Abstract
Background:
Romosozumab is an anabolic agent approved for the treatment of severe osteoporosis in postmenopausal women and in men at high risk of fracture. However, real-world data on its effectiveness, particularly in patients with diabetes mellitus (DM) or prior exposure to antiresorptive therapy, remain limited.
Methods:
Adult patients (⩾18 years) who received romosozumab between January 2021 and May 2024 and had both baseline and post-treatment dual-energy X-ray absorptiometry (DEXA) scans were included. Bone mineral density (BMD) at the lumbar spine, total hip and femoral neck was assessed before and after 12 months of therapy. Subgroup analyses were undertaken according to diabetes status and previous antiresorptive therapy exposure.
Results:
Eighty-seven patients were included (mean age 66.7 ± 13.0 years; 94.3% female). The median percentage increase in lumbar spine BMD was 6.7% (IQR 1.3-12.6), while increases at the total hip and femoral neck were 2.9% (IQR −1.1 to 9.1) and 2.3% (IQR −3.9 to 9.7), respectively. Patients without diabetes demonstrated significantly greater BMD gains than those with diabetes at the lumbar spine (9.9% vs 3.1%; P = .020), total hip (4.1% vs 0.3%; P = .027), and femoral neck (3.9% vs 0.1%; P = .028). Similarly, treatment-naïve patients had greater improvements in total hip BMD compared with those with prior antiresorptive exposure (8.3% vs 2.2%; P = .004).
Conclusion:
Romosozumab significantly increased BMD at the lumbar spine, total hip and femoral neck after 12 months of treatment. The response was more pronounced in patients without diabetes and those who were treatment-naïve, suggesting that metabolic status and previous antiresorptive therapy may influence treatment effectiveness. Prospective studies are warranted to evaluate long-term fracture outcomes and the durability of these effects.
Plain Language Summary
Osteoporosis is a common condition characterised by reduced bone strength and an increased risk of fragility fractures. Romosozumab is approved for individuals at high risk of fracture. Although clinical trials have demonstrated its efficacy, there is comparatively limited real-world evidence, particularly in patients with diabetes mellitus or those previously treated with antiresorptive agents. In this retrospective study, we evaluated adults treated with romosozumab between 2021 and 2024 who had bone density assessments before treatment and after completing 12 months of therapy. Changes in bone density were examined at the lumbar spine, total hip and femoral neck. Overall, bone density increased following treatment, with the greatest gains observed at the lumbar spine and more modest improvements at the hip sites. However, treatment response varied between subgroups. Patients without diabetes showed greater improvements in bone density compared with those with diabetes. In addition, individuals who had not previously received antiresorptive therapy demonstrated larger gains at the hip than those with prior exposure. These findings support the effectiveness of romosozumab in routine clinical practice and suggest that metabolic factors and prior treatment history may influence the magnitude of response. Further prospective studies are required to determine whether these differences translate into variation in fracture outcomes and long-term benefit.
Keywords
Introduction
Osteoporosis is a common skeletal disorder with heterogeneous mechanisms depending on the underlying condition. In postmenopausal women, oestrogen deficiency primarily leads to increased osteoclast activity and accelerated bone resorption, resulting in a higher risk of fragility fractures despite preserved osteoblast function. 1
Diabetes-related bone disease is multifactorial and differs mechanistically according to diabetes type. Shared mechanisms include long standing hyperglycaemia with advanced glycation end-product (AGE) accumulation, oxidative stress and inflammation, osteocyte dysfunction and low-turnover remodelling state, together with the microvascular complications and increased falls risk.2 -4 In Type 1 diabetes, early absolute insulin deficiency impairs the attainment of peak bone mass and reduced the anabolic signalling including growth hormone/IGF-1 pathway. This subsequently leads to lower bone mineral density (BMD) and microarchitectural deficits that worsen with longer disease duration and poor glycaemic control.2,5 In type 2 diabetes, BMD is usually normal or increased; however, fracture risk remains elevated due to impaired bone quality (material properties and microstructure), low turnover with reduced repair of microdamage, adiposity-related marrow fat, inflammatory signalling, and medication effects (eg, thiazolidinediones).2,3,5 This may influence therapeutic response variability in this population. 6 Furthermore, patients with type 2 diabetes mellitus (T2DM) exhibit elevated circulating levels of sclerostin, a glycoprotein produced by osteocytes. 2 Sclerostin has a dual detrimental effect on bone metabolism: it inhibits the Wnt/β-catenin signalling pathway, reducing osteoblast activity and it promotes receptor activator of nuclear factor kappa-Β ligand (RANKL) expression, enhancing osteoclastogenesis and bone resorption.2,7
