Abstract
Background:
While the fast extension of combination antiretroviral therapy (cART) has resulted in significant increases in life expectancy, disorders such as cardiometabolic syndrome (CMetS), which have received less attention, are becoming a major concern in HIV/AIDS patients (PLWHA).
Objectives:
The purpose of this research was to identify biomarkers and determine the prevalence of CMetS in PLWHA using the National Cholesterol Education Program (NCEP) and the International Diabetes Federation (IDF) tools.
Methods:
Between January 2019 and February 2021, a hospital-based study of HIV-infected patients (n = 288) was conducted. The data were analyzed using binary logistic regression. To control the effect of confounders, independent variables with a P-value of <.20 in the bivariate logistic regression were incorporated into multivariate logistic regression. Statistical significance was defined as a 95% confidence interval and a P-value of less than .05.
Results:
The risk of CMetS increased twofold as age increased each year (P = .009), 1.2 times as the age at which cART began increased (P = .015), and 6 times with 1 or more co-morbidities (P = .028), according to the NCEP tool. Furthermore, significant NCEP-CMetS correlations were produced by a rise in diastolic blood pressure (P < .001) and cART duration (P = .006). Male gender was 99.9% less likely to be related to CMetS using the IDF tool, and the risk of CMetS increased fourfold with each unit increase in waist circumference (P < .001). Triglycerides and blood type “A” have been found to have substantial relationships with CMetS using both techniques.
Conclusion:
According to the study, CMetS was found to be common in PLWHA. Age, time on cART, age when cART started, gender, co-morbidities, waist circumference, and diastolic blood pressure were all revealed to be significant predictors of CMetS. Triglycerides and blood type “A” were the only biomarkers found to be significant with CMetS using both the NCEP and IDF tools.
Keywords
Introduction
The introduction of combination antiretroviral therapy (cART) has considerably benefited People Living with HIV/AIDS (PLWHA) in terms of quality of life, increased survival, and psychological boost. However, cART management has been jeopardized due to drug and disease-related issues.1-6 Cardiometabolic syndrome (CMetS) is a group of cardiac and metabolic disorders characterized by changes in common biomarkers such as impaired glucose tolerance, insulin resistance, high serum “triglycerides” (TG), low “high-density lipoprotein” (HDL-C) levels, hypertension, central adiposity, high fasting blood sugar (FBS), and diseases of the heart.6-13
The modified national cholesterol education program adult treatment panel III (NCEP-ATP III)-2005, the international diabetes federation (IDF)-2005, the world health organization (WHO)-1998, the European group for the study of insulin resistance (EGIR)-1999, and the American heart association and the national heart, lung, and blood institute (AHA/NHLBI) definitions are among the definitions used to define CMetS.14-17 The NCEP-ATP III (NCEP-2005 or NCEP) definition does not have any absolute requirements. There is at least one requirement that must be met for the rest. Hypertension, hyperglycemia, dyslipidemia-1 (TG) and dyslipidemia-2 (HDL-C), and waist-circumference are the 5 prerequisites. To define CMetS, 3 out of the 5 requirements must be met. Microalbuminuria measurement is included as an optional criterion in the WHO definition.14,18-22 The detail is available in Appendix Table A1. 14
In medicine, biomarkers are objective indicators of a medical condition that can be measured accurately and reliably. 25 Understanding the spectrum of disease detection, progression, and monitoring through biomarkers is dynamic and powerful. 23 In the past, the biomarker was only associated with fluids, tissues, and chemicals that are related to and used to evaluate disease conditions. 24 In the modern-day, the term biomarker can be used in a variety of ways other than biological samples, as long as it aids in the diagnosis, monitoring, and measurement of disease conditions.25,26
Globally, HIV guidelines tend to focus on treatment and disease monitoring, but they pay little attention to issues like CMetS, which causes the standard of ART care to be inadequate.27-29 Therefore, the present study aims at determining the prevalence and biomarkers of CMetS among HIV-infected adults receiving follow-up care.
Methods
Study design, period, and setting
This was a hospital-based study conducted in HIV-infected people on follow-up treatment at Zewditu Memorial Hospital in Addis Ababa, Ethiopia, from January 2019, to February 2021.
Zewditu Memorial Hospital became Ethiopia’s first hospital to open and offer ART services in July 2003.30,31
The research was part of a major cohort study that was designed to report prevalence data.
Study population
All PLWHA attending the ART follow-up care at Zewditu Memorial Hospital were included in the study. PLWHA over the age of 18 years who were willing to take part in the study were the target population.
Sample size determination
The following equation was used to calculate the sample size: -
Sample size n = [Np (1−p)]/[(d2/z21−α/2*(N−1) +p*(1−p)
Where N (7674) is the total HIV-infected population registered for follow-up care, and P denotes the prevalence of CMetS in the HIV-infected population as determined by published articles. 32 The sample size was 286 with a 5% standard error (d) and a 95% confidence level, and when a 10% contingency (lost to follow-up and defaulters) was taken into account, the study’s ultimate sample size was 314.
