Abstract
Background:
Wilson’s disease (WD) is a rare genetic disorder that impairs copper metabolism, leading to its deposition in various organs, including the liver, brain, and cornea. Endocrine disorders, particularly hyperparathyroidism, are uncommon in WD. Methanol toxicity, a medical emergency, is rarely associated with WD and hyperparathyroidism, making this case particularly unique. We report a rare instance of this complex triad.
Case Presentation:
A 53-year-old male with untreated WD presented with nausea, vomiting, dizziness, and blurred vision after ingesting methanol. Clinical examination revealed optic neuropathy, consistent with methanol toxicity, despite normal fundoscopy. Laboratory investigations revealed significant hypercalcemia and elevated parathyroid hormone (PTH) levels, confirming hyperparathyroidism. Imaging, including a prior technetium-99m sestamibi scan, indicated hyperfunctioning parathyroid tissue. Dialysis was initiated for methanol toxicity, and metabolic acidosis was corrected.
Conclusion:
This case emphasizes the rare coexistence of WD, hyperparathyroidism, and methanol toxicity, presenting significant diagnostic and therapeutic challenges. The pathophysiological interactions between these conditions are not well understood and warrant further research to improve management strategies and clinical outcomes.
Plain Language Summary
Wilson’s Disease is a rare condition where the body cannot properly remove excess copper, leading to liver and brain problems. High calcium levels due to hyperparathyroidism can cause kidney stones and bone issues. Methanol poisoning, often from contaminated alcohol, is dangerous because it can lead to blindness and organ failure. This case report describes a 53-year-old man who was diagnosed with all three conditions at the same time. He had Wilson’s Disease but had never received treatment. After drinking methanol, he developed nausea, vomiting, and vision problems. While his eye exam appeared normal, doctors diagnosed optic nerve damage, a common complication of methanol poisoning. Blood tests showed high calcium and parathyroid hormone levels, leading to a diagnosis of hyperparathyroidism. We used a multidisciplinary approach to manage his condition, including dialysis to remove toxins and treatments for metabolic imbalances. This case is unique because these three conditions are rarely seen together. Understanding their interactions may help doctors better diagnose and treat patients with similar complex cases in the future.
Background
Wilson’s disease (WD), also known as hepatolenticular degeneration, is a rare autosomal recessive disorder that impairs copper metabolism by affecting the ATPase Copper Transporting Beta gene. Estimated prevalence is between 1:30 000 and 1:50 000 with a broad perspective of hepatic, and neuropsychiatric manifestations.1,2
Copper ion secretion to the blood can lead to its deposition in the brain, kidney, cornea and other organs as well. 3 The diagnosis requires a combination of clinical signs and symptoms, along with serum ceruloplasmin measurement, urinary copper excretion, liver biopsy, or molecular testing. WD is also characterized by low levels of blood copper and ceruloplasmin and increased urinary copper excretion and high levels of copper in the liver.2,4
Endocrine disorders are rarely associated with WD. Hypoparathyroidism secondary to WD results from parathyroid gland atrophy due to copper deposition. In addition, Chronic copper deposition may lead to parathyroid dysfunction, with hypoparathyroidism due to glandular atrophy or, rarely, hyperplasia, potentially from compensatory mechanisms.5 -8
Methanol toxicity, caused by ingestion or exposure to methanol-containing substances, is a well-known medical emergency that can lead to severe complications, including metabolic acidosis, visual disturbances, and central nervous system dysfunction. However, the association between methanol toxicity and the simultaneous occurrence of hyperparathyroidism and WD is extremely rare and has not been extensively reported in the medical literature.
In this study, we report a rare case of methanol toxicity in a 53-year-old male with untreated WD and concurrent hyperparathyroidism. The case underscores the unusual coexistence of these conditions, potentially linked to parathyroid hyperplasia from chronic copper deposition, posing significant diagnostic and management challenges. The 4-year treatment gap reflects real-world challenges in WD management. As in Tallis et al’s case, 9 socioeconomic barriers, particularly chelation therapy costs and asymptomatic early-stage disease often delay intervention, 10 increasing complication risks.
