Abstract
Background:
Allergic fungal rhinosinusitis (AFRS) is a noninvasive fungal disease of the sinuses with a very high recurrence rate. A very small number of Japanese cases have been reported.
Material and methods:
The subjects were 6 patients with AFRS out of 429 patients who underwent endoscopic sinus surgery at Kagawa Rosai Hospital between December 2011 and November 2017. We retrospectively examined the clinical features and outcomes of these 6 patients.
Results:
The incidence of AFRS was 1.4% (6/429). Allergic fungal rhinosinusitis was unilateral in 5 cases and bilateral in 1. Computed tomography revealed hyperdense areas representing allergic mucin, but no patient exhibited bone erosion. Magnetic resonance imaging showed hypointense or no signal regions at the locations of allergic mucin. Postoperatively, 1 patient developed recurrence. Because the recurrent patient had no significant symptoms, he refused further surgery and received drug therapy. Preoperative eosinophil counts and total IgE levels were elevated in all patients; postoperatively, both remained high in the patient who developed recurrence. Postoperative treatments included steroid therapy and nasal irrigation.
Conclusions:
Allergic fungal rhinosinusitis is less prevalent in Japan than in Western nations. Peripheral blood eosinophil and serum IgE values may be used as the biomarkers.
Significance:
Allergic fungal rhinosinusitis is prone to recurrence. Postoperative treatment including steroid therapy is important in the management of AFRS.
Introduction
Allergic fungal rhinosinusitis (AFRS) is a noninvasive fungal disease of the sinuses with a very high recurrence rate and has been recognized as a distinct form of chronic rhinosinusitis (CRS).1-3 Allergic fungal rhinosinusitis is characterized by the production of allergic mucin and the development of severe eosinophilic inflammation as a result of a type I/type III allergic reaction to mold/fungus in their environment. Endoscopic sinus surgery (ESS) is considered as the primary method of AFRS treatment for removing fungal debris and eosinophilic mucin in the involved sinuses, and postoperative therapy has been recommended to prevent its recurrence.2,3 Although AFRS has been observed in approximately 5% to 10% of CRS patients who require surgery in the United States,4-6 only a small number of AFRS patients have been reported in Japan. To the best of our knowledge, the incidence of AFRS in Japanese CRS patients remains under investigation. In this work, we performed a retrospective clinical study of the clinical features, test results, types of treatment, and treatment outcomes of 6 AFRS patients treated in our institution.
Materials and Methods
Subjects
The subjects were 6 patients (4 men and 2 women) diagnosed with AFRS, out of 429 patients who underwent ESS for CRS at Kagawa Rosai Hospital between December 2011 and November 2017. Chronic rhinosinusitis was diagnosed by nasal endoscopy and/or computed tomography (CT) of the sinuses, and the diagnosis of AFRS was based on the diagnostic criteria established by the Bent and Kuhn diagnostic criteria3,7 (Table 1). The patients who met all essential criteria were diagnosed with AFRS.
Diagnostic criteria of allergic fungal rhinosinusitis. 5

The ESS involved opening the affected sinus and removing not only the eosinophilic mucin containing the fungus but also any nasal polyps. We evaluated patient age, sex, chief complaints, affected side, affected sinuses, presence of bronchial asthma, preoperative and postoperative eosinophil counts in peripheral blood, preoperative and postoperative IgE levels, fungal culture results, presence of a clinically compromised immune system, and disfiguring facial features such as proptosis or telecanthus. Allergen-specific IgE was measured by an AlaSTAT EIA (Diagnostic Products Corporation, Los Angeles, CA, USA) assay. Computed tomography findings such as bone erosion, bony expansion, bony thinning, septal deviation, and concha bullosa were analyzed. Magnetic resonance imaging (MRI) findings, the postoperative use of oral steroids, steroid nasal sprays and nasal irrigation, recurrence, further surgery, and the follow-up period were also examined. Fungi were identified in all 6 patients by pathologic tests of the mucin extracted during surgery. Preoperative (within 1 month before operation) and postoperative (2-6 months after operation) nasal obstruction, rhinorrhea, and facial pain were evaluated on a simple visual analogue scale from 0 (none) to 6 (extremely severe).
This study was conducted in Kagawa Rosai Hospital under the Declaration of Helsinki and approved by the Ethical Committee of Kagawa Rosai Hospital (H28-1).
Statistical analysis
Statistical analysis was performed using the Wilcoxon signed-rank test between 2 groups. Values of P < .05 were considered statistically significant. All statistical analyses were conducted using the statistical software “EZR” (Easy R). 8
Results
Of the 429 patients diagnosed with CRS who underwent ESS, 6 patients (1.4%) were diagnosed with AFRS (Table 2). They consisted of 4 men (66.7%) and 2 women (33.3%) with a mean age of 41.3 years. The most common symptoms were nasal discharge, nasal congestion, and headache in 3 patients (50%), followed by olfactory disturbance in 2 patients (33.3%). The affected side was unilateral in 5 cases (83.3%) and bilateral in 1 (16.7%). Ethmoid sinus lesions were present in all 6 of them. Only 1 patient suffered from bronchial asthma. No patient exhibited a clinically compromised immune system. The mean preoperative eosinophil count was 462.3 cells/µL and the mean total IgE level was 3572 IU/mL, both values were elevated. Serum fungus-specific IgE antibody was positive for Aspergillus in all the patients (class I or higher). Candida-, Alternaria-, and Trichophyton-specific IgE was positive in 5 patients (83.3%), 4 patients (66.7%), and 4 patients (66.7%), respectively. Serum pollen, mite, dog dander, and/or cat dander-specific IgE was positive in 5 cases (83.3%).
