Abstract
Background:
There is a little knowledge on the extent to which healthcare providers understand and accept the professional recommendations and appropriate dosing strategy regarding metformin XR.
Objectives:
To evaluate UAE community pharmacists’ knowledge, attitude, and practices (KAP) concerning metformin XR.
Methods:
This is a cross-sectional research study conducted amongst licensed community pharmacists. The survey took place via a questionnaire and physical interviews were held. The survey used in this study included questions on demographics and questions on the participants’ attitudes, knowledge and practices concerning metformin XR. The factors influencing KAP regarding metformin XR were examined via simple logistic regression analysis.
Results:
Threehundred fifty-three (n = 353) participants were recruited in the study. Independent pharmacies constituted 57.5% of this study sample and 42.5% were chain pharmacies. The average knowledge score about metformin XR tablets was 42.5% with a confidence interval (CI) of 95% [37.3%, 47.4]. Better knowledge scores on metformin XR tablets was observed in respondents aged ⩾40 years (OR 2.97, 95% CI 1.63-5.4), having greater than 10 years in terms of experience (OR 2.28; 95% CI 1.25-4.16) and pharmacist graduated from Regional or international universities (OR 2.08; 95% CI 1.34-3.24). About 78% (n = 275) of the participants believed that metformin XR tablets have better efficacy and 63.2% (n = 233) indicated that metformin IR was associated with greater adverse effects.
Conclusion:
This study demonstrated a distinct gap in knowledge, attitude and practice pertaining to metformin XR among community pharmacists in the UAE. The community pharmacists need to enhance their practice by receiving accurate and reliable data to support their decision-making on the prescribing of metformin XR. The implementation of novel guidelines and evidence dissemination strategies may help bridge this gap.
Introduction
Diabetes mellitus (DM) is an ongoing concern in public health, with the number of people affected worldwide expected to reach 783 million in 2045. 1 In a 2017 survey of the UAE, 17.3% of the population in the age range of 20 to 79 was found to have type 2 DM, as per to the International Diabetes Federation (IDF). The country thus has more than 1 million people affected by DM, meaning it ranks 15th for age-adjusted comparative DM prevalence. Furthermore, based on the data, the DM prevalence in the UAE is showing the fastest increase in the Middle East and North Africa (MENA) region, and indeed globally. Certain characteristics of the UAE, including its accelerated economic growth and the unhealthy dietary and lifestyle habits of its population, represent significant DM risk factors. Thus, it is expected that the country will see a doubling of the number of diabetics in its population, potentially reaching 2.2 million by 2040 . 2
Treatment for DM primarily involves glycemic control, the prevention of diabetes complications, and reducing the risk of morbidity and mortality. 3 Inadequate patient management generally causes complications and treatment failure. 4 The self-monitoring of blood glucose (SMBG), medications, and lifestyle modification represent the management interventions recommended for patients with DM. 5 Indeed, managing the conditions of any chronic disease is frequently hampered by patients’ non-adherence to their prescribed drug regimen,3,6 which can lead to failure of treatment, increase the risk of hospitalization, and increase the likelihood of patient morbidity and mortality. 7 The main factors causing non-adherence include polypharmacy and complexity in the medication regimen, especially for those patients who have comorbidities and chronic disease conditions.8,9
The commonly prescribed anti-diabetic agent metformin 10 has been found to be very effective in averting diabetes development in high-risk patients, according to the Diabetes Prevention Program (DPP)/DPP Outcomes Study (DPPOS). 11 The role of metformin has been widened, with various preparations being introduced since the beginning of the 21st century. One of these is the extended-release (XR) form, which was developed to replace immediate-release (IR) tablets and thus facilitate a once-daily dosing regimen. 12 The glycemic control efficacy offered by metformin XR is comparable to that of metformin IR,13 -19 while the reduced dosing also enhances compliance.13,16
The research indicates a correlation between metformin XR and a reduction in the number of adverse gastrointestinal (GI) events,14,15 although a randomized trial conducted internationally in 2017 suggested that both the XR and IR formulations share a similar safety profile. 16 Meanwhile, the guidance from the National Institute for Health and Care Excellence (NICE) is to use metformin IR in initial treatments for patients with type 2 DM, while metformin XR should be prescribed for those who experience GI events. 20
