Abstract
Background:
It is postulated that the ability of dipeptidyl peptidase-4 inhibitors (DPP-4-i) to increase circulating endothelial progenitor cells (EPCs) may be at least partly mediated by active stromal cell–derived factor 1α (SDF-1α) (a pivotal mediator of stem cell mobilization from the bone marrow). As other DPP-4-i were demonstrated to increase EPC concentrations, in this study, we sought to investigate the ability of the DPP-4-i alogliptin in modifying EPCs and SDF-1α, in patients with good and poor diabetes control.
Methods:
Two groups of diabetic patients on metformin were divided by hemoglobin A1c (HbA1c): Group A—those with HbA1c ≤6.5% (28 patients) and Group B—those with HbA1c 7.5% to 8.5% (31 patients). Both groups received alogliptin 25 mg/daily for 4 months. At baseline and 4 months later, clinical, laboratory parameters, EPCs, and active SDF-1α were determined.
Results:
After 4-month treatment with alogliptin, either Group A or Group B showed reduced HbA1c levels and concomitant similar increase in EPCs and active SDF-1α.
Conclusions:
Alogliptin showed significant benefits in increasing EPCs and active SDF-1α either in good or poor diabetes control. The study demonstrated that similar to other DPP-4-i, also alogliptin is able to increase EPC concentrations, suggesting the existence of a class effect mediated by SDF-1α. The extent of increase in EPCs is independent from baseline diabetes control.
Introduction
Endothelial progenitors cells (EPCs) are a heterogeneous population of cells in different states of maturation, originated from bone marrow. Since their identification, many studies investigated their self-renewal capability, influence on reparative vascular mechanisms, and neoangiogenesis.1–3 Although EPC isolation and characterization are still debated (which cell phenotype better identifies the “true” circulating EPC remains unsolved), lower levels of EPCs have been detected in the presence of smoking habit, diabetes, hypertension, cardiovascular (CV) disease, and dyslipidemia.4,5 Of note, increased levels of EPCs were found to be associated with a reduced risk of death from CV causes, a first major CV event, revascularization, and hospitalization. 6 A recent meta-analysis aimed to evaluate the prognostic role of the measures of EPCs on CV outcomes and death. The authors selected 21 studies for a total of 4155 patients having acute coronary syndrome, acute myocardial infarction, stroke, elective percutaneous intervention, elective coronary angiography for suspected coronary artery disease, end-stage renal disease, chronic heart failure, and aortic stenosis. Results showed that low vs high levels of EPCs (CD34+, CD133+) predicted CV events, restenosis after endovascular intervention, CV death, and all-cause mortality. 7 As diabetes is considered a coronary heart disease risk equivalent, a number of drugs have been challenged to see whether their use was associated with an increase in EPCs. 8 Pioglitazone, for example, increased early and late outgrowth EPC viability in patients with impaired glucose tolerance. 9 A similar benefit was also demonstrated by a 4-month treatment with add-on insulin. 10 Another study from Taiwan challenged the effects of 2 statins (pitavastatin and atorvastatin) in hypercholesterolemic patients with type 2 diabetes mellitus: although both statins similarly reduced plasma lipids, only pitavastatin increased plasma vascular endothelial growth factor receptor (VEGF) level and circulating EPCs in such high-risk patients. 11 Then, also antihypertensive drugs such as aliskiren and hydrochlorothiazide were investigated in relation to EPCs. The authors observed not only that aliskiren had a favorable effect on endothelial function and EPCs but also that these effects were independent of blood pressure lowering, as they were not observed after the achievement of similar values of blood pressure with hydrochlorothiazide. 12 Among antidiabetic drugs, dipeptidyl peptidase-4 inhibitors (DPP-4-i) look like of particular interest because beyond their glucose-lowering effect, studies suggest that they may have a positive role for the CV system and for induction of mobilization of stem cells.13,14
Stromal cell–derived factor 1α (SDF-1α), a major regulator of progenitor cell kinetics, is a natural substrate of DPP-4, which inactivates it by removing 2 residues at the N-terminus. In a small open-label study, a 4-week therapy with the DPP-4-i sitagliptin in addition to metformin and/or secretagogues, increased plasma SDF-1α concentrations, and circulating EPCs. 15 The most straightforward interpretation displayed by the authors was that DPP-4 inhibition raised SDF-1α concentrations, which mobilized EPCs from the bone marrow. An alternative explanation may be that glucose lowering per se improved the bioavailability of EPCs; however, the short duration of the trial and the loss of correlation between plasma glucose and EPC levels at study end seemed to argue against this hypothesis.