Sampling procedure and enrollment
A systematic random sampling technique was used to recruit study participants. The sample interval (K) was calculated using the formula N/n (7674/314≅24). The first participant was selected using a lottery method.
Data collection and research procedure
Interviews and participant tracking charts were used to gather information from participants. The structured questionnaire used by the WHO Stepwise method to noncommunicable disease risk factor surveillance was adapted for a face-to-face interview (STEPS-2014). 33 The questionnaire included information on sociodemographic and clinical variables. Two trained data collectors administered the questionnaire, took anthropometric measurements, checked blood pressure, and collected blood samples for biomedical testing. Weight (in kilograms), height (in meters), a calculated Body Mass Index (BMI = weight in kg/height in m2), and waist circumference/abdominal circumference (in inches) were taken as anthropometric measurements.
Age, gender, family history, comorbidity, waist circumference, systolic blood pressure (SBP), diastolic blood pressure (DBP), total cholesterol (TC), TG, low-density lipoprotein (LDL-C), tobacco use, alcohol use, coffee use, body mass index, duration with cART, nadir CD4 count, weight gained since cART initiation, frequency of ART change, blood group, Rh Factor, and several other predictor variables were examined to see their impact on the CMetS.
The Type ZT-160 body-weight balance was used to determine height and weight. During appointments, biomedical measurements including PR (per minute), SBP (mmHg), DBP (mmHg), and tests like FBG (mg/dl) and fasting blood cholesterol (mg/dl) were performed. Omron HEM 7203, a completely automatic digital blood pressure monitor, was used to measure BP and PR (Omron Healthcare Co. Ltd., Kyoto, Japan). The devices were calibrated to ensure correct validation. The accuracy of the devices was tested using a mercury sphygmomanometer. Three BP readings were taken from the patients’ right arm with a 5-minute interval between them, and the mean of the 3 readings was used to calculate the final result.34,35
BD FACSPrestoTM and BD FACSCaliburTM were used to determine the absolute CD4 count (AbsCD4) and percent CD4 (percent CD4). The Abbott RealTime HIV-1 test was used to determine viral load. SIEMENS (Dimension EXL 200 Integrated Chemistry System) and Omina Health were used to determine lipid profiles (CS-T240 Auto-Chemistry Analyzer).
Data analysis
IBM SPSS Statistics 25 was used to code, double-enter, and analyze the data. The data were presented using descriptive and inferential statistics. The interactions of the predictors with the outcome variables were determined using logistic regression. To control the influence of confounding, independent variables with a P-value of .20 in the bivariate logistic regression were put into multivariate logistic regression. In the case of pulse rate, SBP, and DBP, the mean of the repeated measures was utilized; for the other variables, a single measurement data was used to determine the effect on the prevalence of CMetS. For statistical analysis, a 95% confidence interval and a level of significance less than .05 were used.
Ethical statement
The study was approved by: (1) the Muhimbili University of Health and Allied Sciences, Office of the Director of Research and Publications (Ref. No. 2018-04-23/AEC//Vol. XII/88), Dar El Salaam, Tanzania. (2) Addis Ababa University, School of Pharmacy, Ethical Review Board (ERB/SOP/41/11/2018), Addis Ababa, Ethiopia. (3) Addis Ababa University, College of Health Sciences, Institutional Review Board (IRB, Meeting number 08/2018), Addis Ababa, Ethiopia. (4) City Government of Addis Ababa Health Bureau, Ethical Clearance Committee (Ref no. A/A/HB/344438/227), Addis Ababa, Ethiopia.
Operational definition
Results
Although 314 individuals were initially enrolled in this hospital-based study, the final sample size used for analysis was (n = 288) HIV-infected people. Twenty-six individuals were excluded from the study for a variety of reasons: two were dropped owing to a change of address, four were dropped due to critical illnesses (one due to HBS, three due to high blood pressure), and the remaining ten were defaulters for unclear reasons (Figure 1).
