Abstract
Background:
It is still unelucidated how hormonal alterations affect developing organisms and their descendants. Particularly, the effects of androgen levels are of clinical relevance as they are usually high in women with Polycystic Ovary Syndrome (PCOS). Moreover, it is still unknown how androgens may affect males’ health and their descendants.
Objectives:
We aimed to evaluate the multigenerational effect of prenatal androgen excess until a second generation at early developmental stages considering both maternal and paternal effects.
Design And Methods:
This is an animal model study. Female rats (F0) were exposed to androgens during pregnancy by injections of 1 mg of testosterone to obtain prenatally hyperandrogenized (PH) animals (F1), leading to a well—known animal model that resembles PCOS features. A control (C) group was obtained by vehicle injections. The PH-F1 animals were crossed with C males (m) or females (f) and C animals were also mated, thus obtaining 3 different mating groups: Cf × Cm, PHf × Cm, Cf × PHm and their offspring (F2).
Results:
F1-PHf presented altered glucose metabolism and lipid profile compared to F1-C females. In addition, F1-PHf showed an increased time to mating with control males compared to the C group. At gestational day 14, we found alterations in glucose and total cholesterol serum levels and in the placental size of the pregnant F1-PHf and Cf mated to F1-PHm. The F2 offspring resulting from F1-PH mothers or fathers showed alterations in their growth, size, and glucose metabolism up to early post-natal development in a sex-dependent manner, being the females born to F1-PHf the most affected ones.
Conclusion:
androgen exposure during intrauterine life leads to programing effects in females and males that affect offspring health in a sex-dependent manner, at least up-to a second generation. In addition, this study suggests paternally mediated effects on the F2 offspring development.
Keywords
Introduction
The developmental origin of health and disease (DOHaD) hypothesis proposes that adverse intrauterine conditions lead to alterations in organisms’ developmental trajectory as a mechanism of adaptation to an anticipated specific post-natal environment. These possible adaptations during development are made based on the parental signals about their health and physical state, including maternal health in pregnancy and paternal and maternal health at the time of conception.1,2 Thus, the environmental influences during early development can lead to a high risk of developing diseases during adulthood, as for example increasing the risk of chronic diseases.1,3 In this regard, it has been shown that epigenetic information is a key factor in intergenerational inheritance and that the epigenome is susceptible to modifications in response to environmental changes and factors such as diets and stress.4,5 It has been reported that epigenetic changes can compromise offspring postnatal health and, both paternal and maternal information seems to play important roles in offspring development and can increase disease susceptibility in the offspring. However, the mechanisms are not yet fully understood. Although it is widely known that the maternal environment during gestation affects offspring development. 5 In the case of paternal inheritance, less is known in this regard. Still, in the last years, it has been described that besides the paternal effects on the placenta that can condition the maternal environment and, thus, affect the developing organism 6 ; sperm epigenome, including DNA methylation marks, histone’s modifications, and mainly non-coding RNAs, can be transmitted to the zygote and contribute to fertilization, embryo development and have been related to offspring postnatal health. In addition, some studies have shown that paternal exposure to different diets or chemical compounds can affect offspring health.4,7-10
Evidence indicates that early life experiences may affect the immediate developing organism and subsequent generations. These intergenerational and transgenerational effects may be, at least, partly explained by nutritional, hormonal and different exposures prior to and during conception due to programing effects. In this regard, different epigenetic and physiological adaptations have been proposed as possible mechanisms.11,12
Polycystic ovary syndrome (PCOS) is one of the main endocrine disorders in women of reproductive age. It negatively impacts fertility and is related to several metabolic alterations, including insulin resistance, which affects up to 70% of PCOS women. 13 PCOS is considered a complex multigenic disorder and although a family aggregation has been described, genetic basis only accounts for up to 10% of its heritability.14,15 Therefore, a strong environmental susceptibility with a possible epigenetic inheritance has been proposed, including mechanisms during fetal and early post-natal life on PCOS susceptibility.14,16 In this regard, it has been reported that daughters born to PCOS mothers may develop PCOS-like phenotypes and related features.17-20 Moreover, recently it has been described that sons of PCOS mothers may also develop PCOS-related alterations such as metabolic and reproductive altered phenotypes.21,22 It has been proposed that these features may be related to programing effects due to an altered in utero milieu, such as androgen excess. 15 Although a maternal contribution to PCOS-like symptoms development in different animal models has been widely studied23-26 a possible paternal contribution to PCOS development is still poorly explored.
