Abstract
Celiac disease (CD) is an autoimmune disorder characterized by the permanent inflammation of the small bowel, triggered by the ingestion of gluten. It is associated with a number of symptoms, the most common being gastrointestinal. The prevalence of this illness worldwide is 1%. One of the main problems of CD is its difficulty to be diagnosed due to the various presentations of the disease. Besides, in many cases, CD is asymptomatic. Celiac disease is a multifactorial disease, HLA-DQ2 and HLA-DQ8 haplotypes are predisposition factors. Nowadays, molecular markers are being studied as diagnostic tools. In this review, we explore CD from its basic concept, manifestations, types, current and future methods of diagnosis, and associated disorders. Before addressing the therapeutic approaches, we also provide a brief overview of CD genetics and treatment.
Introduction
Celiac disease (CD) is an autoimmune disease that is characterized by the permanent inflammation of the small bowel and is triggered by the ingestion of food containing gluten such as wheat, oat, barley, and rye. Small bowel inflammation occurs due to a genetic predisposition of individuals who are carriers of haplotypes HLA-DQ2 and HLA-DQ8. 1 This inflammation affects the proximal region of the small bowel and is conserved in the distal ileum. However, the small bowel has functional reserves, which explains why some individuals do not have mild or malabsorption symptoms. 2
Celiac disease was first described in 1887 by Samuel Gee who described the classic symptoms: diarrhea, poor growth, and fatigue. Gee stated that treatment is based on a gluten-free diet. Later, Paulley et al were the first to analyze biopsy samples of patients with CD and found an extensive flattening of the villi and chronic inflammation of the small bowel cells.1,3
Gluten is a set of proteins that serve as storage contained in the endosperm of certain cereal grains such as wheat, oats, barley, and rye. The endosperm is the raw material used to produce flour for bakery products and gives breads their chewy texture. 4 These proteins can be divided into 2 fractions by their solubility in alcohol: soluble gliadins and insoluble glutenins; each has 2 or 3 different structural domains. 5
The α/β-gliadin and γ-gliadin are the most abundant in foods containing gluten but differ mainly in their C-terminal and N-terminal domains. The α/β-gliadin presents the QPQPFPQQPYP peptide in the N-terminal end, whereas γ-gliadin contains QPQQPFP peptide. The C-terminal does not have repeating units and has fewer glutamine and proline residues compared with the N-terminal. Moreover, glutenins are divided according to their molecular weight in high-molecular-weight (HMW) and low-molecular-weight (LMW) glutenins. The LMW glutenins are mostly present in gluten. 5 Table 1 presents the most common foods that contain gluten and their derivatives as well as a list of gluten-free products.
Food containing gluten, their derivatives, and gluten-free foods.

Gluten triggers the immune response in CD when there is a deficiency of prolyl endopeptidase (PEP) enzymes in the digestive tract. These proteins are not degraded and, as a result, are accumulated in the lumen of the small intestine where they cannot pass the intestinal epithelium. Endogenous and exogenous agents present in the gut lumen affect cell permeability allowing the passage of proteins into the lamina propria where they will be used as a substrate for transglutaminase 2. This enzyme converts glutamine residues to glutamate. The resulting peptides have affinity for the HLA-DQ2/DQ8 molecules (exposed on the antigen-presenting cells). 6
The antigen-presenting cells are found in the lamina propria and present a complex enzyme peptide. As a result, CD4+ T cells are activated triggering a TH1 response. This produces antigliadin antibody (AGA) and anti-transglutaminase antibody. Gamma interferon is released, and presentation of the antigen to the HLA molecules increases. This increases the expression of ligands that are recognized by T cells. 7
Celiac disease also increases the number of CD3+ intraepithelial lymphocytes followed by identification of TH17 T cells involved in the pathogenesis of autoimmune diseases. Interleukin (IL)-23, TH17-associated cytokine, and IL-17A IL1β are also implicated in CD. 8
Types of CDs
Five types of CDs have been identified according to small bowel villous atrophy and positive serologic markers in patients. These include classic, nonclassic, silent, potential CD, and refractory sprue (Table 2). The classic symptomatic CD is characterized by common symptoms of malabsorption and occurs in children between 6 and 24 months of age. 8 Nonclassic symptomatic CD includes a variety of symptoms such as iron deficiency anemia. The anemia occurs due to malabsorption of iron and folate in the jejunum. Nonclassic CD is more common than classic CD. 9 Silent CD has no symptoms. However, the detection of positive celiac-associated antibodies, HLA-DQ2/DQ8, and atrophy of the villi in the small bowel biopsy must be tested. Patients with this type of CD are identified via affected family members or via serum markers belonging to one of the risk groups. 10
Types and symptoms of celiac disease.
