Abstract
Background:
Diabetes mellitus (DM) is a critical risk factor associated with postoperative complications. Preoperative glycemic control, commonly assessed by glycated hemoglobin (HbA1c), may help stratify patients with DM. However, association between DM, HbA1c levels and perioperative outcomes in multi-ethnic Asian populations with distinct cardiometabolic profiles remains underexplored.
Objectives:
This study aimed to study the association between pre-existing DM, suboptimal glycemic control (HbA1c ≥ 7%), and postoperative complications in elective non-cardiac surgical patients, focusing on the role of HbA1c as a risk stratification tool.
Methods:
This secondary analysis included 688 patients from a prospective cohort at Singapore’s largest tertiary hospital. Postoperative complication(s) were assessed using the Comprehensive Complication Index. Patients were categorized into 2 groups—DM (regardless of HbA1c) and no DM (HbA1c ≤ 6%). We used multivariable logistic regression to explore associations within the entire cohort and DM subgroup. The relationship between preoperative HbA1c levels and postoperative complication(s) was also explored.
Results:
The overall incidence of postoperative complication(s) was 20.78%. DM was independently associated with increased postoperative complication(s) (adjusted OR 2.57, 95% CI 1.20-5.50, P = .015). A trend toward a higher likelihood of postoperative complication(s) was observed in patients with DM and suboptimal glycemic control (adjusted OR 1.39, 95% CI 0.56-3.45, P = .482) though this did not reach statistical significance. A noteworthy U-shaped relationship was identified between preoperative HbA1c levels and postoperative complication(s), with increased complications at both low and high HbA1c levels.
Conclusion:
This study highlights a significant association between DM and increased postoperative complications. The observed U-shaped relationship between HbA1c levels and complications underscores the need for comprehensive risk assessment across the full glycemic spectrum. Routine HbA1c screening and tailored perioperative strategies in multi-ethnic Asian populations could enhance surgical outcomes, reduce healthcare costs, and support broader public health goals in DM management.
Keywords
Introduction
Background
The perioperative care landscape is undergoing a paradigm shift, with increasing emphasis on the early identification and management of comorbidities during the preoperative period to optimize patient outcomes. 1 In Singapore, this transformation coincides with a significant increase in surgical procedures, highlighted by a doubling of elderly patients undergoing surgery over the past decade. 2 Concurrently, the rising prevalence of diabetes mellitus (DM)—projected to affect an additional 200 000 individuals over 40 years of age by 2030—adds further complexity to perioperative management. 3
Understanding the impact of DM on postoperative outcomes is particularly critical in Singapore’s unique demographic and healthcare context. While DM is a well-established risk factor for postoperative complications,4-8 preoperative glycemic control—quantified by glycated hemoglobin (HbA1c)—as a potential risk stratification marker remains under-characterized, particularly outside of cardiac surgery. This is especially pertinent in Singapore’s multi-ethnic Asian population, which is predisposed to higher cardiometabolic risks at lower body mass index thresholds compared to Western populations. 9 However, the relationship between glycemic control and surgical outcomes in such populations remains insufficiently studied.
Currently, routine preoperative HbA1c testing for non-cardiac surgical patients is not standard practice in Singapore, representing a potential missed opportunity for early risk stratification and optimization. Moreover, existing literature predominantly relies on retrospective cohorts, 10 limiting the generalizability of findings to real-world practice.
Given the rising burden of DM across Asia and Singapore’s rapidly aging surgical population, there is an urgent need for locally relevant, prospective evidence to inform perioperative DM management strategies, which remain underdeveloped. Addressing these gaps is essential to advancing perioperative care models that integrate chronic disease management into surgical pathways.
Building on our previous local HbA1c epidemiology study, 11 where HbA1c was prospectively measured for all recruited non-cardiac surgical patients, we now undertake an exploratory secondary analysis of postoperative complication(s) within the same cohort. Notably, our previous study revealed that nearly 1 in 5 non-cardiac surgical patients have pre-existing DM, 11 a prevalence more than double the national DM prevalence, 12 further highlighting the significance of perioperative DM management in our setting.