Different androgenized rodent models are used to study the pathophysiology underlying human PCOS, including reproductive and metabolic dysfunction. By using a murine model of prenatal hyperandrogenization, Abruzzese et al have previously shown that in utero testosterone exposure in pregnant rats (F0) leads to 2 different PCOS-like reproductive phenotypes. 25 It has also been shown that the animals exposed to androgens display endocrine, metabolic and reproductive alterations in females’ first immediate exposed generation (F1). These disturbances include altered steroid levels and ovarian steroidogenesis and folliculogenesis,27,28 impaired uterine histomorphology and growth with an increased uterine oxidative stress and inflammatory status.29,30 Together with reproductive alterations, metabolic derangements were also reported as for example systemic dyslipidemia, insulin resistance, and inflammatory status in the adult offspring.30,31 In addition, other authors have also reported that prenatal androgen exposure leads to alterations in the placenta32,33 and neuroendocrine derangements such as an increase in LH pulse frequency and an altered hypothalamic gene expression. 34
Moreover, it has been shown that prenatal androgen exposure affects the male reproductive system displaying altered testis weight, decreased testosterone levels, and sperm count and motility. 35 These findings suggest that prenatal androgen exposure leads to alterations in the first generation’s reproductive system of both females and males. In this regard, the intrauterine period of life is known as a window of susceptibility to programing effects. The developing gonads of the organisms can be affected by environmental factors or alterations in the intrauterine milieu, inducing changes in the germ cells. Thus, in utero exposure in one generation could lead to multigenerational and even transgenerational alterations.2,11,12 Regarding androgenic programing effects, it has been described that prenatal exposure to androgens leads to altered phenotypes during postnatal stages. 15 However, little is known about the possible contribution to subsequent generations.
Taking together this evidence, we hypothesize that prenatal androgen exposure affects the immediate subsequent generation (F1) fertility and leads to multigenerational effects on the second generation (F2) development. Therefore, we aimed to evaluate the multigenerational effect of prenatal androgen until a second generation at early developmental stages. Also, we propose to analyze the impact of androgen exposure on maternal and paternal fertility and its contribution to early offspring development.
Materials and Methods
Experimental animal model and ethics statement
Virgin female rats of Sprague-Dawley strain obtained from the animal facilities from the Faculty of Veterinary Sciences (University of Buenos Aires) were mated with fertile males of the same strain. The experimental designed followed is presented in Figure 1. Three females and one male were housed together under controlled conditions of light (12 hours light: 12 hours dark) and temperature (23°C-25°C). Animals received food (Cooperación SRL, Argentina) and water ad libitum. The vaginal smear was followed up for 7 days before mating to check the estrous cycle. Female animals were mated to males starting at the proestrus or estrous stages for a period no longer than 3 days. The morning when spermatozoa were found in the vaginal smear was designated gestational day 0. Between days 16 and 19 of pregnancy, female rats (Filial zero, F0) were hyperandrogenized as previously described. 25 Briefly, pregnant rats (N = 10) received subcutaneous injections of 1 mg of free testosterone (Sigma Chemical Co. St. Louis, MO, USA) dissolved in 100 µL corn oil on days 16, 17, 18, and 19 of pregnancy, whereas the control group (N = 10) received the same number of injections containing only 100 µL of corn oil as vehicle. Animals were randomly allocated to PH or control groups. The offspring (Filial 1, F1) of females treated with testosterone during pregnancy was designated as the prenatally hyperandrogenized group or PH group (PH females [PHf] and PH males [PHm]), whereas the offspring of animals receiving vehicle composed the control group (C; control females [Cf] or control males [Cm]). The treatments described did not modify the spontaneous term labor, the female-to-male offspring ratio, or the number of pups per litter. Under the conditions of our animal facility, spontaneous term labor occurs on day 22 of gestation. Pups were culled from litters to equalize group sizes (9 pups/mother). Female pups were separated from males at 21 days of age, and all animals were randomly assigned to each assay, which was carried out with the same number of animals from each randomly selected littermate. As previously described, PHf rats displayed 2 phenotypes: irregular ovulatory (49%-50% females with irregular estrous cycle) or anovulatory (38%-43% females presented anovulatory cycles).25,28 Only irregular ovulatory females were used for mating and obtaining the second generation. As already reported, anovulatory animals do not mate without stimulation. 29 Therefore, we let animals mate spontaneously to avoid ovarian stimulation and/or assisted reproductive techniques effects as confounding variables,36,37 we let animals to mate spontaneously. At adulthood, F1-PHm and F1-PHf were mated with control females or males of the same strain, respectively. In addition, F1 control animals (females and males) were mated to obtain the progeny of the control group. Two females and males per litter were selected for mating assays and carefully avoiding breeding animals from a same litter.

Experimental design. Pregnant females (F0) were exposed to androgens or vehicle injections to obtain F1-PH and F1-control animals, respectively. The F1 animals were mated to obtain a second generation and evaluate maternal and paternal contribution, independently.
Pregnant females were maintained in individual cages. Randomly, one group of pregnant females was euthanized at gestational day 14 (GD14) of pregnancy, while another group was left to deliver. The second generation (F2) was euthanized at post-natal day six (PND6) (Figure 1). All the animals were identified per cage and by a color code. Adult animals were anesthetized with carbon dioxide and killed by decapitation. Newborns were killed by decapitation, following guidelines for rodent euthanasia. All the procedures involving animals were conducted in accordance with the 1996 Animal Care and Use Committee of the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET). The present study was approved by the Ethics Committee of the Facultad de Medicina, Universidad de Buenos Aires, Argentina (UBA-Fmed-CICUAL resolution number 3849/2020).
