Abstract
Background:
Non-small cell lung cancer (NSCLC) remains a leading cause of cancer death worldwide, for which better knowledge in molecular prognostic factors is needed to improve clinical outcome. This study aimed to investigate the clinical significance of c-Myc, ALK, ROS1, BRAF, and PD-L1 in NSCLC patients.
Methods:
Formalin-fixed paraffin-embedded tissue specimens were obtained from 124 NSCLC patients. Of these, 66 matched specimens of normal respiratory epithelial and tumor tissue from patients with stages I-III, who underwent surgical resection, and 58 NSCLC specimens from stage IV patients were recruited into this analysis. Immunohistochemistry staining along with semiquantitative criteria were used to evaluate the expression of the interested proteins.
Results:
Of the 66 patients with stages I-III, positive expression of c-Myc was detected in 12 specimens (18.2%) of NSCLC tissue, whereas none of the normal respiratory epithelial tissue was found to have c-Myc expression (P < .001). Of the 66 NSCLC patients, 28 (43.8%) had PD-L1-positive staining on 1%-49% tumor cells and 7 (10.9%) patients expressed PD-L1 in ⩾50% tumor cells. One (2.3%) adenocarcinoma patient was found to have ROS1 rearrangement. Patients with no expression of c-Myc and PD-L1 (co-negative expression) tended to have a better prognosis than other subgroups.
Conclusions:
NSCLC tissue significantly expressed more c-Myc and PD-L1, compared with the matched normal respiratory epithelium, emphasizing the important role of these key drivers in tumorigenesis. Therapeutic approach to precisely inhibit the targetable molecular pathways should be considered on an individual patient basis to improve survival outcome.
Introduction
Non-small cell lung cancer (NSCLC) is one of the most commonly diagnosed cancers worldwide and is the leading cause of cancer-related death. Globally, an estimation of more than 1.8 million new lung cancer cases, accounting for approximately 11.6% of total cancer diagnoses and 18.4% of the total cancer-related deaths, have been made yearly. 1 Survival of NSCLC patients remains unfavorable; therefore, a better understanding of its molecular biology and causality, along with advances in diagnostic methods and multidisciplinary therapeutic approaches, is needed to improve patient survival.
As a transcription and replication factor, c-Myc activates and represses a large number of target genes that promote and inhibit cell growth, respectively. c-Myc deregulation also plays an important role in various cancer types, including NSCLC.2,3 Advances in NSCLC biology research have revealed cellular pathways, and molecules related to cancer development as well as proliferation, making it possible to use targeted therapeutic strategies for patients. The receptor tyrosine kinase inhibitors (TKIs) that have been approved to treat NSCLC patients include those targeting epidermal growth factor receptors (EGFRs), echinoderm microtubule-associated protein-like 4 (EML4)–ALK rearrangement, c-ros oncogene 1 (ROS1), and BRAF.4 -7 Immunotherapy has also been approved as a standard therapeutic option for advanced NSCLC. Immune checkpoint inhibitors, targeting programmed cell death-1 (PD-1), and its ligand (PD-L1) demonstrated the overall survival benefit in this devastating disease. The high level of PD-L1 expression is a predictive biomarker for selecting a patient for first-line therapy, with immune checkpoint inhibitors. 8 This study examined the clinical significance of c-Myc, ALK, ROS1, BRAF, and PD-L1 in NSCLC patients to better understand NSCLC tumorigenesis and explore novel therapeutic strategies to improve patient outcome.
Materials and Methods
Patient data
NSCLC patients who were treated between March 2005 and November 2017 at the Prince of Songkla University Hospital were reviewed and eligible to be enrolled if their archived tissue specimens were available to be examined. Clinical information were obtained and analyzed in relation to the laboratory results. The data comprised the patient characteristics, including patients’ demographic data, smoking status, the NSCLC stage, histology, and survival outcome.
Patient specimens
Formalin-fixed paraffin-embedded (FFPE) tissue specimens were obtained from 124 NSCLC patients. Of these, 66 matched specimens of normal respiratory epithelial and tumor tissue from patients with stages I-III, who underwent surgical resection, and 58 NSCLC specimens from stage IV patients were recruited into this analysis.
Immunohistochemistry
Expression of c-Myc, ALK, ROS1, BRAF, and PD-L1 was determined by immunohistochemistry (IHC) assays. In brief, FFPE tissue of 3-4 μm in thickness and 3 mm in diameter from NSCLC patients was processed for tissue microarrays. To account for the interested protein expressions, IHC assays were performed using the corresponding monoclonal antibodies in conditions specified by the manufacturers (c-Myc [EP121], Cell Marque, Sigma-Aldrich, Burlington, MA, USA; ALK (D5F3), Ventana, Roche Diagnostics, Rotkreuz, Switzerland; ROS1 (D4D6), Cell Signaling Technology, Danvers, Massachusetts, USA; BRAF (VE1), Ventana, Roche Diagnostics, Rotkreuz, Switzerland; PDL-1 (22C3), pharmDx, Dako, Hovedstaden, Denmark). Briefly, the tissue sections were deparaffinized in xylene, rehydrated in graded alcohol, and washed in a wash-buffer solution. Both positive controls and negative reagent controls were included in each staining run as a reference. The epitope retrieval process using Target Retrieval Solution (1:10; Dako) at 105°C for 20 minutes before loading the slides onto the autostainer was applied. The slides were then loaded onto an autostainer, and the following incubations were applied sequentially: 10 minutes peroxide blocking agent (3%), 10 minutes Power Block, 10 minutes proteinase K, primary antibodies to the interested proteins or negative control reagent, and the secondary antibodies. Buffer rinses were performed following each step. The slides were then counterstained with hematoxylin, before being rinsed, gently, in reagent quality water, dehydrated in graded alcohol, cleared in xylene, and cover-slipped.