While the plasma concentration of metformin IR peaks 3 hours after dosing, there is a delay of 7 hours for metformin XR because it is absorbed slowly in the upper gastrointestinal tract after the ingestion of fluid. 12 The administration of this drug should thus be according to its pharmacological properties, and glycemic control requires the proper prescription and guidance from qualified pharmacists and physicians. 21 Although physicians are fundamentally responsible for diagnosing illness and prescribing medication, community pharmacists play an important role in helping patients adhere to treatment regimes, optimizing patient outcomes, and identifying and managing the challenges patients encounter outside the clinical setting. Furthermore, community pharmacists often fulfill the primary point of contact role for patients, supporting and guiding them throughout their illness and treatment regime. It is important to evaluate community pharmacists’ knowledge, attitude, and practices (KAP) of metformin XR for multiple reasons. First, such assessments help to ensure patient safety by confirming that pharmacists have up-to-date and accurate knowledge and understanding of the metformin XR, including the indications and contraindications, recommended dosage, potential side effects, and drug interactions. Second, it helps to ensure effective medication management by evaluating pharmacists’ working knowledge of prescribing practices, dosage requirements, and the latest metformin XR guidelines. Third, it helps to determine if pharmacists have the required knowledge and understanding to offer patients the appropriate advice and education. Assessing pharmacists’ practices can inform the development of interventions that enhance patient adherence to medication and compliance with treatment regimes. Finally, evaluating knowledge gaps assists in developing targeted ongoing education programs, assuring continual professional development for pharmacists. On a holistic level, these assessments enhance patient care and safety by delineating areas for development and implementing the associated remedial plans. Nevertheless, there is as yet little knowledge on the extent to which healthcare providers understand and accept the professional recommendations and appropriate dosing strategy. To fill this gap, this study performs a cross-sectional survey to evaluate knowledge, attitude, and practices (KAP) concerning metformin XR among UAE community pharmacists.
Methods and Materials
Study design and setting
This cross-sectional study seeks to explore UAE community pharmacists’ awareness and perceptions of metformin extended-release tablets (metformin XR), particularly regarding their relevant knowledge, attitudes, and practices. To accomplish this, 5 final-year pharmacy students performed a survey of professional community pharmacists inAbu Dhabi, Dubai and the Northern Emirates. The survey took place via a questionnaire disseminated between August 2022 and December 2022. Thereafter, face-to-face interviews were conducted. Based on our previous experience highlighting the need for comprehensive training of the interviewers to mitigate errors during the survey, the student researchers first received extensive training in interview techniques, handling questionnaires, as well as the necessary scientific terms.
Developing the research instrument with pilot testing
First, a literature review was performed to construct the basis for a structured questionnaire14 -19,22 (Supplemental). Certain parts of the questionnaire were modified to make it more appropriate to the UAE context while retaining the main research points. Subsequently, expert endocrinologists were consulted to ensure that the design of the questionnaire was appropriate and relevant for the purposes of the research. Moreover, 5 faculty members at the Medicine and Clinical Pharmacy Department of Ajman University got consulted to examine the questionnaire to ensure that it was both relevant and appropriate. Some minor amendments were then made to the draft survey prior to the pilot testing, in line with the recommendations of the above experts. For example, the scientific terminology was defined, the question and page numbering was modified, the field name (Sex) was replaced with the name (Gender) throughout, certain questions were linked, and certain answers were chosen to conclude the survey tool.
We utilized Lawshe’s content validity to check the survey’s content validity prior to the pilot testing phase. 23 In this approach, 23 items that have a content validity ratio (CVR) of more than 0.78 are considered acceptable, while those that do not meet this threshold are removed from the instrument. All our items possessed a CVR of more than0.78, meaning that their validity was acceptable. We then assessed the content validity index (CVI) of the final questionnaire based on the means of the items with an acceptable CVR. The CVI was found to be 0.877, and thus the content validity of the final survey got accepted overall. 24
To evaluate the questionnaire’s face validity, we conducted a pilot testing phase on 40 UAE community pharmacists during 10 to 20 August 2022. The data from the pilot respondents were not of the final analysis. Pilot testing revealed that 35 of the respondents could successfully complete the questionnaire. We used the results from the pilot study to examine the reliability of the questionnaire as well as to calculate the sample size that would be needed for the main study. The questionnaire’s reliability was assessed via Cronbach’s α, with the α of 0.79 suggesting that the internal consistency of the instrument was reliable.
