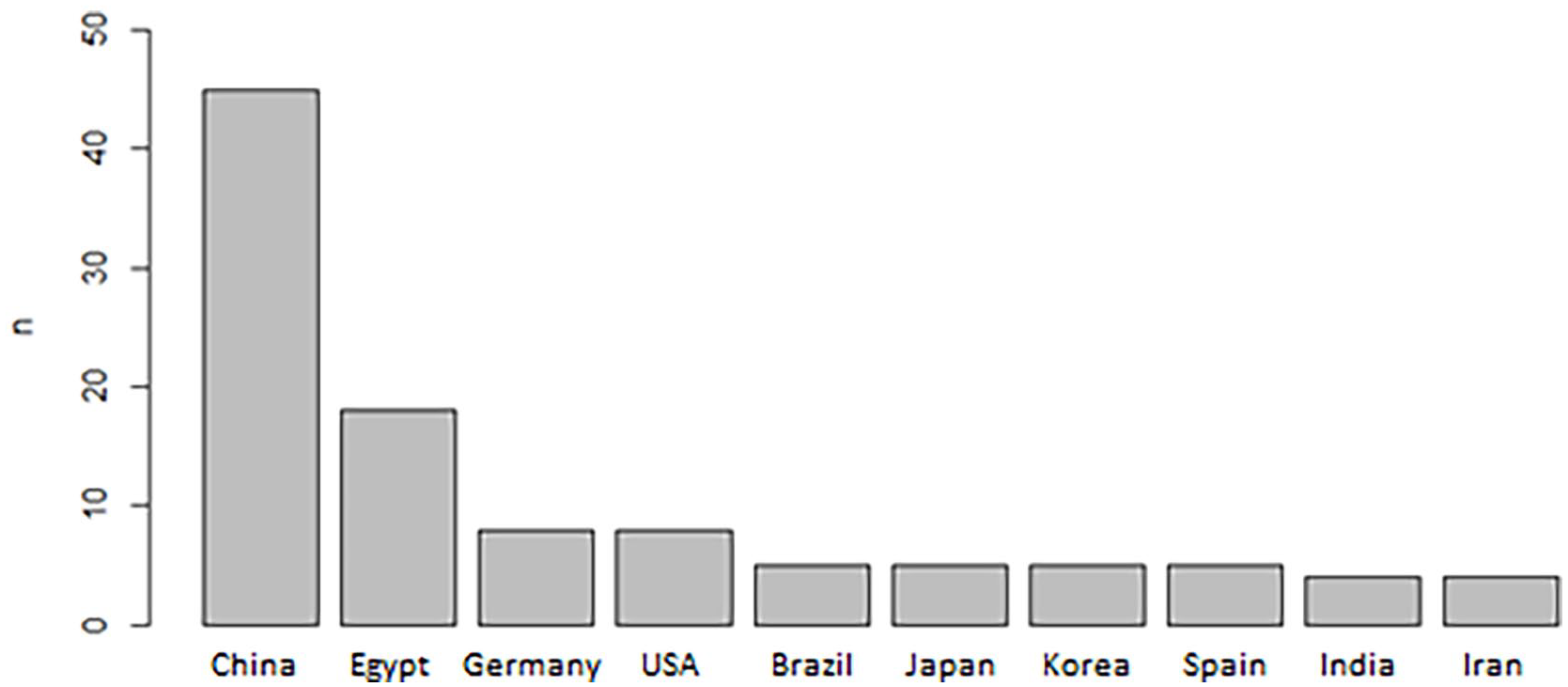
Main countries of origin of articles on BC diagnosis through LB.
The molecular classification of BC (Figure 3) was not described in 59 articles, while 73 articles included patients with various types, based on the molecular classification (luminal A, luminal B, HER2-enriched, and triple-negative). In addition, 2 articles studied only patients with triple-negative BC,101,116 and 2 included patients with HER2-enriched BC.76,95

Molecular classification of BC reported in 136 primary articles.
In reference to the disease stage, 57.4% of the articles detailed the stages that the patients were in at the time. In 43 articles, patients in all stages (I, II, III, and IV) were included, representing a population of 5382 patients with BC (stage II was the most frequent, representing 3230 patients). The details are available in Table 3.
Characterization of primary articles based on the stage of the disease..

The histological grade was identified in 40.4% of the articles, which represents 5102 patients. Patients with all grades (1-3) were included in 51 articles. In 3 articles, grades 2 and 3 were included,68,96,140 and 1 article included only grade 3 patients 120 (Table 4).
