Abstract
Treatment of cancer has transformed with the introduction of checkpoint inhibitors. However, the majority of solid tumor patients do not respond to checkpoint blockade. In contrast, the response rate to programmed cell death 1 (PD-1) blockade in relapsed/refractory classical Hodgkin lymphoma (cHL) is 65% to 84% which is the highest among all cancers. Currently, checkpoint inhibitors are only approved for cHL and primary mediastinal B-cell lymphoma as the responses to single-agent checkpoint blockade in other hematologic malignancies is disappointingly low. Various established biomarkers such as programmed cell death 1 ligand 1 (PD-L1) protein surface expression, mismatch repair (MMR) status, and tumor mutational burden (TMB) are routinely used in clinical decision-making in solid tumors. In this review, we will explore these biomarkers in the context of hematologic malignancies. We review characteristic 9p24.1 structural alteration in cHL and primary mediastinal B-cell lymphoma (PMBCL) as a basis for response to PD-1 inhibition, as well as the role of antigen presentation pathways. We also explore the reported frequencies of MMR deficiency in various hematologic malignancies and investigate TMB as a predictive marker.
Keywords
Introduction
T-cell inhibition through immune checkpoints, notably through cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) and programmed cell death 1 (PD-1) pathways, plays a critical role in cancer evasion of the immune system. Immune checkpoints induce self-tolerance by inhibiting T-cell maturation and dampening peripheral function. In the tumor microenvironment, CTLA-4 is expressed on T-regulatory cells (Tregs) and binds to CD80 and CD86 on antigen-presenting cells (APCs) resulting in decreased co-stimulation of conventional T-cells. 1 CD4+ and CD8+ T-cells become deactivated by programmed cell death 1 ligand 1 (PD-L1) and programmed cell death 1 ligand 2 (PD-L2) expressed on cancer cells. 2
Inhibition of these pathways by monoclonal antibodies has revolutionized the treatment of cancer. For example, treatment of melanoma with ipilimumab and nivolumab demonstrated an overall response rate (ORR) of 50% to 60%.3,4 Importantly, many of the responses were durable. 5 Since the first Food and Drug Administration (FDA) approval of ipilimumab for melanoma in 2010, the use of checkpoint inhibitors has expanded across multiple solid tumors including small-cell and non-small-cell lung cancer (NSCLC), renal cell carcinoma (RCC), hepatocellular carcinoma (HCC), head and neck cancer, urothelial cancer, esophageal cancer, gastric cancer, colorectal cancer, cervical cancer, and endometrial cancer.
Identifying biomarkers that can predict response to checkpoint blockade is of major interest because a significant proportion of patients do not benefit and experience immune-related adverse events or possibly even hyperprogressive disease. 6 These biomarkers are better established in solid tumors and include PD-L1 expression measured by immunohistochemistry (IHC),7,8 PD-L1 amplification, 9 tumor mutational burden (TMB),10,11 microsatellite instability (MSI), and mismatch repair (MMR) status12,13 (Table 1). Pembrolizumab was the first tumor agonistic treatment approved by the FDA based on MSI-high (MSI-H) or mismatch-repair-deficient (dMMR) status. 14 These tumors respond exquisitely to checkpoint blockade due to their high mutational burden and mutation-associated neoantigens. 12 Combinations of 2 or more of the above biomarkers are also being investigated. 15 These biomarkers, most notably PD-L1 expression and TMB, are routinely obtained in the evaluation of solid malignancies and are being incorporated in treatment decision algorithms. 16
Biomarkers routinely used and emerging biomarkers in solid malignancies.

Abbreviations: CPS, combined positive score; dMMR, mismatch repair deficient; FISH, fluorescent in-situ hybridization; IF, immunofluorescence; IHC, immunohistochemistry; MSI-H, microsatellite instability high; NGS, next-generation sequencing; PCR; polymerase chain reaction; PD-L1, programmed cell death 1 ligand 1; PD-L2, programmed cell death 1 ligand 2; RNA, ribonucleic acid; TPS, tumor proportion score.