Research instrument sections
The questionnaire used in this study consisted of the following 2 parts:
Part 1, comprising 6 questions designed to elicit the demographic information of the participants, such as their gender, job, that is, pharmacist in charge or chief pharmacist, their years of working experience, and at which university they had studied.
Part 2, comprising 8 questions on the participants’ attitudes, knowledge and practices concerning metformin XR.
Questionnaire scoring
Three questions were used to assess the participants’ knowledge of metformin XR. They respectively queried participants’ awareness of the dosage range, dosing frequency, and metformin XR timing. Each appropriate answer got a score of 1, and each inappropriate answer scored 0. If a participant gave the correct answers to the 3 questions, their knowledge was considered to be good. The participants’ attitude toward metformin XR was evaluated through questions that assessed whether they perceived any differences between the metformin XR and IR formulations in terms of the side effects and efficacy, as well as which formulation they preferred. Finally, the participants were asked questions pertaining to the circumstances and crucial factors under which they would prescribe metformin XR.
Sample size calculation
The pilot study was the base of the main sample size calculation which recorded an overall response rate of 88%. The pilot study used a question to determine the sample size for the final study, “'Would you choose to prescribe metformin XR?” Around half (50%) of the respondents in the pilot study positively answered this question. This research used an alpha level of 5%, giving a 95% confidence interval (CI). With a precision (D) of 5%, 10% was the maximum width for the 95% CI of 10%. In line with the above, 384 participants was determined to be a sufficient sample size.
Target population
We utilized the following criteria to choose the study sample for the main research. Inclusion criteria were community pharmacists practicing in independent pharmacies or in chains that were registered at the relevant authorities, that is, the UAE Ministry of Health and Prevention, Dubai Health Authority, or the Health Authority Abu Dhabi (HAAD), with at least 3 months’ working experience. Exclusion criteria: not registered at any of the regulatory bodies mentioned above or with less than 3 months’ working experience, including those recently qualified or working in their probation period.
Sampling technique
To ensure a representative sample, a random sampling technique was employed. Survey data gathered in the UAE in 2010 found 2000 professionally active community pharmacies. 25 We identified community pharmacies in the selected study regions and took their contact details, such as their location, from local business directories and the Yellow Pages. This sample was then stratified by assigning the currently active community pharmacies into groups based on their location, producing 3 strata, namely community pharmacies in Dubai, Abu Dhabi and the Northern Emirates, respectively.
Once the community pharmacies had been identified, their details, including name, type, location, email address and phone number, were collected in the sampling framework, which was an Excel spreadsheet. Each community pharmacy was then given a unique ID number. Thereafter, 384 community pharmacies were selected from the list using a simple random selection method. The resulting sample was finally sorted by type and location.
Data collection
The selected community pharmacies were visited by the researchers from 25 August 2022 to 28 December 2022. The researchers informed the pharmacists of the purpose of the research and asked for pharmacies’ email addresses. Lastly, the researchers, who had received thorough training in interview techniques, conducted face-to-face structured interviews with the pharmacists using a questionnaire.
Statistical analysis
The data got analyzed utilizing SPSS Version 26. Mean and standard deviation (±SD) were used to summarize the continuous quantitative variables, and frequencies and percentages (%) were used for the categorical variables. The participants’ knowledge of metformin XR was compared using a chi-square test based on their demographic factors. The factors affecting their knowledge, attitude, and practice regarding metformin XR were examined via simple logistic regression analysis. p-values less than0.05 were considered statistically significant.
Results
Demographic characteristics of the study participants
Three hundred fifty-three (n = 353) participants were recruited in the study. Of the total, 68.3% (n = 241) were female and 31.7% (n = 112) were male. The age groups of the participants were as follows: 38.5% aged 20 to 29 years, 42.2% aged 30 to 39 years and 19.3% aged ⩾40 years. Among the participants, 75 (21.2%) have <1 year of experience, 96 (27.2%) have 1 to 5 years of experience, 67 (19%) have 6 to 10 years of experience and 115 (32.6%) have >10 years of experience. Independent pharmacies constituted 57.5% of the study sample and 42.5% were chain pharmacies. More than half participants (58.6%) graduated from regional/international universities. Of the total participants, 43 (12.2%) worked in Abu Dhabi, 21 (21.5%) worked in Dubai, and 234 (66.3%) worked in Northern Emirates (Table 1).