Romosozumab, a humanised monoclonal antibody against sclerostin, was approved in 2019 for the treatment of postmenopausal osteoporosis. 8 Unlike traditional anabolic therapies such as teriparatide, romosozumab exerts a dual mechanism of action by stimulating osteogenesis via Wnt/β-catenin activation while simultaneously reducing bone resorption through inhibition of RANKL-mediated osteoclast activity.8,9 Although these mechanisms suggest a potential benefit in patients with diabetes-related bone fragility, no randomised controlled trials (RCTs) have specifically evaluated romosozumab in diabetic populations to date. Evidence from post-hoc analyses and real-world studies suggest that antiresorptive and anabolic agents, included are bisphosphonates, denosumab, teriparatide and romosozumab, provide comparable improvements in bone mineral density (BMD) and fracture risk reduction in patients with and without diabetes mellitus.10,11 A recent meta-analysis demonstrated that osteoanabolic agents improve BMD in type 2 diabetes, particularly at the lumbar spine, although fracture data remain limited and underscore the need for diabetes-specific trials with fracture endpoints. 12 However, in the absence of dedicated clinical trials, a persisting knowledge gap regarding optimal osteoporosis management in patients with diabetes remains. The present study aims to describe a single-centre experience and evaluate the effects of romosozumab on bone mineral density, stratified by diabetes status and prior antiresorptive therapy exposure.
Methods
This retrospective observational cohort study was conducted at Tawam Hospital, Al Ain, United Arab Emirates. Ethical approval was obtained from the Tawam Human Research Ethics Committee (REC Approval No. MF2058-2024-1075), with a waiver of informed consent due to the retrospective nature of the study. All adult patients (⩾18 years) who received romosozumab between January 2021 and May 2024 were screened. In the United Arab Emirates, romosozumab is currently approved for the treatment of postmenopausal osteoporosis in women at high risk of fracture, in line with European and United States regulatory approvals. It is not formally approved for male osteoporosis or glucocorticoid-induced osteoporosis in the UAE; however, it can be prescribed off-label in selected high-risk cases based on clinical judgement. The decision to initiate romosozumab was based on patients meeting criteria for severe osteoporosis and/or being at very high fracture risk. This included individuals with severe osteoporosis (T-score ⩽ −2.5 in the presence of a fragility fracture or markedly low BMD such as T-score ⩽ −3.0), multiple vertebral fractures, or documented inadequate response, intolerance or contraindication to previous antiresorptive therapy. Ultimately, treatment decisions were individualised following a comprehensive assessment of fracture risk and the overall clinical context of each patient. A total of 132 patients were identified, of whom 87 had completed 12 months course of treatment and had both baseline (pre-treatment) and follow-up (12-month) dual-energy X-ray absorptiometry (DEXA) scans and were therefore included in the final analysis.
Romosozumab was administered at a dose of 210 mg once monthly (2 subcutaneous injections of 105 mg each). Treatment completion was verified through pharmacy dispensing records and electronic medical records (EMR) documentation, as all injections were administered within the hospital clinic. All patients received routine calcium and vitamin D supplementation in accordance with international guidelines. Bone mineral density (BMD) was measured using a GE Lunar dual-energy X-ray absorptiometry (DXA) system. The instrument underwent regular quality assurance and calibration in accordance with the manufacturer’s specifications, and the same device was used for both baseline and follow-up assessments to maintain measurement consistency. Degenerative changes of the lumbar spine were addressed in line with the International Society for Clinical Densitometry (ISCD) Official Positions. Vertebrae demonstrating significant degenerative changes, artefacts or structural abnormalities were excluded from analysis where appropriate, in accordance with established ISCD criteria. 13 BMD was reported in g/cm2, and T-scores were used for interpretation.
Demographic and clinical data, including age, sex, diabetes mellitus (DM) status and prior exposure to antiresorptive therapy (yes/no), were extracted from the electronic medical records (EMR). The duration of previous antiresorptive therapy were not consistently recorded and were therefore not collected. DM was defined according to a documented diagnosis and use of glucose-lowering medication. All patients in this study had Type 2 DM.