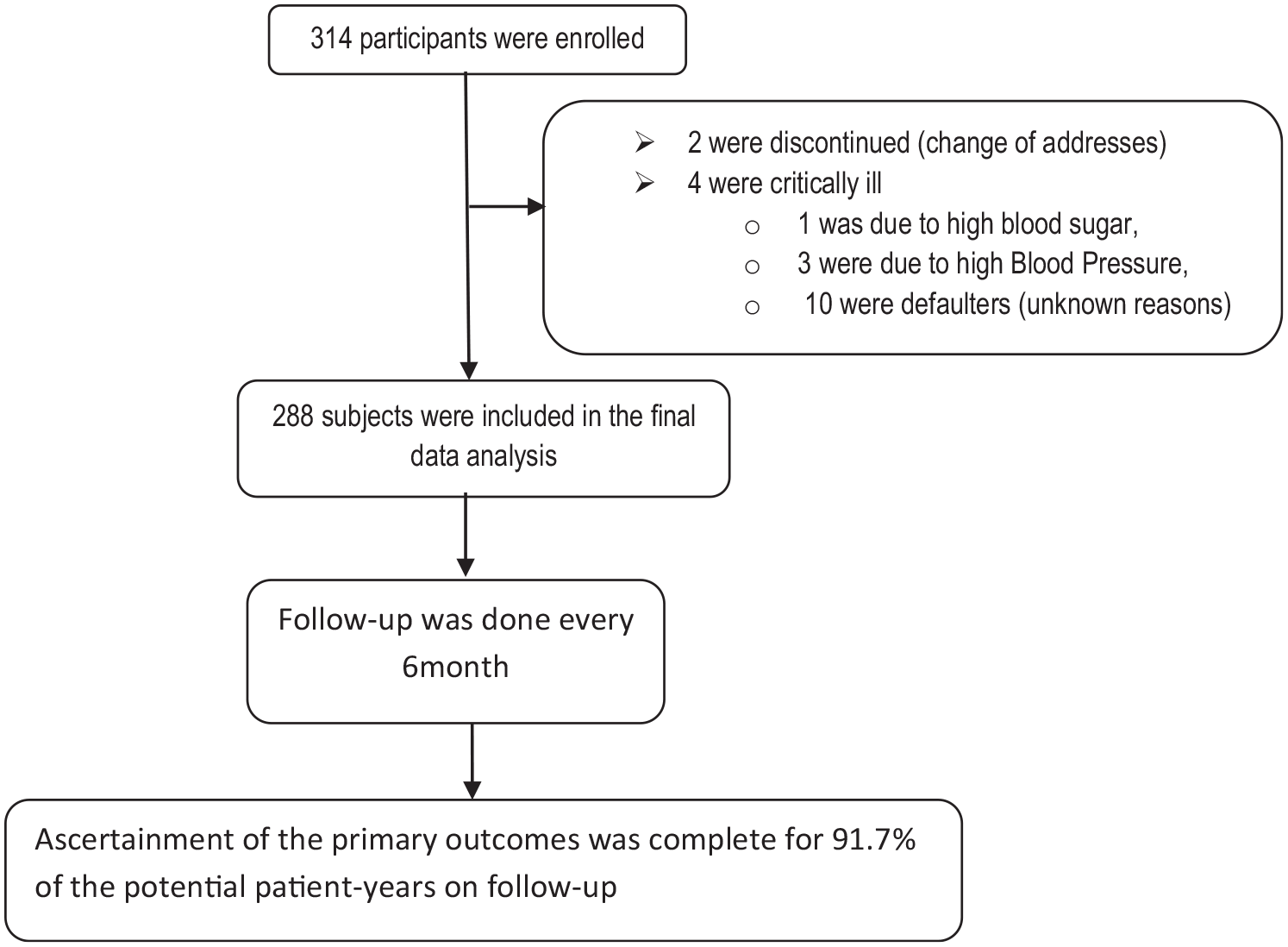
Enrollment, screening, and follow-up.
The demographic data revealed a slight preponderance of females (162, 56.2%), and nearly one in two were married, half 62 (21.5%) were divorced, one in three had completed secondary high school (grades 9-12), and one in four were involved in a small self-employed with employee business. Almost 10% of the population was unemployed, with 4.5% of the population being students. Addis Ababa was represented by 271 people (94.1%) (see Table 1).
Socio-demographic data of research participants: counts, percentages of the total, and cumulative percent; Zewditu Memorial Hospital, Addis Ababa, Ethiopia, January 2021.

Classification is based on ISEC (International Socio-Economic Classes). 36
The majority of patients who have not modified their baseline ART fall into the 1e category, followed by 1c, 1f, and 1d. The majority of patients who have modified their baseline management at least once are now on 1J, 1e, or 2i. Patients who have been on ART for the longest are currently on the 2i regimen. In general, half (139, 48.3%) of the patients were on a 1j (TDF+3TC+DTG) regimen (Figure 2).

The figure shows the current ART regimen, the total years on ART, and the frequency of ART regimen changes from the baseline among study participants, Zewditu Memorial Hospital, Addis Ababa, Ethiopia; January 2021.
Given their clinical backgrounds, the majority of the 131 (44.8%) were classified as stage III of the WHO categorization system, whereas 276 (95.8%) were classified as T1. 235 (81.6%) were on first-line ART treatment, whereas 129 (44.8%) had modified their initial regimen at least once. The prevalence of CMetS using the NCEP-2005 definition was 28.5% (82/288), and it was 43.5% (126/288) using the IDF definition (Table 2).
Clinical information of the study participants: counts and percent of the total; Zewditu Memorial Hospital, Addis Ababa, Ethiopia. N = 288.
