Abstract
Cerebrospinal fluid (CSF) rhinorrhea is rarely reported as the first presenting feature of giant invasive macroprolactinomas. Cerebrospinal fluid rhinorrhea is usually reported as a complication of trauma, neurosurgical, and skull-based procedures (such as pituitary surgery or radiations), and less frequently after medical treatment with dopamine agonists (DAs) for macroprolactinomas. This phenomenon results from fistula creation that communicates between the subarachnoid space and the nasal cavity. Meanwhile, pneumocephalus is another well-recognized complication after transsphenoidal surgery for pituitary macroadenomas. This entity may present with nausea, vomiting, headache, dizziness, and more seriously with seizures and/or a decreased level of consciousness if tension pneumocephalus develops. Case reports about the occurrence of spontaneous pneumocephalus after medical treatment with DAs without prior surgical interventions are scarce in the literature. Our index case is a young man who was recently diagnosed with a giant invasive prolactin-secreting pituitary macroadenoma with skull base destruction. A few months before this diagnosis, he presented with spontaneous CSF rhinorrhea with no history of previous medical or surgical treatment. In this case report, we report an uncommon presentation for giant invasive macroprolactinoma with a CSF leak treated with cabergoline that was subsequently complicated by meningitis and pneumocephalus. This is a very rare complication of cabergoline therapy, which occurred approximately 1 month after treatment initiation.
Keywords
Introduction
Cerebrospinal fluid (CSF) rhinorrhea is a serious entity that is defined as the pathological leak of the CSF through the nose; this occurs due to a bone defect in the skull base, coupled with disruption of the dura and arachnoid membrane. It is typically classified as traumatic and nontraumatic.1,2 Giant invasive macroprolactinomas constitute one of the rare causes of nontraumatic CSF leak. 3 Furthermore, this life-threatening condition is rarely documented as the main presenting symptom of invasive macroprolactinomas. 4 Cerebrospinal fluid leakage may result in catastrophic complications, including meningitis, intracranial abscess formation, pneumocephalus, and intracranial hypotension. Pneumocephalus, which is known as a pneumatocele or intracerebral aerocele as well, is a term used to describe the presence of air within the intracranial compartments—the epidural, subdural, subarachnoid, intracerebral, and/or intraventricular spaces. Pneumocephalus very scarcely presents as a clinical feature of giant invasive macroprolactinoma; it can also complicate the treatment course. Pneumocephalus results from a fistula created by the invasion of a giant macroprolactinoma, which destroys the surrounding bony tissues. The fistula enables the communication between extracranial and intracranial spaces and allows the ambient air to enter the intracerebral compartments when the tumor size has reduced following medical treatment with dopamine agonists (DAs). Very few sporadic cases of pneumocephalus arising from the complication of a giant invasive macroprolactinoma—particularly following the initiation of medical treatment with cabergoline—have been described in the medical literature. Pneumocephalus was first described in 1866 by Thomas 5 following the autopsy in a patient with trauma, and the term was subsequently used in 1914 by Wolff. 6 In addition to neurosurgical and otorhinolaryngological procedures, 7 there are other causes of such complications including trauma, parasellar or paranasal sinus malignancies, infection by gas-forming organisms, and other rarer causes. 8 Postoperative pneumocephalus is usually suspected soon after transsphenoidal surgery and it is confirmed by neuroimaging; conversely, spontaneous pneumocephalus occurs a few weeks after the initiation of treatment with DAs and requires a high index of suspicion.
Case Report
A 24-year-old male patient attended the neurosurgery clinic at our hospital with a history of clear-fluid nasal discharge for 8 months. He was evaluated at a peripheral health care facility where he underwent a computed tomographic (CT) scan of the brain. He was found to have a large suprasellar mass and hyperprolactinemia, at which point he was referred to endocrinology clinic for further evaluation.
The patient was immediately admitted for further assessment and management in our hospital. The nasal discharge was watery, continuous, and worsened when bending forward; the nasal discharge was associated with occasional mild to moderate headaches with mild retro-orbital pain, which rarely required analgesics. A history of decreased libido, occasional morning penile erections, and a few incidents of spontaneous galactorrhea were present along with the loss of facial and axillary hair. However, there was no history of fever, severe headache, vomiting, diplopia, or other visual disturbances. The patient had no complaints of impaired hearing, tinnitus, disorientation, incontinence, hemiparesis, postural symptoms, loss of consciousness, drowsiness, and/or convulsions.