In contrast to solid malignancies, checkpoint inhibitors are only approved for the treatment of 2 hematologic malignancies, classical Hodgkin lymphoma (cHL) and primary mediastinal B-cell lymphoma (PMBCL). The response rate to single-agent PD-1 blockade in relapsed/refractory cHL is 65% to 84%22-36 which is markedly higher than the response rate in any solid tumor. In contrast to cHL, most B-cell non-Hodgkin lymphomas (NHLs), multiple myelomas, and leukemias have disappointingly low response rates to checkpoint blockade. Furthermore, unlike solid tumors, predictive biomarkers have not been clearly defined for hematologic malignancies.
Here, we review the unique immune response biomarkers reported in lymphoma and explore the literature whether any of the markers used in solid malignancies could be applied.
9p24.1 gene alteration
cHL
Chemotherapy with doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) or escalated bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, prednisolone (BEACOPP) is established first-line therapies for advanced-stage cHL. Approximately 30% of patients will relapse or be refractory to frontline therapy. 37 Options for relapsed patients include further cytotoxic chemotherapy, autologous stem cell transplantation, and CD30-directed antibody drug conjugate brentuximab vedotin. 38
Checkpoint inhibitors are now a well-established treatment option in relapsed/refractory cHL with response rates of 65% to 84%, while its role in the frontline setting is currently being investigated (Table 2).22-36 This response rate is the highest reported among all malignancies and is the result of the distinctive biology of cHL. Classical HL is characterized by few malignant Hodgkin Reed-Sternberg (HRS) cells interspersed among ample but ineffective immune infiltrate consisting of eosinophils, macrophages, and lymphocytes which produces abundant cytokines. 