The outcome was the percentage change in BMD at the lumbar spine, total hip and femoral neck after 12 months of romosozumab therapy. Percentage change in BMD was calculated as the difference between 12-month and baseline BMD, divided by baseline BMD and expressed as a percentage. BMD responses were compared between patients with and without diabetes, and between those with and without prior antiresorptive therapy.
Statistical Analysis
Continuous variables were summarised as mean ± standard deviation (SD) for normally distributed data, or as median and interquartile range (IQR) for non-normally distributed data. Categorical variables were presented as frequencies and percentages. Between-group comparisons were performed using the student’s t-test for normally distributed continuous variables, the Mann–Whitney U test for non-normally distributed continuous variables and Fisher’s exact test for categorical variables. All statistical analyses were conducted using IBM SPSS Statistics, version 2024 (IBM Corp., Armonk, NY, USA).
Results
A total of 132 patients received romosozumab during the study period, of whom 87 had complete baseline and post-treatment dual-energy X-ray absorptiometry (DEXA) data and were included in the final analysis. The mean age of the cohort was 66.7 ± 13.0 years, and the majority were female (94.3%). Thirty-four patients (39.1%) had diabetes mellitus (DM). Compared with patients without diabetes, those with diabetes were significantly older (70.2 ± 8.4 vs 64.5 ± 14.9 years, P = .045) and had a higher body mass index (BMI; 31.1 ± 6.9 vs 26.1 ± 6.4 kg/m2, P = .001). Rheumatological disease was more frequent in the non-diabetic group (35.8% vs 11.8%, P = .037). The use of antidiabetic agents—particularly metformin (55.9%), SGLT2 inhibitors (26.5%), sulphonylureas (32.4%), and DPP-4 inhibitors (35.3%) was largely confined to the diabetic subgroup. Biological therapy use was higher among non-diabetic patients (26.4% vs 8.8%, P = .043). No significant between-group differences were observed in nationality, history of fractures or prior osteoporosis treatment (Table 1).
Baseline Characteristics of Patients Receiving Romosozumab, Stratified by Diabetes Status.

Abbreviations: BMI, Body Mass Index; SD, standard deviation; SLE, Systemic Lupus Erythematosus; DPP-4, Dipeptidyl Peptidase-4; GLP-1, Glucagon-Like Peptide-1; SGLT2, Sodium–Glucose Cotransporter-2.
After 12 months of romosozumab therapy (Table 2), significant improvements in bone mineral density were observed across all skeletal sites. The median percentage increase in lumbar spine BMD for the entire cohort was 6.7% (IQR 1.3-12.6), corresponding to a median absolute increase of 0.070 g/cm2, while total hip and femoral neck BMD increased by 2.9% (IQR −1.1 to 9.1) and 2.3% (IQR −3.9 to 9.7), representing median absolute gains of 0.039 and 0.044 g/cm2, respectively.
Changes in Bone Mineral Density and T-Scores After 12 Months of Romosozumab Treatment in Patients With and Without Diabetes.

Abbreviations: BMD, bone mineral density; LS, lumbar spine; SD, standard deviation; IQR, interquartile range; Δ, percentage change.
When stratified by diabetes status, patients without diabetes demonstrated significantly greater BMD gains than those with diabetes. The median percentage increase in lumbar spine BMD was 9.9% (IQR 3.3-13.6) in patients without diabetes compared with 3.1% (IQR −1.2 to 8.6) in patients with diabetes (P = .020), corresponding to median absolute increases of 0.083 and 0.044 g/cm2, respectively. Similarly, total hip and femoral neck BMD increased by 4.1% and 3.9% in patients without diabetes compared with 0.34% and 0.13% in those with diabetes (P = .027 and P = .028, respectively). Mean T-scores improved modestly at all sites but did not differ significantly between groups (Table 2, Figure 1).