On physical examination, the patient was conscious, oriented to time and place, and looking well. He was morbidly obese with a body mass index of 52 kg/m2 with absent facial and axillary hair. The patient’s vital signs were stable with no postural hypotension. Neuro-ophthalmologic examination revealed no visual field deficit with a full range of extraocular muscle movements. Galactorrhea and gynecomastia were noticeable on breast examination. The cranial nerve examination was normal, and the patient had a normal gait and no cerebellar signs.
Genital examination revealed small bilateral testes with size of 6 ml on right and 10 ml on left side and a small phallus. The remaining systemic examination was unremarkable.
Analysis of the nasal fluid confirmed the presence of β2-transferrin and low glucose, indicating CSF rhinorrhea. The pituitary profile was remarkable for central hypothyroidism, hypogonadotropic hypogonadism, and growth hormone (GH) deficiency, but normal for adrenocorticotropic hormone (ACTH) and morning serum cortisol (Table 1).
Laboratory assessment.

Abbreviations: ACTH, adrenocorticotropic hormone; FSH, follicle-stimulating hormone; GH, growth hormone; IGF, insulin-like growth factor; LH, luteinizing hormone; TSH, thyrotropin.
Magnetic resonance imaging of the brain and pituitary (Figures 1 to 3) showed an expansive, irregular lesion in the sellar and suprasellar region, measuring 5.5 cm × 4.4 cm × 5.2 cm, enclosing the right optic nerve and eroding the cavernous sinus and clivus. It extended anteriorly to the posterior ethmoid sinus and superiorly to the right middle cranial fossa, encasing both the internal carotid artery and right middle cerebral artery.

A contrast-enhanced, T1-weighted, sagittal magnetic resonance image showing a huge suprasellar mass (white shaded arrows) extending anteriorly to the posterior ethmoid sinus.
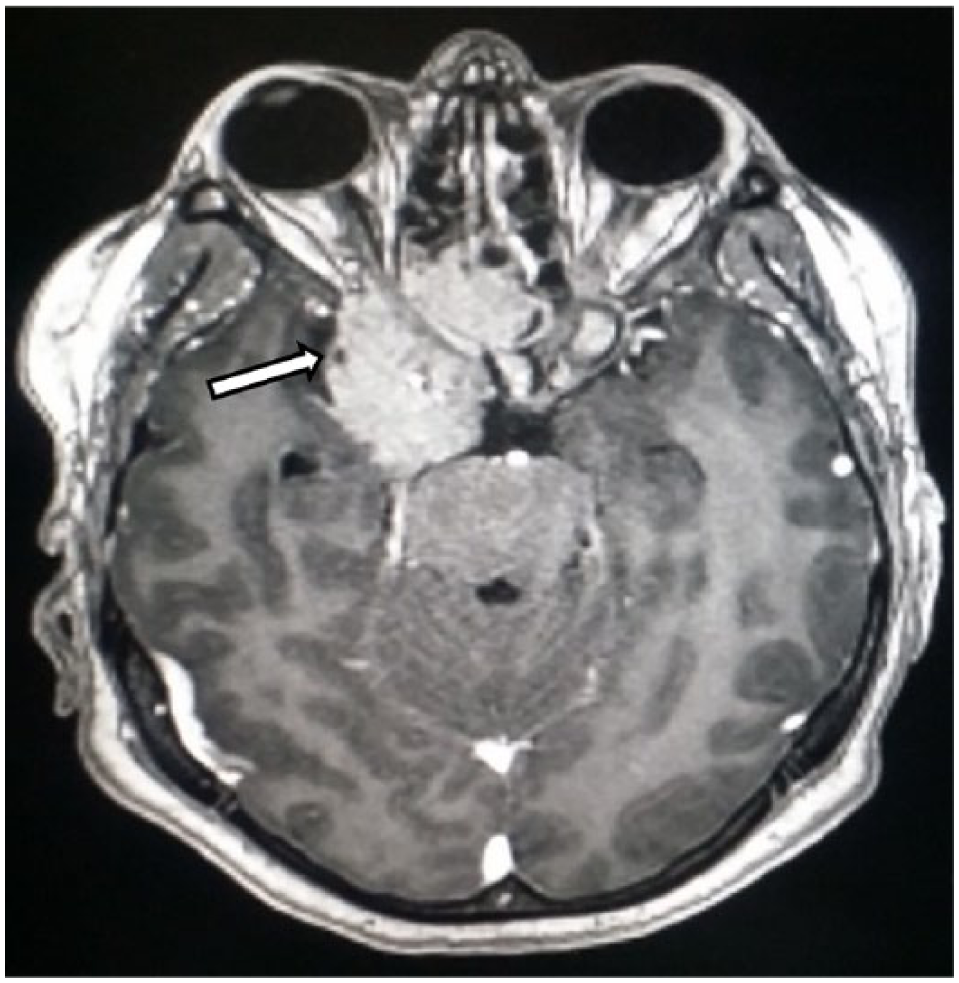
A contrast-enhanced, T1-weighted, axial magnetic resonance image showing the mass (white shaded arrow) encasing the right optic nerve.