As previous studies demonstrated a significant influence of DPP-4-i on EPCs, this study was undertaken to investigate whether also the DPP-4-i alogliptin is able to increase EPCs and SDF-1α concentrations and whether such effect differs in good and poor diabetes control.
Patients and Methods
Subjects
Individuals with type 2 diabetes were recruited in the outpatient clinic of Division of Endocrinology. Eligible subjects were diabetic patients on metformin monotherapy at a dose comprised between 1.5 and 2.5 g/d, having HbA1c <6.5% (Group A) or 7.5% to 8.5% (Group B). Exclusion criteria included any cerebrovascular event, any revascularization procedure, clinically relevant peripheral artery disease, diabetic foot, nephropathy, retinopathy, and clinically relevant neuropathy.
Study design
Eligible subjects in both groups were invited to receive alogliptin 25 mg/daily for 4 months. At baseline, medical history, current therapies, personal history of diabetes and CV disease, smoking, and drinking habits were recorded. At baseline and 4 months later, clinical parameters (body mass index [BMI] and systolic and diastolic blood pressure) were registered and blood was drawn to determine fasting plasma glucose, HbA1c, total cholesterol, triglycerides, high-density lipoprotein cholesterol, creatinine, aspartate aminotransferase, alanine aminotransferase, SDF-1α, and EPC count. All subjects provided written informed consent prior to study entry. The study was approved by Institutional Review Board.
Quantification of circulating EPCs by flow cytometry
To identify and quantify EPCs, we used a standardized protocol: the modified International Society for Hematotherapy and Graft Engineering (ISHAGE) sequential gating strategy, as proposed by Schmidt-Lucke et al. 16 Briefly, 1 mL of whole blood was collected from a forearm vein into EDTA tubes, transported to the cytometry laboratory, and processed within 1 to 2 hours of collection. Hence, 150 μL of whole blood was incubated with the following combination of antihuman monoclonal antibodies: 10 μL of anti-CD133 conjugated with allophycocyanin (APC) (Miltenyi Biotec, Bergisch Gladbach, Germany), 5 μL of anti-CD45 conjugated with APC-H7 (Becton Dickinson, Franklin Lakes, NJ, USA), 10 μL of anti-KDR (also known as type 2 VEGF) conjugated with phycoerythrin (Sigma, Milan, Italy), and 10 μL of anti-CD34 conjugated with fluorescein isothiocyanate (Becton Dickinson) for 30 minutes at 4°C in the dark. Red blood cell lysis was performed using FACS Lysing Solution (BD Biosciences, San Jose, CA, USA) diluted 1:10 (vol/vol) in distilled water and washed with phosphate-buffered saline before flow cytometry acquisition. Data acquisition was performed with a high-performance flow cytometer (FACSCanto II; BD Biosciences). According to the standardized protocol that we used, human circulating EPCs are identified by a minimal antigenic profile that includes at least one marker of stemness/immaturity (CD34 and/or CD133) plus at least one marker of endothelial commitment (KDR). CD45 staining was also performed to exclude cells, such as macrophages, that express “endothelial-like” proteins. 17 The same operator, who was blind to the clinical status of the patients, performed all of the cytometric analyses throughout the study.
Quantification of circulating active SDF-1α
Active SDF-1α was quantified with a custom assay based on the R&D Quantikine kit (R&D Systems, Inc., Minneapolis, MN, USA), following the manufacturer’s instruction except that we used, for detection, an antibody raised against full-length human SDF-1α (22-89) and specific for the N-terminal intact isoform (clone K15C; Chemicon) that was conjugated with horseradish peroxidase using a dedicated kit (ab102890; Abcam). A horseradish peroxidase–labeled antibody was used at final dilution of 1:20 000, based on a titration curve.
Statistical analysis
Comparisons of parameters at baseline between treatment groups were performed using the t test for normally distributed data, the Mann-Whitney test for non-normally distributed variables and the χ2 test for categorical variables. Linear regression analysis was performed using EPCs and SDF-1α (after log-transformation) as the dependent variable after adjusting for age, blood pressure, BMI, lipid levels, and patient group as categorical variables.