Specific Aims and Objectives
In this exploratory secondary analysis of our prospective cohort study, our primary aim was to determine whether pre-existing DM was associated with postoperative complication(s) among non-cardiac surgical patients in one of Singapore’s major tertiary surgical centers, using the Comprehensive Complication Index (CCI) as the outcome measure. Additionally, our secondary aim was to assess the impact of suboptimally controlled DM (HbA1c ≥ 7%) on postoperative complication(s).
By addressing these objectives, our study will provide valuable insights into perioperative care for patients with DM in Singapore. Findings will inform targeted strategies to optimize surgical outcomes and patient care, leveraging the preoperative care period as a strategic opportunity for intervention. This work aligns with broader national health initiatives, such as the War on Diabetes and HealthierSG13,14 and supports the integration of chronic disease management into perioperative care, contributing to Singapore’s evolving population health agenda.
Materials and Methods
Study Design and Setting
This is an exploratory secondary analysis of a prospective, observational study conducted between May 2019 and August 2019 at Singapore General Hospital’s (SGH) Preoperative Assessment Centre (PAC). SGH, the largest academic center in Singapore, is a 1700-bed tertiary referral hospital that conducts approximately 30 000 elective surgical procedures annually. Patients were followed up for 30 days postoperatively to assess for the development of post-operative complications.
Ethics Approval and Written Informed Consent
The study received ethics approval from SingHealth’s Centralised Institutional Review Board (CIRB 2018/3225) and is registered on the clinicaltrials.gov database (NCT04070963). The study protocol adhered to the principles outlined in the Declaration of Helsinki.
Written informed consent was obtained from all participants before their inclusion in the study. Consent forms were provided in English, and translators were available for participants who did not speak English.
Patient Eligibility Criteria
The inclusion criteria encompassed adult patients aged 21 years and older who were scheduled for elective non-cardiac surgery and required preoperative blood tests. Patients were excluded if they were unable to provide informed consent or if their blood samples were insufficient for HbA1c testing. Additionally, patients with pre-existing pre-diabetes (documented in clinical history) and those without a prior diagnosis of dysglycemia but with HbA1c > 6%—which may indicate undiagnosed pre-DM or DM—were excluded, as their risk profiles may differ. For comparison, patients were classified into 2 groups: those with pre-existing DM (irrespective of HbA1c levels) and those without DM (defined as HbA1c ≤ 6%).
Data Collection
Baseline demographic data, American Society of Anesthesiologists (ASA) physical status classification, medical comorbidities and HbA1c performed at PAC were collected. The Charlson Comorbidity Index, an instrument that calculates a score based on specific medical conditions to reflect mortality risk, was calculated to quantify the overall comorbidity burden. 15 Surgical details, including type and nature of surgery and surgical table codes, were documented. Postoperative complication(s) within 30 days postoperatively were assessed using the Comprehensive Complication Index (CCI).
Data were extracted from the institution’s clinical information system (Sunrise Clinical Manager [SCM], Allscripts, Illinois, USA) and stored in the enterprise data repository and analytics system (SingHealth-iHiS Electronic Health Intelligence System), which integrates information from multiple healthcare systems.
Primary Outcome: Postoperative Complication(s) Defined by Comprehensive Complication Index (CCI)
The primary outcome measure was the CCI, a widely accepted metric for quantifying the severity and burden postoperative complication(s). 16 The CCI integrates various postoperative complications, including infections, organ failure, reoperation, and other clinically relevant postoperative events, with each complication weighted according to its severity. Assessed for 30 days postoperatively, the CCI was selected as the primary outcome measure because it is more sensitive and superior to traditional morbidity endpoints, thus reducing the sample size required to detect significant differences between study groups. 17
HbA1c Testing and Cutoffs
HbA1c testing was added to routine preoperative blood tests for all participants. HbA1c levels were measured using an immunoassay on the Roche Cobas c501 analyzer (Roche Diagnostics), accredited by the National Glycoprotein Standardization Program (NGSP) and standardized to the Diabetes Control and Complications Trial (DCCT) assay.