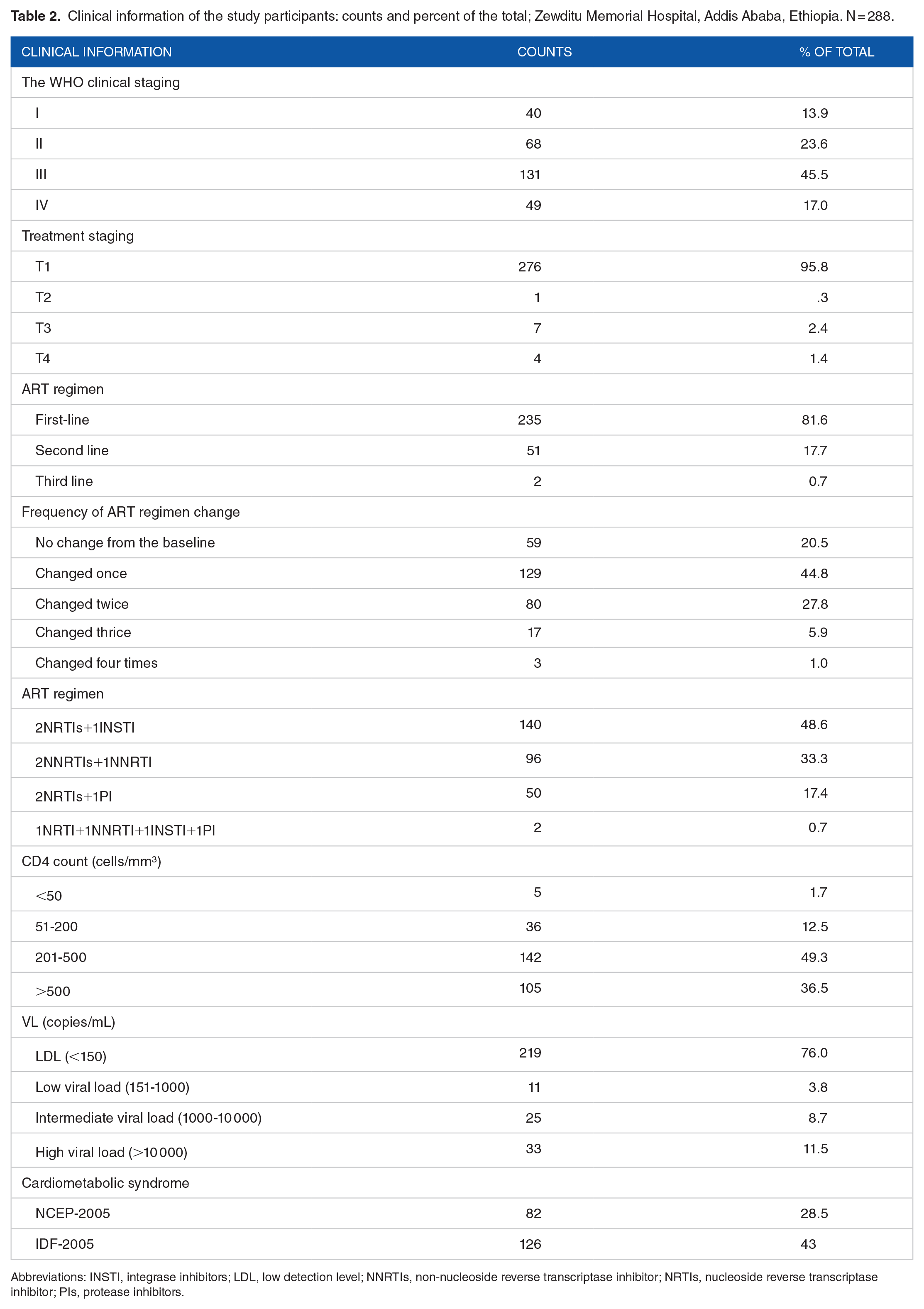
Abbreviations: INSTI, integrase inhibitors; LDL, low detection level; NNRTIs, non-nucleoside reverse transcriptase inhibitor; NRTIs, nucleoside reverse transcriptase inhibitor; PIs, protease inhibitors.
The average age of the population was 43.51 ± 11.273, with mean and a standard deviation (SD) of 40.7 ± 10.05 for women and 47.1 ± 11.4 for males, respectively. At the time of HIV confirmation, the average age was 32.7 ± 11.1, with women accounting for 30.2 ± 10.4 years and men for 35.9 ± 11.4 years.
The total number of years spent on cART was 9.81 ± 4.67 (mean ± SD). The youngest participant was 7 years old at the time of HIV confirmation and cART initiation, and the oldest participant was 63 years old for HIV confirmation and 65 years old for ART initiation. The short-term ART length was 0.46 years, while the long-term ART duration was 22.2 years. Table 3 provides a more detailed look at the patients’ features.
Characteristics of subjects (N = 288) enrolled in the study, overall and by gender, Zewditu Memorial Hospital, January 2021, Addis Ababa, Ethiopia.

For jobless participants with no stable income, the minimum monthly income appears to be zero. K = 1000.