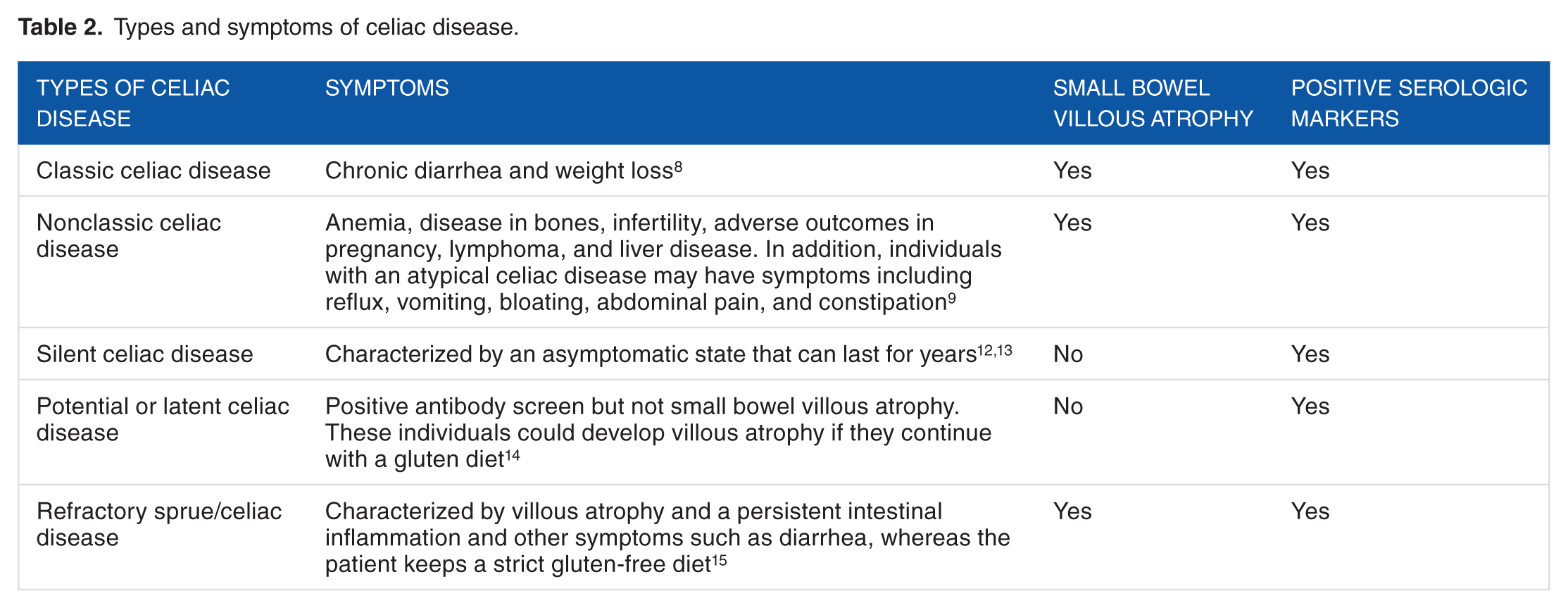
Potential CD has a normal small bowel biopsy and positive serum markers. The continuous ingestion of gluten can result in villous atrophy. Refractory sprue/CD is divided into 2 groups: primary refractory sprue (when the patients do not have a good response to a gluten-free diet) and secondary refractory sprue (when patients respond well to a gluten-free diet but subsequently relapse). These 5 types of CDs establish the celiac iceberg, which represents the complexity of the disease that, in some cases, remains undiagnosed 8 (Figure 1).
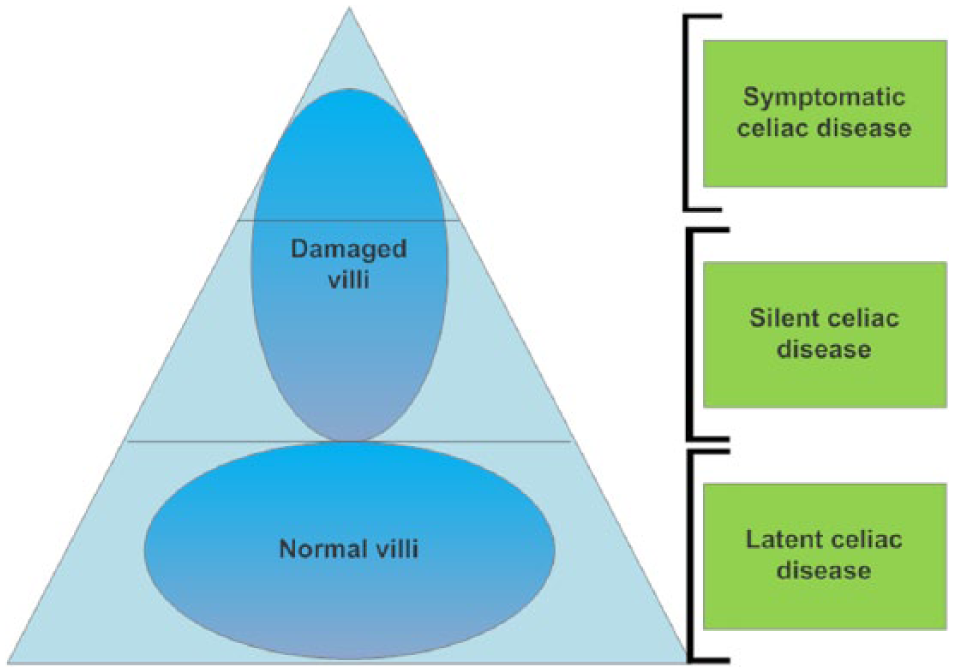
Graphical representation of the types of celiac disease “celiac iceberg.” The tip of the iceberg represents symptomatic cases, whereas the rest of the iceberg are silent cases and latent cases that are undiagnosed. 8
Gastrointestinal and Nongastrointestinal Manifestations
Celiac disease is characterized by a high level of variability in patients and their clinical manifestations. The most characteristic symptoms are diarrhea, steatorrhea, extreme lethargy, bloating, edema, and atrophy of the villi of the small bowel. 2 Other symptoms may occur in a lower percentage of individuals such as anemia, bone diseases, weight loss, and abdominal pain. 8 Table 3 presents other symptoms besides the gastrointestinal findings including certain atypical symptoms and their association with other diseases. 11
Clinical manifestations of celiac disease and their association with other diseases.

