Abstract
Background:
Type 1 diabetes (T1D) is an autoimmune disease characterized by insulin deficiency and impaired glucose regulation. While sex hormones are known to influence insulin sensitivity, their specific roles in T1D remain underexplored. Prolactin and progesterone have been associated with glucose metabolism, yet their influence in individuals with T1D, particularly men, has not been well studied. This study investigated the relationship between key sex hormones and glycemic control in men and women with T1D.
Methods:
Seventy-eight adults with T1D (25 men, 53 women) were recruited for a cross-sectional study examining associations between circulating sex hormone concentrations and hemoglobin A1c (HbA1c). Participants underwent anthropometric and body composition assessments and provided fasting blood samples for measurement of estradiol, progesterone, prolactin, total testosterone, free testosterone, and HbA1c. Pearson’s correlations were used to evaluate associations between hormone levels and HbA1c.
Results:
In men, higher concentrations of prolactin (r = −.571, P = .003) and progesterone (r = −.434, P = .030) were significantly associated with lower HbA1c. No such associations were observed in women for prolactin, progesterone, estradiol, or testosterone. Similarly, in men, estradiol, total testosterone, and free testosterone were not significantly correlated with HbA1c.
Conclusions:
This study provides novel evidence that prolactin and progesterone may be linked to improved glycemic control in men with T1D. These associations were not observed in women. The findings underscore the importance of sex-specific approaches in endocrine and metabolic research. Further longitudinal and mechanistic studies are needed to confirm these relationships and explore the potential of prolactin and progesterone as therapeutic targets in T1D management.
Plain Language Summary
Type 1 diabetes (T1D) is a lifelong condition where the body can’t produce insulin, a hormone that helps control blood sugar. Managing blood sugar is important to prevent long-term health problems. While we know that sex hormones like estrogen and testosterone affect how the body handles sugar, their role in people with T1D, especially men, hasn’t been well understood. In this study, we looked at whether certain hormones are linked to blood sugar control in adults with T1D. We measured sex-hormones and a marker of long-term blood sugar control called hemoglobin A1c (HbA1c) in 78 adults (25 men and 53 women). We found that in men, higher prolactin and progesterone were linked to better blood sugar control (lower HbA1c). These relationships were not seen in women. Other hormones like estrogen and testosterone didn’t show any clear link to blood sugar control in either group. This study is the first to show that prolactin and progesterone might play a helpful role in blood sugar management for men with T1D. We didn’t see the same pattern in women. These findings suggest that hormones beyond insulin may influence blood sugar, and that these effects can differ between men and women. Understanding these differences could lead to more personalized treatment strategies for people with T1D. More research is needed to explore why these hormones seem to help and whether they could be used in future therapies.
Background
Type 1 diabetes (T1D) is an autoimmune condition characterized by the destruction of insulin-producing beta cells in the pancreas, resulting in lifelong insulin dependency and impaired glucose regulation. 1 Currently 1.25 million individuals in America are impacted by T1D, with 8.7 million people affected globally. Despite advancements in insulin therapy, the pathophysiology of T1D remains complex, with ongoing research investigating various factors that may contribute to disease progression and complications.
The role of sex hormones in glucose metabolism is well established, with estradiol, progesterone, testosterone, and prolactin each influencing insulin sensitivity in distinct ways. Estradiol and progesterone are predominately referred to as female sex hormones. Estradiol generally enhances insulin sensitivity and promotes glucose uptake into cells, 2 while progesterone has the opposite effect of increasing insulin resistance and potentially raising blood glucose levels, particularly in individuals with underlying metabolic dysfunction. 3 Testosterone, a hormone typically associated with males, plays a key role in glucose regulation, in a sex-dependent manner. In men, normal concentrations of testosterone enhance insulin sensitivity, whereas low testosterone is linked to increased insulin resistance and a higher risk of type 2 diabetes. 4 In addition, high concentrations of testosterone in men can disrupt glucose regulation and contribute to an increased the risk of metabolic disorders. 5 In women, although testosterone is present in lower concentrations, elevated concentrations of testosterone is associated with insulin resistance, contributing to higher circulating glucose and an increased risk of metabolic syndrome. 6 Thus, maintaining homeostasis in concentrations of testosterone appears to be crucial for supporting healthy glucose metabolism in both men and women.