Age, comorbidity, waist circumference, SBP, DBP, TG, LDL-C, body mass index, time since starting ART, and blood group “A” all demonstrated significant associations with the NCEP-CMetS using bivariate analysis. Whereas, age, comorbidity, waist circumference, DBP, TG, LDL-C, cigarette smoking, body mass index, time since starting ART, blood group “A,” and “age at which ART was begun” were all found to have a significant association using multivariate analysis (Table 4).
The impact of cardiometabolic predictors on the outcome variables using the NCEP Adult Treatment Panel III (NCEP ATP III-2005) criteria. Data extrapolated from a cohort study of HIV-positive persons receiving follow-up care at Zewditu Memorial Hospital in Addis Ababa, Ethiopia, in the year 2021.

Abbreviations: BMI, Body Mass Index; DBP, diastolic blood pressure; HDL-C, high-density lipoprotein; LDL-C, low density lipoprotein; SBP, systolic blood pressure; TC, total cholesterol; TGs, triglyceride.
Significant values. **P < .01
The risk of CMetS increased by 1.7 times as age increased each year, according to multivariate analysis (AOR: 1.780, 95% CI (1.647, 2.940), P = .009). People with one or more comorbidities were 6 times more likely than those without to acquire CMetS (AOR: 5.962, 95% CI (1.214, 29.271), P = .028). Increases in diastolic (AOR: 1.133, 95% CI (1.062, 1.209), P < .001) and TG levels (AOR: 1.014, 95% CI (1.008, 1.019), P < .001), as well as an increase in body mass index (AOR: 1.259, 95% CI (1.078, 1.470), P = .004), time on ART (AOR: 1.023, 95% CI (1.007, 1.039), P = .006), and age at which ART began (AOR: 1.251, 95% CI (1.044, 1.499), P = .015), all elevated the incidence of CMetS (Table 4).
Gender, family history, comorbidities, waist circumference, SBP, DBP, body mass index, weight gained while on ART, and blood group “A” all had significant bivariate correlations with the IDF-CMetS. Only gender, waist size, TG, and blood group “A” were found to have a significant link using multivariate analysis (Table 5).
Shows the impact of cardiometabolic predictors on the outcome of cardiometabolic syndrome, as defined by the International Diabetes Federation (IDF-2005). Data from a cohort study of HIV-infected people in Zewditu Memorial Hospital’s follow-up care in Addis Ababa, Ethiopia, in 2021.
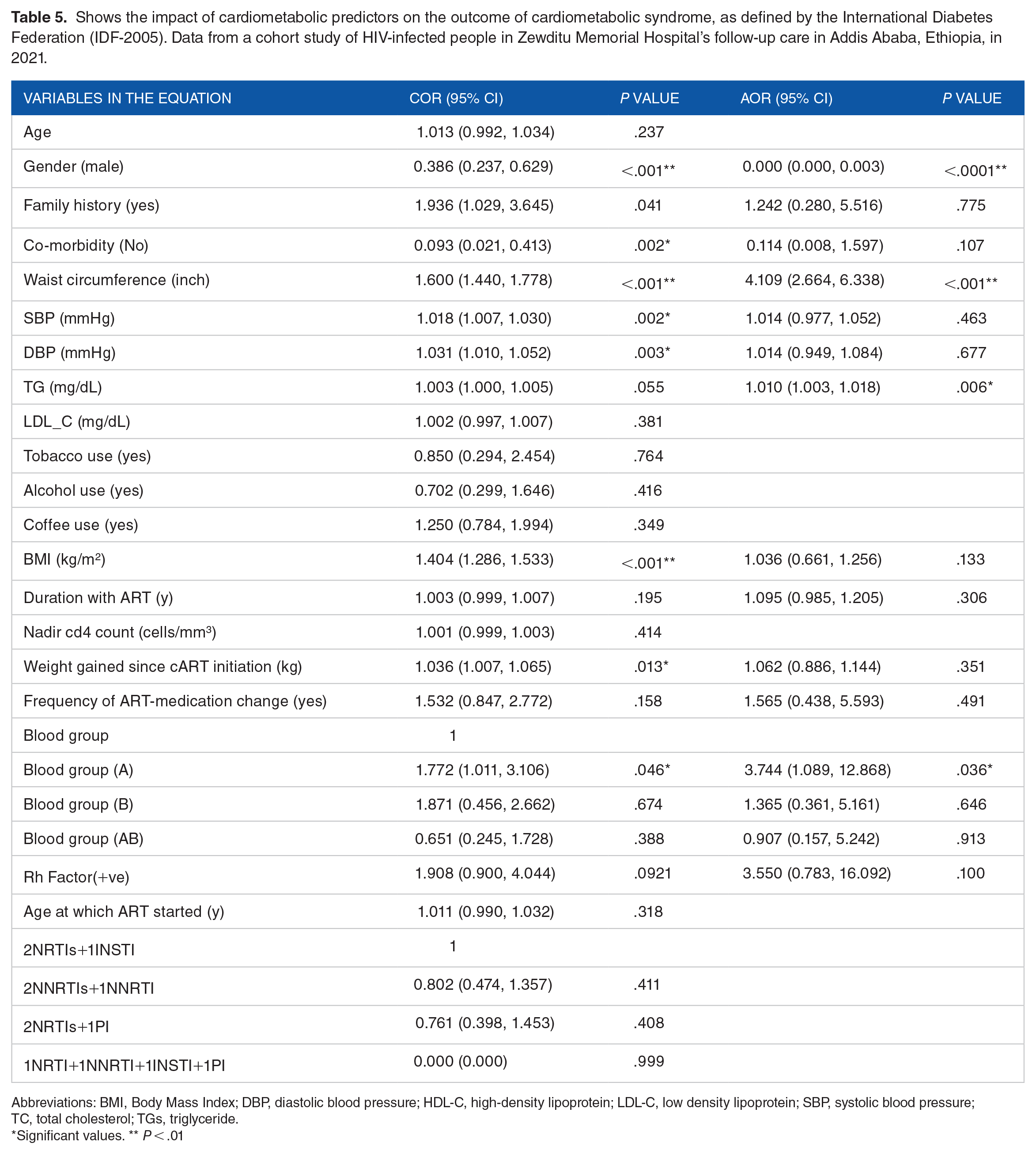
Abbreviations: BMI, Body Mass Index; DBP, diastolic blood pressure; HDL-C, high-density lipoprotein; LDL-C, low density lipoprotein; SBP, systolic blood pressure; TC, total cholesterol; TGs, triglyceride.
Significant values. ** P < .01
Males were 99.9% less likely to get CMetS, and each inch increase in belly circumference increases the risk by 4 times. When compared to blood group “O,” blood group “A” indicated 1.3 times higher risk (Table 5).
Discussion
Antiretroviral therapy has slowed HIV progression, increased survival, and improved quality of life in people living with HIV.37,38 This enormous benefit, however, is not without drawbacks. In recent years, PLWHA is at an increased risk of cardiometabolic disorders.39-42