Case Presentation
In September 2022, a 53-year-old male was admitted to our medical department presenting with nausea, vomiting, lethargy, loss of appetite, dizziness, and blurred vision, which he described as black and white. These symptoms developed approximately 4 hours following the accidental ingestion of methanol-containing solvent. The patient reported vomiting 3 times but denied any episodes of diarrhea, abdominal tenderness, cough, or shortness of breath.
The patient’s medical history was notable for fatty liver disease, depression, and a diagnosis of WD 4 years prior. He was non-adherent to chelation therapy due to socioeconomic constraints, despite the confirmed diagnosis. His medication regimen included sertraline 150 mg, propranolol 10 mg, and clonazepam 1 mg daily. His family history was significant for Parkinson’s disease in his mother and diabetes mellitus in his siblings.
Physical examination revealed bilateral mid-sized pupils non-reactive to light, though visual acuity remained unimpaired and fundoscopy was normal. Despite normal fundoscopy, further ophthalmologic assessment revealed optic neuropathy, consistent with methanol toxicity, and a poor visual prognosis (Figure 1).

Fundoscopic examination revealing optic neuropathy in a 53-year-old male with methanol toxicity and untreated WD.
During his hospital stay, the patient experienced multiple episodes of hematemesis. An esophagogastroduodenoscopy revealed a mucosal tear at the lower esophageal sphincter suggestive of a Mallory-Weiss tear, alongside antral gastritis (Figure 2).

Esophagogastroduodenoscopy revealed a mucosal tear at the lower esophageal sphincter suggestive of a Mallory-Weiss tear in our patient.
Laboratory investigations at the time of admission showed significant hypercalcemia, prompting a suspicion of hyperparathyroidism. Table 1. Persistent hypercalcemia and elevated PTH levels (197.1 pg/mL) predating acute renal dysfunction by 4 years, alongside prior technetium-99 m sestamibi scan confirmation of parathyroid adenoma, supported primary hyperparathyroidism. A review of prior lab and imaging results from 5 years earlier revealed a total serum calcium of 11.8 mg/dL, elevated alkaline phosphatase (555 U/L), aspartate aminotransferase (127 IU/L), and alanine aminotransferase (145 IU/L), which had initially gone uninvestigated. Further endocrine evaluation identified a PTH level of 197.1 pg/mL 4 years prior, suggesting primary hyperparathyroidism. Also, a parathyroid technetium-99m sestamibi scan confirmed hyperfunctioning parathyroid tissue beneath the left thyroid lobe. Figure 3.
Laboratory Results at Admission.

Abbreviations: ALP, alkaline phosphatase; ALT, alanine amino transferase; AST, aspartate amino transferase; CRP, C reactive protein; INR, International Normalized Ratio; LDH, lactate dehydrogenase; PCO2, partial pressure of carbon dioxide; pH, potential of hydrogen; PO2, partial pressure of oxygen; RDW-CV, red cell distribution width - coefficient of variation; TSH, thyroid stimulating hormone.

Technetium-99m sestamibi scan showing persistent radiotracer uptake beneath the left thyroid lobe, suggestive of hyperfunctioning parathyroid tissue from 5 years prior. (A) 15 minutes. (B) 180 minutes.
Pathology results from 5 years earlier further indicated elevated serum glutamic-oxaloacetic transaminase and serum glutamic-pyruvic transaminase levels, measuring 53 and 81 U/L, respectively. The patient also exhibited decreased ceruloplasmin, increased urinary copper excretion, and the presence of a Kayser-Fleischer ring, consistent with WD. A liver needle biopsy at that time revealed mild portal and lobular inflammation, graded as 3 with a stage of 1.
Additionally, investigations for WD revealed a 24-hour urinary copper excretion of 350 µg (reference range: 15-70 µg), a ceruloplasmin level of 38 mg/dL (reference range: 15-60 mg/dL), and a blood copper level of 122 µg/dL (reference range: 70-150 µg/dL). These findings were consistent with untreated WD.