Analyses of IHC-stained slides were performed using a 10× magnification objective in light microscopy. A semiquantitative criterion was used for the interested proteins interpretation. In detail, the percentage of positive tumor cells per slide (0%-100%) was multiplied by the dominant intensity pattern of staining (0 = negative or trace; 1+ = weak; 2+ = moderate; 3+ = intense); therefore, the overall score ranged from 0 to 300. Specimens with IHC 2/3+ ⩾75%, of the interested cells or the overall score ⩾150 were considered positive for c-Myc, BRAF expression, or ALK and ROS1 rearrangements. Specimens with 1/2/3+ for PD-L1 were categorized as either a low (1%-49% positive) or a high (⩾50% positive) level of PD-L1 expression.
Statistical analysis
Chi-square or Fisher exact test was used to analyze the association between clinicopathologic characteristics and the expression of proteins. Kaplan-Meier plots were carried out to estimate median survival times in each subgroup and the log rank test was used to compare their differences. Cox proportional hazard model was used to evaluate prognostic significance of protein markers in NSCLC patients.
Results
Clinicopathologic characteristics of NSCLC patients
One hundred twenty-four NSCLC patients, 66 in stages I-III and 58 in stage IV, were enrolled. The clinicopathologic characteristics of the patients are shown in Table 1. Of the 124 recruited patients, 68 were men and 56 were women, with the mean age being 61.3 ± 12 years. There were 100 (80.6%) patients with adenocarcinoma and 24 (19.4%) patients with squamous cell carcinoma. All patients with stages I-III diseases underwent radical surgery as an initial treatment, from which the paired tissue from their normal respiratory epithelium specimens was available to be co-investigated. Patients with stage IV were more likely to have a history of smoking than patients with stages I-III (53.4% vs 47%, P = .055). In addition, stage IV patients significantly had a higher proportion of adenocarcinoma histology than patients with stages I-III (P = .002, Table 1).
Clinicopathologic characteristics of patients with non-small cell lung cancer by stages (N = 124).

Chi-square or Fisher exact test.
Expression of ALK, ROS1, and BRAF in NSCLC tissue
Protein expression of the interested molecules was categorized as positive or negative, based on IHC semiquantitative scores. Figure 1 represents ALK, ROS1, and BRAF expression in squamous and adenocarcinoma NSCLC patients. One (2.3%) adenocarcinoma patient was found to have ROS1 rearrangement. None of the NSCLC patients from this study appeared to have ALK kinase-fusion or BRAF mutation (Table 2).
Biomarkers of patients with non-small cell lung cancer (N = 124).
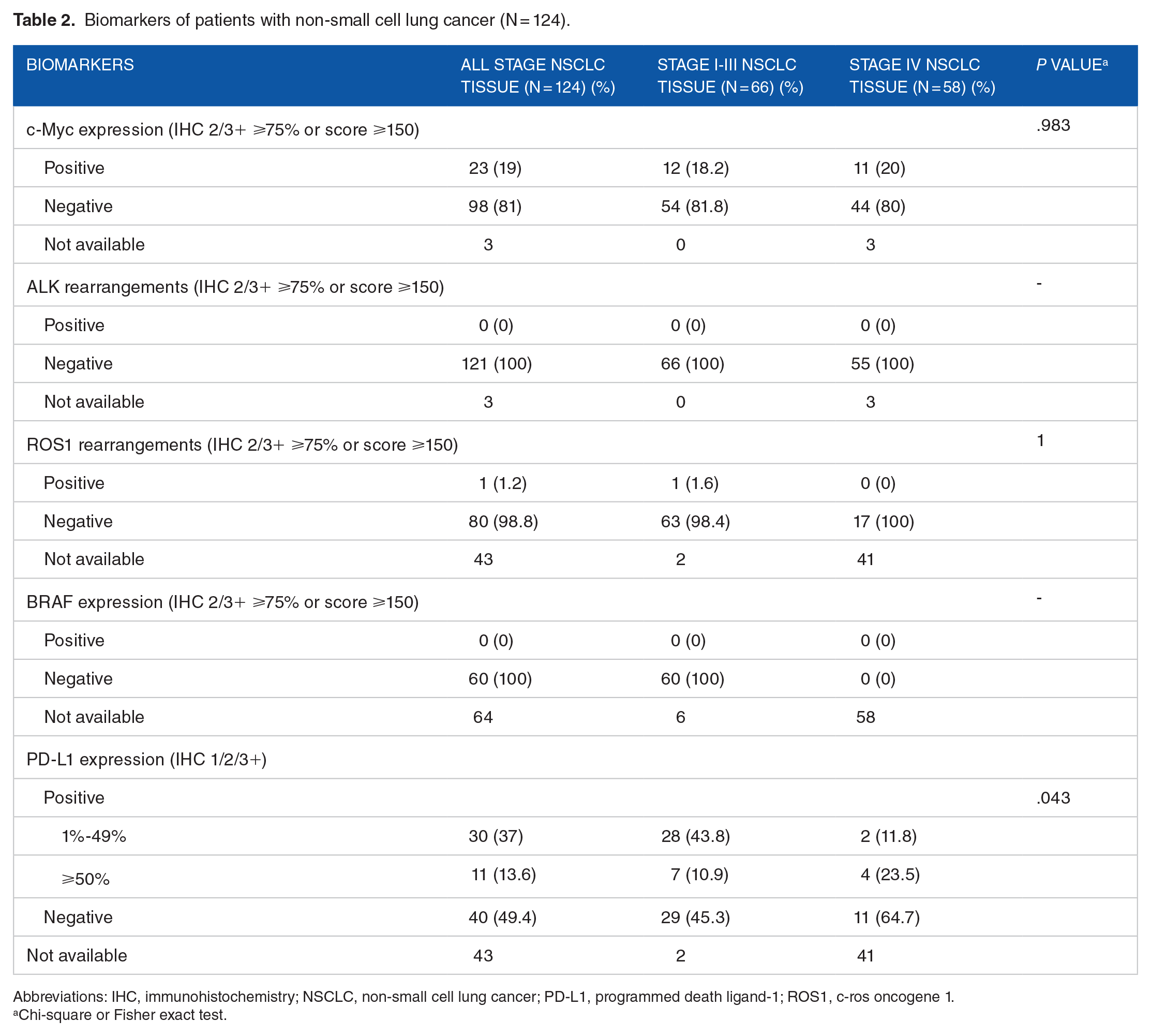
Abbreviations: IHC, immunohistochemistry; NSCLC, non-small cell lung cancer; PD-L1, programmed death ligand-1; ROS1, c-ros oncogene 1.