Characterization of primary articles based on the histological grade.

Diagnostic role of LB in BC
The body fluids in which LB was analyzed are detailed in Table 5 emphasizing that in 96.3% of the studies, it was performed in peripheral blood. Additional body fluids analyzed included urine69,74,86 and saliva.71,120
Characterization of LB analyzed in primary articles.
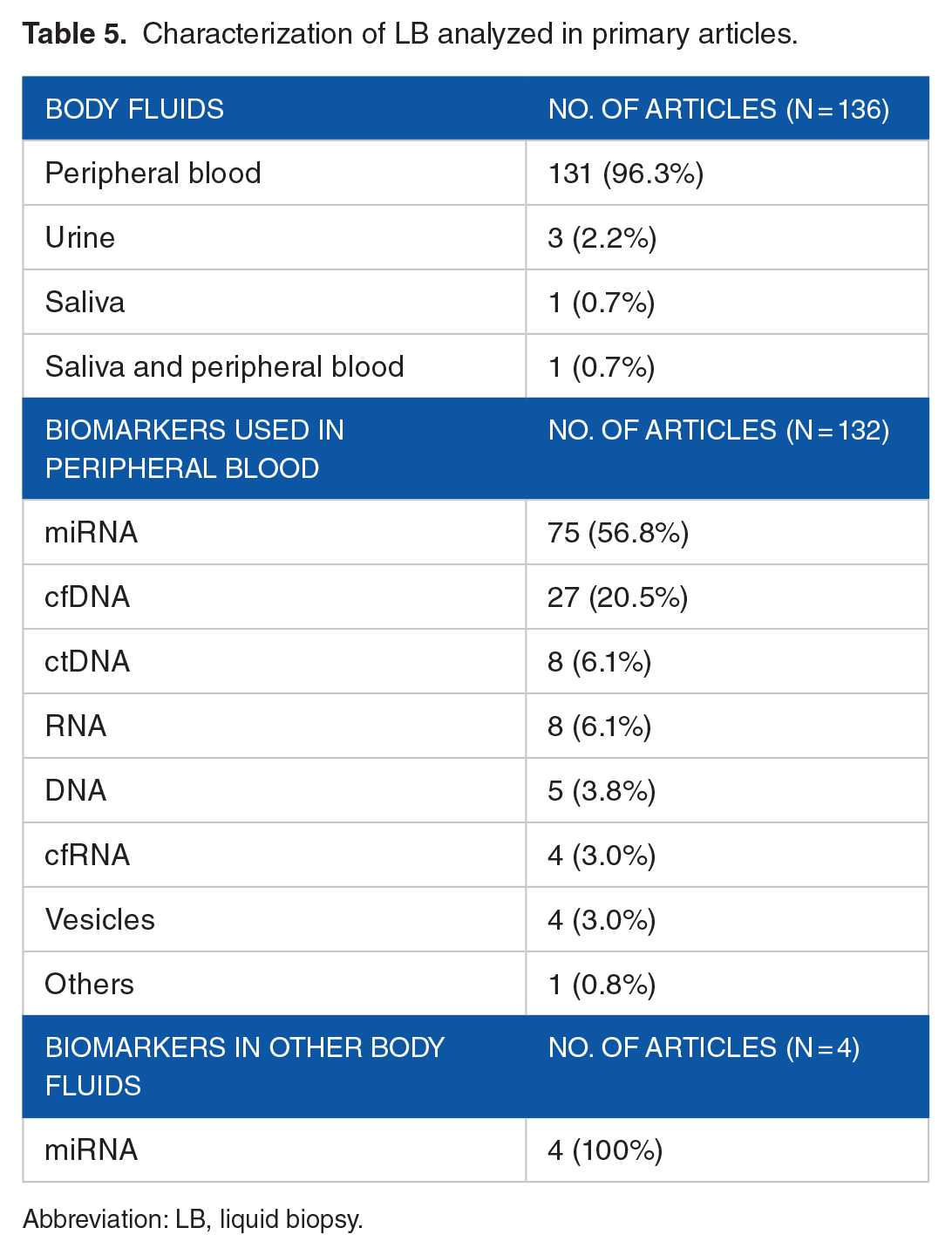
Abbreviation: LB, liquid biopsy.
Regarding the different LB methods, miRNA analysis (56.8%) predominated, followed by cfDNA (20.5%), in the studies that used blood samples. In those that analyzed saliva or urine, the diagnosis was performed by miRNA analysis.