Patients with HbA1c ≤ 6% were classified as the unexposed group (no DM or pre-DM), consistent with Singapore’s Handbook for the Chronic Disease Management Programme 18 and a prior local study validating HbA1c as a screening tool in our multiethnic population. 19 While this differs from the American Diabetes Association (ADA) guidelines, 20 it reflects national practice and population-specific evidence. This contextualized threshold may also be relevant to other multiethnic Asian populations.
To define suboptimally controlled DM, we used a cutoff of HbA1c ≥ 7%, consistent with the ADA and Singapore’s Ministry of Health Clinical Practice Guidelines on Diabetes Mellitus recommendations for long-term DM management.20,21 Although thresholds for predicting postoperative complications vary and remain uncertain,22-24 HbA1c ≥ 7% is among the more lenient cut-offs, selected to align with long-term DM management, maintain comparability with prior studies, and capture a broader at-risk population.
Sample Size and Post-hoc Power Calculation
The sample size for this study comprised all eligible patients from the prospective cohort. Recruitment was originally powered to estimate the prevalence of DM, rather than to detect differences in postoperative complications between patients with and without DM. Therefore, the maximum number of patients available for analysis was constrained by this initial study design and eligibility criteria. To assess whether the study was adequately powered to evaluate the association between DM and postoperative complications, a post-hoc power calculation was performed. Statistical power of 80% or more was considered sufficient.
Statistical Analyses
To explore the association between DM and postoperative complication(s), patients were categorized into 2 groups: those with pre-existing DM (regardless of HbA1c levels) and those without DM (defined as HbA1c ≤ 6%).
Descriptive statistics were used to characterize the patients with and without DM. Continuous variables were presented as mean (standard deviation) for normally distributed data and median (interquartile range) for skewed data, while categorical variables were expressed as frequencies (percentages). Univariate analyses were also done to compare variables between patients with and without DM. Continuous variables were assessed using either 2-tailed unpaired T-tests or Mann-Whitney U tests, depending on the normality of the data. Meanwhile, categorical variables were assessed using either Pearson’s chi-square test or the Kruskal-Wallis test based on the observed cell counts.
The primary outcome, the Comprehensive Complication Index (CCI), was dichotomized into a binary outcome: a CCI of 0 indicated no postoperative complication, while a CCI above 0 indicated the presence of postoperative complication(s). Differences between patients with and without postoperative complication(s) were assessed using simple logistic regression, with crude odds ratios (OR) and corresponding P-values presented.
We then assessed the independent association of pre-existing DM with postoperative complication(s) with multivariable logistic regression, where adjusted OR and corresponding P-values were presented. To avoid overfitting, scores related to age and DM were excluded from the Charlson Comorbidity Index. The model incorporated known confounders such as demographics, comorbidities assessed via the Charlson Comorbidity Index, and surgical complexity, where surgical table codes served as a surrogate. Table codes 1A-3C indicate low surgical complexity, Table codes 4A-5B indicate intermediate surgical complexity, and Table codes 6A-7C indicate high surgical complexity. ASA physical status classification was omitted due to collinearity with the Charlson Comorbidity Index.
To investigate the relationship between preoperative HbA1c and postoperative complication(s), a quadratic term of HbA1c*HbA1c was added to the multivariable logistic regression model and the relationship between preoperative HbA1c and predicted postoperative complication probability was illustrated based on this model. The observed relationship was further corroborated using actual data.
The performance and fit of the multivariable logistic regression models, both with and without the quadratic term, was evaluated with the Bayesian information criterion (BIC), Hosmer-Lemeshow goodness-of-fit test, classification statistics, and area under the receiver operating curve (ROC).
For the secondary outcome where we assessed the association of suboptimally controlled DM (HbA1c ≥ 7%) and postoperative complication(s), a subgroup analysis was conducted among patients with DM. The independent variable of interest, HbA1c ≥ 7%, was included in the multivariable logistic regression model, adjusting for similar known confounders.
Statistical significance was achieved with P-value < 0.05.
All statistical analyses were performed using Stata statistical software (StataSE version 18.0 for Mac, StataCorp, College Station, TX).