Chi-square or Fisher exact test.
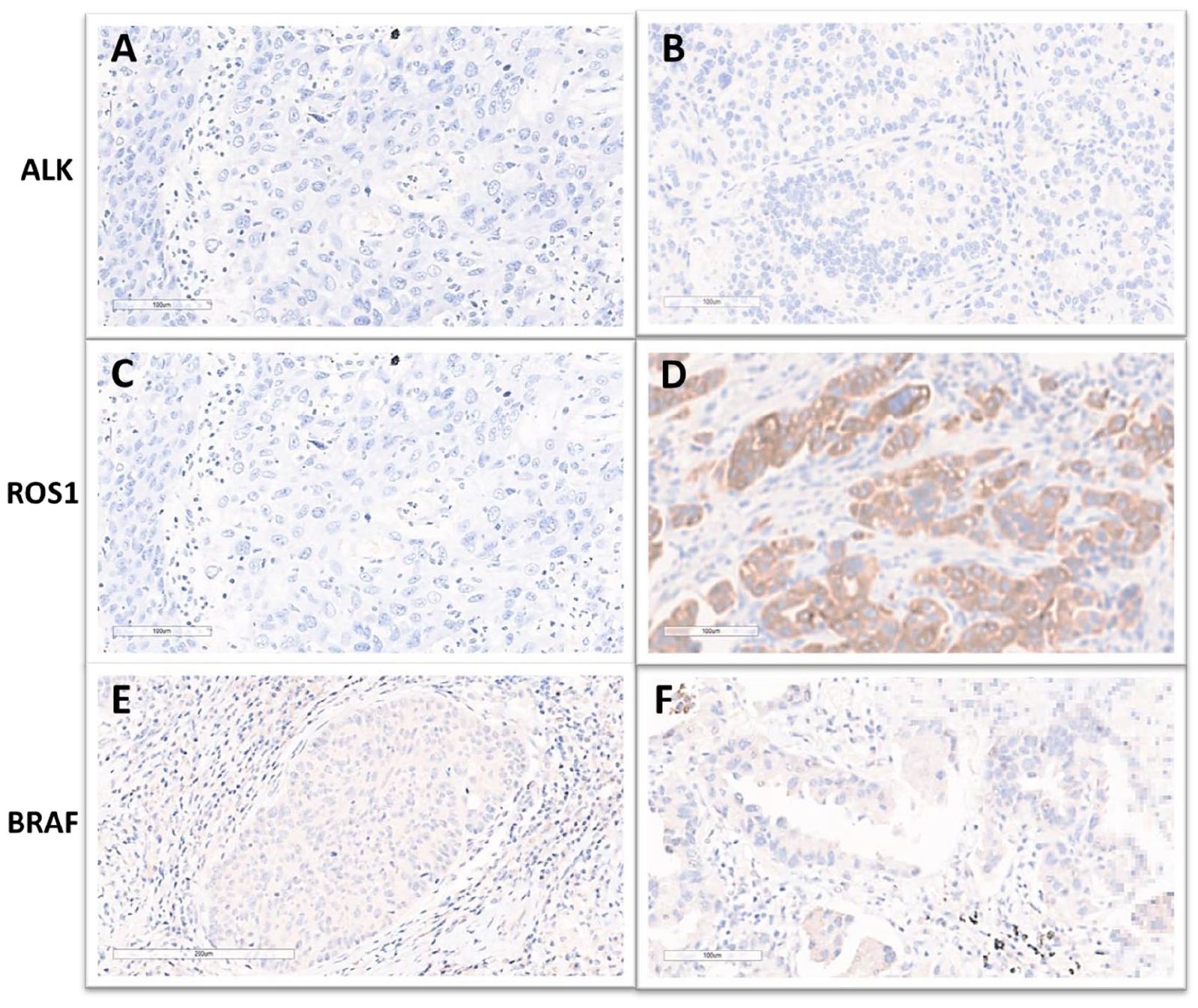
ALK, ROS1, and BRAF expression in squamous and adenocarcinoma NSCLC patients. Representative images (10×) on squamous carcinomas (A, C, and E) and adenocarcinomas (B, D, and F) showed negative staining for ALK, ROS1 (squamous carcinoma, C), and BRAF, respectively. Positive staining for ROS1 (adenocarcinoma) is shown in D. ROS1 indicates c-ros oncogene 1.
Expression of c-Myc in NSCLC tissue
From 66 NSCLC patients with stages I-III, positive expression of c-Myc was detected in 12 (18.2%) patients, which was insignificantly different from that of 11 from 55 (20%) available tumor tissues from patients with stage IV (P = .983) (Table 2). As shown in Figure 2, expression of c-Myc was observed as nuclear and/or cytoplasmic staining, yet mainly in the nucleus of tumor cells. On univariate analysis, the expression of c-Myc was significantly more frequent in smokers. In patients with a smoking history, 17 of 23 (73.9%) patients were positive for c-Myc. This was significantly higher than those who never smoked or had an unknown smoking history (6 in 23, 26%; P = .026). In addition, patients with squamous carcinoma tended to have more c-Myc expression than adenocarcinoma histology (P = .077). The expression of c-Myc tended to be less frequent in female patients (P = .066) (Table 3).