39 Despite the proinflammatory tumor microenvironment, the immune system is incapable of clearing HRS cells. Mechanisms of HRS cell immune evasion include PD-L1/PD-L2 overexpression from 9p24.1 structural alterations, mutations in antigen presentation pathways, and alterations in nuclear factor kappa-light-chain-enhancer of activated B-cells (NF-κB) signaling pathway. 40
Prospective trials in classical Hodgkin lymphoma using checkpoint inhibitors.
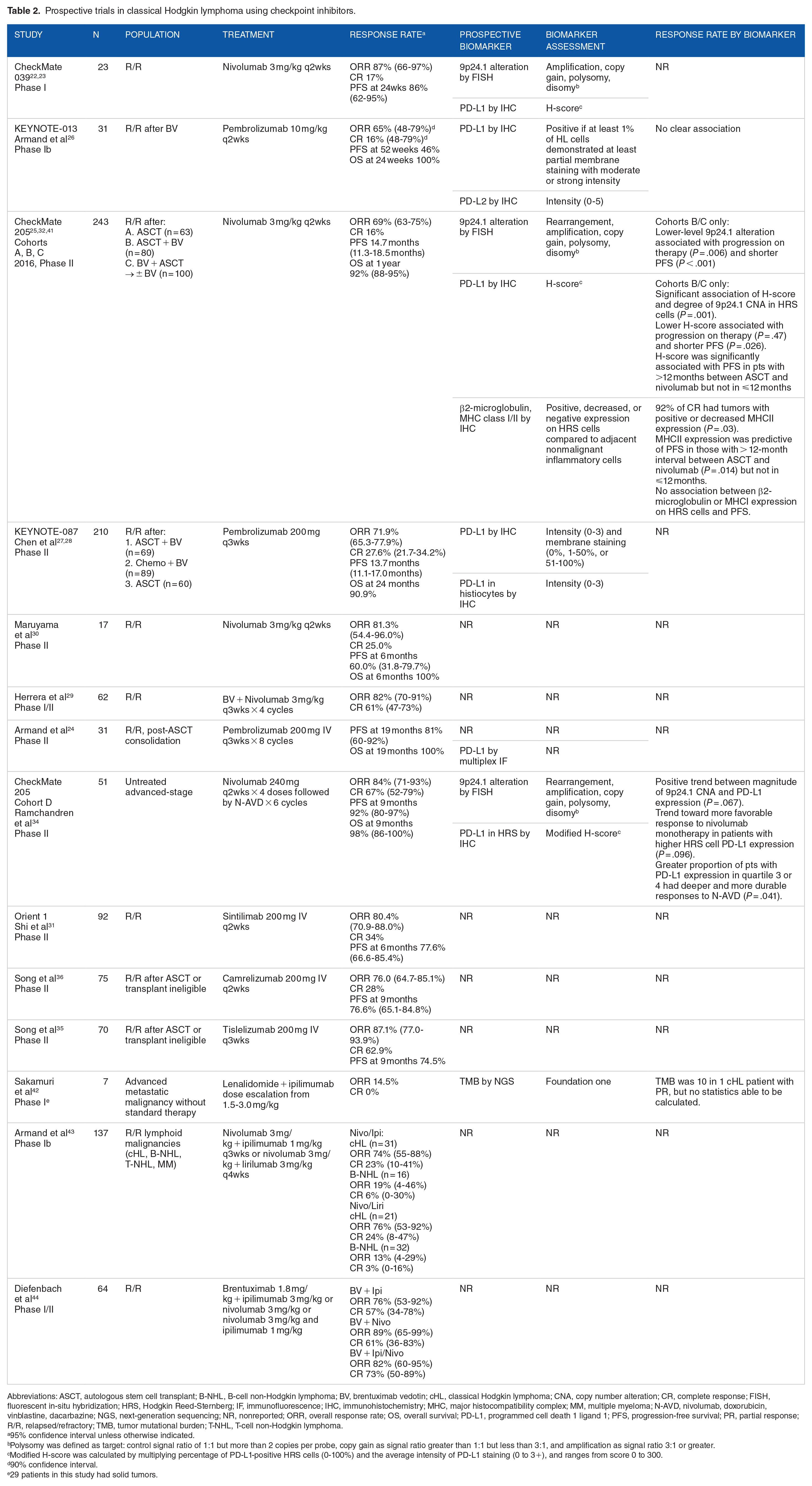
Abbreviations: ASCT, autologous stem cell transplant; B-NHL, B-cell non-Hodgkin lymphoma; BV, brentuximab vedotin; cHL, classical Hodgkin lymphoma; CNA, copy number alteration; CR, complete response; FISH, fluorescent in-situ hybridization; HRS, Hodgkin Reed-Sternberg; IF, immunofluorescence; IHC, immunohistochemistry; MHC, major histocompatibility complex; MM, multiple myeloma; N-AVD, nivolumab, doxorubicin, vinblastine, dacarbazine; NGS, next-generation sequencing; NR, nonreported; ORR, overall response rate; OS, overall survival; PD-L1, programmed cell death 1 ligand 1; PFS, progression-free survival; PR, partial response; R/R, relapsed/refractory; TMB, tumor mutational burden; T-NHL, T-cell non-Hodgkin lymphoma.
95% confidence interval unless otherwise indicated.
Polysomy was defined as target: control signal ratio of 1:1 but more than 2 copies per probe, copy gain as signal ratio greater than 1:1 but less than 3:1, and amplification as signal ratio 3:1 or greater.
Modified H-score was calculated by multiplying percentage of PD-L1-positive HRS cells (0-100%) and the average intensity of PD-L1 staining (0 to 3+), and ranges from score 0 to 300.
90% confidence interval.
29 patients in this study had solid tumors.