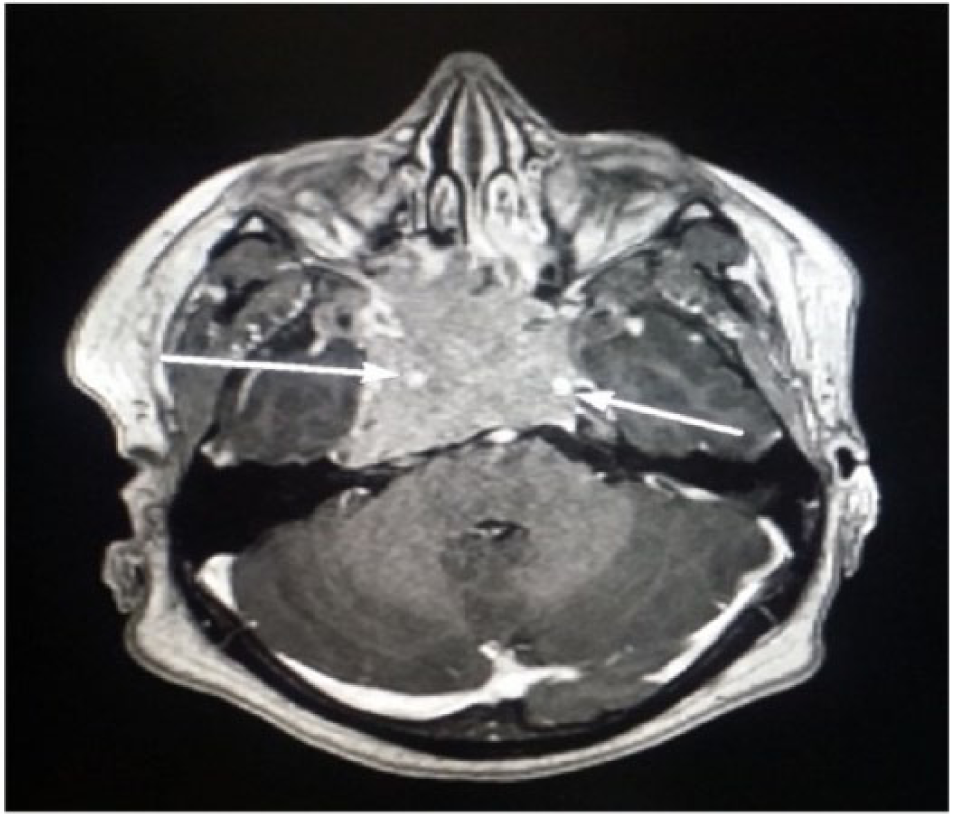
A contrast-enhanced, T1-weighted, axial magnetic resonance image showing the tumor encasing both the internal carotid arteries (the long, white arrows, bilateral).
A final diagnosis of giant invasive macroprolactinoma was made and treatment initiated with 0.25 mg of oral cabergoline twice weekly. Levothyroxine 50 μg once daily started orally after ensuring a normal cortisol response to Synacthen stimulation test. He was discharged home in clinically stable condition with instructions to attend emergency department in case of worsening CSF leak, headache, visual disturbances or fever and given 6 week outdoor follow-up appointment.
Patient was readmitted after 5 weeks of cabergoline therapy with severe headache, nausea and worsening of CSF leak. An urgent CT scan revealed extensive pneumocephalus in addition to the previously noted invasive and destructive giant macroprolactinoma, with no evidence of acute territorial infarction or intracerebral hemorrhage (Figures 4 and 5).

Axial computed tomographic images of the brain without contrast showing extensive pneumocephalus (the arrows are pointing to very-low-density areas, representing air) in the basal and suprasellar cisterns extending to the ventricular system and cerebrospinal fluid spaces.