Univariate analyses: Association between c-Myc and clinicopathologic characteristics (n = 121).

Chi-square or Fisher exact test.

c-Myc expression in squamous and adenocarcinoma NSCLC patients. Representative images (10×) on squamous carcinomas (A & B) and adenocarcinomas (C & D) showed c-Myc-negative staining (A & C) and positive staining (B & D).
Association of clinicopathologic characteristics and c-Myc with overall survival
According to disease stages of NSCLC, median overall survival duration in 43 patients with stage I was not reached (95% confidence interval [CI]: 58.1; -), whereas in 20 patients with stage II, this was 33.4 months (95% CI: 16.2-76.2). There were only 3 patients with stage III whose median overall survival was excluded from this analysis regarding the outlier characteristic. Fifty-eight patients with stage IV were demonstrated to have a median overall survival duration of 14.8 months (95% CI: 22.3-38.5) (Figure 3A).

Overall survival duration in NSCLC patients. (A) When stratified by stages of disease, median overall survival in stage I (43 patients) was not reached (95% CI: 58.1; -), 33.4 months (95% CI: 16.2-76.2) in stage II (20 patients), and 14.8 months (95% CI: 22.3-38.5) in stage IV (58 patients). Three patients with stage III were excluded from this analysis regarding their outlier characteristics. (B) When stratified by c-Myc expression, median overall survival in c-Myc negative (98 patients) was 32.5 months (95% CI: 23.0-42.2) and in c-Myc positive (23 patients) was 19.5 months (95% CI: 8.2; not reached). (C) When stratified by PD-L1 level of expression, median overall survival in PD-L1 negative (40 patients) was 59 months (95% CI: 21; -), 54.2 months (95% CI: 33.4; -) in PD-L1 low-positive (1%-49%, 30 patients) and 31.5 months (95% CI: 8.8; -) in PD-L1 high-positive (⩾50%, 11 patients). (D) When stratified by c-Myc and PD-L1 expression, median overall survival in c-Myc and PD-L1 co-negative (33 patients) was 59.0 months (95% CI: 23.0; -), in negative c-Myc and positive PD-L1 subgroup (31 patients) was 42.2 months (95% CI: 33.4; -), in c-Myc and PD-L1 co-positive expression (10 patients) was 31.5 months (95% CI: 5.4; -), and in positive c-Myc and negative PD-L1 subgroup (6 patients) was 15.7 months (95% CI: 2.8; -). PD-L1 indicates programmed death ligand-1.
Multivariate analyses of c-Myc and clinicopathologic characteristics for overall survival revealed that age and disease stage were significant, independent prognostic parameters (Table 4). Patients with disease stage IV were significantly associated with a poorer overall survival outcome than those in stage I, with a hazard ratio of 12.3 (95% CI: 5.97-25.36). Ninety-eight NSCLC patients with no expression of c-Myc tended to have a longer median overall survival duration (32.5 months, 95% CI: 23.0-42.2) than that of 23 patients with c-Myc expression (19.5 months, 95% CI: 8.2; -) (P = .388, Figure 3B).
Multivariate Cox regression analyses of c-Myc and clinicopathologic characteristics for overall survival in stage I-IV non-small cell lung cancer patients (N = 124).

Abbreviations: CI, confidence interval; HR, hazard ratio; LR, likelihood ratio.
Expression of PD-L1 in NSCLC tissue
Regarding PD-L1, 64 patients with stages I-III had available tissue for PD-L1 IHC testing. Of 64 (43.8%) NSCLC patients, 28 had 1%-49% of tumor cells positive for PD-L1 and 7 (10.9%) patients expressed PD-L1 in ⩾50% tumor cells. Seventeen patients with stage IV disease had tumor specimens available for PD-L1 staining and this revealed a lower number of PD-L1 positivity than those in stages I-III, significantly: 35.3% versus 54.7% (P = .043) (Table 2). As shown in Figure 4, the positive expression of PD-L1 mainly occurred in the cell membrane, although it occasionally appeared in the cytoplasm of tumor cells.

PD-L1 expression in squamous and adenocarcinoma NSCLC patients. Representative images (10×) on squamous carcinomas (A, C, and E) and adenocarcinomas (B, D, and F) showed negative, 1%-49%, and ⩾50% positive staining for PD-L1, respectively.
Association of PD-L1 with overall survival
According to the univariate analyses, PD-L1 was significantly more expressed in male patients and squamous carcinoma subtype (P = .006, Table 5). On multivariate analyses of PD-L1, and clinicopathologic characteristics for overall survival in stages I-IV NSCLC patients, disease stage was a significant, independent prognostic parameter (Table 6). Patients with disease stage IV were significantly associated with a poorer overall survival outcome when compared with those in stage I, with a hazard ratio of 17.69 (95% CI: 6.61-47.37).
Univariate analyses: Association between PD-L1 and clinicopathologic characteristics (n = 81).

Chi-square or Fisher exact test.
Multivariate Cox regression analyses of PD-L1 and clinicopathologic characteristics for overall survival in stage I-IV non-small cell lung cancer patients (n = 81).
