Abstract
Hypercalcemia can result from either hyperparathyroidism or non-parathyroid conditions. When hypercalcemia is accompanied by undetectable parathyroid hormone (PTH) levels, hyperparathyroidism is rarely considered the diagnosis. Herein, we report the case of a 65-year-old Caucasian woman referred to our hospital for further evaluation of hypercalcemia. Her symptoms included fatigue and brain fog, with undetectable PTH levels. A comprehensive workup, including a series of laboratory and imaging tests, excluded common non-parathyroid causes such as malignancy and familial hypocalciuric hypercalcemia. Ultrasound identified a likely enlarged parathyroid gland, which was further confirmed by a sestamibi scan. After 2 weeks of cinacalcet treatment, the patient’s calcium levels decreased, indicating the parathyroid gland as the likely source of hypercalcemia. Parathyroidectomy was subsequently performed, revealing a 1927 mg adenoma. Postoperatively, the patient’s calcium levels normalized, PTH levels became detectable within the normal range, and her symptoms resolved, with a marked improvement in energy. This case demonstrates that primary hyperparathyroidism can present with hypercalcemia and undetectable PTH. A genetic mutation in the PTH gene within the adenoma may explain the undetectable PTH levels preoperatively.
Plain Language Summary
This case report discusses a 65-year-old woman who was found to have high calcium levels in her blood, a condition known as hypercalcemia. Typically, hypercalcemia can result from either a condition where the parathyroid glands produce too much parathyroid hormone (PTH) or from other non-parathyroid-related issues like cancer. However, when hypercalcemia occurs with undetectable levels of PTH, it is unusual for hyperparathyroidism to be the cause. In this patient’s case, she experienced symptoms like fatigue and brain fog, but her initial tests showed undetectable PTH levels. Further tests ruled out other common causes of high calcium, such as cancer or a genetic condition called familial hypocalciuric hypercalcemia. An ultrasound and a special scan called a sestamibi scan revealed an enlarged parathyroid gland, suggesting the presence of a parathyroid adenoma, a non-cancerous tumor. To confirm this, the doctors gave the patient a medication called cinacalcet, which is known to lower calcium levels if the parathyroid glands are involved. After two weeks of treatment, her calcium levels dropped, indicating that the parathyroid gland was indeed the source of the problem. The patient underwent surgery to remove the adenoma, which weighed nearly two grams. Following the surgery, her calcium levels returned to normal, and her PTH levels became detectable again within the normal range. Her symptoms, including fatigue, improved significantly, and she reported feeling more energetic. This case is unusual because it demonstrates that primary hyperparathyroidism can still be the cause of hypercalcemia even when PTH levels are initially undetectable. The doctors suspect that a genetic mutation in the adenoma might explain why PTH levels were undetectable before surgery. This case emphasizes the importance of thorough clinical evaluation, even when initial test results are not typical.
Introduction
Hypercalcemia affects approximately 1% to 4% of the general population, with primary hyperparathyroidism being the most prevalent cause.1,2 The hallmark of primary hyperparathyroidism is the overproduction of parathyroid hormone (PTH) by the parathyroid chief cells. Therefore, the diagnosis of primary hyperparathyroidism is confirmed when an elevated or inappropriately normal PTH level is present. Single parathyroid adenomas account for the majority of primary hyperparathyroidism cases, and parathyroidectomy offers a cure rate exceeding 95%, significantly reducing the risk of complications of hyperparathyroidism.3 -5
In cases where hypercalcemia arises from non-parathyroid sources, patients typically present with lower-than-normal PTH levels due to suppressed parathyroid glands. 6 These patients often require an extensive work-up because multiple organ systems, and sometimes an occult malignancy, may be involved. Despite comprehensive investigations, the etiology of hypercalcemia may remain elusive, necessitating repeated examinations, increasing healthcare costs, and causing considerable anxiety for patients. 7
When a patient presents with undetectable PTH and an unrevealing work-up for non-parathyroid causes of hypercalcemia, it is uncommon for physicians to reconsider primary hyperparathyroidism as a potential diagnosis. 6 While the risks associated with surgical exploration of the parathyroid glands must be weighed carefully, the operation may provide further diagnostic clarity and a potential cure.8,9 Herein, we report a case of hypercalcemia with undetectable PTH that was successfully treated with parathyroidectomy after a negative evaluation for non-parathyroid origins of hypercalcemia.