CMetS prevalence data among HIV-positive people varies from nation to country. In our study, the prevalence was 28.5% (82/288) using the NCEP-2005 definition and 43.5% (126/288) using the IDF definition. According to a compilation of studies, the prevalence of CMetS in HIV-positive people was 20.6% using NCEP-2005 and 31.3% using the IDF-2005. 32 Using both the NCEP-2005 (X2 = 121.94, df = 1, P < .001) and the IDF-2005 models (X2=32.99, df =1, P < .001) as a reference, the chi-square goodness of fit test (2X2) result suggested that the prevalence in our study was significantly higher. 32 However, when compared to individual study reports, our prevalence is within the range of previously published studies,43-46 and it is slightly higher than the Polish, 47 SHIVA (France), 48 Australian, 49 South Korean, 50 Ethiopian,8,46,51 and global meta-analysis 52 studies; while it is lower than those reported from Nigeria 53 and China. 54 Variations in study design, sample size, population genetics, study duration, duration with HIV and ART, treatment regimen or switch of therapy, and socio-demographic differences could all contribute to this heterogeneity.
Sex, age, weight, body mass index, sedentary lifestyle, central obesity, and cigarette smoking were all found to have an impact on the prevalence, etiology, and progression of CMetS in several investigations.51,55 The European AIDS Clinical Society has stated unequivocally that the risk of getting CMetS increases with age. 51 This is because older people are more impacted by the constellation of cardiovascular risk factors that make up the CMetS. Hence, the aging of the world’s population is a major contribution to the rising prevalence of the illness. 56 According to the NCEP tool, the risk of CMetS increased 2 times with each year of age in our sample (AOR: 1.780, 95% CI (1.647, 2.940), P = .009), which is consistent with prior research. 56
According to certain research, CMetS is more common between the ages of 45 and 65, 57 and the prevalence of CMetS may then diminish. 58 For several reasons, people aged 65 and up have fewer CMetS episodes than their younger counterparts. 59 One reason could be that people who developed CMetS before the age of 60 died prematurely before reaching 65 and up; another reason could be that diet has an impact on CMetS, and elders are frequently malnourished. 60 Many countries’ life expectancies are below 65, which can influence the outcome. 61 Only 9 (3%) of the participants in our study were beyond the age of 65, indicating that the preceding argument may be valid.
In addition to age, the age at which ART was initiated may have an impact on CMetS outcomes. In our analysis, we discovered that as ART start age increased, the probability of developing CMetS increased as well (NCEP-2005: OR = 1.27, CI 1.031-1.564, P = .025), indicating that people who started ART therapy later in life are more likely to develop CMetS than those who started earlier. In the non-HIV infected sample, however, the CMetS to infancy connection was symmetrical. 62
Gender is another key factor to consider when it comes to CMetS. Even though both male and female sex hormones have a role in the development of obesity and the regulation of insulin homeostasis and blood pressure, 63 gender-based CMetS occurrences are reported in a variety of ways. Using the IDF-2005 model, the male gender was 99.9% less likely to be related to CMetS in our research. This is supported by research conducted in Latin America,58,64 and Ethiopia. 65 Males, on the other hand, had a higher incidence, according to a study from South Africa. 66 This inconsistency could be attributed to differences in gender proportions, ages, lifestyle implications, and the CMetS evaluation tools used.