Intragroup differences within variables before and after treatment during the study have been analyzed using a general linear model for repeated measures. Values are provided as mean ± SD. Data were analyzed using the SPSS version 15 statistical package (SPSS; IBM Corporation, Armonk, NY, USA). Corrected P values are reported for significant results (2-tailed significance, P < .05).
Results
Since January 2013 to December 2014, 72 subjects satisfied the inclusion criteria. About 28 of 41 patients with HbA1c <6.5% and all the 31 patients with HbA1c 7.5% to 8.5% agreed to participate. At the end of the study, data were available for 28 patients in Group A and 31 patients in Group B. At baseline, Group A and Group B were similar in age, sex, smoking habit, BMI, and duration of diabetes. Also, liver and kidney function were similar, as well as concomitant drugs (antihypertensive, lipid-lowering, antiplatelet, metformin). Hemoglobin A1c, blood glucose, and triglycerides were significantly higher in Group B vs Group A but similar cholesterol levels (Table 1). After 4 months, we observed the following: (1) similarly reduced HbA1c (by 9.6% in Group A; by 10% in Group B) and (2) similarly increased EPCs (by 52% CD45−CD133+KDR+, by 47% CD45−CD34+KDR in Group A; by 62% CD45−CD133+KDR+, by 47% CD45−CD34+KDR in Group B) and SDF-1α concentrations (by 95% in Group A; by 106% in Group B). The extent of EPCs or SDF-1α changes was not related to HbA1c variations (Table 2).
Baseline characteristics of patients.

Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; HBA1c, hemoglobin A1c; HDL, high-density lipoprotein; NS, not significant.
Data are expressed as mean ± SD.
HbA1c, SDF-1α, EPCs at baseline and 4 months after treatment with alogliptin.
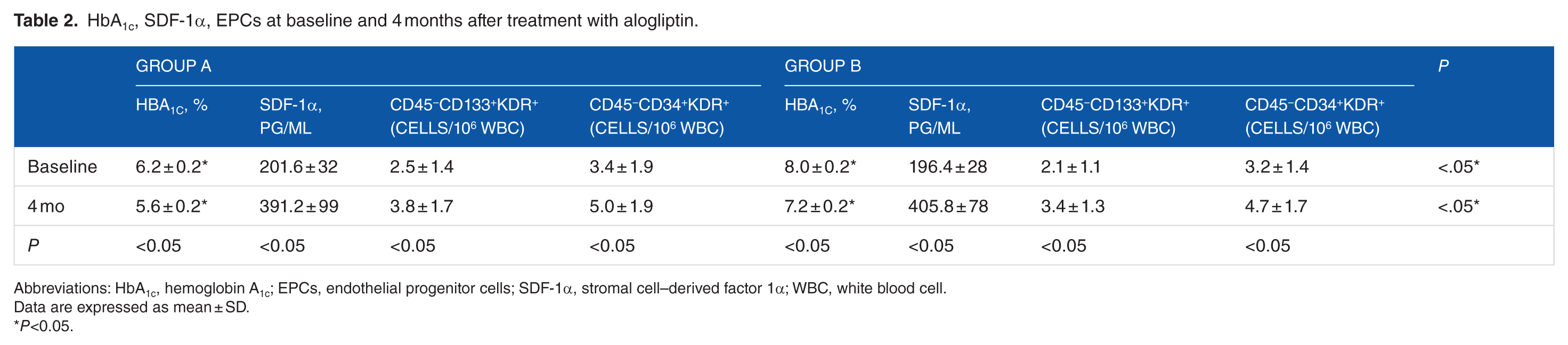
Abbreviations: HbA1c, hemoglobin A1c; EPCs, endothelial progenitor cells; SDF-1α, stromal cell–derived factor 1α; WBC, white blood cell.
Data are expressed as mean ± SD.
P<0.05.
Discussion
Our findings show that the 4-month treatment with alogliptin induced a significant increase in active SDF-1α. This effect was accompanied by a similar increase in EPCs and a similar reduction in HbA1c both in those with good and poor diabetes control. It still has to be elucidated whether increased EPC concentration is attributable to improved glycemic control, to upregulated SDF-1α (as a specific DPP-4-i mechanism of action), or both. The aim of the study was to investigate whether alogliptin, similar to other DPP-4-i, was able to increase EPC concentration. Indeed, the study was not designed to ascertain the relative weight of improved blood glucose control and SDF-1α upregulation.