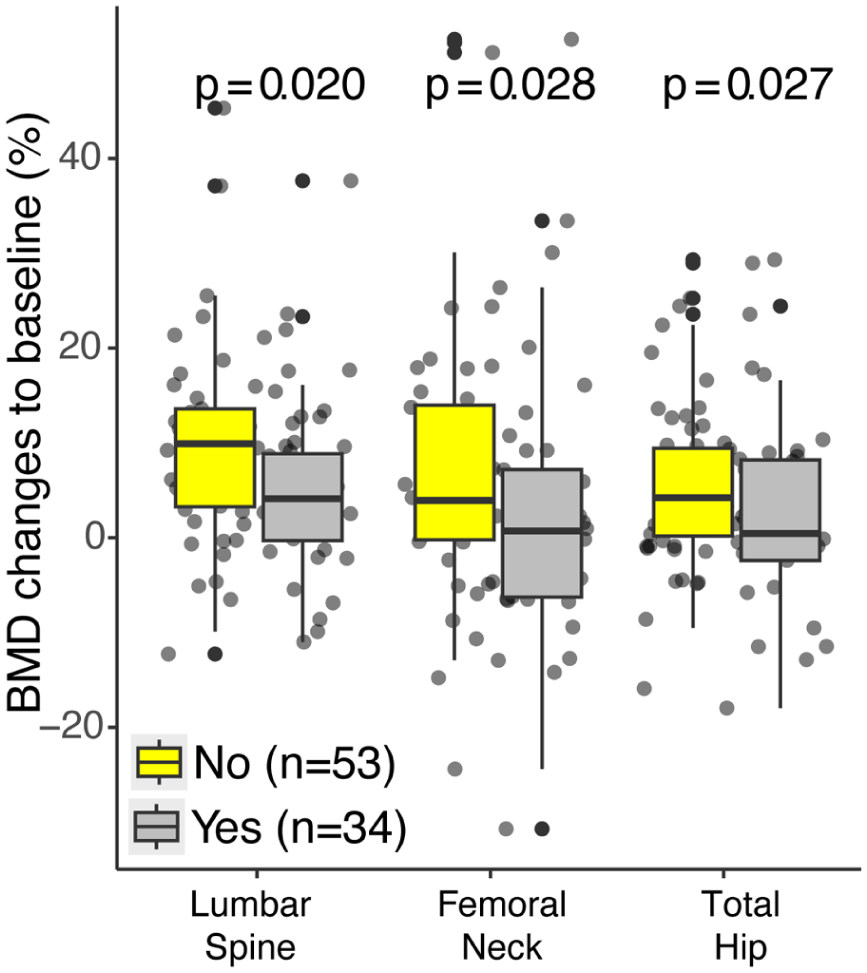
Percentage change in bone mineral density (BMD) at the lumbar spine, total hip and femoral neck after 12 months of romosozumab therapy in patients with and without diabetes mellitus (DM). Data are presented as median with interquartile range. Significant differences between groups were observed at all skeletal sites (P < .05).
Among the 83 patients with available data (Table 3), 32 were treatment-naïve and 51 had prior exposure to antiresorptive therapy (bisphosphonates or denosumab). Both groups experienced increases in BMD following 12 months of romosozumab therapy. Median lumbar spine BMD gains were 8.6% (IQR 0-12.1) in treatment-naïve patients and 6.7% (IQR 1.5-12.8) in previously treated patients (P = .820), corresponding to median absolute increases of 0.065 and 0.072 g/cm2, respectively.
Bone Mineral Density Changes After 12 months of Romosozumab in Patients With and Without Prior Antiresorptive Therapy.

Abbreviations: BMD, bone mineral density; LS, lumbar spine; SD, standard deviation; IQR, interquartile range; Δ, percentage change; g/cm2, grams per square centimetre.
Prior antiresorptive therapy includes bisphosphonates (oral or IV) or denosumab. Patients previously treated with teriparatide were excluded from this comparison.
Notably, treatment-naïve patients achieved significantly greater improvements in total hip BMD compared with those previously treated (median 8.3% vs 2.2%, P = .004). Differences in femoral neck BMD change did not reach statistical significance (6.5% vs 1.6%, P = .110). Overall, romosozumab produced clinically acceptable BMD gains across skeletal sites regardless of prior therapy status, although the magnitude of response was attenuated in previously treated individuals (Table 3, Figure 2).