A non-contrast brain MRI showed intra-axial high signal intensity foci in the right frontal lobe (20 × 12 mm) and bilateral occipital lobes (right: 16 and 9 mm; left: 18 mm) on T2 fluid-attenuated inversion recovery, with corresponding signal voids on Apparent Diffusion Coefficient maps, suggestive of acute ischemic infarctions. The MRI also noted a 23 mm retention cyst in the right maxillary sinus. An orbital MRI was unremarkable.
Given the patient’s acute renal dysfunction and hypercalcemia, dialysis was initiated. Despite these interventions, the patient developed leukocytosis and was empirically started on vancomycin (1 g IV every 4 hours).
The concurrence of methanol toxicity, WD, and primary hyperparathyroidism in this patient presents a highly complex and rare clinical scenario, demanding careful consideration in management and treatment strategies.
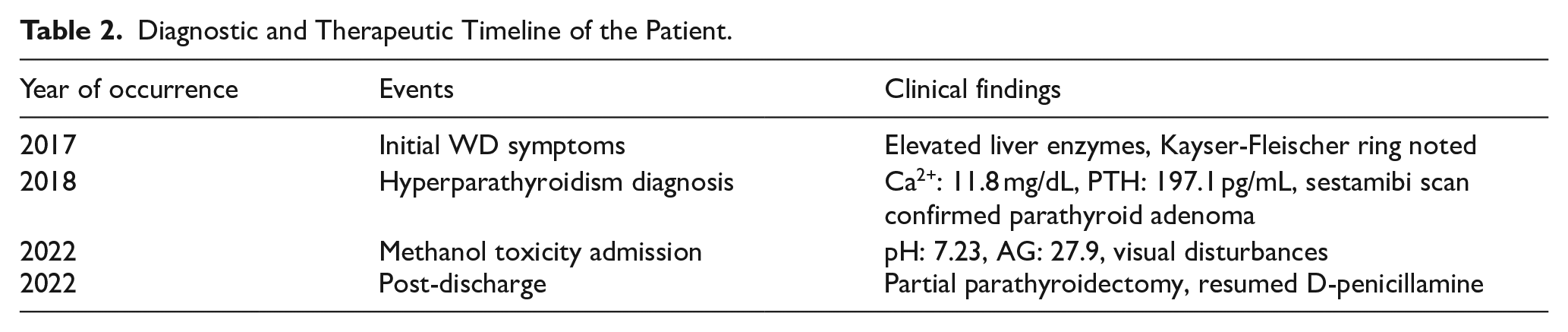
Following discharge, the patient was scheduled for regular multidisciplinary follow-up. Despite initial non-adherence, he resumed chelation therapy with D-penicillamine and underwent partial parathyroidectomy. At 6-month follow-up, serum calcium had normalized to 9.1 mg/dL, though optic neuropathy remained irreversible. Liver function tests showed improvement (ALT 68 U/L), and 24-hour urinary copper excretion decreased to 150 µg/day. The diagnostic and therapeutic timeline, including key clinical events and findings, is summarized in Table 2.
Diagnostic and Therapeutic Timeline of the Patient.
Discussion
This case report highlights the exceptionally rare and complex clinical presentation of concurrent hyperparathyroidism and WD in a patient with methanol toxicity. To our knowledge, this combination has been scarcely reported in the literature, with only one similar case documented by Tallis et al in 1990. 9 The interplay between these conditions presents significant diagnostic and therapeutic challenges, which are worth exploring in depth.