Metabolic assessment of the F1 animals before mating
To evaluate the metabolic status of the F1 at 90 days of life, the animals were weighted, and an intraperitoneal glucose tolerance test (IPGTT) was done. IPGTT was analyzed in females and males in both F1-C and F1-PH animals. Blood samples were taken after 0, 30, 60, 90, and 120 minutes post intraperitoneal injection of 2 g dextrose/kg body weight.
Moreover, before mating, in order to assess female’s health outcomes, we evaluated female’s anogenital distance and lipid profile. In this regard, serum cholesterol and triglycerides from females from the F1-C and F1-PH groups were analyzed using a colorimetric-enzymatic assay of Weiner Lab (Argentina) following manufacturer instructions.
Mating, fertility and fecundity index of F1
At adult age (90 days of life), F1 females from the F1-C and F1-PH ovulatory groups were mated with control males. In addition, males from the F1-PH group were mated with control females. The morning when spermatozoa were found in the vaginal smear was designated gestational day 0. The vaginal smear was followed up for 7 days, starting in the proestrus stage. A mating index was determined by monitoring the presence of vaginal plugs the morning after mating. A mating time was determined as the period (in days) until we observed the presence of vaginal plugs during the coupling.
The fertility index was determined as the number of females that delivered live pups 22 days after mating (presence of spermatozoa in the vaginal smear) over the total of females. The fecundity index was determined by as the number of females that presented a vaginal plug after mating and delivered live pups, over the total of females mated.
Maternal and F2 embryos parameters during gestation period until gestational day 14 (GD14)
On day 14 of pregnancy, a group of F1 pregnant females from each of the 3 experimental mating groups (Cf × Cm; PHf × Cm: Cf × PHm) were randomly selected, anesthetized with CO2 and euthanized. The blood from the mothers was collected to evaluate serum cholesterol and triglyceride levels by colorimetric-enzymatic assays (Weiner Lab, Argentina). The uterus was removed to determine the uterine weight, the number of implantation sites, the resorption sites (to assess the resorption rate), the embryos weight, the embryos crown to rump distance and the placental size.
Morphometric studies
On day 14 during pregnancy, embryos and placentas were photographed and were accurately measured to assess development, utilizing tools (distance and area) of the Amscope Software 3.7 (United Scope LLC, Irvine, CA, USA). The mean areas of each placental zone were evaluated in square centimeters (cm2). The mean longitudes of each embryo were expressed in centimeter.
Post implantation loss rate
The post implantation loss rate was calculated at day 14 of pregnancy by the following formula: number of implantation sites (viable and absorbed) − number of viable fetuses × 100/number of implantation sites (viable and absorbed). 38
Maternal parameters until day 6 after parturition
Another group of F1 pregnant females from each of the 3 experimental groups obtained after mating (Cf × Cm; PHf × Cm: Cf × PHm) were followed up during pregnancy, delivery and until day six after parturition. We determined the maternal weight and glucose levels. In the mornings of days 0, 7, 14, and 20, pregnant females were weighted, and blood glucose levels (food ad libitum overnight) were monitored using a specific glucose meter (OneTouch Ultra—Johnson & Johnson), and the values were expressed in milligrams per deciliter (mg/dL).
In addition, after delivery (post-partum), on day 6, the mothers were euthanized and the blood was collected to evaluate serum cholesterol and triglycerides levels by colorimetric-enzymatic assays (Weiner Lab, Argentina). We also assessed the maternal blood glucose levels by a colorimetric assay (Weiner Lab, Argentina) and registered the maternal body weight and the ovarian and uterine weights.
Maternal capacity was also assessed. This parameter is an evaluation of the ability of the mother as a caregiver, and it was evaluated by assessing the survival of the pups during the first 96 hours after birth.
F2 offspring assessment until post-natal day six (PND6)
After birth, F2 neonates were euthanized at PND6 by decapitation with surgical scissors. The size of the offspring was evaluated, and we followed up post-natal death until day 6. Pups were weighted on post-natal days 3 and 6. Several parameters were evaluated on PND6 in the offspring, after determination of the sex by visual examination. We assessed the F2 offspring basal glucose levels by colorimetric-enzymatic kit (Weiner Lab, Argentina), the length of the pups, the anogenital distance, and metabolic features as the serum levels of cholesterol and triglycerides by colorimetric-enzymatic assays (Weiner lab, Argentina).
Statistical analysis
Data are presented as mean ± standard error of mean deviation (SEM). In Table 2, for a better visualization of the phenomena, some parameters are expressed as follows: “pups born” and “post-natal death” parameters are expressed as mean and maximum-minimum range. In addition, the data corresponding to “maternal capacity” is presented as median and maximum-minimum range. Data normality was checked using the Shapiro-Wilks test. The equality of variance was tested using the Levene’s test. Outliers analysis was performed by box-plot data visualization. The Log10 transformation was used to normalize the distribution of data allowing the statistical analysis when required. Data were backtransformed for presentation in the figures. The t-students test or one-way ANOVA with post hoc Tukey test were used to compare between 2 or 3 groups, respectively. Differences were considered statistically significant at P < .05. All experiments were performed with at least 4 to 5 animals per group based on previous results from our lab for the animal model. All analyses were performed using the Instat program (GraphPad Software, San Diego, CA, USA) and Infostat 39 (Universidad de Córdoba, Argentina).