Results
Cohort Characteristics
A total of 723 patients were assessed for eligibility. Thirty-five patients were excluded due to either a documented history of pre-diabetes or HbA1c > 6% without a prior diagnosis of dysglycemia, indicating possible undiagnosed pre-diabetes or diabetes. The final study cohort comprised 688 elective non-cardiac surgical patients, of whom 140 (20.35%) having pre-existing DM and 548 (79.65%) without DM (HbA1c ≤ 6%). Table 1 summarizes the perioperative variables.
Perioperative variables of the whole cohort.
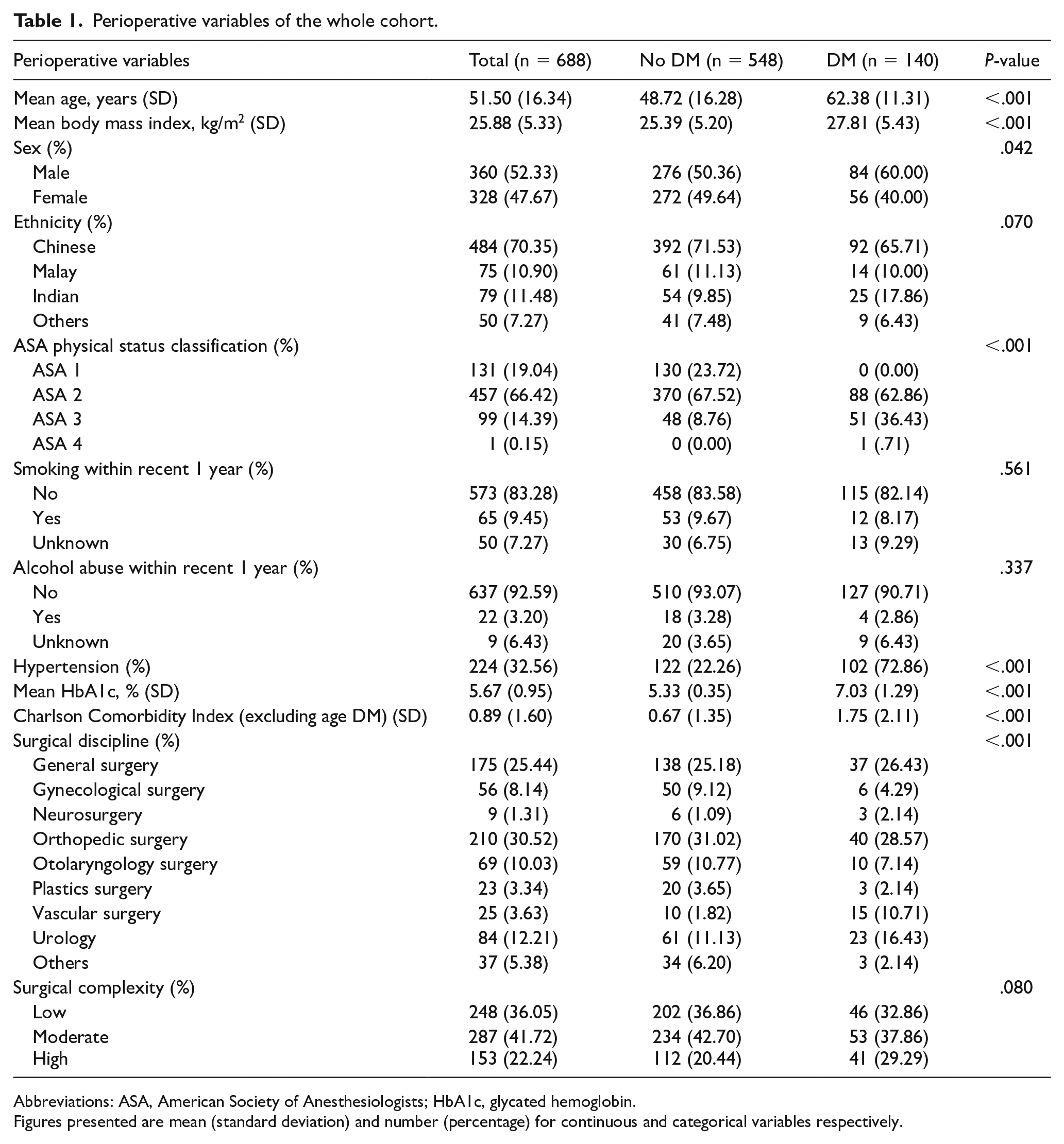
Abbreviations: ASA, American Society of Anesthesiologists; HbA1c, glycated hemoglobin.