Number and percentages of the questions on demographics (n = 353).

Regional universities: serve the local population within the GCC countries; International universities, institutions with a global presence.
Knowledge, attitude and practice of the pharmacists about metformin XR tablets
The average knowledge score about metformin XR tablets was 42.5% with a 95% confidence interval (CI) [37.3%, 47.4]. Better knowledge scores on metformin XR tablets was observed in participants aged ⩾40 years (OR 2.97; 95% CI 1.63-5.4), having more than 10 years of experience (OR 2.28; 95% CI 1.25-4.16) and pharmacist graduated from Regional/international universities (OR 2.08; 95% CI 1.34-3.24) (Table 2).
Knowledge about metformin XR tablets according to demographic characteristics.

Abbreviations: OR, odds ratio; CI, confidence interval.
P-values less than .05 were considered statistically significant.
Regarding the attitude about metformin XR tablets, 78% (n = 275) of the participants believed that metformin XR tablets have better efficacy and 63.2% (n = 233) indicated that metformin IR was associated with greater adverse effects. About 10% (n = 35) of the participants recognized the similarity in efficacy profile between the metformin IR and metformin XR tablets. Moreover, the majority of the study participants acknowledged the metformin XR in terms of safety and efficacy. Patients preferring not to take multiple dosing daily (59.5%) was the most common identified reason for choosing the metformin XR and patients’ preference (66%) was the most common factor for prescribing metformin XR among community pharmacists. The results of each question related to knowledge, attitude and practice were shown in Table 3.
Number and percentages of the questions on knowledge, attitude and practice items (n = 353).
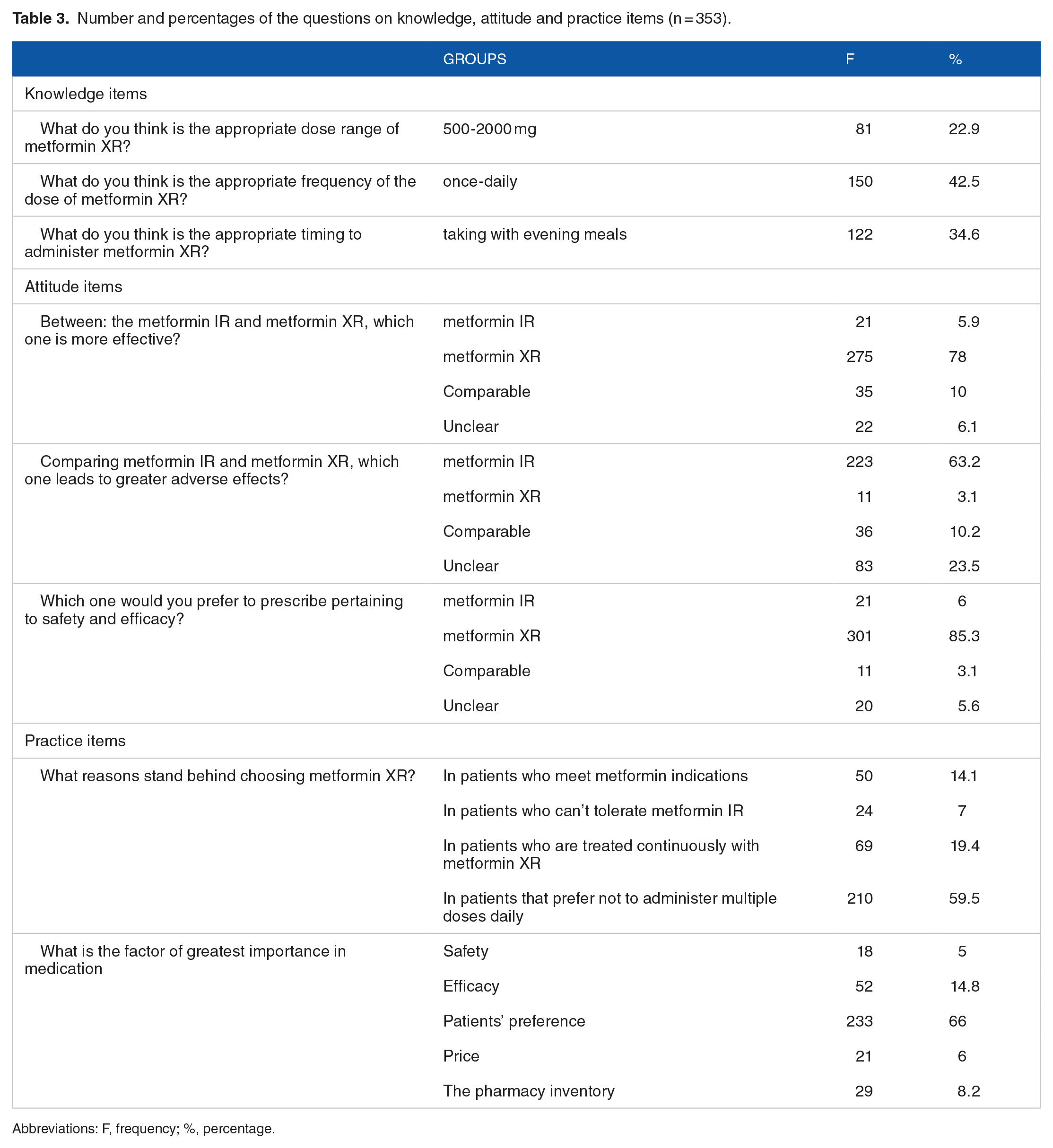
Abbreviations: F, frequency; %, percentage.
Discussion
This study conducted a cross-sectional survey to evaluate UAE community pharmacists’ knowledge, attitude, and practices (KAP) concerning professional recommendations and appropriate dosing strategy of metformin XR. This study is important as dearth of KAP about the management of DM can result in improper control of the disease. To our knowledge and based on exhaustive perusal of existing research, this is the first KAP study of metformin XR among UAE community pharmacists. In this study, demographic analysis showed that over half of the respondents were females, worked in independent pharmacies, graduated from regional/international universities, and worked in Northern Emirates. Moreover the bulk of participants were aged between 30 and 39. Based on the average knowledge score on metformin XR tablets at 42.5%, it is evident that community pharmacists have poor knowledge about DM medication. Less than half of the respondents correctly responded to the appropriate dosage, frequency, and timing of metformin XR. Similarly, Chen et al, reported that physicians, especially non-endocrinologists, are not at a professional level for prescribing metformin. 26 This clearly implies a deficiency of the KAP toward metformin XR among the target population, in spite of the fact that metformin is one of the most utilized antidiabetics among the patients with type 2 diabetes. 27 Nonetheless, better knowledge scores on metformin XR were also observed in participants aged ⩾40 years, having more than 10 years of experience, and pharmacist graduated from regional/international universities.