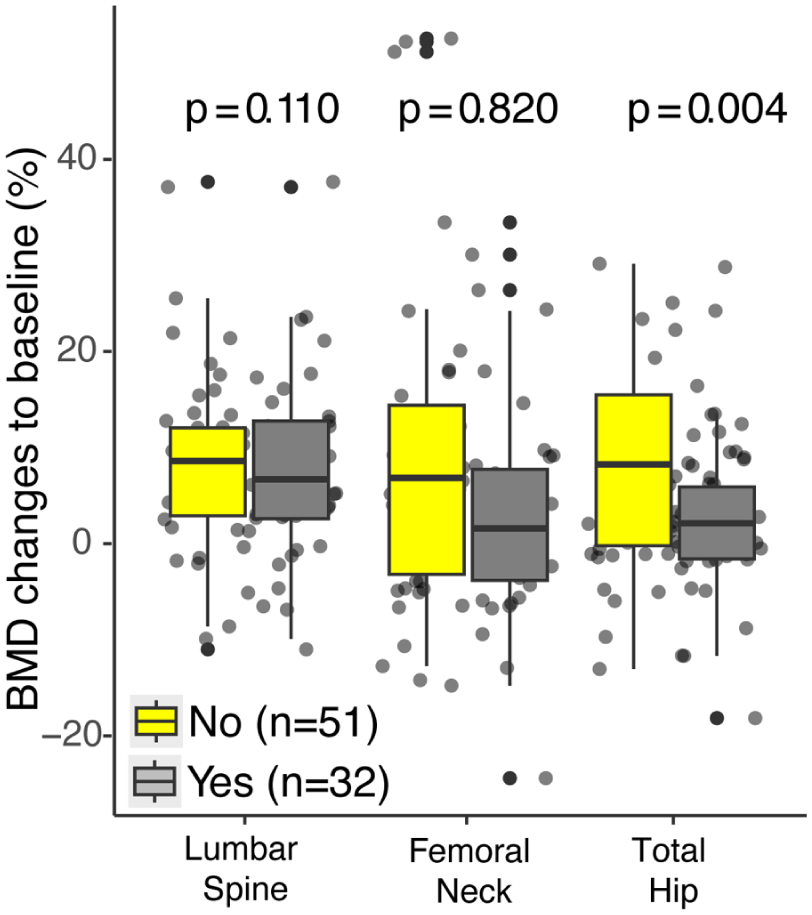
Percentage change in bone mineral density (BMD) at the lumbar spine, total hip and femoral neck after 12 months of romosozumab treatment in treatment-naïve patients compared with those previously treated with antiresorptive agents. Data are presented as median (interquartile range). A significantly greater increase in total hip BMD was observed in treatment-naïve patients compared with previously treated individuals (P = .004).
Discussion
In this retrospective study of 87 patients (62% Emirati and 94% women) treated with romosozumab for 12 months, statistically significant improvements in BMD were observed across the lumbar spine, total hip and femoral neck.
These findings reaffirm the dual anabolic and antiresorptive effects of romosozumab and add to growing evidence on differential treatment responses according to diabetes status and prior antiresorptive therapy in both Emirati and expatriate populations in the United Arab Emirates.
In the overall cohort (86% postmenopausal), the median percentage increase in lumbar spine BMD was 6.7%, while total hip and femoral neck BMD increased by 2.9% and 2.3%, respectively. Previous studies have reported lumbar spine BMD gains ranging from 10.3% to 14.65% and total hip gains from 3.1% to 6.9% after 12 months of romosozumab therapy.1,14 -16 Although slightly lower, our findings are broadly aligned with results from landmark phase III trials such as FRAME (Fracture Study in Postmenopausal Women with Osteoporosis) and ARCH (romosozumab or alendronate for Fracture Prevention in Women with Osteoporosis).1,14 The modest differences likely reflect real-world variability in age, comorbidities and prior antiresorptive therapy, clinical factors that are strictly controlled in most clinical trials. These improvements remain clinically consequential; however, clinicians should remain cautious when applying population-level data to individual patients, particularly considering the efficacy and safety profiles of anabolic therapies. 17 It is also important to note that not all anti-fracture agents exert uniform effects across skeletal sites, as demonstrated with drugs such as raloxifene and ibandronate. 18
The coexistence of DM and osteoporosis presents a complex clinical challenge due to their bidirectional relationship and increased fracture risk. Chronic hyperglycaemia is known to impair bone integrity through the accumulation of advanced glycation end-products (AGEs), which disrupt collagen cross-linking and bone mineralisation. 19 Certain antidiabetic medications may adversely affect bone, while others such as metformin and GLP-1 receptor agonists have protective skeletal effects. 20 In this study, patients with DM demonstrated significantly lower BMD gains than non-diabetic individuals: lumbar spine (3.1% vs 9.9%; P = .020), total hip (0.3% vs 4.1%; P = .027), and femoral neck (0.1% vs 3.9%; P = .028). These findings are consistent with established mechanisms of diabetic bone disease, including low bone turnover and increased AGE deposition.21,22 Despite blunted responses, romosozumab still produced measurable BMD improvements in the diabetic subgroup, suggesting efficacy even in metabolically compromised bone.19,23 Romosozumab has also been shown to improve bone microarchitecture and strength in patients with diabetes and diabetic kidney disease.23,24 Given the limited clinical data specific to diabetic populations, 23 these results contribute meaningful real-world evidence. They also support the need for individualised treatment strategies, closer cardiovascular monitoring and potentially longer treatment courses in patients with diabetes. 9
Prior antiresorptive therapy influenced treatment response in this cohort. Patients previously treated with antiresorptives exhibited smaller BMD gains than treatment-naïve individuals, particularly at the total hip (2.2% vs 8.3%; P = .004). A multicentre observational study reported lumbar spine and femoral neck BMD increases of 13.6% and 4.2%, respectively, in treatment-naïve patients after only 6 months of romosozumab, suggesting early bone turnover markers may predict treatment response. 25 Although treatment sequencing was not evaluated in this study, existing clinical evidence supports initiating therapy with anabolic agents followed by antiresorptives, as this approach yields superior BMD gains and fracture risk reduction compared with the reverse sequence.9,16,26,27 Additionally, longer duration of prior antiresorptive therapy has been shown to correlate negatively with subsequent BMD gains in both the lumbar spine and hip. 16