Results
Prenatal hyperandrogenism effects on F1 metabolic features
Prenatal hyperandrogenization affected F1-PHf response to glucose oral test. The IPGTT showed that prenatal hyperandrogenism altered glucose in F1-PHf as compared to F1-Cf (Figure 2A, P < .05),thus confirming our previous results.25,30 No differences were found when comparing F1-PHm to F1-Cm (Figure 2B, P > .05). In addition, the area under the curve was increased for the F1-PHf as compared to F1-Cf animals (Figure 2C, P < .05) but no differences were found for the area under the curve of the IPGTT between F1-PHm and F1-Cm (Figure 2D, P > .05). In addition, no differences were found regarding body weight, nor in the anogenital distance in adulthood (90 days of age) in F1-PHf nor in F1-PHm as compared to animals of the control group (Figure 2E-H P > .05).
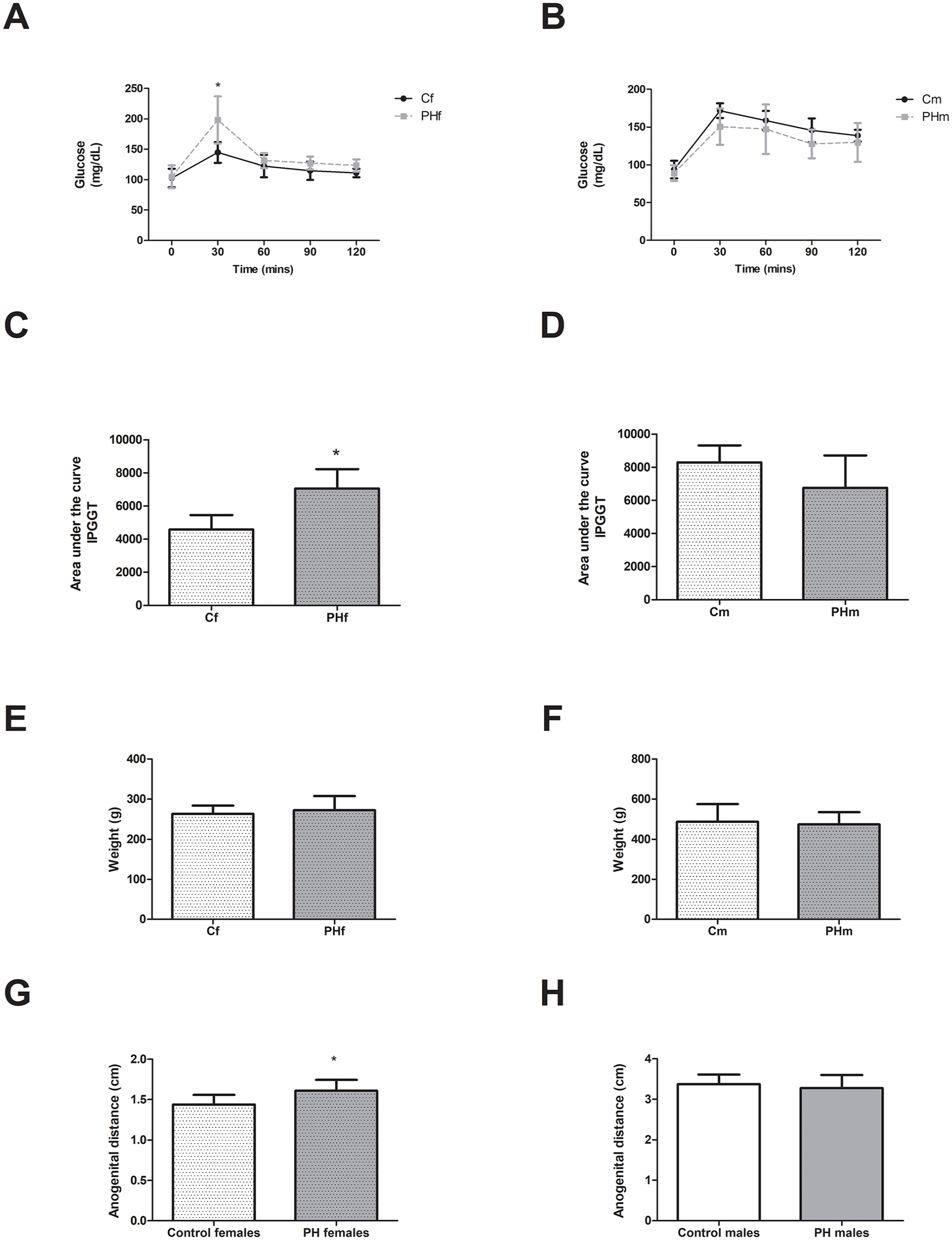
Prenatal hyperandrogenism effects on F1 animals’ glucose metabolism and body weight (A) IPGTT evaluated in F1 female (f) offspring of control (C) and prenatally hyperandrogenized (PH) groups. Blood samples followed by intraperitoneal injection of 2 g dextrose/kg body weight were collected at 0, 30, 60, 90, and 120 minutes post injection for IPGTT (=4-5 per group). (B) IPGTT evaluated in F1 male (m) offspring of C and PH groups. Blood samples followed by intraperitoneal injection of 2 g dextrose/kg body weight were collected at 0, 30, 60, 90, and 120 minutes post injection for IPGTT (=4-5 per group). (C) Area under the curve after IPGTT evaluated in F1 female (f) offspring of C and PH groups (=4-5 per group). (D) Area under the curve after IPGTT evaluated in F1 male (m) offspring of C and PH groups (=4-5 per group). (E) Weight at 90 days of age of F1 females (n = 7 per group). (F) Weight at 90 days of age of F1 males (n = 7 per group). (G) anogenital distance F1 females (n = 7 per group). (H) anogenital distance of F1 males (n = 7 per group). *P < .05. Values are mean ± SEM by t-student test.