A defining characteristic of HRS cells is the 9p24.1 gene alteration/amplification, which contains genes CD274 (PD-L1), PDCD1LG2 (PD-L2), and Janus Kinase 2 (JAK2).41,45 In the phase II CheckMate 205 study evaluating single-agent nivolumab in relapsed/refractory cHL, all HRS cells had either copy number alterations (CNA) or translocations involving 9p24.1 locus.25,32,34 Ten percent of tumors had polysomy, 60% had copy gain, 27% had amplification, 2% had balanced re-arrangement, and 1% had unbalanced rearrangement of 9p24.1 as their highest level of genetic alteration. 41
Higher level of structural variants of 9p24.1 is associated with a poor prognosis in cHL. 45 Increased expression of genes in this locus results in increased protein expression of PD-L1/PD-L2 both directly and indirectly by upregulating JAK/signal transducer and activator of transcription (STAT) signaling pathway. 46 Expectedly, higher PD-L1/PD-L2 IHC expression measured by the product of percentage of positive malignant cells and the intensity of signal (modified PD-L1 H-score) is associated with higher response rates and progression-free survival (PFS) to anti-PD-1 therapy.32,34,41
In a post hoc analysis of cohorts B and C of CheckMate 205, 41 patients who had progression of disease on therapy had lower level 9p24.1 alteration (P = .006) and lower PD-L1 H-score (P = .047). Those who had lower-level 9p24.1 alteration and lower PD-L1 H-score also had lower PFS (P = .026). Interestingly, 2 patients with balanced rearrangements of PD-L2 had rapidly progressive disease. In cohort D of CheckMate 205, 34 a phase II trial of nivolumab combined with doxorubicin, vinblastine, and dacarbazine (AVD) in newly diagnosed advanced cHL, a greater proportion of patients in the third and fourth quartile for PD-L1 H-score had deeper and more durable responses (P = .041). Although rare in solid tumors (0.7%), PD-L1 amplification is associated with a response rate of 66.7% to PD-1 inhibition, approaching what is seen in cHL. 9
PD-L1 H-score may be a surrogate marker of level of 9p24.1 gene alteration. A major limitation is that above studies classified PD-L1 H-score to quartiles instead of defining an absolute cut off value for high level of PD-L1 expression. Further studies will need to demonstrate the predictive value of absolute PD-L1 H-score in cHL to anti-PD-1 therapy.
PMBCL
PMBCL is a subtype of diffuse B-cell lymphoma (DLBCL) which has similar genetic signatures and clinical characteristics to cHL. The most commonly used frontline therapies for PMBCL are dose-adjusted rituximab, etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin (DA-R-EPOCH) or rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) followed by mediastinal radiation. Autologous transplant, brentuximab vedotin, or checkpoint inhibitors are used for relapsed/refractory disease. 47
Alterations of 9p24.1 have been reported in 63% to 75% of cases of PMBCL.46,48,49 Interestingly, there is larger change in transcript abundance of PD-L2 than PD-L1, correlating with higher PD-L2 surface expression. 49 In one series, PD-L1 IHC was positive in 53% while PD-L2 was positive in 78% of lymphoma cells. 50 The significance of this finding is unknown. There are also several care reports of chromosomal rearrangements involving 9p24.1 locus, which result in high surface expression of PD-L1. 51
Similar to cHL, PMBCL is very sensitive to PD-L1 inhibition, with ORR of 45% to 73% (Table 3).33,52,53 KEYNOTE-017, 52 a phase II study of pembrolizumab monotherapy in 53 patients with relapsed refractory PMBCL, demonstrated an ORR of 45% with complete response (CR) of 13%. Patients who achieved CR had durable response at a median follow-up of 12.5 months: 76% of patients who responded had response durations greater than 12 months. Ninety-five percent of patients had 9p24.1 structural variation, including 4 patients with translocations. The degree of CNA was significantly associated with PD-L1 protein expression, which was significantly associated with PFS. Although limited to one prospective study, PD-L1 H-score is also a promising biomarker in patients with PMBCL predicting response to checkpoint inhibition.
Prospective trials in non-Hodgkin lymphoma using checkpoint inhibitors.

Abbreviations: ASCT, autologous stem cell transplant; BV, brentuximab vedotin; CLL, chronic lymphocytic leukemia; CR, complete response; CXCR5, C-X-C motif chemokine receptor 5; DLBCL, diffuse large B-cell lymphoma; ELISA, enzyme-linked immunosorbent assay; FISH, fluorescent in-situ hybridization; FL, follicular lymphoma; HR, hazard ratio; HSCT, hematopoietic stem cell transplant; IHC, immunohistochemistry; LAG3, lymphocyte activating 3; MHC, major histocompatibility complex; MDS, myelodysplastic syndrome; MM, multiple myeloma; MPN, myeloproliferative neoplasm; NE, not estimable; NHL, non-Hodgkin lymphoma; NR, nonreported; ORR, overall response rate; OS, overall survival; PD-1, programmed cell death protein 1; PD-L1, programmed cell death 1 ligand 1; PD-L2, programmed cell death 1 ligand 2; PFS, progression-free survival; PMBCL, primary mediastinal B-cell lymphoma; PR, partial response; RNA, ribonucleic acid; R/R, relapsed/refractory; SD, stable disease; SLL, small lymphocytic lymphoma; sMICA, soluble major histocompatibility complex class I polypeptide-related sequence A; TCL, T-cell lymphoma.