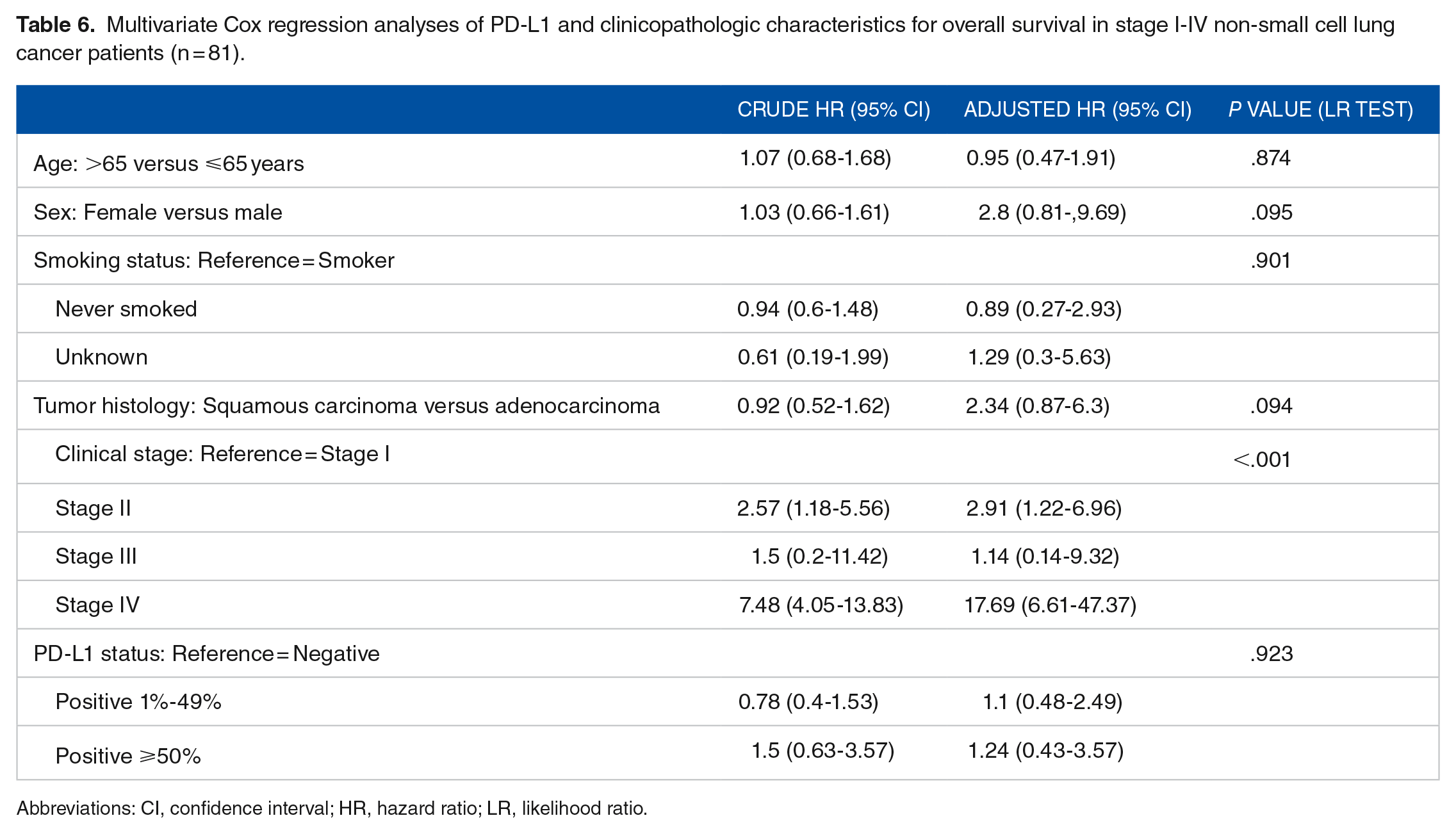
Abbreviations: CI, confidence interval; HR, hazard ratio; LR, likelihood ratio.
When stratified upon the level of PD-L1 expression, NSCLC patients with lower degrees of PD-L1 expression (1%-49%) tended to live longer than those with a higher degree of this protein expression (⩾50%). Median overall survival duration (40 patients) was 59 months (95% CI: 21; -) in PD-L1 negative, 54.2 months (95% CI: 33.4; -) in PD-L1 low-positive (1%-49%, 30 patients), and 31.5 months (95% CI: 8.8; -) in PD-L1 high-positive (⩾ 50%, 11 patients) (Figure 3C).
None of the NSCLC patients in this study were treated with either anti-PD1 or anti-PD-L1 immunotherapy. The major reasons were the unavailability of the immune checkpoint inhibitors at that time, the reimbursement criterion, and an unaffordability of the patients to the self-paid immune checkpoint inhibitors.
Co-expression of c-Myc and PD-L1 in NSCLC tissue
We tested for both c-Myc and PD-L1 co-expression in NSCLC patients. Multivariate analyses of c-Myc, and PD-L1 and clinicopathologic characteristics for overall survival in NSCLC patients revealed that age and disease stage were significant, independent prognostic parameters (Table 7). Patients with disease stage IV were significantly associated with a poorer overall survival outcome than those in stage I, with a hazard ratio of 15.43 (95% CI: 6.42-37.07).
Multivariate analyses of c-Myc and PD-L1, and clinicopathologic characteristics for overall survival in stage I-IV non-small cell lung cancer patients (N = 124).