Figures presented are mean (standard deviation) and number (percentage) for continuous and categorical variables respectively.
Patients with DM were older (P < .001), more likely to be male (P = .042), had hypertension (P < .001), and higher BMI (P < .001), ASA physical status classification (P < .001), and Charlson Comorbidity Index (excluding age and DM; P < .001) compared to those without DM. A greater proportion of patients with DM underwent vascular surgery (10.71% vs 1.82%, P < .001). While a higher percentage of patients with DM underwent the most complex surgeries compared to those without DM (29.29% vs 20.44%), this difference was not statistically significant (P = .080). Additionally, the length of hospitalization was longer for patients with DM by 1.19 ± 3.21 days (P < .001).
Primary Outcome: Postoperative Complication(s)
The overall incidence of postoperative complication(s) was 20.78% (n = 143/688). Patients with DM had a significantly higher incidence of complications (32.14%) compared to those without DM (17.88%, P < .001), underscoring the impact of DM on surgical outcomes. Post-hoc power analysis confirmed that the study was adequately powered (99.2%) to detect this difference. Simple logistic regression (Table 2) showed a significant association between DM and postoperative complication(s), with a crude OR of 2.18 (95% CI 1.43-3.30, P < .001).
Univariate analyses for the whole cohort.

Abbreviations: ASA, American Society of Anesthesiologists; CCI, Comprehensive Complication Index; CI, confidence interval; HbA1c, glycated hemoglobin.
Figures presented are mean (standard deviation) and number (percentage) for continuous and categorical variables respectively.
Other variables associated with higher crude odds of postoperative complication(s) included older age (P < .001), higher ASA classification (P < .001), higher preoperative HbA1c levels (P = .042), hypertension (P = .002), higher Charlson Comorbidity Index (excluding age and DM; P < .001), undergoing neurosurgery (P = .004) or plastic surgery (P = .005), and high surgical complexity (P < .001; Table 2).
The occurrence of major end-organ complication(s) and infections within the cohort was infrequent. Specifically, pulmonary complication(s) occurred in 3 patients, cardiovascular complication(s) in 2 patients, renal complication(s) in 6 patients, and infections in 8 patients. There were no documented cases of neurological complication(s) or in-hospital mortality.
Patients with postoperative complication(s) had significantly longer hospital stays (6.32 ± 5.33 vs 2.48 ± 1.55 days, P < .001) and higher 30-day readmission rates (0.01 ± 0.14 vs 0.12 ± 0.35, P < .001), underscoring the clinical and economic burden of these events.
Multivariable Logistic Regression Analysis
In the final multivariable logistic regression model, which adjusted for demographics, comorbidities, and surgical complexity, pre-existing DM remained independently associated with postoperative complication(s), with an adjusted OR of 2.57 (95% CI 1.20-5.50, P = .015; Table 3).
Final multivariable logistic regression for the whole cohort.

Abbreviations: CI, confidence interval; DM, diabetes mellitus; HbA1c, glycated hemoglobin; OR, odds ratio.
HbA1c*HbA1c is a quadratic term added in the multivariable logistic regression model to reflect the U-shaped relationship between preoperative HbA1c levels and postoperative complication(s).
Other independent risk factors for postoperative complication(s) included female sex (adjusted OR 1.62, 95% CI 1.04-2.54, P = .034), Indian ethnicity (adjusted OR 1.68, 95% CI 1.08-2.62, P = .022), higher Charlson Comorbidity Index (excluding age and DM; adjusted OR 1.32, 95% CI 1.16-1.49, P < .001), and high surgical complexity (adjusted OR 2.44, 95% CI 1.35-4.41, P = .003; Table 3).