95% confidence interval unless otherwise indicated.
Polysomy was defined as target: control signal ratio of 1:1 but more than 2 copies per probe, copy gain as signal ratio greater than 1:1 but less than 3:1, and amplification as signal ratio 3:1 or greater.
Modified H-score was calculated by multiplying percentage of PD-L1-positive malignant cells (0-100%) and the average intensity of PD-L1 staining (0 to 3+), and ranges from score 0 to 300.
Target of pidilizumab is currently unclear.
90% confidence interval.
Number of patients indicated are those with hematologic malignancies, and excluding patients with solid malignancies.
DLBCL
DLBCL is the most common type of aggressive non-Hodgkin lymphoma and is typically treated with R-CHOP. Approximately 30% to 40% of patients will relapse or be refractory to frontline chemoimmunotherapy, and 20% to 50% of those can be cured with autologous stem cell transplantation if there disease is chemosensitive.67,68 Outcomes are poor for patients with chemorefractory disease. 69 Recently, chimeric antigen receptor (CAR) T-cell therapy was approved for the treatment of this population with 40% of patients achieving durable remissions. 70 Unfortunately, the majority of patients are not able to receive CAR T-cell due to cost, availability, and/or performance status.
Response rates to single-agent PD-1 blockade in DLBCL have been disappointingly low. Unlike cHL and PMBCL, there is a low incidence (10-27%) of structural variations of PD-L1/PD-L2 in DLBCL.54,71-73 Interestingly, these genetic alterations are more commonly seen in activated B-cell-like (ABC)/nongerminal center B (GCB)-type DLBCL, a subtype of DLBCL associated with a poor prognosis. 72 Tumors with 9p24.1 amplification had similar genomic profiles to PMBCL. 73 In a cohort of 1253 patients with DLBCL, PD-L1 protein expression was significantly associated with non-GCB subtype and Epstein-Barr virus (EBV) positivity and showed a trend toward inferior overall survival. 74 A meta-analysis demonstrated that 27% to 61% of DLBCL had PD-L1 expression, which was significantly associated with poor prognosis. 75 Checkpoint blockade in DLBCL has been of great interest; however, the response rate in unselected patients has only been 9% to 36% (Table 3).54,59,62 Given the association between 9p24.1 structural alteration and response to PD-1 inhibition seen in cHL and PMBCL, future studies should focus on DLBCL tumors with these alterations.
Significant PD-L1/PD-L2 gene arrangement has also been observed in extranodal subtypes of DLBCL—primary testicular lymphoma (PTL) and primary central nervous system lymphoma (PCNSL). In one series, 54% of PTL had 9p24.1 CNA and 4% had translocation involving PD-L1, while 52% of EBV-negative PCNSL had 9p24.1 CNA alterations and 6% had translocation involving PD-L2. 76 Structural variants of PD-L1 and PD-L2 correlated with surface expression of respective proteins. 76 The surprisingly high rate of PD-L1 and PD-L2 alteration seen on PTL and PCNSL suggest that these subsets of DLBCL may respond favorably to checkpoint inhibitors. In fact, a 5-patient case series of relapsed/refractory PCNSL and PTL who were treated with nivolumab demonstrated a response in all patients, 4 with CR and 1 with partial response. 77 Many of these responses were durable, ongoing at 13 to 17 months follow-up. There are several studies currently underway investigating the role of checkpoint inhibitors in relapsed refractory PTL and PCNSL (NCT02857426, NCT03770416, NCT03798314, and NCT04022980).
Gray zone lymphoma
Gray zone lymphoma (GZL) presents with features resembling both cHL and PMBCL. It can often present with mediastinal mass and possess morphological and immunophenotypical features of both lymphomas. 78 Although the optimal treatment strategy is not well established, it is treated similarly to aggressive large B-cell lymphoma. 79
The frequency of 9p24.1 alteration is not clearly defined in this rare entity; however, appears to be in significant proportion of tumors. One small series of 33 patients reported JAK2 and PDL2 alterations in 55% of cases. 78 Whether these patients will respond to checkpoint blockade is unclear. A case series of 3 patients in relapsed/refractory GZL demonstrated complete and durable responses in all 3 patients. 80 A clinical trial investigating the role of checkpoint inhibitors in GZL is ongoing (NCT03255018).