Prolactin is primarily known for its role in lactation; however, it also affects glucose metabolism. Elevated concentrations of prolactin have been associated with increased insulin resistance, impaired glucose uptake, and higher circulating glucose concentrations.7 -9 Additionally, prolactin can increase insulin secretion while simultaneously promoting hepatic glucose production, reducing skeletal muscle glucose uptake, and encouraging fat accumulation, all of which contribute to insulin resistance. 10 Its effects on hypothalamic pathways may further influence appetite regulation and energy expenditure, disrupting glucose homeostasis, especially during pregnancy and lactation, periods of naturally elevated concentrations of prolactin. 11 Indeed, chronic elevation of prolactin can therefore impair normal glucose regulation and increase the risk of developing metabolic disorders. 12 In fact, emerging evidence suggests that prolactin may play a role in the pathophysiology of T1D. Evidence indicates that prolactin modulates immune responses, potentially contributing to the autoimmune destruction of beta cells. 13 In addition, studies utilizing male mouse models of T1D demonstrate that elevated prolactin exacerbates insulin resistance and impairs the capacity of insulin secretion.9,12 In addition, prolactin may influence immune cell function, beta cell regeneration, and insulin sensitivity, all processes that are relevant to T1D progression and disease severity. 14 Despite growing interest of the role of prolactin in the pathogenesis of T1D, data documenting the role of prolactin on dysglycemia in humans is limited and poorly understood. Thus, the present study aims to explore the relationship between various sex hormones and glycemic control in individuals with T1D. By investigating these hormonal influences, this research may provide insights into the metabolic factors contributing to T1D progression and potential therapeutic targets.
Methods
Experimental Design
All participants attended a study visit at the Laboratory of Integrative Vascular and Exercise Physiology (LIVEP) within the Georgia Prevention Institute at Augusta University. After providing informed consent, they underwent anthropometric and body composition assessments. Height and weight were measured using a stadiometer and a calibrated platform scale (CN20, DETECTO, Webb City, MO) to calculate body mass index (BMI), and total body fat was quantified by dual-energy X-ray absorptiometry (QDR-4500W; Hologic, Waltham, MA). Participants arrived in the morning after an overnight fast and abstained for at least 12 hours from caffeine, tobacco products, and moderate-to-vigorous physical activity. Individuals with type 1 diabetes were instructed to continue their usual basal insulin regimen during the visit.
Participant Characteristics
A total of 25 men and 53 women with clinically diagnosed type 1 diabetes were enrolled from the Augusta University Department of Endocrinology and through community word-of-mouth recruitment. Individuals were excluded if they had a history of hepatic, renal, or cardiovascular disease; uncontrolled hypertension (systolic or diastolic blood pressure >140/90 mm Hg); proteinuria; HbA1c greater than 12%; or any vascular complications of diabetes. To minimize hormonal variability, all female participants were evaluated during the menses phase of their menstrual cycle. Study procedures were approved by the Augusta University Institutional Review Board (IRB #989225; approval date: February 15, 2017) and registered at ClinicalTrials.gov (NCT03436992). Participant enrollment and data collection were completed over a 5-year period.
Clinical Laboratory Values
A venous blood sample was collected to assess glycated hemoglobin (HbA1c) (%) and circulating concentrations of various sex hormones including estradiol (pg/mL), prolactin (ng/mL), progesterone (ng/mL), total testosterone (ng/dL), and free testosterone (pg/mL) (Laboratory Corporation of America Holdings, Birmingham, AL).
Statistical Analysis
Statistical analyses were performed using SPSS 29.0. All data are expressed as mean ± standard error of mean (SEM) unless otherwise noted. For demographic and baseline clinical variables, independent t-tests were used to examine differences. Pearson’s correlations were performed to evaluate the relationship between hormones and HbA1c. Linear regression analysis was performed to evaluate the associations between hormone concentrations and HbA1c while adjusting for potential confounding variables. Statistical significance was set at P < .05.
Results
Participant Characteristics
Demographics and clinical laboratory values are presented in Table 1. Women had a higher percent body fat than men (P < .001). Men had higher total testosterone (P < .001) and free testosterone (P < .001) than women.
Overall Participant Characteristics.

Abbreviations: BMI, Body Mass Index, HbA1c, Hemoglobin A1c.
Data are presented as mean ± SD; independent samples t test. Bold values indicate statistically significant.