Prenatal hyperandrogenism effects on F1 maternal parameters
Table 1 shows the analyzed parameters of the F1 females before mating. The F1-PHf presented increased serum triglycerides and total cholesterol levels as compared to F1-Cf (P < .05). In addition, the F1-PHf also displayed increased anogenital distance compared to F1-Cf (P < .05).
Metabolic maternal parameters of F1 female animals before conception.

Data presented as mean ± SEM, *P < .05 by t-student test. N = 7 animals per group. Statistically significant differences are highlighted in bold font.
Prenatal hyperandrogenism effects on F1 parental reproductive parameters
To evaluate the effects of prenatal androgen exposure on F1 female and male fertility we assessed the mating, fecundity and fertility indexes and the time to mating for the F1 considering PHf, PHm and control animals to obtain the F2 (Figure 3).

Prenatal hyperandrogenism effects on F1 animals’ reproductive outcomes. (A) Mating index was determined as the number of females with vaginal plug over the total of females used in each experiment. (B) Fecundity index was determined by as the number of females that presented a vaginal plug after mating and delivered live pups, over the total of females mated. (C) Fertility index was determined as the number of females that delivered live pups over the total of females used in each experiment. (D) Mating time was determined as the period (in days) until we observe the presence of vaginal plugs during the coupling.
F1-PHf and F1-PHm showed no differences in the mating index nor in fecundity and fertility index as compared to the F1-C animals (Figure 3A-C, respectively, P > .05). Our results showed that F1-PHf showed an increased time to mating with control males as compared to the control group (Figure 2D, P < .05). In the case of F1-PHm no differences in the mating time with control females was found as compared with the control group (Cf × Cm) (Figure 3D).
Maternal and paternal effects on metabolic outcomes during gestation
Prenatal hyperandrogenism affected maternal metabolism during gestation. We found no alterations in weight gain during pregnancy in F1-PH females mated to control males (PHf × Cm) nor a paternal contribution to the females mated to F1-PH males (Cf × PHm) as compared to the control group (Cf × Cf) (Figure 4A, P > .05). Regarding glucose levels during pregnancy, we found no differences at the start of the pregnancy and at days 7 and 20 of pregnancy in maternal glucose levels in either of the studied groups. However, at day 14 of pregnancy, F1-PH females mated to control males (PHf × Cm) showed increased glucose levels as compared to control females that were mated with both F1-PHm and Cm (Figure 4B P < .05).

Maternal and paternal effects on metabolic outcomes during gestation. (A) The curve represents the mean growth rates of the analyzed groups: Cf × Cm, PHf × Cm, Cf × PHm (n = 6 per group). (B) Glucose levels analyzed at different timepoints during gestation (0, 7, 14, and 20 days) (n = 5-6 per group); *P < .05 by ANOVA test in the PHf × Cm as compared to control group (Cf × Cm). (C) Serum levels of total cholesterol, a versus b P < .05 by ANOVA test (n = 5-6 per group), (D) Serum levels of triglycerides (n = 5-6 per group). Values are mean ± SEM and analyzed by ANOVA test with post-hoc Tukey test.
Regarding lipid profile, at gestational day 14, F1-PH females mated to control males (PHf × Cm) and control females that were mated with F1-PH males (Cf × PHm) presented increased serum levels of total cholesterol as compared to those control females mated to control males (Cf × Cm) (Figure 4C, P < .05). No differences were found regarding triglycerides levels in either of the analyzed groups (Figure 4D, P > .05).
Maternal and paternal effects on pregnancy development
No maternal nor paternal effects were found when evaluating implantation sites number or resorption sites number in females mated to F1-Cm (PHf × Cm) or F1-PHm (Cf × PHm) as compared to the control group (Cf × Cm) (Figure 5A and B, respectively, P > .05). In addition, no differences were found regarding the ratio between uterine weight and the number of implantation sites among the 3 analyzed groups (Figure 5C, P > .05).