The results indicate a misapprehension of the features of metformin XR is common among community pharmacists in Dubai, Abu Dhabi, and the Northern Emirates, which may bring about inappropriate prescription and instability of long-term glucose control. 22 Less than half of the pharmacists provided correct responses to the timing of the drug, based on the drug label. 28 This finding confirms the potential inappropriate knowledge and practice of metformin XR use, which requires additional investigation. Comparably low KAP scores have been reported for both clinicians and adults with type 2 diabetes in China. 29 On the contrary, related studies 30 have argued for the use of metformin in patients with newly diagnosed T2DM, particularly those without cardiovascular risk factors or unaffected by advanced kidney failure, based on the confirmed beneficial effects of the medication. Therefore, novel techniques need to be incorporated into dissemination strategies in order to enhance the delivery of metformin XR by healthcare providers and patients. For instance, electronic health record systems can incorporate structured guidelines and the clinical decision support system (CDSS) to improve this condition without the need for additional work or study load. 31
As reported on previous studies,32 -35 good adherence to metformin generally enhances clinical outcomes. Earlier randomized controlled trials and methodical reviews reported that the XR formulation improves drug adherence by 10% because of its non-complicated use.16 -19 However, as regards practice of metformin XR, the pharmacists obviously neglected this advantage by choosing the medication in patients that prefer not to administer multiple doses daily, and prescribing the medication mostly due to patients’ preference rather than efficacy and safety of the drug. This indicates a potential lack of direction in adapting international evidence to prescribing settings in the UAE with respect to metformin XR, indicating a weak localization of international standards as implied in a previous report. 36
In addition, the statistical significant relationship between the poor knowledge of the prescribing of Metformin XR tablets and university of graduation. This calls for evidence-based continuous education at all levels in the study area. This deficiency of KAP of metformin XR is compatible with findings reported in previous studies performed among patients living with type 2 DM and clinicians . 22 Although there have been calls for continuous education of clinicians and prescription audits by clinical pharmacists, 33 this study demonstrates that the KAP of pharmacists can be similarly insufficient. Hence, evidence dissemination strategy may necessitate novel approaches and restructuring to methodically enhance the practical performance of pharmacists. This deficiency might also be attributed to poor adaptation of international guidelines to local situations. 36 The implementation of appropriate guidelines and the clinical decision support system (CDSS) may prevent this misconception among community pharmacists in UAE.