Abbreviation: NR, not reported.
From 6 to 24 months of age, children present symptoms such as impaired growth, abdominal distention, abnormal stools, fatigue, problems with the nervous system and musculoskeletal systems, as well as dermatological, hematological, endocrinologists, oncological, neurological, and pulmonary findings.10,16 Regarding clinical manifestations, classic CD is more common in children, whereas silent CD is more common in adults. 17 In addition, adults have a much higher delay in diagnosis (10 ± 9 years) compared with children (1 ± 2 years). Adults are frequently associated with other autoimmune diseases. There is not a significant difference between children and adults in terms of genetic biomarkers. 18
Infertility is also common in adult women. Studies have shown that 2.65% of women with infertility are CD-positive via endomysial antibody (EMA) test.20,21 Singh et al 22 confirmed that CD is more prevalent in women with all types of infertility than in the general population. According to Lasa et al, 23 when infertility is unknown, it is advisable to conduct CD tests. A gluten-free diet could improve the likelihood of conception. Men could also be affected with teratozoospermia and asthenozoospermia. 24 In addition, children of male patients with CD can have a shorter gestational age and lower birthweight. 15
Furthermore, CD is associated with autoimmune diseases—mainly with type 1 diabetes mellitus that occurs in 3.5% to 5% of patients with CD. Another prevalent condition is hypothyroidism, which is 10-fold more common in celiac patients. Liver diseases such as autoimmune hepatitis and sclerosing cholangitis transaminitis are common. Finally, Crohn disease, alopecia areata, Sjögren’s syndrome, hypopituitarism, ulcerative colitis, scleroderma, Addison’s disease, dermatomyositis microscopic colitis, systemic lupus, psoriasis, erythematosus, and hypoparathyroidism are also associated with CD. 25 Table 4 summarizes the risk of developing each disease depending on health conditions.7,8
Diseases with increased risk of CD.

Abbreviation: CD, celiac disease.
Genetics of CD
The pathogenesis of CD remains incompletely understood, but adaptive and innate immune responses have major roles. Genetics is a contributing factor to CD disease susceptibility, but environmental factors such as gluten are also determinant.26,27 The HLA complex is mainly related to CD, and it consists of 47 Mb on chromosome 6p21. It contains approximately 200 genes, of which more than half have an immune purpose. The main function is the presentation of exogenous peptide antigens to the helper T cells. 28 In addition, many genetic linkage analyses have identified susceptible loci in certain chromosomes, ie, in chromosomes 2, 5, 6, 9, 15, and 9. This reveals the multifactorial complexity of CD that is associated with the presence of the HLA heterodimers.29,30
The HLA-DQ2*5 molecule is encoded by DQA1*05:01 and DQB1*02:01 alleles in cis-configuration in the DR3 haplotype. In general, up to 95% of patients with CD are positive to HLA-DQ2 (DQA1*0501/DQB1*0201), and the remaining 5% are positive to HLA-DQ8 (HLA-DQB1*0302) haplotypes. 31 HLA-DQ2/DQ8 common haplotypes have been shown to elevate disease risk by 6-fold. However, HLA status alone cannot initiate or promote the development of CD in individuals. 32 Moreover, HLA-DQ2 or HLA-DQ8 is positive in 40% of Europeans, but only 3% of them develop CD. 26 The CD-related HLA DQ antigens (DQ2) are present in 5% to 10% of Chinese and sub-Saharan Africans versus 5% to 20% in Western Europeans. The DQ8 is positive in 5% to 10% of English, Tunisians, and Iranians and in <5% of Eastern Europeans, Americans, and Asians. 33
Recent studies have characterized CD as highly heritable. However, the development of the disease is also related to environmental factors. 34 Moreover, the frequency of the disease in first-degree relatives is high, but inheritance has not yet been completely confirmed. It is known that the HLA genes have moderate impact on CD heritability and have Mendelian inheritance with incomplete penetrance.8,35
The recent fine mapping and genome-wide association studies (GWAS) have identified up to 57 non-HLA CD susceptibility single-nucleotide polymorphisms (SNPs), most of which are noncoding variants lacking any functional annotation. 26 Therefore, rather than showing these unknown variants, we focused on Banaganapalli et al, 27 who adopted a multidimensional computational approach for uncovering the plausible mechanisms through which these GWAS SNPs are connected to CD pathogenesis. The functional annotations of 57 CD lead variants and their strongly linked 1008 variants was conducted with the 1000 genome project data of Central European Populations (CEU). The LD variant consists of 939 SNPs, 28 insertions, and 41 deletions. At the initial phase, they identified that 25 (43.85%) out of 57 CD-SNPs lie in evolutionarily constrained genetic element regions.