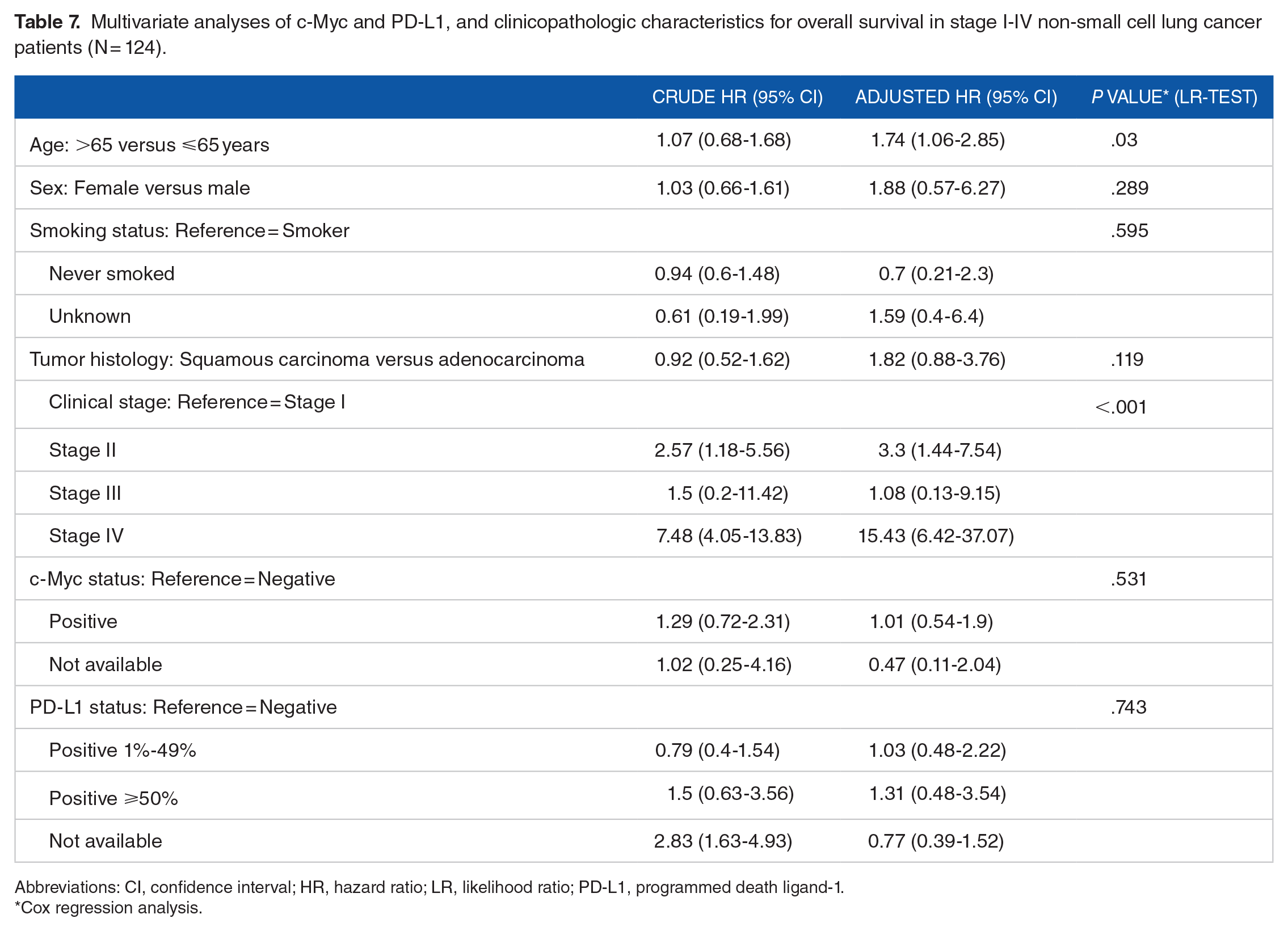
Abbreviations: CI, confidence interval; HR, hazard ratio; LR, likelihood ratio; PD-L1, programmed death ligand-1.
Cox regression analysis.
The survival of NSCLC patients, whose tumors were eligible to be tested for both c-Myc and PD-L1 expression, was demonstrated in Figure 3D. Patients with no expression of c-Myc and PD-L1 (co-negative expression) tended to have a better prognosis than other subgroups. Among 33 patients with c-Myc and PD-L1 co-negative expression, the median overall survival was 59.0 months (95% CI: 23.0; -), longer than that of 42.2 months (95% CI: 33.4; -) in the negative c-Myc and positive PD-L1 subgroup (31 patients). Ten patients with co-positive expression of c-Myc and PD-L1 had a median overall survival of 31.5 months (95% CI: 5.4; -) compared with that of 15.7 months (95% CI: 2.8; -) in 6 patients with positive c-Myc and the negative PD-L1 subgroup.
Biomarkers between paired-normal respiratory epithelium and NSCLC tissue
In NSCLC patients with stages I-III, 66 matched specimens of normal respiratory epithelial and tumor tissue were recruited into this analysis. All patients underwent surgical resection. As shown in Figure 2, positive staining of c-Myc was observed mainly in the nucleus of tumor cells. Positive expression of c-Myc was detected in 12 (18.2%) specimens of NSCLC tissue, whereas none of the normal respiratory epithelial tissue was found to have c-Myc expression (P < .001, Table 8). In patients with adenocarcinoma histology, 4 of 46 (8.7%) patients were positive for c-Myc, significantly less than those with squamous carcinoma (8 in 20, 40%; P = .005).
Biomarkers between normal respiratory epithelium and non-small cell lung cancer tissue (n = 66).

Abbreviations: IHC, immunohistochemistry; NSCLC, non-small cell lung cancer.
McNemar test.
Chi-square or Fisher exact test.
Of 64 (43.8%) NSCLC patients, whose tumor tissue were available for PD-L1 testing, 28 had 1%-49% of tumor cells positive for PD-L1 and 7 patients (10.9%) expressed PD-L1 in ⩾50% tumor cells (Table 8). The PD-L1 expression was also detected in 36 of 62 (58.1%) matched, normal respiratory epithelial tissue, but with less degree of positivity than in tumor specimens (P = .456). Squamous carcinoma showed a higher number of PD-L1 expression than adenocarcinoma histology; 80% versus 43.1%, respectively (P = .006).