Relationship Between HbA1c and Postoperative Complication(s)
The relationship between preoperative HbA1c levels and postoperative complication(s)was explored using multivariable logistic regression, which included a quadratic term (HbA1c*HbA1c). The results, illustrated in Figure 1, showed a U-shaped relationship between the predicted probability of postoperative complication(s) and preoperative HbA1c levels. Specifically, the predicted probability of postoperative complication(s) was higher both at the lower and higher ends of the HbA1c spectrum, with a nadir noted between HbA1c 5.1% and 6%. This U-shaped relationship was further corroborated by actual data where the incidence of postoperative complication(s) was plotted against preoperative HbA1c levels (Figure 2).

Scatterplot and quadratic fit curve of predicted probability of postoperative complication(s) versus preoperative HbA1c values based on the multivariable logistic regression model presented in Table 3. Quadratic fit curve shows a U-shaped relationship between increasing HbA1c and predicted probability of postoperative complication(s).

Incidence of postoperative complication(s), defined as Comprehensive Complication Index > 0, versus preoperative HbA1c in our cohort. U-shaped relationship seen between increasing HbA1c and incidence of postoperative complication, with nadir noted at HbA1c 5.1% to 6%.
Model Performance and Fit
The fit of the multivariable logistic regression models, with and without the quadratic term (HbA1c*HbA1c), was assessed. The model with the quadratic term showed a slightly better fit, with a lower BIC of 656.44 compared to 654.99 for the model without the quadratic term. The Hosmer-Lemeshow goodness-of-fit test indicated adequate fit for both models (P = .1173 and P = .1384, respectively). Correct classification rates were similar (79.71% with and 80.19% without the quadratic term). The area under ROC was slightly higher for the model with the quadratic term (0.7343 vs 0.7225), suggesting a better discrimination ability.
Secondary Outcome: Impact of Suboptimally Controlled DM (HbA1c ≥7%)
Among patients with pre-existing DM, 67.86% (n = 95) were managed with oral hypoglycemic agent(s) (OHGA), 3.57% (n = 5) with insulin, 10.00% (n = 14) with both OHGA and insulin, and 18.57% (n = 26) through diet control. Univariate analysis showed no significant association between treatment modality DM and development of postoperative complication(s) (P = .487).
Additionally, among patients with pre-existing DM, 44.29% (n = 62 out of 140) had suboptimally controlled DM (HbA1c ≥ 7%).
Subgroup analysis with a multivariable logistic regression model (Table 4) showed that suboptimally controlled DM, indicated by HbA1c ≥ 7%, was associated with a higher odd of developing postoperative complication(s) (adjusted OR 1.39, 95% CI 0.56-3.45, P = .482), though this did not reach statistical significance. High surgical complexity was independently associated with postoperative complication(s) among patients with DM (adjusted OR 6.38, 95% CI 1.80-22.59, P = .004; Table 4).
Multivariable logistic regression for patients with DM.

Abbreviations: CI, confidence interval; DM, diabetes mellitus; HbA1c, glycated hemoglobin; OR, odds ratio.
Discussion
Building on our local HbA1c epidemiology study, this is one of the first studies in Southeast Asia to examine the association between pre-existing DM and postoperative complication(s) in a diverse non-cardiac surgical population at Singapore’s largest tertiary hospital. To our knowledge, it is the first in the region to evaluate these associations using systematically collected perioperative data, including universal HbA1c screening in all surgical patients, enabling comprehensive assessment of glycemic status.
Pre-existing DM—a major endocrine and cardiometabolic risk factor—was significantly associated with increased odds of postoperative complication(s) in our cohort. This finding aligns with international studies, including a recent meta-analysis of over 3 million patients which reported significantly elevated odds of postoperative complications among patients with DM (OR 1.65, 95% CI 1.49-1.84). 4 Our study extends this evidence by confirming the association in a multi-ethnic Southeast Asian population, thereby enhancing the global generalizability of this relationship. The strength and consistency of this finding across diverse populations reinforces the biological plausibility of DM as a perioperative risk factor for postoperative complications.
In Singapore, the relevance of this association is amplified by the disproportionately high burden of DM among surgical patients, shaped by demographic transitions such as population aging and the rising prevalence of cardiometabolic disease. The observed surgical DM prevalence of 19.1% 11 underscores a substantial clinical and systems-level challenge. Without targeted intervention, the adverse impact of DM on surgical outcomes and acute care utilization will likely intensify. 25 Our findings support the timely integration of perioperative DM management, including early identification and tailored glycemic risk assessment, into existing surgical workflows. This is especially critical in high-volume health systems where surgical demand is expected to grow.