In the follow-up phases, the authors used computational algorithms (CADD, GWAVA, and FATHMM) and deleterious intensity measurements to discover that 42 (3.94%) out of 1065 variants (57 CD lead and 1008-linked SNPs; r2 ≥ 0.8) are differentially deleterious or possibly deleterious in nature to CD. Data analysis of deleterious variants revealed that 12 allelic variants (3 rank I + 9 rank II SNPs) localized to 7 genes have significant differential expression in nonimmune cell sources. Five SNPs (rs34505903, rs6441961, rs6441962, rs6441972, and rs6771900) showed significant expression quantitative trait loci (eQTL) for CCR2 gene in skin tissue. The significant eQTLs were also observed for 2 rank II SNPs (rs10797440 and rs4648562) for MMEL1 gene in blood. The other genes with significant eQTL expression were PLEK (rs3816281—blood), UBE2L3 (rs11089620—blood), ELMO1 (rs60600003 gene—intestinal tissue), and LMAN1L (rs4886619 gene—esophagus tissue). 27
Deleterious SNPs of CCR2 gene influences its expression levels and may also elicit a cascade of T cell–mediated immunologic events leading to intestinal gluten intolerance in genetically susceptible individuals (Table 5). This study demonstrates the utility of integrated in silico analysis of annotations, gene expression, and pathways in prioritizing the potential complex disease variants from large-scale open-source genomic data. 27
Multiple functional annotations and prediction analysis of the 12 CD susceptibility SNPs revealed by the eQTL analysis.

Abbreviations: CD, celiac disease; eQTL, expression quantitative trait loci; SNP, single-nucleotide polymorphism.
Rank 1: deleterious variants; rank 2: possibly deleterious variants.
Prevalence
Studies of CD around the world are shown in Table 6. Celiac disease is almost exclusively seen in European populations. Several serological studies of people from America, Australia, Asia, and Africa have shown that the prevalence in these regions is 0.5% to 1%.1,36 Thus, it was found that CD is a worldwide condition and is probably the most common disorder related to food intolerance.37,38
Worldwide celiac disease prevalence.

Abbreviations: EMA, Anti-endomysial antibody; AGA, antigliadin antibody; ELISA, enzyme-linked immunosorbent assay; IgA, immunoglobulin A; IgG, immunoglobulin G; tTG, anti-tissue transglutaminase.
Fasano et al 36 studied a North American population and found that the prevalence of CD is similar among different age groups (children, adolescents, adults, and the elderly) (0.8%). Risk populations include patients with CD, symptomatic patients, and people with type 1 diabetes mellitus, infertility, osteoporosis, anemia, short stature, arthritis, and Down syndrome. These have the highest prevalence (1.2%-4.8%). 36 However, only 17% of CD individuals have been diagnosed. 8
Data on the prevalence of CD are scarce in Latin America. It has been estimated that Argentina, Brazil, and Chile have a prevalence of 0.6% in adults, 0.15% in blood donors, and 4.76% in risk populations.41–43 Moreover, in Central America (Cuba) in 1981, cases of children with CD were reported with a prevalence of 2.3%. 44
In Europe, CD has been extensively studied along with its prevalence. In general, the prevalence in Europe is 1%, but this varies from country to country. In subjects aged 30 to 64 years, the prevalence is 2.4% in Finland and Sweden, 0.3% in Germany, and 0.7% in Italy. 45
Although CD seems absent in Asian populations, approximately 3 to 10 cases remain undiagnosed. Other studies have shown that the prevalence of CD is high. In India (Punjab), the prevalence in children is 0.3%. 58 In the northern part of India, the general prevalence is 1.04%. 59 In other locations in Asia, the prevalence of CD remains unknown. Turkey, Iran, Syria, and Israel have values between 0.2% and 2%.60–63 In Japanese and Chinese populations, CD is believed to be practically nonexistent. However, there are studies in adult descendants of Chinese and Japanese families who migrated to Canada showing positive CD results. 73 In fact, a sample of 62 Chinese patients with chronic diarrhea showed that 4 were diagnosed with CD suggesting that the disease may be more common than it was believed to be by Asians. 64 According to Cummis and Roberts-Thomson, 65 the prevalence in Japan is 0.005%.