Maternal and paternal effects on pregnancy development. (A) Number of implantation sites evaluated at gestational day 14 (GD14); (B) embryo resorption at GD14 was assessed as the number of implantation loss sites over the total number of visualized implantation sites; (C) uterine weight to number of implantation sites ratio; (D) placental area; (E) embryo crown rump length; (F) embryo weight. Values are mean ± SEM and analyzed by ANOVA test with post-hoc Tukey test, a versus b P < .05. N = 5-6 per group.
Prenatal hyperandrogenism affected the placental area. Our results showed that the placental area was affected by both maternal and paternal effect in the groups PHf × Cm and Cf × PHm, which showed higher placental area than that of the control group (Cf × Cm) (Figure 5D, P < .05).
The analysis of embryo parameters showed that embryos from F1-PH males display a longer embryo crown rump length than embryos of F1-PH mothers and F1 control parents (Figure 5E, P < .05). Regarding embryonic weight, no differences were found among the analyzed groups (Figure 5F, P > .05).
Post-partum and maternal parameters
After parturition, at day six post-partum (PND6), we analyzed different maternal parameters. We found no differences among the 3 groups in F1 maternal weight (Figure 6A, P > .05). Glucose levels and serum triglycerides levels were increased in the F1-PHf group as compared to F1-Cf that were mated to both C and PH males (Figure 6B and C, respectively, P < .05). Regarding total cholesterol levels, no differences were found in the analyzed females after parturition (Figure 6D, P > .05). Moreover, no differences were found in ovarian and uterine weight of the analyzed females (Figure 6E and F, P > .05).

Post-partum maternal (F1 animals) parameters. Maternal parameters evaluated at day 6 post-partum: (A) weight, (B) glucose levels, (C) serum levels of triglycerides, (D) serum levels of total cholesterol, (E) average ovarian weight, and (F) uterine weight. All the parameters were analyzed in F1 females corresponding to the different groups of breeding as previously described: Cf × Cm, PHf × Cm and Cf × PHm. Values are mean ± SEM. a versus b P < .05 by ANOVA test with post-hoc Tukey test.
Furthermore, we found no differences in the number of pups born per mother, the post-implantation loss and the post-natal death of the offspring among the analyzed groups (Table 2). When analyzing maternal capacity, we found no differences among the groups. However, the F1-PHf showed a tendency to a decreased maternal capacity as compared to F1-Cf (P = .0521).
Maternal (F1) and litter (F2) evaluation.

Abbreviations: C, control; f, female; m, male; PH, prenatally hyperandrogenized.
Data presented as mean and maximum to minimum range for pups born and post-natal death parameters. In the case of maternal capacity, data is presented as the median and maximum to minimum range. The post-implantation loss is expressed as mean ± SEM. Statistical tendency is highlighted in bold font.
P < .05 by ANOVA and post-hoc Tukey test.
F2 parameters
We studied the intergenerational effect of prenatal androgen exposure to a second generation (F2). We found no differences in body weight at birth among the F2 offspring (Figure 7A, P > .05). At PND3, we found that the average litter weight of the offspring of F1-PHf was lower than the offspring of the F1-C group and F1-PHm (Figure 7B, P < .05).

F2 litter parameters at early post-natal development. Effects of prenatal androgenization on the second generation (F2) (A) average litter weight at birth; (B) average animals’ weight at post-natal day three (PND3). All the parameters were analyzed on the litters’ of F1 mating groups as previously described: Cf × Cm, PHf × Cm and Cf × PHm. Values are mean ± SEM. a versus b P < .05 by ANOVA test. N = 5-6 per group.
At PND6, we studied both female and male F2 offspring. The proportion of males and females born to F1-PHf, F1-PHm and F1-C animals showed no alterations (data not shown). We found that both female and male offspring of the F1-PHf showed decreased body weight as compared to the offspring of F1-C animals (Figure 8A, P < .05). Moreover, the male offspring of F1-PHm showed a decreased body weight as compared to the males born to control animals (Figure 8A, P < .05).

Effects of prenatal androgenization on the second generation (F2) at early post-natal development. The figure shows the evaluated parameters of the F2 offspring at post-natal day 6 (PND6) considering the designed mating groups (F1 Cf × F1 Cm; F1 PHf × F1 Cm; F1 Cf × F1 PHm, N = 5-6 per group.). All the data was analyzed separately considering the F2 sex (females and males): (A) body weight, (B) body length, (C) anogenital distance, (D) glucose levels, (E) total serum cholesterol, and (F) serum triglycerides. Values are mean ± SEM. *Indicates P < .05 by ANOVA test with post-hoc Tukey test.
The offspring (F2) of F1-PH mothers showed decreased body length as compared to the offspring of the control group and those of F1-PH fathers (Figure 8B, P < .05) and the F2 female offspring of F1-PHf mothers showed decreased anogenital distance as compared to the offspring of the control group (Figure 8C, P < .05).
Regarding glucose levels, we found that the female offspring of the F1-PHf showed higher glucose levels than the offspring of the F1 control group (Figure 8D, P < .05). No differences were found when analyzing the male offspring of F1-PHf and F1-PHm as compared to the male offspring of the control animals (Figure 8D, P > .05).