MHC expression
A key step in T-cell activation is antigen presentation on major histocompatibility complex (MHC) molecules. Any disturbance in this pathway may decrease the tumor neoantigen presentation and T-cell activation. Defects in antigen presentation have been proposed as a mechanism of immune evasion in both lymphoma and solid tumors. 81
Beta-2-microglobulin (B2M), a subunit of MHC class I molecule, plays a crucial role in transport of MHC class I molecules to the cell surface. B2M is mutated in 39% of cHL patients. 40 MHC class I expression is decreased to negative in 79% of cases of HRS cells, and is associated with inferior survival. 82 Interestingly, MHC class II expression is also significantly decreased in 67% of malignant cells. However, unlike MHC class I, this was not associated with inferior outcomes. The same group later reported in a post hoc analysis of CheckMate-205, 41 92% of tumors that achieved CR had no expression of B2M and MHC class I, suggesting that activation of MHC class I is not critical for a response to PD-1 inhibitor. MHC class II expression in the available samples were negative in 29% of patients, decreased in 32% of patients, and positive in 39% of patients, and interestingly, its expression was predictive of response to anti-PD-1 therapy. Notably, B2M mutation is seen in approximately 30% to 40% of tumors of PMBCL but approximately 10% in DLBCL.48,83 Overall, these findings propose that responses to PD-1 inhibition in cHL may rely on CD4+ T-cell activation.
In addition to B2M, several other genes involved in antigen presentation are frequently mutated in cHL and PMBCL. Deletion of 6p21.32, the locus for MHCI and MHCII genes, decreases surface expression of MHC class II, and is recurrently found in both cHL (43%) 40 and PMBCL (32%), 83 while it is only rarely found (12%) in DLBCL. 84 Structural variants of the class II MHC transactivator (CIITA) is a frequent gene fusion partner in lymphoma, and its alteration is found in 15% to 38% of PMBCL, 15% of cHL, and <5% of DLBCL.48,83,85
The above findings suggest that MHC class I and/or II downregulation may be a resistance mechanism to PD-1/PD-L1 blockade in cHL. Whether the activity of CD4+ T-cell or CD8+ T-cell alone or both are implicated should be further studied. In melanoma, decrease in MHC class I expression by B2M mutation has been shown to be a resistance mechanism to PD-1 therapy. 86 Selected population of patients may require combination approach with PD-1 and CTLA-4 blockade or CAR T-cell therapy that bypass T-cells receptor and MHC interaction.
TMB
Cancer usually develops due to acquired somatic mutations. High nonsynonymous mutational burden leads to cancer-associated neoantigen presentation that may be more readily recognized by the immune system. High TMB correlates with increased response to PD-1/PD-L1 blockade, especially in melanoma and NSCLC.10,11,87,88 When analyzing patients who received immunotherapy across different cancer types, 20% of patients with low (1-5 mutations/mb) to intermediate TMB (6-19 mutations/mb) had response to immunotherapy while 58% of patients with high TMB (>20 mutations/mb) had responses. 10 As such, TMB is a useful marker in predicting response to checkpoint inhibitors in solid malignancies.
Unlike solid malignancies, TMB in hematologic malignancies is not clearly defined, only reported in few series. Median TMB of cHL is 7.66 mutations/mb with notably higher TMB in EBV-negative cHL, level similar to NSCLC. 40 Median TMB of PMBCL is 7.0 mutations/mb. 83 Although not characterized by each lymphoma subtype, another study demonstrated that B-cell NHL had higher mutational burden compared to other hematological malignancies (TMB of cHL was not reported). 89 The relationship of TMB in hematologic malignancies to response to checkpoint inhibition has not been established. Studies characterizing the mutational load across hematologic malignancies are urgently warranted.