In North Africa, a high incidence of CD has been found (0.25%-5.6%) in the general population. These data are very close to those reported in European countries. 62
In addition, a study on Saharawi children was conducted to detect CD using antiendomysial and anti-transglutaminase (anti-tTG) tests. As a result, a prevalence of 5% to 6% was obtained. 45 Although the reason for the high prevalence in these regions is not clear, the genetic background could be a link. Moreover, it is believed that this high prevalence is an evolutionary advantage to obtain fewer differentiated enterocytes necessary for the accession of microorganisms in the duodenum. Therefore, CD gives a selective advantage to protect individuals from gastrointestinal infections and parasites such as Giardia lamblia or Vibrio cholerae. 68
Three studies of CD in Oceania have been reported. The first was done in 2000 in Christchurch, New Zealand, resulting in a 1.2% general prevalence. 71 The second one was conducted in 2001 in West Australia with a prevalence of 0.23%. The third study was a screening using anti-tissue transglutaminase (tTG) antibody assays in 3011 subjects showing a prevalence of 0.56% to 0.96%. 72
According to Clot and Babron, there is a genetic predisposition in certain individuals, which increases the prevalence of the disease. For example, between monozygotic twins, there is a high rate (70%), whereas among first-grade relatives the value is 10% to 15%; in dizygotic twins, it is 30%. 32
Diagnostics
It is important to mention that clinical manifestations are not sufficient for a true diagnosis. Rather, a combination of several complementary studies such as clinical, hematological, serological, genetics, results from biopsies of the duodenum, and the response to a gluten-free diet is required. In addition, other groups are more susceptible to developing CD. These include first-degree relatives of patients who have already been diagnosed, patients with Down syndrome, type 1 diabetes mellitus, autoimmune thyroiditis, selective IgA deficit, digestive diseases, autoimmune diseases, neurological and psychiatric disorders, or other conditions such as fibromyalgia and Turner syndrome. 74
However, there are several strategies for diagnosis. The typical diagnosis includes a blood count to determine clotting times, which in positive patients can present alterations. A complete biochemistry panel includes ferric levels, serum transferrin levels, transferrin saturation, and a liver test. 75 Symptoms that are related to CD, other associated diseases, and the risk groups should be studied for a better diagnostic strategy. Symptomatic patients differ in their symptoms according to their age; ie, children show irritability, diarrhea, abdominal pain, decay, lack of appetite, malnutrition, anemia, and muscular hypotrophy. 74 In adult patients, digestive problems are usually not seen; however, other symptoms such as abdominal pain, delayed menarche or irregular menstrual cycles, iron deficiency anemia, short height, and muscular weakness have been reported. Youth and adolescents usually present dermatitis herpetiformis. 75
Clinical diagnois: antibody testing
The diagnosis of CD from serological tests changed the perspective of the disease because its impact was much greater than expected. Serological tests can determine the condition in most classical symptoms. 74 Different serological markers are used. The most widely used are the AGA and immunoglobulins (IgA and IgG) acting against α-gliadin antigens. Other antibodies include tissue transglutaminase (tTG), antiendomysium (EMA) and gliadin peptides. 75
Table 7 shows the specificity and sensibility of the antibodies used in the diagnosis of CD. On the other hand, EMA have a higher efficiency (95%). 74 Nevertheless, some problems have been identified with this technique. At the end of the 20th century, tissue transglutaminase tTG auto-antigens reacting against the EMA was described. The tTG has been widely used due to its determination by enzyme-linked immunosorbent assay with ≥90% efficiency. However, the sensitivity and accuracy of the tTG depend specifically on the intensity of the duodenal lesion. 75
Sensitivity and specificity of serum antibodies. 8
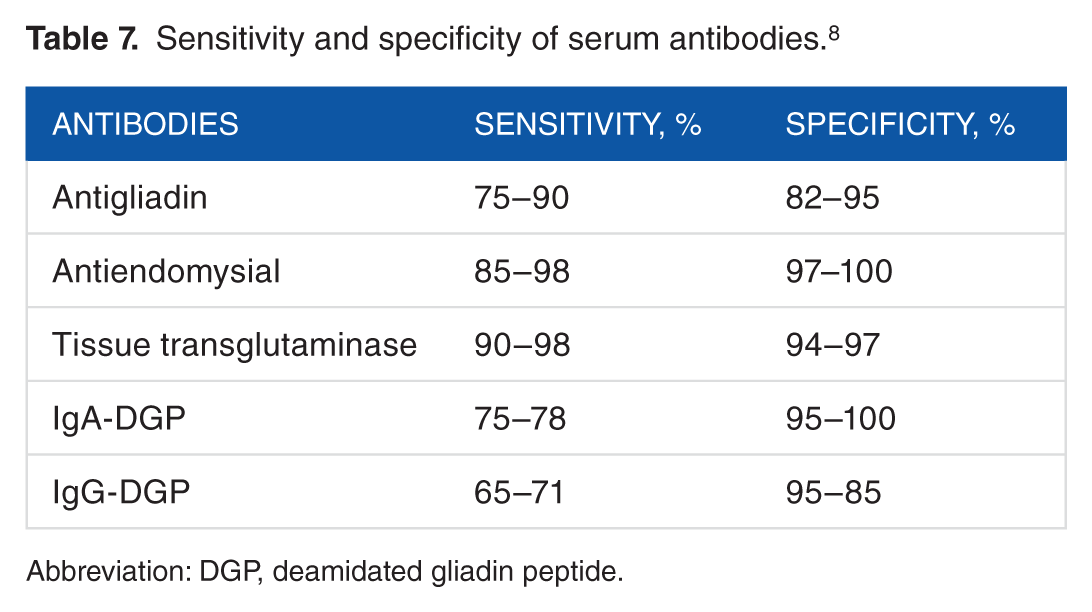
Abbreviation: DGP, deamidated gliadin peptide.