WD and Endocrine Manifestations
Although WD is mainly known for its hepatic and neuropsychiatric manifestations due to abnormal copper deposition, its association with endocrine disorders like hyperparathyroidism is rare, though reported in some case studies. 7 Hypoparathyroidism is more commonly described in WD due to copper deposition causing glandular atrophy and dysfunction, but cases of hyperparathyroidism in WD have also been documented, suggesting that both conditions can coexist in some cases. 11
As demonstrated in our case and Tallis et al’s 9 report, WD’s endocrine manifestations extend beyond classical hepatic/neurological features. The mechanisms behind this relationship are not fully understood, but chronic copper accumulation in the parathyroid glands could contribute to their dysfunction.12,13
In our patient, hyperparathyroidism was diagnosed years before Wilson’s disease (WD), with elevated PTH and hypercalcemia observed 4 years prior to the WD diagnosis, suggesting that copper-induced parathyroid dysfunction may precede the classic WD symptoms. In contrast, hypoparathyroidism typically develops later due to copper accumulation in the parathyroid glands, leading to glandular atrophy. This comparison highlights the complex and evolving nature of endocrine dysfunction in WD.
Recent studies suggest that copper accumulation in the parathyroid glands may not only lead to atrophy but could also trigger parathyroid hyperplasia. Copper’s role in this process may involve its effects on oxidative stress, calcium-sensing receptor (CaSR) signaling, or mitogenic pathways that promote parathyroid cell proliferation. Animal models of Wilson’s disease have shown that copper deposition can disrupt normal calcium homeostasis and alter parathyroid function, potentially contributing to hyperparathyroidism.12,14 -16 These studies highlight the complexity of copper’s effects on endocrine tissues and support the hypothesis that long-term copper exposure could lead to parathyroid hyperplasia.
Tallis et al 9 documented the only other known case of WD with hyperparathyroidism. In their patient, hyperparathyroidism manifested as a parathyroid adenoma after 8 years of penicillamine therapy for established WD. In contrast, our patient’s hypercalcemia and elevated PTH were documented 4 years prior to WD diagnosis, suggesting either independent development or that copper-mediated parathyroid dysfunction (possibly via hyperplasia) may precede classical WD manifestations. Both cases implicate copper in parathyroid pathology, but highlight different temporal relationships and potential mechanisms. While copper deposition in WD classically causes parathyroid atrophy and hypoparathyroidism, our case and Tallis et al’s report suggest that chronic copper overload may also trigger parathyroid hyperplasia or adenoma formation. 9 Proposed mechanisms include copper-mediated oxidative stress disrupting calcium-sensing receptor (CaSR) signaling or direct stimulation of parathyroid cell proliferation via mitogenic pathways.9,17 -19
In our patient, WD was confirmed by characteristic biochemical findings, including elevated urinary copper excretion and decreased serum ceruloplasmin levels. Concurrent hyperparathyroidism, diagnosed years before the current presentation, was suggested by persistent hypercalcemia and elevated PTH levels. This case highlights the importance of endocrine evaluations in patients with WD, as both hypoparathyroidism and hyperparathyroidism may be overlooked, leading to delayed diagnosis and management of associated complications.5,20
Methanol Toxicity and Its Exacerbating Factors
Methanol toxicity is a well-known medical emergency characterized by metabolic acidosis, optic neuropathy, and central nervous system depression.21,22 Our patient presented with classic symptoms of methanol toxicity, including nausea, vomiting, and visual disturbances, which rapidly progressed to optic neuropathy. The severity of the patient’s metabolic acidosis and optic neuropathy might have been exacerbated by his underlying conditions, including WD and untreated hyperparathyroidism, which may have contributed to an altered metabolic response to methanol.23,24
The concurrence of these conditions reflects clinically significant interactions. While hyperparathyroidism-induced hypercalcemia primarily affects renal calcium handling, potentially leading to nephrolithiasis and renal dysfunction, its potential interaction with WD remains theoretically important but not well-documented. More established is the dual mitochondrial toxicity. Copper accumulation impairs cytochrome c oxidase 25 while methanol’s formate metabolites disrupt oxidative phosphorylation 26 likely compounding metabolic disturbances. Tallis et al’s 9 case similarly demonstrated that WD can involve unexpected endocrine manifestations, though through different mechanisms.