Case Description
A 65-year-old Caucasian woman with a history of hypothyroidism and hypertension was referred first to the endocrinology clinic for evaluation due to persistently elevated calcium levels. She complained of fatigue and brain fog for the past 3 years. She denied joint pain, kidney stones or fractures and had no history of surgery. The patient was not taking any medications known to affect calcium levels, apart from over-the-counter vitamin D supplements, and she did not use biotin. Her family history was negative for hypercalcemia or other endocrinopathies. Notably, her medical history included residence near Chernobyl in 1988. She was 180 cm tall, weighed 89.8 kg, with a BMI of 27.7 km/m2. Physical examination revealed a supple neck with no evidence of thyromegaly or palpable masses.
Initial laboratory evaluation revealed an albumin-adjusted serum calcium level of 10.9 mg/dl (reference range, 8.4-10.4 mg/dl) (Table 1), an intact PTH level of < 1.0 pg/ml (reference range, 12-88 pg/ml), and a vitamin D level of 39 ng/ml (reference range, 20-100 ng/ml). The calcium/creatinine clearance ratio was 0.027 (normal > 0.01). Creatinine and thyroid function tests were within normal limits. Neck ultrasound identified a likely enlarged parathyroid gland, measuring 1.8 × 1.3 × 1.1 cm, located posterior to the right thyroid lobe.
Laboratory studies.

Abbreviations: FLC, free light chain; PTH, parathyroid hormone; PTHrP, parathyroid hormone-related peptide; Serial dilution, dilution factor from 11 to 501; TSH, thyroid-stimulating hormone. The following formula is used to calculate the calcium creatinine clearance ratio: [Urine Calcium × Serum Creatinine] ÷ [Serum Calcium × Urine Creatinine].
The patient was advised to discontinue vitamin D supplements, but repeat laboratory tests continued to show elevated calcium at 10.9 mg/dl with an undetectable PTH level (<1.0 pg/ml), confirmed by an external laboratory. Ionized calcium was 1.46 mmol/l (reference range, 1.12-1.32 mmol/l). Parathyroid hormone-related protein (PTHrP) was negative (0.7 pmol/l, normal ⩽ 4.2 pmol/l). The glomerular filtration rate was normal at 82 ml/min/1.73 m² (normal ⩾ 60 ml/min/1.73 m²). A Technetium-99 m-sestamibi scan showed increased uptake in the same region as the likely parathyroid adenoma on ultrasound. Hematologic malignancy tests, including Kappa free light chain (FLC) levels at 20.4 mg/l (reference range, 3.3-19.6 mg/l) and an FLC ratio of 1.82 (reference range, 0.26-1.65), were conducted. Despite these slight elevations, the absence of an M spike in serum protein electrophoresis (SPEP) allowed for the exclusion of multiple myeloma or other hematological disorders, as confirmed by a hematologist. A CT scan of the chest, abdomen, and pelvis to search for occult malignancy contributing to hypercalcemia was noncontributory. The patient was referred to an endocrine surgeon for further evaluation of potential hyperparathyroidism.
Although imaging results were indicative of a parathyroid adenoma and findings for other sources of hypercalcemia were negative, the surgeon remained uncertain whether parathyroidectomy would resolve the hypercalcemia. To confirm the parathyroid gland as the source of hypercalcemia, cinacalcet was prescribed at a dosage of 30 mg twice daily for 2 weeks. Cinacalcet, a calcimimetic, increases the sensitivity of calcium-sensing receptors on the parathyroid chief cells, theoretically lowering calcium levels if the parathyroid gland is the source of the condition. The typical starting dose was chosen, with a 2-week follow-up scheduled to assess its effect. At the follow-up visit, the patient’s calcium levels had decreased to 10.0 mg/dl, supporting the involvement of the parathyroid gland, and cinacalcet was discontinued.
With clinical and imaging evidence indicating primary hyperparathyroidism, an extensive informed consent discussion was undertaken, and the patient consented to radioguided parathyroidectomy. During the operation, a 1927 mg right superior parathyroid adenoma was excised from behind the lower right thyroid lobe (Figure 1). Intraoperative gamma probe measurements showed a background count of 255, with the excised adenoma having an ex vivo count of 1098, indicating radiocolloid enrichment. Post-resection intraoperative PTH measurements remained at 1.0 pg/ml at 5, 10, and 15 minutes. Histopathological analysis confirmed the adenoma diagnosis. The patient was re-evaluated 2 weeks postoperatively, at which time her serum calcium had decreased to 9.0 mg/dl, and her PTH level had risen to 15.8 pg/ml. The incision healed well, and she experienced no tingling or hoarseness. The patient reported a significant improvement in energy levels, better sleep, and fewer aches and pains. Six months after surgery, her calcium level was stable at 9.6 mg/dl, and her PTH level was 19.7 pg/ml, confirming the surgical cure. The patient reported sustained improvements in energy.