When patients follow a gluten-free diet, antibodies are no longer present in the blood because their production is not needed and their diagnosis is not effective. In this case, individuals may consume food containing gluten for at least 2 weeks before the antibody test. 76
Histologic diagnosis
Duodenal endoscopy findings can be helpful for the diagnosis of CD. One of them is the “pattern in mosaic,” characterized by a reduction or disappearance of the folds of Kerckring. The other refers to the “scalloped pattern” describing the circular folds of the duodenum that acquire a toothlike configuration. Currently, diagnosis through Marsh classification from an upper endoscopy in adults has been shown to be the most widely used technique. Marsh allows the identification of the entire spectrum of histologic lesions (Table 8). However, a combination with antibody testing may be required in some cases. This technique could fail to detect histologic changes when the patient is under a gluten-free diet or there are patchy mucosal lesions, peptic changes, incorrect orientation of the slide during the microscope analysis, a limited number of samples taken, or latent CD. 8
Modified Marsh classification. 77
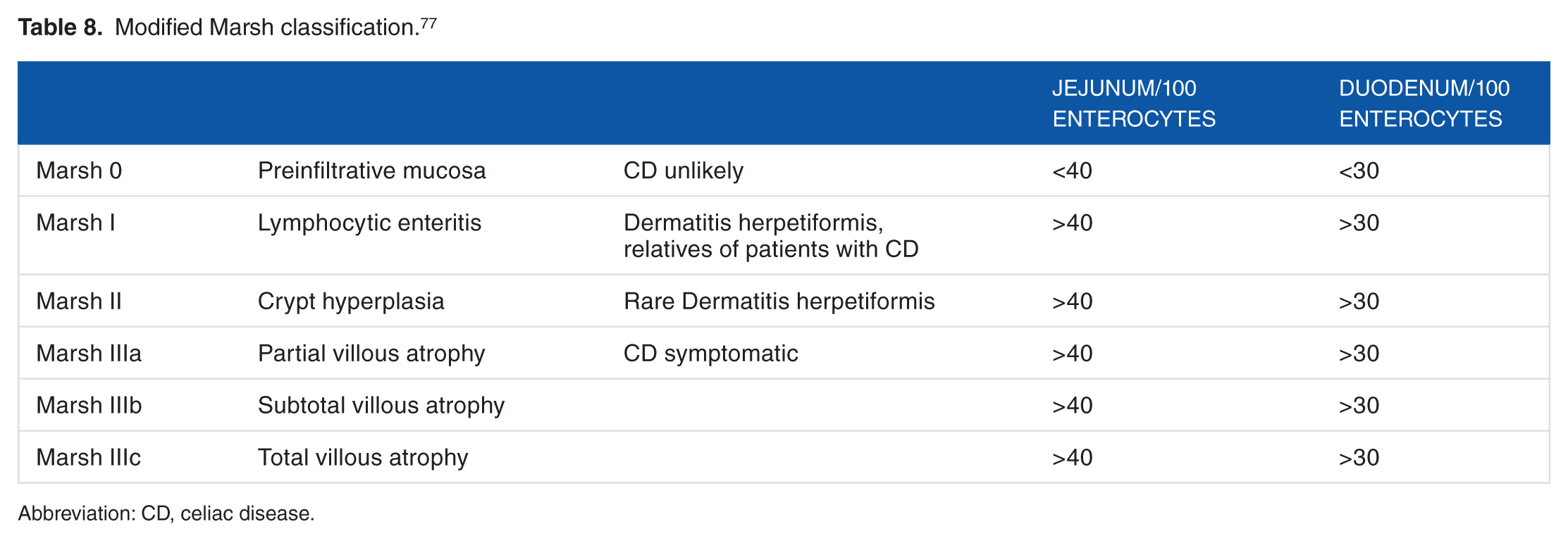
Abbreviation: CD, celiac disease.
In Marsh I cases (lymphocytic enteritis), either a subsequent diagnosis with hematoxylin-eosin staining or immunohistochemical techniques with monoclonal antibodies are recommended because it can cover the same symptomatology of patients with villous atrophy. 78 Up to 95% of cases diagnosed with CD during childhood can have a complete recovery of the intestinal mucosa within 2 years after starting a gluten-free diet. 79 However, in adults, the recovery rate of the mucosa is less effective and requires more than 12 months of a strict gluten-free diet. 76 However, a minority of patients without symptoms but with persistent atrophy of the villi of the mucosa may develop refractory CD that opens the possibility to other complications over time. 80
Genetic testing
Currently, there are genetic markers for the diagnosis of CD based on the identification of alleles coding for HLA-DQA1, HLA-DQ2, HLA-DQB1, or HLA-DQ8. The presence of HLA alleles could determine the susceptibility to CD. 8 The HLA-DQA1 codes for the α chain of HLA heterodimers, whereas HLA-DQB1 codes the β chain. Approximately 90% of the people with CD express the HLA-DQ2 heterodimer, which compromises 2 gene variants: HLA-DQA1 *0501/*0505 and HLA-DQB1*0201/*020*. Patients express only 1 of the variants—not both. The HLA-DQ8 heterodimer is expressed in 5% to 10% of the cases.74,81
However, there is evidence that non-HLA genes are associated with CD susceptibility. van Heel et al 28 affirms that HLA genes contribute to 30% to the development of CD and non-HLA to 70% in identical twins.