Clinical Interpretation
Untreated hyperparathyroidism, particularly with hypercalcemia, can worsen renal function by impairing calcium handling in the kidneys, increasing the risk of nephrolithiasis and reducing renal clearance. In this case, the patient’s elevated PTH and hypercalcemia likely compounded renal dysfunction, making him more susceptible to the nephrotoxic effects of methanol.27,28 Methanol’s metabolites exacerbate renal and metabolic disturbances, suggesting that hyperparathyroidism may not only impair baseline renal function but also increase vulnerability to methanol toxicity, emphasizing the need for early management of hyperparathyroidism to prevent such complications.29,30
Diagnostic Challenges and Management Strategies
The diagnostic complexity of this case cannot be overstated. Each of the conditions including WD, hyperparathyroidism, and methanol toxicity, presents with overlapping metabolic disturbances, which complicates the clinical assessment. The patient’s persistent hypercalcemia and elevated PTH levels were key indicators of hyperparathyroidism, yet these findings were only uncovered retrospectively. This case highlights the need for thorough and early biochemical screening in patients with complex clinical presentations, particularly those with a history of chronic metabolic disorders.
Management of such a rare triad of conditions requires a coordinated approach, involving multiple specialties, including hepatology, endocrinology, nephrology, and toxicology. The decision to initiate dialysis in this patient was critical in managing the acute renal dysfunction and severe hypercalcemia, and it underscores the importance of rapid intervention in such complex cases.
Implications for Clinical Practice and Future Research
The rarity of this case makes it a valuable contribution to the existing literature, emphasizing the need for clinicians to consider rare but possible coexisting conditions in patients with atypical presentations. While the exact pathophysiological interactions between WD, hyperparathyroidism, and methanol toxicity remain to be fully elucidated, this case suggests that the presence of one condition may exacerbate the others, leading to a more severe clinical presentation. Further research is warranted to explore the underlying mechanisms linking these conditions and to develop evidence-based guidelines for managing such complex cases. In particular, studies focusing on the long-term outcomes of patients with similar presentations could provide valuable insights into the best practices for diagnosis, management, and prognosis.
Conclusion
The simultaneous occurrence of hyperparathyroidism and WD in a patient with methanol toxicity is an exceptionally rare phenomenon. This case report serves to increase awareness among clinicians about this unique triad and emphasizes the importance of considering these conditions in the differential diagnosis of patients presenting with methanol toxicity. Further research and case studies are needed to elucidate the underlying mechanisms and explore optimal management.
Footnotes
Acknowledgements
Special thanks to all the fellow authors who contributed to this article.
Abbreviations
WD: Wilson’s Disease
PTH: Parathyroid Hormone
MRI: Magnetic Resonance Imaging
CaSR: Calcium-sensing receptor
Ethical Considerations
The study and all experimental protocols were approved by the Professional Ethics Committee of the Golestan University of Medical Sciences. All methods were carried out in accordance with relevant guidelines and regulations.
Consent to Participate
An informed written consent was also obtained from the patient for the implementation of this project.
Consent for Publication
The patient provided written informed consent for publication of anonymized case details and accompanying images, including fundoscopy and endoscopy findings.
Author Contributions
Fatemeh Mohammadzadeh: Conceptualization, Investigation, Methodology, Project administration, Resources, Validation, Writing—review & editing. Alireza Fatemi: Conceptualization, Data curation, Formal analysis, Methodology, Supervision, Visualization, Writing—original draft, Writing—review & editing. Yasmin Yazdooei: Conceptualization, Data curation, Methodology, Resources, Validation, Writing—original draft. Alireza Norouzi: Conceptualization, Data curation, Visualization, Writing—review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Guarantor Author
Alireza Fatemi accepts full responsibility for the study, including the conduct of the research, having access to the data, and controlling the decision to publish.
Preprint Disclosure
A previous version of this manuscript was submitted as a preprint to SSRN (SSRN 4983830). The current manuscript has been revised and expanded with additional clinical details.