Relationship Between HbA1c and Sex Hormones in Men With T1D
The relationship between HbA1c and prolactin and progesterone in men are illustrated in Figure 1. There was a negative relationship between HbA1c and prolactin (Figure 1A; r = −.571; P = .003) and progesterone (Figure 1B; r = −.434; P = .030). These relationships persisted after controlling for BMI, weight, and body fat percentage (all P < .05). There was no relationship between HbA1c and estradiol (r = .033; P = .876), total testosterone (r = −.188; P = .368) or free testosterone (r = −.188; P = .379) in men. There was no relationship between HbA1c and duration of T1D (r = .079; P = .703) or insulin concentration (r = .113; P = .590).
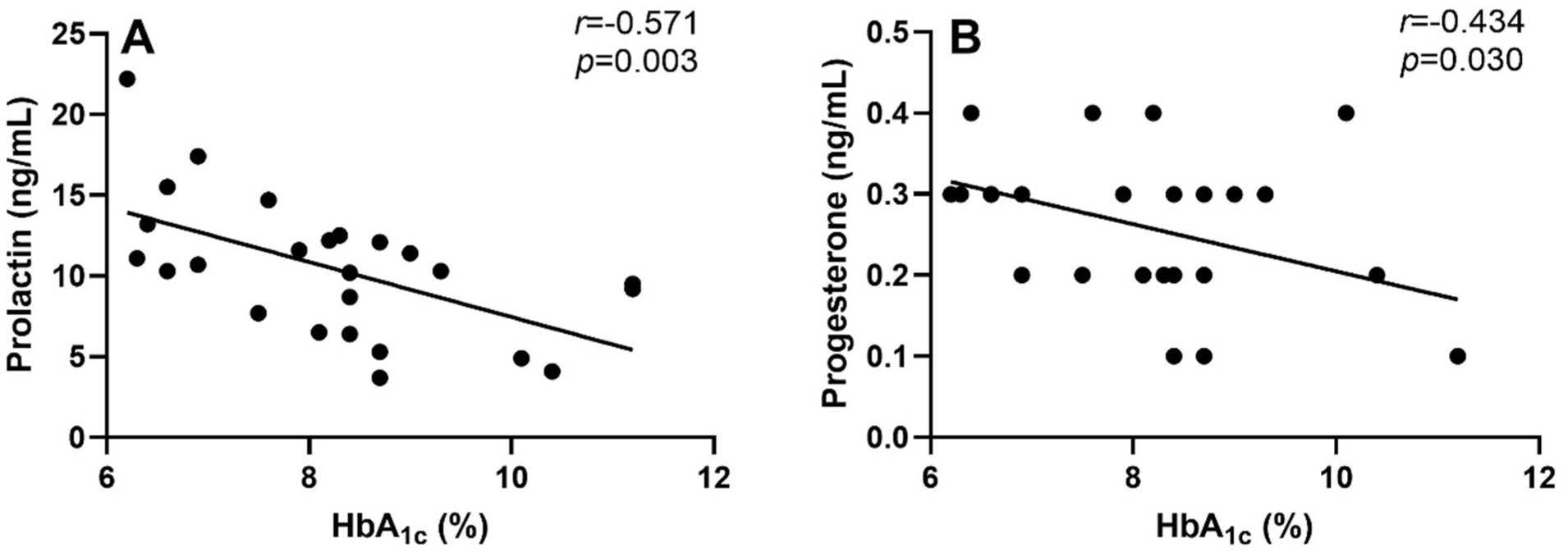
Relationship between hemoglobin A1c (HbA1c) and (A) prolactin and (B) progesterone in men with type 1 diabetes; n = 25. Pearson’s Correlation. Data are presented as mean ± SEM.
Relationship Between HbA1c and Sex Hormones in Women With T1D
The relationship between HbA1c and prolactin and progesterone are illustrated in Figure 2. There was no relationship between HbA1c and prolactin (Figure 2A; r = −.175; P = .211), progesterone (Figure 2B; r = −.234; P = .092), estradiol (r = −.023; P = .871), total testosterone (r = −.140; P = .318), or free testosterone (r = −.100; P = .475). There was no relationship between HbA1c and duration of T1D (r = .118; P = .368) or insulin concentration (r = −.168; P = .229).

Relationship between hemoglobin A1c (HbA1c) and (A) prolactin and (B) progesterone in women with type 1 diabetes; n = 53. Pearson’s Correlation. Data are presented as mean ± SEM.