Genetic testing is recommended for patients who have a well-founded clinical suspicion and present anomalies in their duodenal histologic findings. An individual is considered genetically predisposed to CD if DQ2 and/or DQ8 are positive. 8
A definitive diagnosis for CD could be confirmed when the person has a positive result on biopsy and has a clinical and histologic improvement after a gluten-free diet. In cases of refractory sprue suspicion, imaging, immunohistochemical, and TCR gene rearrangements as well as the evaluation of T-cell lymphoma studies should be performed. 25
New and future diagnostic strategies
Because the prevalence of CD has increased in the last years, early diagnosis is imperative for risk families and undiagnosed patients. 82 To achieve this, new diagnostic methods and treatments for CD are being tested including salivary diagnostic tests, gene expression panels, reduced-gluten grains, oral enzyme therapeutics, and DQ2 blockers.83,84
There is a possibility of diagnosing CD through a saliva sample. Rujner et al 85 presented results regarding the sensitivity of salivary tests IgA-EMA (antiendomysium) and IgG-AGA (antigliadin). In 2011, a CD screening was done using saliva samples from Italian children. Anti-tTG and immunoglobulin (IgA) assays were performed using fluid-phase radioimmunoprecipitation. 86
Capsule endoscopy is a recent and less aggressive method that scans the entire digestive tract. With this methodology, anomalies and histologic changes in the duodenal field could be analyzed. The capsule endoscopy represents an effective technique for the diagnosis of CD and other diseases associated with recurrent gastrointestinal hemorrhage and malignant tumors in the bowel, but it cannot obtain biopsies. 75
Chromoendoscopy offers high specificity and sensitivity that determines the villous changes using specific dyes. 82 However, this technique needs a magnification endoscopy to show improved yields in CD gastroscopy. In addition, narrow-band imaging uses filtered light that is absorbed by hemoglobin to show intestinal mucosa vascular organization. This could detect partial damages in patients with CD. 87 A new diagnostic technique is the biopsy-based organ culture. Recent research has shown that celiac antibodies could be detected in small-intestinal organ cultures of patients with CD. 88 However, these techniques are laborious, time-consuming, and require special skilled and experienced personnel.
Future biomarkers candidates
Galatola et al 82 stated that CD could be predicted using small panels of genes including KIAA, TAGAP, and SH2B3 9 months before clinical or serological signs. Trynka et al 89 estimate 40 known loci that are related to the development of CD. In addition, Lie et al 90 showed that there are genes involved in pathogenesis of both CD and type 1 diabetes that are located within or near the HLA.
Other genes unrelated to the HLA complex have a relationship with the development of CD. These include Myoxin IX, which increases the risk of developing CD 2.3-fold due to the cytoskeleton remodeling and cell permeability in the duodenal wall epithelium functions. 8
IL2-IL21 is another non–HLA-related gene region associated with susceptibility to CD. This explains the 3% to 4% of inheritance of the disease. However, near this region, related SNPs associated with the CD are found and demonstrate the genetic relationship of the region. 91
Genes coding for signaling molecules that play a role in the secondary activation of T lymphocytes have also been correlated to the development of CD in Finnish families. These include CTLA4 (cytotoxic T lymphocyte associated), CD28, and ICOS (inducible co-stimulator). They are all located on chromosome 2q33. However, according to Haimila et al, 92 the link to CTLA4 is still under study.
In a study of 12 000 patients with CD GWAS studies, CD susceptibility is related to 43 loci (including HLA locus). Moreover, SNPs in the non-HLA loci represent approximately 15% of the disease risk. Interestingly, most of the CD-associated SNPs do not imply protein changes. The immune response of CD is also determined by SH2B3, CCR3, IL18RAP, RGS1, IL12A, and TAGAP SNPs. The rs3184504 in the SH2B3 gene has specifically shown disease susceptibility. The IL10 allele variants are related to CD due to the reduction in the production of anti-inflammatory cytokines involved in the response of severe inflammatory lesions caused in the early stages of the disease.8,93–95
In addition, there is a positive relation between CD and MICA SNPs. MICA-A5 transmembrane SNP is associated with atypical CD. Lopez-Vazquez et al 96 demonstrated high expression of this gene in biopsies. Similarly, the allele MICB0106 is significantly associated with the disease. 36 Therefore, MICA and MICB molecules are overexpressed in CD 7 ; CD major susceptibility genes can be found in Table 9. Finally, HLA genes are highly polymorphic with more than 7500 common SNPs. Thus, the genotyping of SNPs could present an efficient diagnosis of CD risk groups and patients. 97 However, there are differences between worldwide populations.37,98,99
Major genes related to CD.