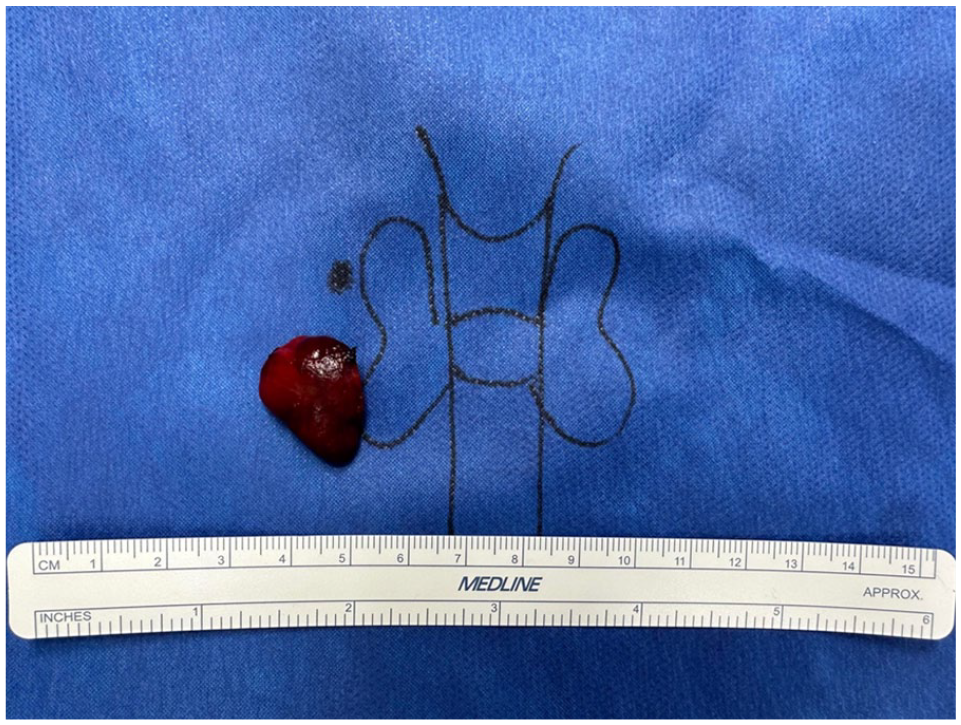
Right inferior parathyroid adenoma with the size of 2.1 × 1.4 × 0.6 cm.
Discussion
In this case, we describe a patient with hypercalcemia who exhibited undetectable PTH levels and a negative non-parathyroid workup. Her medication history did not suggest a drug-induced etiology for hypercalcemia. The calcium/creatinine clearance ratio effectively excluded familial hypocalciuric hypercalcemia, and with a normal glomerular filtration rate (GFR), impaired renal function as a potential cause of low urinary calcium excretion was also ruled out. The absence of malignancy was confirmed through negative PTHrP levels and a normal CT scan, while hematological studies excluded multiple myeloma. Additionally, normal calcitriol levels made lymphoproliferative and granulomatous disorders an unlikely cause of her condition. 6 Despite the unusual laboratory findings, the imaging results and reduction in calcium levels following cinacalcet administration suggested possible hyperparathyroidism, leading to the discovery of a large parathyroid adenoma during surgery. Postoperatively, the patient’s PTH levels returned to the normal range when reassessed with the same PTH assay that initially failed to detect them.