Romosozumab has demonstrated robust anti-fracture efficacy, including a 73% reduction in new vertebral fractures compared with placebo 1 and a 48% reduction compared with alendronate after 12 months of therapy. 14 Meta-analyses indicate that a 2% to 6% increase in total hip BMD corresponds to a 16% to 40% reduction in hip fracture risk. 28 Although fracture outcomes were not assessed in this study, the observed BMD improvements are likely to translate into meaningful reductions in fracture risk. This reinforces that DEXA-derived BMD change remains a valid surrogate marker for fracture risk reduction in the absence of clinical fracture data. 17 However, it has to be noted that although BMD is an established surrogate marker of treatment response, it does not fully explain fracture risk reduction. Improvements in bone quality, including trabecular microarchitecture and TBS, may be particularly relevant in diabetes, where fracture risk is often disproportionate to measured BMD. 6
Strengths and Limitations
This study provides real-world data on the use of romosozumab in a Middle Eastern population, with complete 12-month follow-up and verified treatment administration through hospital-based dispensing. However, several limitations should be acknowledged. As this was a retrospective study including all eligible patients treated during the study period, no formal sample size calculation was performed, which may have limited the statistical power, particularly for subgroup analyses. Additionally, there was no fracture outcomes or bone turnover markers in this study. Residual confounding related to the severity and duration of diabetes, degree of glycaemic control and the presence of diabetes-related complications may have influenced our findings, as these variables were not collected in the dataset for analysis. The duration of prior antiresorptive therapy was not recorded, which may have influenced treatment response. Furthermore, some patients with diabetes had previously received antiresorptive therapy before commencing romosozumab, introducing potential confounding.
Conclusion
This study demonstrates that 12 months of romosozumab therapy results in significant increases in BMD at the lumbar spine, total hip and femoral neck. Although patients with diabetes mellitus and those with previous exposure to antiresorptive therapy showed a blunted response compared with treatment-naïve individuals, the gains remained clinically meaningful. These findings support an individualised approach to management, particularly in patients with diabetes and reinforce the role of initiating anabolic therapy, such as romosozumab, in individuals at high fracture risk. Prospective studies are required to determine the durability of these BMD improvements, their translation into fracture risk reduction and the long-term cardiometabolic safety of romosozumab, particularly in diverse populations and those with metabolic comorbidities.
Footnotes
Acknowledgements
The authors used OpenAI’s ChatGPT exclusively for English language refinement. All scientific content, analysis and conclusions were developed independently by the authors.
Ethical Considerations
The study was approved by the Tawam Human Research Ethics Committee (MF2058-2024-1075).
Consent to Participate
The requirement for informed consent was waived due to the retrospective study design, anonymised data collection and absence of patient interventions.
Consent for publication
Not applicable.
Author Contributions
Hind Alshamsi: Conceptualizsation; Data curation; Resources; Writing – original draft. Duha Alnaqbi: Conceptualisation; Data curation; Writing – original draft. Charu Sharma: Formal analysis; Methodology; Writing – original draft. Romona D Govender: Investigation; Methodology; Writing – review & editing. Raya Almazrouei: Conceptualisation; Investigation; Methodology; Supervision; Writing – review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Due to confidentiality considerations, the original patient data cannot be shared. All relevant findings are fully presented within this manuscript.