Abbreviations: CD, celiac disease; GTPase, guanosine triphosphatase.
We suggest considering Banaganapalli et al 27 because the presence of any of these 12 SNPs is deleterious. Other biomarker candidates are being studied for CD diagnosis including serum intestinal fatty acid–binding protein, CYP3A4-catalyzed simvastatin metabolism, and gluten-reactive CD4+ T cells in the blood. 97 Intestinal fatty acid–binding protein is presented in the epithelial cells of the intestine and released into blood on mucosal injury and presented in patients with CD. 102 CYP3A4 metabolizes simvastatin—a cholesterol-lowering drug. In patients with CD, CYP3A4 expression and activity are reduced. 103 Finally, gluten-reactive CD4+ T cells were found in higher concentrations in the circulation of patients with CD who are untreated and under a gluten-free diet.
Treatment
The only treatment that works with excellent results is a permanent gluten-free diet. In children, this treatment eliminates gastrointestinal symptoms, stabilizes nutritional measures, improves growth, and stabilizes weight and hematological/biochemical parameters. However, sticking to a gluten-free diet is very difficult because many products that claim to be gluten-free actually have traces of gluten. It has been shown that small amounts of gluten are sufficient to cause changes in the intestinal mucosa. 8
Therapeutic Approaches
In low-income countries, the dietary treatment could be expensive and difficult to achieve. Consequently, new treatments are being investigated. 97 This includes oral enzyme therapeutics. The objective of this strategy is to reduce the amount of gluten that reaches the small intestine to prevent the immune response. The human digestive system lacks PEP enzymes capable of degrading peptides found in gluten. The problem with this strategy is the large amount of enzymes needed to cover a gluten detoxification of a daily intake of 20 g. Furthermore, the stomach environment could decrease enzyme function. 83 In contrast, minimizing gluten absorption can use HMW polymers with affinity for gluten binding. Thus, gluten is not absorbed by the epithelium and does not exert toxicity. 104
Alternatively, de-sensitization treatment involves repeatedly dosing selected gliadin immunogenic peptides of wheat, barley, and rye that inhibit the proliferation of T cells and expression of pro-inflammatory cytokines. As a result, gluten tolerance is recovered. 84 Finally, one way to prevent the interaction with gluten is blocking the synergy of HLA molecules with gluten peptides—this requires high affinity blockers. 19
However, proteins present in gluten have been investigated, and there are several strategies to remove CD activator proteins in gluten-containing grains. One of these is via genetic manipulation and selective breeding. However, one concern is that the desirable features of flour will be lost. 83
Finally, a disease-modifying therapeutic approach for CD is being developed. This intradermal therapeutic vaccine claims to return a HLA-DQ2.5-positive patient to a normal diet with peptides that trigger a pro-inflammatory response and stop the immune defensive activity to gluten antigens. 105 However, this method is under investigation.
Patients could also have the symptoms of the disease despite a gluten-free diet. In this case, food cross-contamination in commercial products such as canned food, millet, sorghum, frozen foods, ham, bacon, and meat cured products should be tested.106,107 The Alimentarius Codex has established a limit of 20 ppm (mg/kg) of gluten in food to categorize food as gluten free. 108 However, Collin et al estimated that the residual amount of gluten suitable for celiac patient consumption was 100 ppm. Moreover, the daily flour intake for long-term mucosa recovery was 80 g. In conclusion, the 100 ppm contained in 830 mg of gluten was shown to be safe for celiac patients. 109
There are also over-the-counter (OTC) medications with active and inactive ingredients, but consumers do not always have access to such information. It is important to note that both starch and hydrolyzed starch, as well as wheat, are common ingredients in OTC medicines. In addition, inactive ingredients and their sources are usually variable between original and generic drugs. 110
Morbidity and Mortality
The mortality of CD increases from 1.9 to 3.8 times due to the development of malignancies such as lymphomas, mouth and pharynx squamous cell carcinomas, and small intestine adenocarcinoma. However, patients who follow a strict gluten-free diet significantly reduced the occurrence of these conditions. 25
Ludvigsson et al found an increased mortality and latent CD in their study. The mortality rate found was 10.4 per thousand people per year, whereas in the latent CD, it was 6.7. Thus, they concluded that the risk of death from CD has a relatively high value. 111 According to Peters et al, cardiovascular disease is the leading cause with 39.4% in patients with CD, followed by malignant neoplasms with 19.4%, digestive diseases with 12.1%, and respiratory diseases with 9.8%. 112
Finally, although CD is more common than thought, its symptoms can easily be confused with other diseases or it can be misdiagnosed. A variety of genes are involved in the predisposition to this disorder. Therefore, further studies are needed to identify a more reliable, effective, and less invasive diagnostic method. The prevalence of the disease varies from country to country but is seen in people of different ethnicities. 113
Footnotes
Peer review:
Three peer reviewers contributed to the peer review report. Reviewers’ reports totaled 829 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Research: CS; Contributed to the writing of the manuscript: CS and JMG; Jointly developed the structure and arguments for the paper: CS and JMG; Made a critical revisions and approved final version: CP. All authors reviewed and approved of the final manuscript.