Several years ago, Fadini et al 17 in a nonrandomized clinical trial comparing 4-week sitagliptin vs no additional treatment in addition to metformin and/or secretagogues observed an increase in circulating EPCs, accompanied by a concomitant upregulation of SDF-1α. The same group in a randomized, crossover, placebo-controlled trial assessed the effect of another DPP-4-i, linagliptin on EPCs in type 2 diabetic patients with or without chronic kidney disease. 18 The study demonstrated that linagliptin acutely (4 days) was able to increase EPCs and anti-inflammatory cells and suggested that a direct effect of DPP-4 inhibition may be important to lower vascular risk in diabetes, especially in the presence of chronic kidney disease. Two other papers challenged the influence of DPP-4-i on EPC count. In one of them, sitagliptin more than glimepiride was associated with a significant increase in EPCs—phenotypically characterized as CD34+/CXCR-4+ cells in 30 patients with type 2 diabetes in poor glucose control with metformin and/or sulfonylurea. 19 However, as sitagliptin obtained a better glucose control than glimepiride, the study did not clarify whether the obtained results were mainly due to a DPP-4-i class effect or to a glucose-lowering effect. In the other study, Dei Cas et al 20 tried to address the issue on whether the positive increase in EPCs is a benefit induced by DPP-4-i per se or it is secondary to improved glucose control. The authors compared the effect of vildagliptin vs glimepiride on top of metformin; once obtained similar HbA1c levels, vildagliptin but not glimepiride exerted a significant increase in EPCs at 12 months of follow-up. This finding suggests that SDF-1α, as major regulator of progenitor cell kinetics, more than improving glycemic control, may play a pivotal role in EPC circulating levels. The same study strongly suggests a long-term beneficial effect of this therapy on the endothelial repair process and its counterbalancing role to endothelial injury. In another mid-term study (3 months), saxagliptin and metformin equally improved the number of EPCs and flow-mediated dilation in newly diagnosed type 2 diabetic patients. 21 Although recent evidence proved that in patients with type 2 diabetes, a reduced baseline level of circulating CD34+ stem cells predicts adverse CV outcomes up to 6 years later, it is not known whether a reduction in blood stem cells causes CV events per se or whether it represents a bystander of inflammation, hematopoietic expansion, and bone marrow abnormalities, which in turn promote atherosclerosis. 22
Of note, intervention trials have failed to demonstrate any additional protective CV effect of this class of drugs compared with active comparators.23–25 These trials have been conducted in diabetic subjects at elevated CV risk: in the SAVOR-TIMI study (saxagliptin), about 80% of patients had a history of CV disease; in the EXAMINE study (alogliptin), diabetic patients had recent acute coronary syndrome; then, in the TECOS study (sitagliptin), patients had established CV disease. It still has to be elucidated whether DPP-4-i may exert a preventive role in the absence of CV risk factors. Indeed, various clinical trials have shown that cardiac function improved in patients with acute myocardial infarction who underwent bone marrow–derived stem cell therapy.26,27 These findings highlight the vasculoprotective effects of the EPCs, suggesting that in diabetic patients without established CV disease, a microvascular improvement induced by DPP-4-i may in turn prevent future macrovascular adverse events.
Although the present data confirm that DPP-4-i improved glycemic control and influenced EPC concentration also by upregulation of SDF-1α, we did not find an inverse correlation between HbA1c and EPCs or SDF-1α. Probably, confounders such as hypertension/antihypertensive drugs, dyslipidemia/lipid-lowering drugs, use of antiplatelet drugs, and smoking may have an influence on such parameters.
In conclusion, after 4-month treatment with alogliptin, either patients with good HbA1c or those with elevated HbA1c at baseline showed reduced HbA1c and concomitant similar increase in EPCs and active SDF-1α. The extent of increase in EPCs was independent from baseline diabetes control. These results are in accordance with previous studies investigating the influence of DPP-4-i on EPCs and confirm that also for alogliptin, the increase in EPC count is mediated by SDF-1α. Such effect is shared by all the DPP-4-i and has to be considered a class effect.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
RN was responsible for the design of the study, and drafting the manuscript. ELG was responsible for conducting the study. ELG was responsible for data and laboratory analysis.