Discussion
Sex hormones are increasingly recognized as critical modulators of metabolic health, with emerging evidence suggesting sex-specific effects of different hormones on glycemic regulation. However, little is known about how these hormones influence glucose metabolism in individuals with T1D, a population characterized by the absence of endogenous insulin production. The present study contributes novel findings by demonstrating that in men with T1D, higher concentrations of prolactin and progesterone are associated with a lower HbA1c, an association that was not observed in women. These sex-specific relationships were unexpected and warrant further investigation, as prior research has generally emphasized the metabolic effects of these hormones in women or in broader populations without stratifying by sex or T1D status.
Prolactin and Glycemic Status
Prolactin, traditionally associated with lactation, is regulated through a dopaminergic feedback loop, whereby dopamine inhibits prolactin release. 15 However, the metabolic actions of prolactin appear bidirectional. Chronic hyperprolactinemia can reduce insulin sensitivity through peripheral mechanisms and via secondary hypogonadism, 16 whereas very low prolactin may fail to adequately stimulate prolactin receptor (PRLR) signaling in β-cells, impairing insulin secretion and islet trophism. 17 This duality aligns with the concept of a “Homeo-Fit prolactin window,” in which modest, transient increases in prolactin are associated with improved glycemic regulation. 18
In the current study, an inverse relationship was observed between concentrations of prolactin and HbA1c in men with T1D. This aligns with studies showing that low concentrations of prolactin have been linked to increased risk of diabetes, particularly in men. 19 Indeed, others show that prolactin is associated with improved insulin sensitivity and favorable glycemic profiles in individuals with type 2 diabetes. 20
Elevated prolactin levels have been shown to impair glucose uptake in skeletal muscle, increase hepatic glucose production, and promote adipose tissue dysfunction, while also influencing hypothalamic appetite regulation and energy expenditure. 21 Importantly, the concentrations of prolactin of the participants in the current study fell within clinically normal limits, supporting the idea that subtle, within-range variation can favorably influence glycemia. Even after controlling for covariates, the inverse association between prolactin and HbA1c in men remained significant, supporting a robust observed relationship between prolactin and glycemic control.
These findings are preliminary and should be interpreted with caution given the cross-sectional nature of the analysis. Nonetheless, they raise important hypotheses about prolactin’s potential as a biomarker, or even therapeutic target, for glycemic control in men with T1D. Future studies should evaluate whether prolactin concentrations track with glycemic fluctuations over time, respond to insulin dosing, or influence diabetes-related complications. Longitudinal and mechanistic studies, including dopaminergic pathway analyses, could further illuminate the causal direction and biological underpinnings of these associations.
While dopamine was not measured here, the inverse physiological relationship between prolactin and dopamine suggests a possible indirect pathway whereby prolactin-driven dopaminergic changes could influence insulin sensitivity. If validated in larger and more diverse cohorts, these associations could prompt a re-evaluation of how hormonal regulation is incorporated into diabetes care models for men.
In contrast, no association was found between prolactin and glycemic control in women with T1D. This absence may reflect the hormonal complexity in women, particularly the cyclical and dynamic fluctuations of sex steroids such as estrogen and progesterone. These may mask or override the metabolic effects of prolactin or introduce greater interindividual variability that limits detectable associations. Future studies stratified by menstrual phase, hormonal contraceptive use, or menopausal status will be critical for elucidating whether prolactin plays a more nuanced role in women’s glucose regulation. Taken together, these preliminary findings support a “Homeo-Fit prolactin window” framework and raise the possibility that prolactin may act as a novel, sex-specific hormonal modulator of glycemic control in T1D.
Progesterone and Glycemic Status
Though commonly considered a female hormone, progesterone also plays important roles in men, including testosterone synthesis and estrogen balance. 22 Prior studies have shown that progesterone can impair glucose regulation by reducing insulin sensitivity in adipose tissue and skeletal muscle, leading to increased concentrations of blood glucose. 3 Additionally, data from animal models demonstrate that progesterone can impair insulin signaling and increase hepatic glucose production in the context of diabetes.23,24 However, in the present investigation, higher progesterone concentrations were associated with lower HbA1c in men with T1D, a novel and counterintuitive finding. This suggests that in the context of exogenous insulin use, progesterone may influence glucose metabolism via alternative, possibly insulin-independent mechanisms. For instance, while male and female progesterone receptor knockout animal models suggest improved glucose tolerance via increased beta-cell proliferation, 25 other studies have demonstrated that progesterone inhibits glucose oxidation in isolated adipocytes. 26 In support, the present findings identified a significant inverse association between progesterone and HbA1c in men, even when controlling for weight, BMI, and body fat percentage, indicating that this relationship may be independent of these common metabolic confounding variables. These preliminary results warrant further investigation into whether progesterone modulates insulin action or glucose uptake in unique ways in men with T1D. Future studies using longitudinal or mechanistic designs are needed to determine causality and assess the therapeutic relevance of this association.