Discussion
The NSCLC remains a leading cause of cancer-related death worldwide, urging a better understanding in molecular tumorigenesis and multidisciplinary therapeutic approaches to improve survival outcome. To explore the incidence of potentially targetable NSCLC patients, with the available potent anti-cancer therapy, we conducted this study including patients in stages I-III who underwent radical resection and patients in stage IV. In the cohort of stages I-III patients, IHC was performed and the expressing protein profiles of the tumor as well as the matched normal respiratory epithelium samples were analyzed to provide better understanding and knowledge of NSCLC tumorigenesis as well as to develop effective therapeutic strategies further on. In addition to investigating the preexisting molecular targets for therapy in NSCLC patients, this study also examined the genetic aberrations potentially targetable as novel therapeutic strategies for better treatment outcome.
The c-Myc, as an onco-protein, is responsible mainly in the transcriptional process of several genes and regulates the development and progression of various tumors, including NSCLC. 9 c-Myc expression has been reported to be an independent, unfavorable prognostic factor in adenocarcinoma NSCLC patients. 10 Gain in the copy number of Myc was found to be associated with lung adenocarcinoma in nonsmokers. 11 Increased activation of Myc has been observed in lung squamous cell carcinomas, compared with the premalignant lesions within the same patients. 12 This finding is concordant to our study, in which squamous carcinoma tended to have more c-Myc expression than adenocarcinoma histology.
In this study, expression of c-Myc was significantly more demonstrable in tumorous tissue than normal respiratory tissue, confirming the major role of c-Myc in NSCLC development. Of all disease stages, the expression of c-Myc was significantly more frequent in smokers and tended to be less frequent in female patients and adenocarcinoma subtype. In addition, NSCLC patients, who overexpressed c-Myc, had a tendency to have poorer overall survival than those who did not, highlighting the significance of this molecule during stepwise lung carcinogenesis.
As other cancer types, NSCLC is characterized by both clinical and molecular heterogeneity. Precision therapeutic approaches, based on molecular genetic characterization, to improve response and prolong survival have been implemented into NSCLC treatments. Molecular indicators of good prognosis include EGFR mutation and positive kinase rearrangement status, mainly EML4-ALK and ROS1, especially in NSCLC patients receiving EGFR, ALK, and ROS1-targeted therapy, respectively, as compared with conventional, standard chemotherapy.4 -7,13 Minority of NSCLC patients, who have BRAF mutation, might be beneficial to small molecule inhibitors targeting the RAF proliferative pathway.14,15 In addition, immune checkpoint inhibitors, against either PD-1 or PD-L1, have been demonstrated to significantly prolong duration of disease progression and overall survival in advanced stages of NSCLC.16 -21 In this study, alterations of expression of ROS1, and PD-L1 were more significantly prevalent in tumorous tissue than normal respiratory tissue, confirming the major role of both proteins in NSCLC tumorigenesis.
Translocation of ALK and its most common fusion partner EML4 has been validated as a favorable predictor to ALK-TKIs efficacy in NSCLC.22 -24 ALK rearrangements occurs in approximately 4%-17% of NSCLC patients, depending mainly on nonsmoking status and adenocarcinoma histology.4,25,26 In randomized phase III trials, ALK-targeted TKIs, namely, crizotinib, ceritinib, and alectinib, have been shown to provide superiority in response rates, survival, and quality of life, to platinum-doublet chemotherapy in NSCLC patients harboring ALK rearrangement. This has established them as a standard, first-line therapy.4,5,22,23,27 -29
Two ALK laboratory methodologies, fluorescence in situ hybridization (FISH) and IHC, have interchangeably been approved to be diagnostic tools for EML4-ALK fusions, and for selection of NSCLC patients eligible for ALK-targeting therapy. The IHC has been considered a standard, alternative assay to FISH in evaluating ALK translocations; therefore, IHC can be used as a screening test for this genetic aberration in NSCLC patients.5,30,31 In this study, we used the IHC assay to detect EML4-ALK fusion protein. However, none of the NSCLC patients in our cohort was found to have EML4-ALK translocation. The IHC, using D5F3 antibodies, is qualified as a screening tool and revealed a comparable sensitivity to FISH to detect ALK-rearranged NSCLC, with a positive rate ranging from 82.4% to 94.5%. Moreover, in ALK-rearranged NSCLC patients who received crizotinib, an ALK-targeting TKI, the overall response rate, disease control rate, and progression-free survival were not statistically different between the FISH- and the IHC-detectable groups. 32
Pilling et al 33 reported the Myc signaling network to be critical for crizotinib, an ALK-targeting TKI, sensitivity and that Myc activity was regulated in an ALK-dependent manner in ALK rearranged NSCLC cell lines. However, ALK rearranged NSCLC appears to be a minority subset of NSCLC and none of the patients in this study was found to be positive for ALK, limiting any further exploratory analysis on this relationship between both biomarkers from this cohort.
The other targetable genetic aberration in NSCLC therapy is the ROS1 translocation, especially with CD74. The ROS1 is a receptor tyrosine kinase of the insulin receptor family. Fusion of ROS1 in NSCLC is approximately identifiable in 1% to 2%, especially in nonsmoking patients with adenocarcinomas.34 -37 Similar to ALK fusion, the ROS1 translocations can be identified by a FISH break-apart assay and the IHC of ROS1 has been available to screen for this genetic aberration. 38 Impressively shown in the previous reports, advanced NSCLC patients whose tumors contained the ROS1 translocations responded up to 72% to crizotinib, a multitargeted TKI, which resulted in a better treatment outcome in NSCLC harboring this genetic aberration.39,40 In this study, 1 patient with adenocarcinoma NSCLC was detected to harbor ROS1 fusion protein by IHC.