Sagittal computed tomographic images of the brain without contrast showing extensive pneumocephalus (the arrows are pointing to very low-density areas, representing air) in the basal and suprasellar cisterns extending to the ventricular system and cerebrospinal fluid spaces.
The patient was started on prophylactic antibiotics and hydrocortisone, and he underwent endoscopic transsphenoidal pituitary macroadenoma debulking and skull base reconstruction using neuronavigation, fascia latae, and a fat graft.
Immunostaining for pituitary hormones was positive for prolactin with no expression of GH, ACTH, thyrotropin, luteinizing hormone, or follicle-stimulating hormone, whereas p53 staining was negative. The Ki67 (MIB-1) labeling index was weakly positive.
Cabergoline therapy was continued at a dose of 0.25 mg twice weekly. The patient had an unremarkable recovery but was continued on hydrocortisone and discharged home without complications.
One month later, the patient had his third admission for recurrent CSF leak and meningitis necessitating antibiotics and endoscopic skull base repair. However, the patient recovered fully with no neurological sequelae. His clinical course remained uneventful without any further recurrence of the CSF leak. At 9-month follow-up visit at the endocrinology clinic, he was stable with a prolactin level of 6600 mIU/L (311.3 μg/L).
Discussion
Giant invasive macroprolactinomas are very rare types of pituitary adenomas that require special attention due to the challenges they pose in diagnosis and management. 9 These macroprolactinoma subtypes account for 0.5% to 4.4% of all pituitary tumors. Giant invasive macroprolactinomas are predominantly found in young men.10,11 They are defined as pituitary adenomas with a diameter of 4 cm or more, 12 remarkable extrasellar extension, and significantly elevated prolactin levels (usually >1000 µg/L), 13 with no concomitant GH or ACTH secretion. Prolactinomas typically present with features of hyperprolactinemia and/or hypogonadism, but these symptoms are usually overlooked in men. Men usually present with larger tumor sizes, more aggressive tumors, and they might present very late with symptoms of space-occupying lesions. 14
Spontaneous CSF rhinorrhea is considered an atypical presentation for giant invasive macroprolactinoma, 15 indicating a large tumor size and extension to the surrounding intracranial structures with the invasion of the sphenoid and/or ethmoid sinuses. It is very uncommon in patients with untreated giant prolactinomas and is mostly induced by transsphenoidal surgery or by DA therapy, which causes rapid tumor shrinkage.15,16
In a literature review, Lam et al 17 reported 52 patients (between 1980 and 2011) with a pituitary adenoma and with nonsurgically-induced CSF leaks. Of these patients, 42 (81%) had prolactin-secreting tumors. There were no reports on the exact sizes of the tumors, except for a few cases, but the prolactin concentrations were >1000 µg/L in 37 cases and >10 000 µg/L in 13 cases, thus suggesting the presence of a giant prolactinoma in many of these patients. Interestingly, a large proportion of the prolactinoma-associated CSF rhinorrheas (86%) occurred following the initiation of DA treatment with bromocriptine or cabergoline, and only 6 patients had CSF rhinorrhea as the presenting symptom of their prolactinoma. The average initial prolactin level in patients with spontaneous CSF leakage was 9169 ng/mL compared with 4917 ng/mL in those with DA-induced leakage.