CMetS was 6 times more likely to develop in people with one or more comorbidities (NCEP-2005: AOR = 5.962, 95% CI (1.214, 29.271), P = .028). Type 2 diabetes mellitus, heart failure, dyslipidemia, obesity, and high blood pressure are only a few of the comorbidities that have been linked to the formation of CMetS.67,68 Comorbidities enhanced the likelihood of CMetS, as indicated by many resources. Comorbidity screening should also be included in normal CMetS care.69-71
CMetS was more common in people who had been on ART for a longer time (NCEP-2005: AOR = 1.023, 95% CI (1.007, 1.039), P = .006). This finding is comparable to one from Malawi, 72 in which a longer period of ART was linked to a higher level of CMetS. Furthermore, a Kenyan study discovered that as the duration of ART increased, CMetS increased significantly. 73 This could be linked to the fact that long-term ART usage has been shown to increase metabolic risk.74-76
Although waist circumference is an important component of metabolic syndrome, the IDF criterion of a raised waist circumference should be used with caution because many men with normal waist circumference have a higher mortality risk. 77 In another way, when comparing the IDF to the NCEP, the IDF utilizes lower obesity cut-off values than the NCEP, which could also lead to false-positive results. Using the IDF-2005 tool, we discovered that an increase in waist circumference (central adiposity) was linked to the prevalence of CMetS (AOR = 4.109, 95% CI (2.664, 6.338), P < .001). Several more research42,50,78-82 have confirmed this finding.
The waist circumference, not the BMI, is regarded to be a stronger indicator of cardiovascular risk.83,84 We have positive relationships with CMetS (NCEP-2005: AOR = 1.259, 95% CI (1.078, 1.470), P = .004), even though several studies have shown that BMI is insufficient for predicting cardiovascular risk. A positive connection between BMI and CMetS has also been reported in several other studies.82,85-87
Another criterion for defining metabolic syndrome is systolic and DBPs. 86 Among all the variables that constitute up CMetS, hypertension is the most critical factor contributing to a significant increase in cardiovascular mortality. It’s noteworthy that the prevalence of both hypertension and CMetS increases with age, according to a previous study.82,88,89 In our sample, only an increase in DBP caused a significant difference in CMetS prevalence (NCEP-2005: AOR: 1.133, 95% CI (1.062, 1.209), P < .001). The rationale behind this could be that DBP is more important in middle-aged adults aged 45 to 55, but SBP is more important in older people aged 55 and up in terms of cardiometabolic and vascular risks. 90
Lipid profiles, just as critical as blood pressure, play a vital influence in the course and severity of CMetS. TG and HDL-Cs are 2 lipids that are included in the key CMetS defining tools. Though the correlation was not as strong to predict in mg/dL of TG in our study, the cumulative effect could impact associated with other irregularities using both the NCEP-2005 (AOR: 1.014, 95% CI (1.008, 1.019), P < .001) and the IDF-2005 (AOR = 1.010, 95 % CI (1.003, 1.018), P = .006) models. Several other studies have discovered that TG has a substantial impact on CMetS outcomes.40,81,82,91,92 TG, for example, can indicate future cardiovascular risks, according to one study, 93 and the presence of TG, according to another study, can potentially make CMetS progression more difficult. 94
Even though LDL-C was not listed as a criterion in the CMetS definition, multiple resources have demonstrated evidence of its significance in the progression of CMetS.90,93 As a result, additional CMetS-defining illnesses, such as TG and HDL-C, may be hampered by its development. It’s also worth noting that many of the lipid-lowering medicines on the market target LDL-C to treat dyslipidemia, and disrupting this molecule could have major effects in the course of CMetS. 93 We discovered that those with higher low-density lipoprotein were also more likely to have CMetS in our study utilizing the NCEP-2005 tool. According to information from multiple published sources, PLWHA may develop severe management crises and die young if lipid abnormalities are not addressed as aggressively as they are in the general population.92,93
The blood type “A” has been substantially linked to CMetS. However, we do not have any articles that tackle the subject of CMetS. The association could be linked to the prevalence of the blood type, which was the most prevalent, accounting for 35.8% of all cases.