The validity of the different tools for early diagnosis of BC described by the primary studies (markers obtained in DNA or RNA) is noted in Tables 6 to 8. It is noteworthy that the sensitivity of most is higher than 70% (Table 6).
Individually tested biomarkers for early diagnosis of BC in peripheral blood.
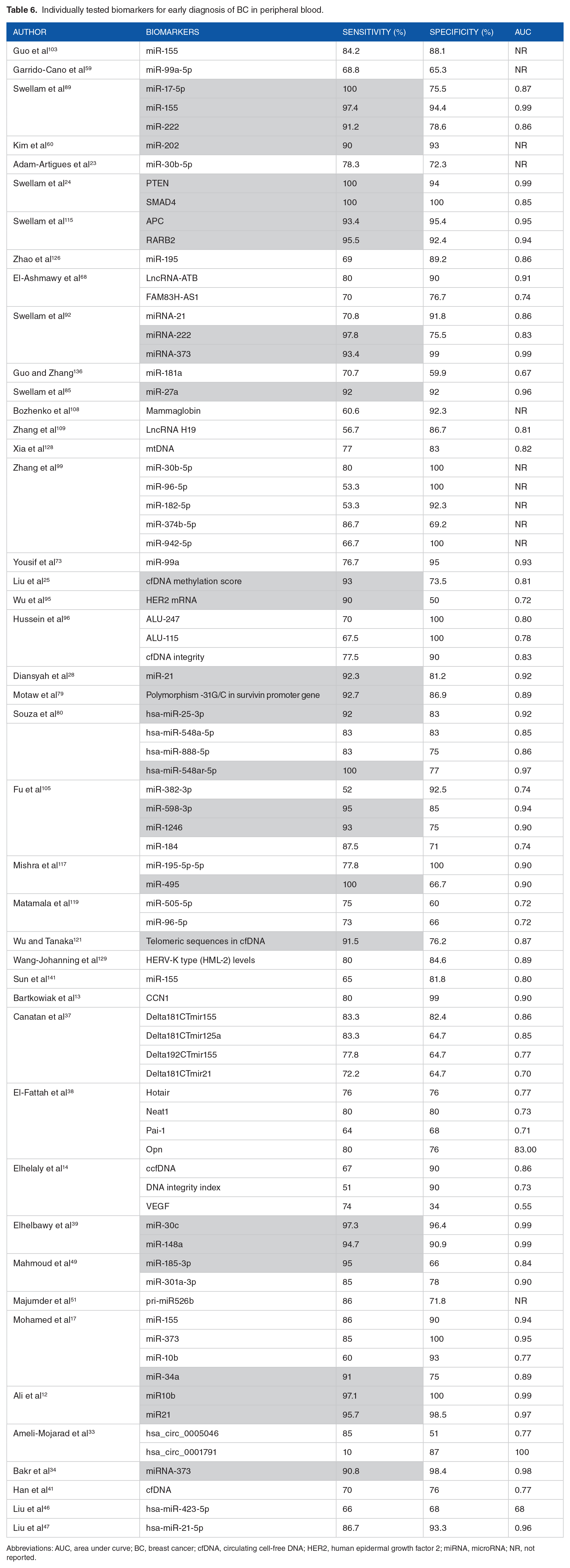
Abbreviations: AUC, area under curve; BC, breast cancer; cfDNA, circulating cell-free DNA; HER2, human epidermal growth factor 2; miRNA, microRNA; NR, not reported.
As shown in Table 6, highlighted in gray, 28 biomarkers presenting a Sensitivity greater than 90% were identified: miR-17-5p, miR-155, miR-222, 89 miR-202, 60 PTEN, SMAD4, 24 APC, RARB2, 115 miRNA-222, miRNA-373, 92 miR-27a, 85 cfDNA methylation score, 25 HER2 mRNA, 95 miR-21, 28 polymorphism -31G/C in survivin promoter gene, 79 hsa-miR25-3p, hsa-miR-548ar-5p, 80 miR-598-3p, miR-1246, 105 miR-495, 117 telomeric sequences in cfDNA, 121 miR-30c, miR-148a, 39 miR-185-3p, 49 miR-34a, 17 miR10b, miR21, 12 and miRNA-373. 34
As shown in Table 7, highlighted in gray, 16 biomarker panels in peripheral blood can be observed19,22,27,29,30,42,44,54,61,81,93,98,106,111,112,123 and 2 biomarker panels in saliva74,86 with a sensitivity greater than 90%.