In addition, no differences were found when analyzing serum total cholesterol and triglycerides levels of the offspring of F1-PHf or F1-PHm as compared to the F2 offspring of control animals (Figure 8E and F, respectively, P > .05).
Discussion
Adverse intrauterine conditions can affect the developing organisms leading to negative effects on the offspring health and contributing to post-natal disorders. It has been proposed that some complex pathologies of rising prevalence in modern societies, such as insulin resistance, type 2 diabetes and cardiometabolic diseases, have a strong environmental influence and that the intrauterine period of life plays a crucial role in their development. 40 Androgen excess is recognized as a major contributing factor to the development of PCOS, 41 which is related to several metabolic alterations, including altered glucose, insulin and lipid metabolism.42,43 It has been widely studied in different animal models (including monkeys, sheep, rats and mice) that a direct exposition during intrauterine life to androgen excess leads to the development of PCOS-like features together with related-metabolic derangements.23-25,30,44-46 During intrauterine life, if the developing gonads of the organisms are affected by environmental factors or alterations in the intrauterine milieu, changes in the germ cells could be induced and lead to multigenerational alterations affecting also the following generation. Even more, transgenerational effects have also been described in developmental programing models. 47
Regarding PCOS and androgen excess-related inheritance, it has been recently reported transgenerational effects by maternal inheritance in an animal model of prenatal androgen excess.46,48 However, a possible paternal inheritance related to the syndrome remains poorly explored. Recently, in other animal models of PCOS, it has also been shown a possible transgenerational transmission of PCOS-like features.49,50 Thus, this evidence suggests that PCOS-related features could be transmitted across generations and that androgens can act as programing factors leading to multigenerational and even transgenerational effects.
Different animal models have been proposed to simulate PCOS features. However, the prenatally androgenized animal models are among the most accepted as they lead to permanent reproductive and metabolic features as seen in PCOS women and propose a possible mechanistic approach to PCOS development. 12 In this regard, different animal models of fetal programing by androgen excess have been developed to evaluate the impact of prenatal testosterone and DHT, a non-aromatizable androgen, on the offspring. Evidence shows that those animal models lead to several differences regarding the programing effects, which may be related to the fact that DHT is a more potent androgen and that as testosterone could be converted into estradiol, thus, some effects related to PCOS-like features could be estradiol-dependent51,52 as has been suggested by different reports. 26 In addition, it has also been reported that the effects on the animal phenotypes are androgen-dose dependent. 53 Therefore, in this study, we proposed to evaluate the effects of prenatal testosterone exposure on multigenerational effects by both maternal and paternal lineage independently by exposing animals (F0) to a dose of 1 mg of testosterone between days 16 to 19 of pregnancy. As previously reported, this dose leads to PCOS-like phenotypes in females and does not alter vaginal opening nor results in defeminization, which has been reported in animal models using higher doses of this androgen.25,27,29,54 Furthermore, we also aimed to analyze the effects on the second generation (F2) development during intrauterine life and at early development. The results of this study could contribute to evaluating early signs of possible multigenerational developmental programing effects caused by androgens affecting offspring health, thereby opening a possible window of opportunity for clinical interventions.
By using a well-established model of prenatal hyperandrogenization,25,27-29 we evaluated the effects of prenatal androgen exposure on female and male offspring of the first generation (F1). Although it has been widely studied the direct effects of prenatal androgen excess in females,25,45,55 the effects on males are less studied, but some studies have reported metabolic and reproductive alterations.35,56 Our results showed that in females, prenatal androgen excess affected the glucose tolerance and lipid profile without affecting their body weight, thus, confirming our previous results.25,28,30 When studying the F1 male offspring, we found no effects of prenatal testosterone exposure on F1-males’ glucose tolerance or body weight. These results are in accordance with previous reports that show that prenatal testosterone exposure did not affect male offspring’s body weight in adulthood. 35 Therefore, these results suggest that prenatal hyperandrogenization effects are sex-dependent and more significant in females than males. These findings are in agreement with those reported by Lazic and colleagues, 57 who have already suggested that androgen developmental programing effects of some metabolic traits would be sex-specific. In a recent study, contrary to our results, Risal et al 58 have reported that exposure to the androgen DHT in utero leads to obesity and an altered metabolic profile including and altered glucose tolerance. Thus, these results suggest that, as also reported for females, DHT fetal programing effects lead to a worse metabolic profile than those caused by prenatal testosterone exposure.
Besides the effects of androgen excess on metabolic outcomes, its impact on reproductive traits and fertility has already been described. Therefore, we evaluated different fertility-related outcomes in both F1-PH females and males. As it has been already reported that anovulatory PHf could not achieve natural conception, 29 we only worked with ovulatory females for this study. In addition, to avoid possible confounding variables, in this study, we only looked for natural conception as it has been reported that the use of assisted reproductive technologies and artificial hormonal stimulation lead to developmental programing effects.59,60 Our results showed that both PH females and males could mate and get a successful pregnancy. However, F1-PHf showed an increased time to mating compared to control females in order to achieve conception. This could be explained as these animals present irregular estrous cycles and altered hormonal parameters that could affect and delay the mating.