In another large retrospective review, Suliman et al 4 reviewed 114 patients with macroprolactinoma (from 1985 to 2004). The incidence of spontaneous CSF rhinorrhea in those cases was 8.7%. Among those patients, 6.1% (7 patients) developed a DA-induced CSF leak, whereas 2.6% (3 patients) developed spontaneous rhinorrhea. In this review, it was found that DA resistance was more common in macroprolactinomas with CSF leak compared with those without rhinorrhea (30% [n = 10] versus 5% [n = 104], P = .003). 4
The CSF rhinorrhea in our patient can be explained by the direct extension of the tumor through the diaphragma sellae and erosion of the skull base into the sphenoid sinus, resulting in communicating fistula. Unfortunately, the rhinorrhea had recurred and was complicated with bacterial meningitis. It is estimated that meningitis may develop in approximately 15% to 25% of patients with unrepaired CSF leak, and 10% of them consequently die.18,19
Our patient was initially started on a low dose of cabergoline (0.25 mg twice weekly) to avoid worsening of CSF leak. Considering long history of CSF leak along with patients refusal for surgical intervention, Neurosurgery team opted for close monitoring and observation. Cabergoline is a widely used and relatively safe DA. Its therapeutic effect in treating prolactinomas is well-documented in the literature. 20 Generally, it has well-tolerated side effects, even at larger doses. Cabergoline is the first-line medication used to treat microprolactinomas, nearly all cases of macroprolactinomas, and most of the cases of giant invasive macroprolactinomas. 21
Giant invasive macroprolactinomas can have a remarkable response following the initiation of cabergoline treatment, which can be associated with tumor shrinkage and a significant reduction in or normalization of prolactin levels.9,22,23 Cerebrospinal fluid leaks can occur in cases of invasive macroprolactinoma following treatment with DA17,24 or, spontaneously, as either the primary symptom or an associated symptom,4,17 as occurred in our case. Although the standard management of CSF rhinorrhea is surgical repair in 71% of cases, 4 cases of spontaneous resolution following medical treatment with DA have also been reported in the medical literature.24,25 However, our patient did not undergo surgical repair initially and was not started on prophylactic antibiotics due to the debatable data pertaining to the rules of preventing meningitis development. 26
In addition, the pneumocephalus that occurred in this case represents a rare and serious complication of DA treatment for invasive prolactinomas. Cabergoline-induced tumor shrinkage exposes the erosion of the sella floor and allows the CSF leakage and/or pneumocephalus to occur.
Pneumocephalus can develop due to the disruption of the skull base and CSF fistulas, 27 and two hypotheses may explain its development. The first pertains to the one-way valve, otherwise known as the “ball-valve” mechanism, which requires positive pressure events, such as coughing, sneezing, and Valsalva maneuvers. This event allows the air to enter through the cranial defect where it is trapped, leading to tension pneumocephalus. 28 The second hypothesis is the inverted bottle theory, according to which CSF drainage creates a negative intracranial pressure gradient, which is relieved by the influx of air. 29
Tension pneumocephalus, which is a more serious form of this phenomenon, is the collection of air that increases intracranial pressure and mimics features of space occupying lesion. It may manifest as a wide range of clinical presentations, including severe restlessness, deteriorating consciousness, focal neurological deficits, and even cardiac arrest. 8 It is an unusual neurosurgical emergency that can lead to severe neurological damage and disastrous consequences and can be life threatening if it goes unnoticed.
The detection of pneumocephalus with evidence of its mass effect on the surrounding structures is the primary feature that differentiates tension pneumocephalus from benign pneumocephalus. The “Mount Fuji” sign is the most widely reported radiologic sign that specifically indicates the presence of tension pneumocephalus. 30 It is observed when the presence of subdural free air causes compression and separation of the frontal lobes. The displacement of the frontal lobes and the widening of the interhemispheric space simulate the silhouette of Mount Fuji. Although this sign is specific to the pressure effect of subdural air, a large volume of free air might also accumulate in other intracranial compartments (eg, in the subarachnoid and intraventricular spaces) and cause similar mass effects. 31 Postoperative pneumocephalus is usually suspected in soon after transsphenoidal surgery and is confirmed by neuroimaging. A very high index of clinical suspicion is required for its prompt diagnosis and management. Overall, the management of pneumocephalus requires urgent neurosurgical evaluation and/or intervention to decrease intracranial pressure and repair the skull base disruption to prevent further morbidity and mortality.
Conclusions
Spontaneous CSF rhinorrhea and pneumocephalus are rare and unusual types of complications related to the presence of giant invasive macroprolactinomas. Pneumocephalus and CSF rhinorrhea can occur after neurosurgical approaches or they rarely ensue following the initiation of DA, such as cabergoline, and rarely as an initial presenting feature, as in this reported case. Although the patient was initially declined the surgical procedure, it is thought that if this treatment option had been employed, they might have decreased the risk of meningitis and pneumocephalus development. We believe that a low threshold of suspicion for these complications and watchful monitoring could help in the early assessment and prompt management of this phenomenon while also preventing serious neurologic sequelae. A multidisciplinary care team is essential, and close follow-up after any treatment is necessary.
Footnotes
Acknowledgements
English language editing of this manuscript was done by Journal Prep.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SSE wrote the first draft of the manuscript. MMA, MHA, and SQQ contributed to the writing of the manuscript. SSE and MHA made contributions to the acquisition of the clinical data. SSE, MMA, MHA, and SQQ agreed with the manuscript results and conclusions. SSE, MMA, and MHA jointly developed the structure and arguments of the paper. SSE and MHA made critical revisions and approved the final version of the manuscript. All authors reviewed and approved the final manuscript.