In women, no association was observed between progesterone and HbA1c. This may reflect hormonal variability due to the menstrual cycle, contraceptive use, or premenopausal status, which could obscure metabolic effects in cross-sectional analysis. Future research stratified by cycle phase and hormonal status may help clarify progesterone’s role in female glucose regulation. Together, these findings introduce a potentially novel hormonal pathway relevant to glycemic control in T1D, particularly in men, and lay the groundwork for future studies to build on this early signal.
Other Sex Hormones and Glycemic Status
Estrogen and testosterone are widely studied in relation to metabolic outcomes. Estrogen has been linked to increased cardiovascular risk in women with T1D, 27 while testosterone is associated with improved insulin sensitivity in men. 28 Indeed, the relationship between testosterone and dysglycemia is also non-linear. Both hypogonadism and supraphysiologic concentrations of androgens can promote insulin resistance and metabolic dysfunction through different pathways, supporting how low testosterone may produce effects reminiscent of hyperandrogenism. 29 Lower concentrations of free testosterone are commonly observed in men with T1D and may contribute to metabolic dysfunction 30 ; however, in the present study, no association was observed between HbA1c and concentrations of estradiol, total testosterone, or free testosterone in either sex. The absence of associations may indicate that prolactin and progesterone play a more prominent role in glycemic regulation in the context of T1D. Alternatively, these null findings may reflect smaller effect sizes, hormone-binding variations, or the influence of other unmeasured metabolic factors. These results highlight the importance of continuing to explore nontraditional hormonal influences on glucose metabolism in this population.
Implications and Future Directions
The findings from the current investigation are clinically relevant for several reasons. First, these findings suggest that concentrations of circulating prolactin and progesterone could represent potential metabolic modulators in men with T1D; however, this interpretation is hypothesis-generating and based on cross-sectional correlations. Confirmation will require longitudinal and mechanistic studies. Second, the lack of associations between sex-hormones and HbA1c in women, where hormonal interactions are inherently more complex, highlights the need for more granular, sex-specific, and phase-specific endocrine research. Finally, these results challenge the conventional dogma about the uniform effects of sex hormones across different populations (ie, healthy to diseased). Specifically, the role of sex hormones in T1D may differ substantially from those in the general population or even in individuals with type 2 diabetes, which most of the research has been conducted in thus far. While the present findings suggest that prolactin and progesterone may influence glycemic control, the underlying biological mechanisms remain speculative. In addition, although the present investigation utilized a cross-sectional experimental design, the observed associations cannot establish causality. Future studies employing longitudinal and interventional designs in a larger cohort are needed to confirm the present findings, clarify the specific molecular pathways involved, and determine the overall clinical relevance.
Conclusion
Findings from the present investigation document that higher concentrations of prolactin and progesterone are associated with lower HbA1c in men with T1D. These associations were not observed in women, possibly due to the complex and fluctuating hormonal landscape in female physiology. These findings underscore the need for future mechanistic and longitudinal studies to explore the hormonal modulation of glucose metabolism in sex-specific contexts and to assess the therapeutic potential of targeting prolactin and progesterone pathways in T1D management.
Footnotes
Acknowledgements
The authors would like to thank the participants for their time.
Ethical Considerations
All study protocols were approved by the Institutional Review Board at Augusta University.
Consent to Participate
All participants gave written consent to participate.
Author Contributions
Abigayle B. Simon: Conceptualization; Data curation; Formal analysis; Writing—original draft; Writing—review & editing. Dhruti Trivedi: Formal analysis; Writing—original draft; Writing—review & editing. Jeffrey Thomas: Data curation; Formal analysis; Writing—review & editing. Matthew S. Nicholson: Data curation; Methodology; Writing—review & editing. Ryan A. Harris: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by 1R01HL137087 (RAH).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used during the current study are available from the corresponding author on reasonable request.