Regarding the attitude of the community pharmacists selected for the study, majority of the participants believed that the metformin XR tablets were more efficacious, while metformin IR are associated with greater adverse effects. 85.3% of the respondents preferred to prescribe metformin XR according to safety and efficacy, which is inconsistent with recent evidence .16 -19
The management of diabetes in a community pharmacy has resulted in better diabetes control and considerable enhancement of patients’ healthcare. 37 Community pharmacists can ensure regular screening, assessment, health education, and referral. 38 Given that patients with chronic illness visit pharmacies more regularly for their medicine refill, pharmacists can seize the opportunity to advise and educate the patient concerning his/her condition more proficiently. Thus, there is need for the pharmacist to be equipped with adequate knowledge about DM and its control since it is a vital requirement for the provision of sufficient data for patients. The contribution of pharmacists in patient care has lowered the volume of hospital admissions and patient visitations for medical emergencies. This explains the benefits of having appropriate knowledge, attitude and practice regarding the administering of metformin XR to DM patients. The role of pharmacists in diabetes management is critical for the diagnosis of patients, evaluation, referral, education and constant monitoring. 37
Limitations
Despite the negative outcomes, this study is encumbered by a number of limitations. This study is first limited by the fact that it focuses only on community pharmacists in the UAE. The settings can differ in other countries, which might affect the research outcomes. Thus, there is the need for external validation in other regions and countries. Nonetheless, this study clearly showed a gap in the knowledge of community pharmacists managing patients with type 2 DM using metformin XR. Secondly, the response rate is inaccessible due to the randomized sampling technique used. Moreover, accurate responses to the questionnaire are dependent on the honesty and trust of the respondents, which could affect the responses as they may be subject to certain bias. Regardless of these constraints, this study covers community pharmacists across Dubai, Abu Dhabi, and the Northern Emirates with no effect from a subgroup on the answers
Conclusion
Given their strategic position in the healthcare system, community pharmacists can directly monitor and advise rural patients with DM. This exerts a great chance for the community pharmacists to get involved deeply in the management as well as follow-up of patients with DM. However, adequate knowledge, attitude, and practice are required to effectively play this role. This study demonstrated a distinct gap in knowledge, attitude and practice pertaining to metformin XR among community pharmacists in the UAE. The good knowledge of the prescribing of Metformin XR tablets was significantly related to participants aged ⩾40 years with more than 10 years of experience. The community pharmacists need to enhance their practice by receiving accurate and reliable data to support their decision-making on the prescribing of metformin XR. The implementation of novel guidelines and evidence dissemination strategies may help bridge this gap.
Supplemental Material
sj-docx-1-end-10.1177_11795514231203913 – Supplemental material for Insights Into Metformin XR Pharmacotherapy Knowledge Among Community Pharmacists: A Cross-Sectional Study
Supplemental material, sj-docx-1-end-10.1177_11795514231203913 for Insights Into Metformin XR Pharmacotherapy Knowledge Among Community Pharmacists: A Cross-Sectional Study by Ammar Abdulrahman Jairoun, Sabaa Saleh Al-Hemyari, Moyad Shahwan, Nageeb Hassan, Sa’ed H Zyoud, Ammar Ali Saleh Jaber and Tariq Al-Qirim in Clinical Medicine Insights: Endocrinology and Diabetes
Footnotes
Acknowledgements
We would like to thank our colleagues for their participation in this study and their support of our work in this way; they helped us obtain results of better quality.
Declarations
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