What makes this case particularly rare is the unusual laboratory finding of hyperparathyroidism with undetectable PTH levels before surgery. We considered two hypotheses: either the assay failed to recognize the PTH molecule due to a mutation, or it recognized the molecule but could not successfully bind for positive detection due to inhibition. The intact PTH assay, used in our analysis (Beckman Coulter, Brea, CA), employs chemiluminescent immunoassay technology. 10 This method utilizes two types of antibodies that attach to specific segments of the PTH molecule—namely, the segments from amino acids 39 to 84 and 15 to 20, out of the total 84 amino acids comprising the hormone. However, if the PTH molecule is altered by genetic mutations, these antibodies may not effectively bind to their target sites, leading to inaccuracies in hormone detection. A similar mechanism is observed in Hepatitis B surface antigen escape, where a single nucleotide mutation alters the conformation of the antigenic determinant, thereby inhibiting neutralizing antibody binding. 11
Au et al 12 documented a patient with hyperparathyroidism who had hypercalcemia and undetectable PTH levels due to genetic mutation (Table 2). A large, 4.98 g adenoma was excised during parathyroidectomy. Despite the adenoma testing PTH-negative on immunohistochemistry, adjacent normal tissue stained positively. Microarray analysis confirmed PTH mRNA upregulation, and DNA sequencing revealed a Cytosine to Thymidine substitution, resulting in premature termination of translation at the 52nd amino acid, preventing the formation of the mature 84 amino acid peptide.13,14 Since the capture antibody of the PTH assay targets the 39 to 84 portion of the PTH molecule, it is plausible that the truncated PTH would not be recognized by the assay. There are notable parallels between the case described by Au et al and ours. Both involved large adenomas, significantly larger than average, 15 and both showed undetectable PTH levels preoperatively that normalized post-surgery. Although we did not identify direct evidence of a genetic mutation in our patient, her history of residing near Chernobyl may increase the risk of such mutations due to potential radiation exposure, as studies have shown that adults exposed to nuclear accidents have a significantly increased prevalence of hyperparathyroidism.16,17
Review of cases with hypercalcemia and undetectable parathyroid hormone.

Abbreviations: PTH, parathyroid hormone; M, man; W, woman.
Antibody interference can also lead to undetectable PTH levels in assays, even when the PTH molecule itself is not mutated. This phenomenon was highlighted in a case reported by Shand et al, 18 where a patient with normal calcium levels had undetectable PTH. Further testing, including proportional mixing and affinity binding assays, indicated the presence of excess immunoglobulin, which obscured accurate PTH measurement. In our patient, antibody interference appears to be an unlikely cause. There was no monoclonal spike observed, and the serum FLC ratio was only marginally raised. Crucially, antibody interference would not account for the post-surgical normalization of PTH levels following parathyroidectomy. Hence, we concluded that antibody interference was not a significant factor in this particular case.
Besides antibody interference, we considered the hook effect as another possible explanation for the undetectable PTH levels. 19 The hook effect is a potential pitfall in two-antibody sandwich immunoassays, such as the intact PTH assay. In this assay, a capture antibody is anchored to a solid surface to bind the analyte, while a labeled antibody binds to another site on the analyte. Normally, the analyte is “sandwiched” between the two antibodies, allowing for signal detection. However, when PTH concentrations vastly outnumber the antibodies available, the detection antibodies may bind to unattached analytes rather than those captured, leading to falsely low PTH readings. Nonetheless, when serum is diluted and the hook effect is present, PTH levels should increase. Our tests showed undetectable PTH even after dilution from 11-fold up to 501-fold, contradicting the hook effect scenario. Reviews of several case reports have also discounted the hook effect as a cause.12,18,20,21 Typically, PTH levels are detectable even in the thousands in patients with renal impairment, and the analyte concentration must be exceedingly high to trigger the hook effect. Given our patient’s mild hypercalcemia and presentation with fatigue and brain fog as the dominant symptoms, an excessively high PTH level which might lead to the hook effect is quite improbable.
Another intriguing question about this case is why PTH was detectable using the same assay 2 weeks after surgery. When the foci of abnormal PTH production were resected, it allowed the other parathyroid glands to gradually return back to normal function (which explains undetectable PTH immediately after resection of adenoma) and produce detectable levels of PTH. The re-detection of PTH after surgery also excludes a non-parathyroid origin, as under such conditions, PTH would remain suppressed due to uncorrected calcium levels and would not respond to parathyroid surgery.
Conclusions
In summary, our report presents a case of primary hyperparathyroidism caused by a surgically confirmed adenoma, where PTH levels were undetectable preoperatively but normalized postoperatively. The exact cause remains uncertain, though a mutation in the PTH gene within the adenoma is suspected. This case highlights the importance of diagnosing primary hyperparathyroidism based on clinical assessment, even when PTH levels are undetectably low, after thoroughly excluding other non-parathyroid causes of hypercalcemia.