A noteworthy finding from our study was the U-shaped relationship between preoperative HbA1c levels and postoperative complication(s), with increased complication risk at both low and high ends of the glycemic spectrum relative to mid-range values (5.1%-6%). This mirrors earlier observations by O’Sullivan et al in patients undergoing vascular surgery, 26 but diverges from the literature which focuses on mortality outcomes or populations with cardiac comorbidities.27,28 Importantly, our analysis contributes new evidence on this relationship within a diverse non-cardiac surgical population. Our results suggest that optimal preoperative glycemia may lie within a narrower target range than previously assumed, although further research is needed to validate and understand the implications of this relationship in the non-cardiac surgical setting.
Our findings challenge the traditional emphasis on hyperglycemia alone as perioperative risk factor. The increased risk observed at lower HbA1c levels is likely multifactorial. In patients with DM, excessively tight glycemic control may predispose to hypoglycemia, which is associated with neuronal injury and adverse perioperative outcomes.29,30 Among patients without DM, low HbA1c levels may reflect frailty, malnutrition and early stages of chronic disease, 31 all of which impair surgical recovery.32,33 These insights highlight the need for perioperative risk stratification strategies assessment strategies that account for both extremes of the glycemic spectrum.
Subgroup analysis further showed a trend toward increased postoperative complications in patients with suboptimally controlled DM (HbA1c ≥ 7%). While our study was not powered to detect definitive thresholds, this direction of association is biologically plausible and consistent with the upper limb of the U-shaped curve observed in the overall cohort. It also corroborates with prior evidence showing that elevated HbA1c (≥7-8%) increased postoperative complication(s) in abdominal, spine and joint replacement surgeries.22-24 However, the lack of a universally accepted HbA1c threshold reflects ongoing uncertainty in this area. Our findings suggest that future research should focus on assessing the predictive utility of these thresholds and determining which surgical populations would benefit most from preoperative optimization.
In our cohort, 44.3% of patients with DM had suboptimally controlled glycemia (HbA1c ≥ 7%), raising important considerations about the feasibility of strict glycemic control mandates preoperatively. Implementing a universal policy to achieve HbA1c < 7% may not be practical in high-volume surgical systems. A more sustainable approach may be targeted optimization for patients undergoing high-risk or complex surgeries, balancing clinical benefit, safety, procedural timeliness and resource allocation.
Our study has several key strengths. First, this is one of the few studies globally to demonstrate a U-shaped relationship between preoperative HbA1c levels and postoperative complications in a diverse non-cardiac surgical cohort, addressing an important gap in perioperative risk stratification. Secondly, as an exploratory secondary analysis of a prospective cohort, its benefits from the systematic data collection inherent in the primary study design, enhancing reliability and internal validity. The use of universal HbA1c screening in all surgical patients, irrespective of baseline risk, minimizes selection bias commonly observed in retrospective studies where HbA1c measurement is often limited to higher-risk patients, potentially overestimating postoperative complication(s). This approach also enabled accurate classification of glycemic status, improving the precision of our findings. Additionally, the use of a validated outcome measure ensures that our assessment of postoperative complication(s) is robust and consistent. Lastly, the inclusion of a large, diverse patient cohort improves the external validity and generalizability of our results to non-cardiac surgical populations in similar healthcare settings.
Nonetheless, several limitations warrant consideration. First, postoperative complications were analyzed categorically using the CCI, which precluded assessing their severity on a continuous scale. Secondly, despite comprehensive adjustment for known variables, the observational nature of our study introduces the possibility of residual confounding from unmeasured factors that could influence the relationship between DM status and postoperative complication(s), including perioperative glucose control, which has been shown to impact postoperative complication(s). 29 Third, our study did not differentiate between type 1 and type 2 DM patients, and individuals with undiagnosed DM or pre-DM were excluded from the analysis. Although the number of such latter individuals was small, potentially limiting the impact of their exclusion, these constraints may limit insights into risk heterogeneity across glycemic subtypes. 4 Fourth, the post-hoc power analysis was based on the observed effect size, which may overestimate the precision and robustness of the findings. While the calculated power exceeded the conventional threshold, prospective sample size power calculations remain the preferred methodological approach. Lastly, the study’s single-center nature may affect external validity. However, we note that Singapore’s national healthcare system is relatively integrated, and tertiary institutions share similar patient demographics and clinical practice patterns, which may mitigate this limitation.