MSI and MMR
MSI-High or dMMR tumors have defects in DNA repair which result in higher mutational load. Similar to tumors with high TMB, there is an increase in cancer-associated neoantigen presentation leading to increased recognition by the T-cells. Unselected tumors with MSI-High or dMMR status have very high response rates to checkpoint inhibitiors12,13 which subsequently led to the FDA approval of pembrolizumab based on MSI status agnostic of tumor type. MSI and MMR status have not been defined in various hematologic malignancies, but deficient status is seen in approximately 9% in cHL, 40 8% in PMBCL, 83 and 0.3% to 3.2% in DLBCL.84,90 Their association with response to checkpoint blockade is currently unknown in lymphoma as no patients with lymphoma were represented in the trials involving MSI-H/dMMR cancers.
Conclusion
Although checkpoint inhibitors have changed the treatment paradigm for numerous solid malignancies, their benefit in hematologic malignancies has been limited to cHL and PMBCL. The genetic alterations of 9p24.1 locus containing PD-L1 and PD-L2 gene and antigen presentation pathways is the basis for checkpoint blockade response in cHL and PMBCL. However, there is still significant subset of patients with cHL and PMBCL who do not benefit from checkpoint blockade. Furthermore, patients with DLBCL and other indolent lymphomas largely do not benefit from PD-1 inhibitors, although a small subset of patients have durable response. It is therefore imperative that appropriate patients are selected based on available biomarkers and to understand resistance mechanisms.
A proposed algorithm for treatment decisions involving PD-1 inhibitors in relapsed or refractory lymphoma is presented in Figure 1. Classical HL or PMBCL with low PD-L1 H-score (although absolute score cutoff is to be determined) may predict a suboptimal response to checkpoint inhibition. Furthermore, an absence of MHC class II expression may be a possible resistance mechanism. However, standardized ways to measure MHC class II expression in the clinic are currently lacking. Combination chemoimmunotherapy or alternative therapies such as CAR T-cell could overcome checkpoint blockade resistance.

Proposed algorithm for anti-PD-1 inhibitor use in (A) cHL and PMBCL, and (B) in DLBCL and GZL.
Checkpoint inhibitors may be reasonable considerations in NHL with high TMB (particularly non-GCB/ABC subtype of DLBCL), high PD-L1 IHC expression, MSI/dMMR, or 9p24.1 structural alteration. Compared to cHL or PMBCL, NHLs exhibit lower PD-L1 surface expression and preliminary evidence of low TMB. Gene expression profiling has demonstrated that the majority of NHLs lack an inflamed gene signature, a characteristic that may be needed for successful checkpoint therapy. 91 Investigating these mechanisms will help explore ways to overcome resistance.
There are several areas of future interest. Structural alterations in 9p24.1, TMB, MSI/MMR status are not characterized for other types of NHLs or myeloid malignancies, and absolute cut-off value of the PD-L1 H-score should be established and validated in different lymphoma subtypes in prospective studies. Promising data suggest checkpoint blockade is efficacious for relapse postallogeneic stem cell transplantation.58,66 However, as Davids and colleagues demonstrated, increased risk of immune-related adverse events and fatal graft versus host disease is a serious concern in this setting and biomarker-based approaches may help better select appropriate patients. 64 The effect of PD-1/PD-L1 inhibition is also thought to synergize with CAR-T therapy by decreasing T-cell exhaustion. However data are limited to case series which report significant off-target immune effects. 92 Alternatively, preclinical data suggest that CAR-T cells engineered to secrete PD-1 blocking single change variable fragments may provide equal or increased efficacy but decreased toxicity. 93 Finally, checkpoint blockade may sensitize lymphoma to subsequent therapy. Carreau and colleagues showed that the overall response to subsequent therapy after checkpoint inhibition was 62% in 81 patients with relapsed refractory Hodgkin lymphoma 94 and 51% in 59 patients with relapsed refractory non-Hodgkin lymphoma. 95
With further studies, we are hopeful that biomarkers including PD-L1 expression, TMB, and MSI status will be used to best-select patients with hematologic malignancies for treatment with checkpoint blockade who are most likely to benefit.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.M.G. receives consulting and speaking fees from Seattle Genetics and consulting fees from EUSA Pharma.
Author Contributions
A.J and A.M.G drafted the manuscript. E.D.B and A.M.G reviewed and edited the manuscript. All authors approve the final manuscript.