BRAF mutations were present in 0.3%-4.9% of NSCLC patients, with a majority in the V600E mutational subgroup. BRAF V600E mutations are more prevalent in the female gender and are associated with poorer survival outcome. 41 The minority of NSCLC patients who harbor the BRAF mutation might be beneficial to small molecule inhibitors targeting the RAF proliferative pathway.14,15 However, no patients in our study were demonstrated to have the BRAF V600E mutation, as detected by IHC assay.
Tumor cells exploit the activated PD-1/PD-Ls signaling pathway to evade the physiological immune checkpoint control. 42 Currently, the emerging role of immunotherapeutic agents, especially the checkpoint inhibitors against PD-1 and PD-L1, has been established, providing another therapeutic option for advanced NSCLC patients. 7 IHC for PD-L1 expression, on NSCLC cells and tumor-infiltrating immune cells, has been extensively assessed and reported to correlate with benefit from respective PD-1 or PD-L1 inhibitory therapy.16,43 -45
The PD-L1, a major negative-regulatory molecule in tumor immunogenic pathways, has been demonstrated to be expressed in various tumor types, including NSCLC, and is associated with an unfavorable outcome.46 -51 However, in this study, no statistical difference in overall survival was demonstrated when patients were stratified by PD-L1 expression status. Similar to our study, other researchers reported no definitive prognostic significance of PD-L1 expression in NSCLC, possibly partly relating to the smaller number of the investigated patients.52,53
The PD-L1 expression has also been reported to be driven by EGFR mutation or ALK kinase fusion and the treatment correlated with EGFR- or ALK-TKIs may downregulate the PD-L1 protein expression. This resulted in poorer survival outcome in NSCLC patients harboring these targetable genetic aberrations.54,55 Recommendation from standard cancer treatment guidelines includes PD-L1 IHC testing in all NSCLC patients to precisely suggest the most beneficial therapeutic agent. Our study showed comparable results of PD-L1 positivity in NSCLC patients with the previous reports. 51 The results from our study revealed that NSCLC patients with squamous carcinoma histology were more likely to express PD-L1 than those with adenocarcinoma subtype. This finding concurred with a previous report from Zhou et al, 56 who reported a higher proportion of squamous carcinoma NSCLC patients expressing PD-L1, when compared to those with adenocarcinoma subtype.
Preclinical investigations confirmed the regulatory role of c-Myc to PD-L1 and also revealed the role of both molecules in promoting immune escape through the tumor immune response effectors in several tumor types, including lung cancer.57 -60 In clinical settings, a recent report by Zhou et al 56 revealed that PD-L1 expression was positively correlated with c-Myc expression and that PD-L1 and c-Myc co-expression was an unfavorable prognostic factor in NSCLC patients, which is similar to our study. In this study, the Kaplan-Meier plotter demonstrated relatively shorter median over survival duration in the c-Myc-positive NSCLC patients, than that of the c-Myc-negative group. Moreover, the survival time in the PD-L1 and c-Myc double-negative patients tended to be better than that of other subgroups. Therefore, a conjoint analysis of the PD-L1 and c-Myc expression is a potential approach to better determine prognosis in NSCLC patients.
Molecularly targetable laboratory results of NSCLC specimens may provide more precise guidance for further or subsequent therapeutic strategies. The results of this study illustrated a prognostic significance of overlapping biomarkers in NSCLC patients, c-Myc and PD-L1 expression, for example, providing a potential therapeutic strategy development for precision medicine, using anti-PD-1/PD-L1 immune checkpoint inhibitors as single agents, or in combination with potential targeted therapy.
There are some limitations of this study to declare. First, this was a retrospective study, containing a limited number of patient cohorts. However, we have made our best attempt to minimize the possible bias. In this study, the consecutive NSCLC patients with available specimens were recruited for the analysis. Most NSCLC patients who underwent surgery were stage I-II, while fewer number of stage III patients were eligible to receive surgery. Most of the stage III NSCLC patients were treated with chemotherapy and radiation. Therefore, the findings from the study might have been underrepresenting the stage III patients. In addition, considering the few number of stage III NSCLC patients, they were excluded from the survival analysis regarding their outlier characteristics, as an implication of the sensitivity analysis consideration. The small sample size in addition to a limit of diagnostic tests of targeted molecules may not adequately reveal the large perspective of genetic aberrations in NSCLC. As such, further validation in a larger cohort is encouraged to be conducted, especially in a prospective manner. It should also correlate the patient characteristics and treatment outcome with the corresponding molecular targeted therapy, when available. Second, related to the study’s retrospective nature, bias in patient cohort selection and the treatment paradigm, that each patient received, might have been introduced. This was different from data obtained from a prespecified prospective study or clinical trials. However, this study provides a useful benchmarking data for evaluating and suggesting an appropriate diagnostic molecular test panel, in selection for the best treatment for advance NSCLC further on.
Conclusions
The NSCLC tissue significantly expressed more c-Myc and PD-L1, compared with the matched normal respiratory epithelium, emphasizing the important role of these key drivers in tumorigenesis. We found that NSCLC patients with the co-expression of c-Myc and PD-L1 had a poor survival outcome. Therapeutic approaches to precisely inhibit the targetable molecular pathways responsible for NSCLC development and progression should be considered on an individual patient basis to improve survival outcome.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Faculty of Medicine, Prince of Songkla University, Songkhla, Thailand.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
PS contributed to conceptualization. PS and Pat T contributed to methodology. Par T contributed to validation. PS and Pat T contributed to investigation. PS contributed to writing—original draft preparation. PT and RC contributed to writing—review and editing. Par T and SS contributed to supervision.
Ethics Approval
The study was approved by Faculty of Medicine, Prince of Songkla University Institutional Review Board (Protocol number REC 60-283-14-1).