Biomarker panels tested in LB for early diagnosis of BC.

Abbreviations: AUC, area under curve; BC, breast cancer; cfDNA, circulating cell-free DNA; LB, liquid biopsy; NR, not reported.
As shown in Table 8, highlighted in gray, 8 individual biomarkers and 7 panel biomarkers taken from peripheral blood had statistically significant area under the curve (AUC) values (greater than 0.80). However, 21 studies (20 in peripheral blood and 1 in urine) presented statistically significant P values (less than .05).
Analysis through AUC values or P values of different individual biomarkers and panel biomarkers in peripheral blood and urine. (Sensitivity and specificity were not reported in these primary articles.).

AUC, area under curve; cfDNA, circulating cell-free DNA; EV, extracellular vesicles; miRNA, microRNA.
Sensitivity, specificity, AUC, or P value of the test analyzed was not described in 17 of the articles studied. These articles, however, did address frequencies and associations with patients’ clinical pathological characteristics.
Methodological quality
The average MQ of the articles was 24.7 points (Table 9). Most of the articles were cases and control studies. None of the studies validate the sample size used, and from a methodological standpoint, there is a lack of homogeneity throughout all of the articles reviewed. Furthermore, not all studies describe the inclusion and exclusion criteria; and in some, the study population involves less than 100 patients. However, the study test is described in sufficient detail in all of these studies, and regardless of the results, the same reference standard was applied to all study subjects. Furthermore, in most of the articles, the objectives of the study were clear and precise.
MQ scores of the primary articles studied by year (n = 136).

Abbreviations: MQ, methodological quality; SD, standard deviation.
Discussion
There are 3 SRs related to this issue. One of them studied circulating tumor ctDNA with disease-free survival in patients with BC, 148 another described the clinical uses of LB in BC, 149 and the last one reported the validity of HER2/ERBB2 copy number variation in LB from BC patients. 150 This is the first SR aimed at establishing the main biomarkers obtained by LB, useful for the early diagnosis of BC. This evidence is highly relevant because the identification of biomarkers in the early diagnosis of BC would undoubtedly be valuable in reducing mortality rates resulting from this neoplasm.
The LB approach shows promise, given that the standard BC screening technology is limited. For instance, the sensitivity of mammography depends on age, ethnic origin, personal history, the experience of the radiologist, and the quality of the technique applied. 151 In addition, ultrasound imaging of the breast also depends on the radiologist’s expertise. 148
The primary use of serum markers CA-153, CA27-29, CA-125, and CEA is applied to monitor response to treatment. However, these markers are not recommended as screening methods in light of their low diagnostic sensitivity in early disease, and their lack of specificity. 148 Despite scientific technological advancements, LB has not yet been standardized as a routine diagnostic method in the clinical setting.
It is expected that the sequencing of the genetic material obtained through LB and the significant amount of research being conducted in this area will prompt the implementation of this diagnostic tool for diagnosis, early detection, and follow-up of BC patients.
Our study found that only 4 primary articles researched the determination of biomarkers in urine and saliva, in such a way that although the use of LB in different bodily fluids has been described in BC, peripheral blood is still the most frequently used.