Future work should aim to validate our findings and further refine preoperative glycemic control management strategies with larger, adequately powered studies across diverse populations and surgical settings. Prospective studies should validate the differential threshold effects of HbA1c on postoperative complication(s) and investigate the biological mechanisms underpinning the U-shaped relationship between HbA1c and postoperative complication(s). Identifying specific patient subgroups, such as those with high-risk comorbidities or undergoing complex surgeries, who would benefit most from preoperative glycemic control optimization will be critical for informing resource allocation. Prospective evaluation of perioperative glycemic control strategies in both patients with and without DM, including those with low HbA1c levels, will also be valuable. This comprehensive approach will help develop evidence-based guidelines that are broadly applicable while also offering tailored recommendations for patients with higher risk profiles or those undergoing more complex surgeries.
In light of our findings, we propose that all surgical patients with pre-existing DM undergo routine preoperative HbA1c testing. This simple, proactive measure enables early identification of at-risk individuals and facilitates stratified perioperative planning. Given the substantial prevalence of suboptimally controlled DM, this strategy holds potential for both improving short-term surgical outcomes and generating long-term population health benefits by leveraging the preoperative period as a window of health opportunity.
To complement this strategy, further studies should also explore strategies for detecting and managing undiagnosed DM and pre-DM in surgical populations. These individuals may exhibit different risk trajectories34,35 and therefore require tailored perioperative approaches.
Conclusion
In summary, pre-existing DM, a prominent cardiometabolic risk factor in our Asian population, was significantly associated with increased postoperative complication(s) among elective non-cardiac surgical patients at Singapore’s largest tertiary center. Patients with suboptimally controlled DM (HbA1c levels ≥ 7%) appear to face heightened risks of postoperative complication(s). Moreover, the observed U-shaped relationship between preoperative HbA1c levels and postoperative complication(s) highlights the need for comprehensive perioperative risk assessment across the full glycemic spectrum.
We recommend routine HbA1c screening for all patients with pre-existing DM and advocate for further research to refine glycemic optimization strategies tailored to surgical risk profiles. By advancing evidence-based perioperative practices, we can improve surgical outcomes, promote a healthier population and contribute to a more resilient healthcare system equipped to meet the needs of Singapore’s aging and increasingly cardiometabolically burdened society.
Footnotes
ORCID iDs
Ethical Considerations
SingHealth Centralised Institutional Review Board CIRB 2018/3225.
Consent to Participate
Written informed consent was obtained from all eligible participants.
Author Contributions
RRGN, MF, and HRA conceptualized the study and developed the methodology. Data curation was performed by RRGN and BPYT. RRGN and CXWK conducted the formal analysis, managed the software, and created the visualizations. Funding acquisition was handled by RRGN and HRA. The investigation was carried out by RRGN, CXWK, YK, BPYT, MF, and HRA. Project administration responsibilities were shared by RRGN, CXWK, BPYT, and HRA. Resources were provided by HRA. Supervision was provided by MF and HRA. The original draft was written by RRGN, with review and editing contributions from RRGN, CXWK, YK, BPYT, MF, and HRA.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our work was supported by the SingHealth-Duke NUS Anaesthesiology Academic Clinical Program Pilot Research Grant 2019 (ANAESPRG19/04). Roderica Rui Ge Ng is supported by the National Medical Research Council Research Training Fellowship, Ministry of Health, Singapore (MOH-RTF21nov-0003 and MOH-RTF24jan-0022). Hairil Rizal Abdullah is supported by the National Medical Research Council Population Health and Health Services Research Clinician Scientist Award – Investigator, Ministry of Health, Singapore (HCSAINV22jul-0002). The funding sources have no role in our study design and the analysis and interpretation of our results.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author.