In terms of ART treatment, 48.6% of the people in our research were on a 2NRTIs+1INSTI regimen, 33.3% were on 2NRTIs+1NNRTI, 17.4% were on 2NRTIs+1PI, and 0.7% was on a 1NRTI+1NNRTI+1INSTI+1PI regimen. Several other research49,62,95 found a similar regimen pattern. Furthermore, the significance of ART in the development and progression of CMetS has been investigated in various other studies, although ours yielded no significant results.80,96 ART regimens such as PIs and NNRTIs are thought to play a substantial role in the development and progression of CMetS in HIV-infected people, according to several studies. 97
In general, measuring and monitoring cardiometabolic indicators has been used as a universal technique for identifying CMetS. 98 We used the IDF and NCEP tools to report the prevalence of CMetS and provide the same results, combining them simply improves the yield beyond that of the single tool. 97 Studies that are widely available also produce and publish conflicting outcomes. The study design, sample selection, the duration of the study, the definition of tools used, and the sociodemographic characteristics of the study population all play a role in the variation in the report.
Limitations of the Study
The study cannot be used to represent all HIV-positive people in Ethiopia because the data comes from a single institution. Since the study did not include a control group, the ability to predict severity in contrast to the general population may be hampered.
Conclusions
CMetS was shown to be prevalent in PLWHA, according to the study. Age, time on ART, age when ART began, gender, co-morbidities, waist circumference, DBP, and body mass index (BMI) were all found to be significant predictors of CMetS. The only biomarkers identified to be significant with CMetS using both the NCEP and IDF tools were TG and blood type “A.” Combining the IDF and NCEP techniques to report CMetS prevalence could increase the yield above what could be achieved with either tool alone.
Recommendations
It is suggested that a multicenter, global study with bigger sample size and longer follow-up time be conducted. For HIV patients, strategies for decreasing hypertriglyceridemia and increasing HDL cholesterol levels are advantageous, and they can be directed at first by lifestyle changes and the proper selection of ART medicines.
Footnotes
Appendix
Definitions and criteria for CMetS. 14

| Description | NCEP ATP III (2005 revision) | WHO (1998) | EGIR (1999) | IDF (2005) |
|---|---|---|---|---|
| Absolutely required | None | Insulin resistance* (IGT, IFG, T2DM or other evidence of IR) | Hyperinsulinemia ‡ (plasma insulin >75th percentile) | Central obesity (waist circumference§ ): ⩾94 cm (M), ⩾80 cm (F) |
| Criteria | Any three of the five criteria below | Insulin resistance or diabetes, plus two of the five criteria below | Hyperinsulinemia, plus two of the four criteria below | Obesity, plus two of the four criteria below |
| Obesity | Waist circumference: >40 inches (M), >35 inches (F) | Waist/hip ratio: >0.90 (M), >0.85 (F); or BMI >30 kg/m2 | Waist circumference: ⩾94 cm (M), ⩾80 cm (F) | Central obesity already required |
| Hyperglycemia | Fasting glucose ⩾100 mg/dL or Rx | Insulin resistance already required | Insulin resistance already required | Fasting glucose ⩾100 mg/dL |
| Dyslipidemia | TG ⩾150 mg/dL or Rx | TG ⩾150 mg/dl or HDL-C:<35 mg/dL (M), <39 mg/dL (F) | TG ⩾177 mg/dL or HDL-C <39 mg/dL | TG ⩾150 mg/dL or Rx |
| Dyslipidemia (second, separate criteria) | HDL cholesterol:<40 mg/dL (M), <50 mg/dL (F); or Rx | HDL cholesterol: <40 mg/dl (M), <50 mg/dl (F); or Rx | ||
| Hypertension | >130 mmHg systolic or >85 mmHg diastolic or Rx | ⩾140/90 mmHg | ⩾140/90 mmHg or Rx | >130 mmHg systolic or >85 mmHg diastolic or Rx |
| Other criteria | Microalbuminuria † |
IGT, impaired glucose tolerance; IFG, impaired fasting glucose; T2DM, type 2 diabetes; IR, insulin resistance; other evidence includes euglycemic clamp studies.
Urinary albumin excretion of ⩾20 µg/min or albumin-to-creatinine ratio of ⩾30 mg/g.
Reliable only in patients without T2DM.
Criteria for central obesity (waist circumference) are specific for each population; values given are for European men and women.
Rx, pharmacologic treatment.
Acknowledgements
We would like to thank the Muhimbili University of Health and Allied Sciences (MUHAS), Addis Ababa University (AAU), the DAAD, Zewditu memorial Hospital (ZMH), and the EDCTP. We would like to thank all the research participants in the study. We would also like to forward our appreciation for our research assistants Sr. Tizita Woldeyesus Tadesse, BSc, and Mr. Chalachew Teshome Tiruneh, MPH. We are also grateful for statistical resources support from Dr. Todd Grande, Dr. Jacob Mays, Dr. Brandon Foltz, and Dr. Mike Crowson.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The German Academic Exchange Service (DAAD) provided funding for both study and research supplies. The data collectors’ honorarium was also funded by the European & Developing Countries Clinical Trials Partnership (EDCTP).
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
Can be retrieved via the Addis Ababa University repository system at the request of the study team.
Author Contributions
Informed Consent and Consent for Publication
Prior to data collection, all study participants gave their informed consent. Permission for publication was not required because no aspect of the description was meant to identify an individual’s identity.