Contrary to what we reported in our previous review, in which most of the primary articles applied the determination of biomarkers using ctDNA, 149 in this SR, miRNA expressions were researched in 56 studies, with the aim of identifying biomarkers that differentiated between tumor tissue, healthy tissue, benign tumor breast tissue, and BC. This could be explained because the levels of cfDNA and ctDNA are significantly low in the preclinical stages, which reduces the sensitivity for screening. 152 Thus, the Yong Tay study determined that although ctDNA had a specificity greater than 99% for detecting BC, its sensitivity was only 33%. 153
The explanation may be related to the miRNA biomarker normal signals derived from active metabolic processes occurring in all living, growing cells, increasing the pool of cellular biomarkers in earlier stages. The expressions obtained from cfDNA originate from tumor cells that detach from a tumor at an advanced stage of its development. 154
In 29 articles,23-25,28,59,60,68,73,79,80,85,89,92,95,96,99,103,105,108,109,115,117,119,121,126,128,129,136,141 sensitivity and specificity were reported individually for a single biomarker. In 21 of these, sensitivity was greater than 70%. However, in 10 of the studies in which sensitivity was less than 70%, and in one, 80% greater specificity was reported. In contrast, 26 articles reported sensitivity and specificity figures greater than 70% for combined biomarkers in the form of panels27,29,30,61,76,77,81,82,87,90,93,94,97,98,101,104,106,111-114,118,123,124,132,147 leaving only 6 panels with figures lower than 70%.61,76,93,114,124,132 Finally, 27 articles reported AUC and P values,26,62,63,67,71,72,78,83,84,88,102,110,116,120,122,125,127,130,133,135,137,139,140,142,143,145,146 and of these, AUC was lower than 0.767,133,137 only in 3 articles. In contrast, 2 articles reported AUC values above 0.926,88
A study worth noting is by Hua Zhao, in which 31 miRNA biomarkers were found in White patients, and 18 in African Americans, all with adequate sensitivity and specificity to discriminate between BC and healthy subjects. 145 Despite the above, to be considered useful, a biomarker must meet a set of analyses and clinical criteria. The benefit provided by the biomarker is underscored in the clinical setting to reduce mortality from BC and clinical validity (the ability to accurately identify a patient with BC).155,156
Consequently, even though research results are increasingly promising, the use of biomarkers for the early diagnosis of BC requires time to better understand the mechanisms related to circulating tumor material and to achieve adequate reproducibility.157,158
The studies by Ming et al 70 and Yoshinami et al 64 also evaluated gene profiles and the presence of mutations, coinciding with Jimenez et al 75 and Duque et al, 149 in which the most frequently found mutations affected these loci: PIK3CA, TP53, and AKT1.
MiRNA-34a expression was low and miRNA-155 expression was elevated in BC vs controls with a significant P value. In addition, a correlation was demonstrated between the expression of miRNA-155 or miRNA-34a and TNM, presence of nodes, and histological grade. 107 Similarly, the Nadeem study agrees with this result by showing that low miRNA-195 expression was correlated with clinical stage, nodes, and histological grade. 100
However, the studies of Delmonico et al 120 and Ritter et al, 69 which analyzed methylation promoters in DNA in saliva and blood, as well as miRNA in urine and blood, did not find significant associations.
As is noted in Figure 2, 45 studies from China (33%), 18 from Egypt, 8 from the United States, and 8 from Germany representing more than half of the primary articles were found. The figures clearly indicate considerable interest in ongoing research by these countries, regarding this area.
Finally, and in reference to the MQ analysis of the primary studies (applying the MInCir-Dg scale), 11 it is important to emphasize that the median score was 25 points (17-34 points) and the average was 24.7 points, which represents a regular MQ. It should be highlighted that the lowest scores were associated with the type of design (most of the studies correspond to cases and controls) and not having estimated the sample size, which determines that the level of evidence of the primary articles is 2b and 3b for diagnostic studies, with a grade B recommendation. 159
Regarding the limitations of this study, the heterogeneity of the primary studies should be highlighted, as various methods are used, both for the identification of different biomarkers through LB (CTCs, ctDNA, cfDNA, miRNA, and EV-RNA), as well as the fact that some studies evaluate biomarkers individually, while others do so through combinations, establishing biomarker panels under evaluation. Another important limitation of the study was that none of the primary studies performed a sample size calculation, and in the articles, the number of participants varied and was inconsistent. In addition, some primary studies established sensitivity and specificity, while others only reported AUC values, and some only reported P values. These variables are made for a difficult analysis and comparison. Despite these limitations, the strengths of the primary studies are that a significant number maintained a methodological strategy to perform the analyses in test cohorts, and then in validation cohorts, maintaining groups of cases and control groups in each of the studies.
Conclusions
Integrating LB in clinical practice as part of the process for early diagnosis of BC is a promising alternative. The biomarkers, obtained from samples obtained through LB, consisting of miRNA molecules, were the most frequently investigated biomarkers in the early diagnosis of BC. MiR-21, miR-155, and miR-195 have the greatest potential to discriminate between healthy individuals, BC, and benign breast tumors. There are panels of combined biomarkers, with the potential to increase diagnostic sensitivity. Our results reflect that LB has a high sensitivity and specificity for the diagnosis of BC, especially in early stages.
Footnotes
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
GD and CM contributed to the concept and design of the research. TO reviewed and approved the study design. GD, CA, JPH, and BG performed the selection process for article recruitment. GD, CA, DP, JPH, and BG extracted the data from the studies included, and MM and LA verified the extracted data. GD, CM, CA, and BG collaborated in the analysis of the results and presentation of data. GD and BG contributed to final revisions. Data sharing is not applicable to this article as no new data were created or analyzed in this study. All authors contributed in the drafting of the article.