Although all animals could conceive, some metabolic parameters were altered in pregnant females. In this regard, the F1-PHf presented increased glucose levels at GD14 (equivalent to human second trimester 61 ). In addition, we found that the total cholesterol levels during pregnancy were increased in both F1-PHf and those females mated to F1-PHm (belonging to the mating group Cf × PHm). It is known that cholesterol levels rise during pregnancy in humans and rats, 62 and that this is related to sex-steroid production. 63 Moreover, increased cholesterol levels have been related to pregnancy-related complications such as preeclampsia and gestational diabetes, which are seen in PCOS pregnant women.63,64 During pregnancy, the placenta is the major organ involved in the control of oxygen and nutrient exchange but is also involved in steroid production. 65 Surprisingly, our results showed that PH females and females mated to PH males presented an increased placental area compared to control animals. It has been described that large placental size is related to adverse intrauterine conditions and has also been associated with gestational diabetes and altered offspring parameters.66,67 Remarkably, besides PH females, who display PCOS-like features as previously reported,28,29 we showed that control females mated to F1-PHm also present alterations during pregnancy. These results suggest that there is a paternal-mediated effect on the maternal environment. In recent years, attention has been paid to epigenetic effects in the inheritance related to programing effects across different generations. In addition, in the case of maternal inheritance, it has also been described that alterations in the maternal environment while pregnant can lead to alterations in the placenta and thus, to alterations in nutrient and hormone exchange with the developing fetuses, thus suggesting a possible mechanism that involved placental adaptations in programing effects. 68 In the case of paternal mediated programing effects, epigenetic mechanisms could transmit these effects through male gametes in those males exposed to androgens during prenatal life. In this regard, it has already been reported that paternal experiences, previous to conception, can affect the offspring health and impact placental and fetal growth.7,69,70 In accordance with this evidence, we also found that embryos that resulted from a mating between F1-PHm and control females presented a longer crown-rump length than that of the embryos of control animals mating, thus suggesting a paternal-mediated inheritance. In addition, a recent study, 58 showed that males prenatally exposed to androgens and their descendants up to the 2 following generations present a different pattern of sperm small-non coding RNAs as compared to control males. Despite the possible direct male-derived epigenetic effects on the offspring, it has also been described that there is a paternal effect, probably mediated by epigenetic factors, on placental functions. Thus, alterations inherited from fathers to offspring could also be related to placental-mediated effects. 6 Despite the recent advances related to the mechanisms of inheritance related to programing effects, further studies are needed to elucidate these phenomena.
After delivery, females from the PH group presented altered metabolic parameters, including increased triglycerides and glucose levels and we found a tendency (P = .0521, Table 2) to decrease maternal capacity. Thus, these results suggest that the altered metabolic profile of these females persists even during lactation. As early post-natal development is another window of programing, it would be important to further analyze the possible alterations of these metabolic traits on the F2 offspring development. In this regard, our results showed that the offspring of F1-PHf was smaller in body weight than the offspring of control animals at day 3 of post-natal life. In addition, both F2-males and F2-females born to F1-PHf showed a decreased body weight and length at PND6, thus showing altered developmental trajectories of these animals. Only F2-females born to PH mothers presented altered glucose levels and anogenital distance. Therefore, confirming a sex-specific action of developmental programing effects that persists during 2 generations. In the case of the F2 animals born to F1-PHm, although they displayed an increased length during embryonic development, no signs were detected in post-natal life. However, F2-males born to PH fathers showed a high body weight in early post-natal life.
Taking together, the results of this work show that androgen exposure during prenatal life differentially affects the analyzed parameters of F1-offpsring in a sex-dependent manner. To the best of our knowledge, this is the first report that evaluates the programing effects caused by prenatal testosterone exposure up to a second generation and considers maternal and paternal effects independently. In this regard, our results suggest that there are maternal and paternal effects that differently affect the development of the second generation. In agreement with our results, Zhang et al 49 have shown, by exposing animals to the androgen DHEA during postnatal life, that maternal and paternal effects differentially affect offspring development. In addition, Zhang et al 49 showed that the offspring resulting from the mating of females and males ancestrally exposed to the androgen DHEA (during postnatal life) presented more reproductive and metabolic alterations as compared to the animals born to only one parent ancestrally exposed to DHEA. Taken together, these results suggest a possible synergic effect between maternal and paternal inheritance in the context of parental androgenization. In this study, we aimed to evaluate the maternal and paternal effects in independently. Thus, further studies are required to deepen the possible synergic effects of offspring born to mothers and fathers prenatally exposed to androgens.
In addition, more studies are needed to clarify the possible epigenetic and physiological adaptations that could contribute to the multigenerational inheritance effects mediated by androgen exposure.
In conclusion, the results of this study show that in utero exposure to androgen excess leads to programing effects in both females and males, which impact the animals’ health in a sex-dependent manner for at least 2 generations and also affects pregnancy and developmental outcomes. These results also highlight the important role of paternal inheritance in growth trajectories affected by an exposure to intrauterine androgen excess.