Clinical features of patients with allergic fungal rhinosinusitis.
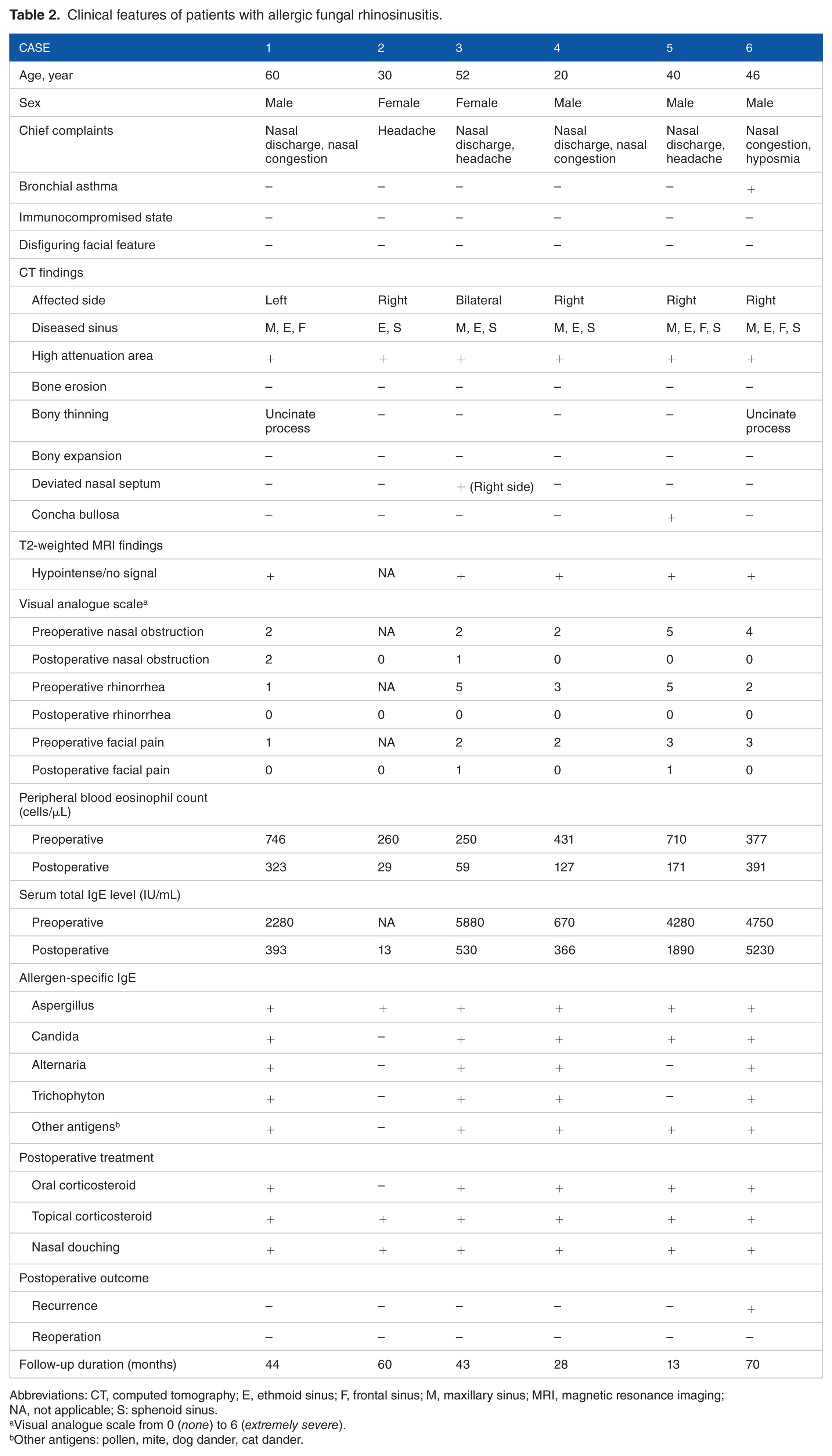
Abbreviations: CT, computed tomography; E, ethmoid sinus; F, frontal sinus; M, maxillary sinus; MRI, magnetic resonance imaging; NA, not applicable; S: sphenoid sinus.
Visual analogue scale from 0 (none) to 6 (extremely severe).
Other antigens: pollen, mite, dog dander, cat dander.
Hyper-attenuating soft tissue masses were revealed by CT in all of the patients. No bone erosion or bony expansion was observed. There was bony thinning of the uncinate process in 2 cases. One case had a deviated nasal septum to the affected side. There was concha bullosa of the affected side in 1 case. Hypointense or no signal regions on T2-weighted MRI at the locations of allergic mucin were observed in all 5 patients who underwent preoperative MRI. None of the cases had a disfiguring facial feature such as proptosis or telecanthus. Figure 1 shows preoperative CT and MRI pictures, and preoperative and postoperative endoscopic views of the nasal cavity in case 4.
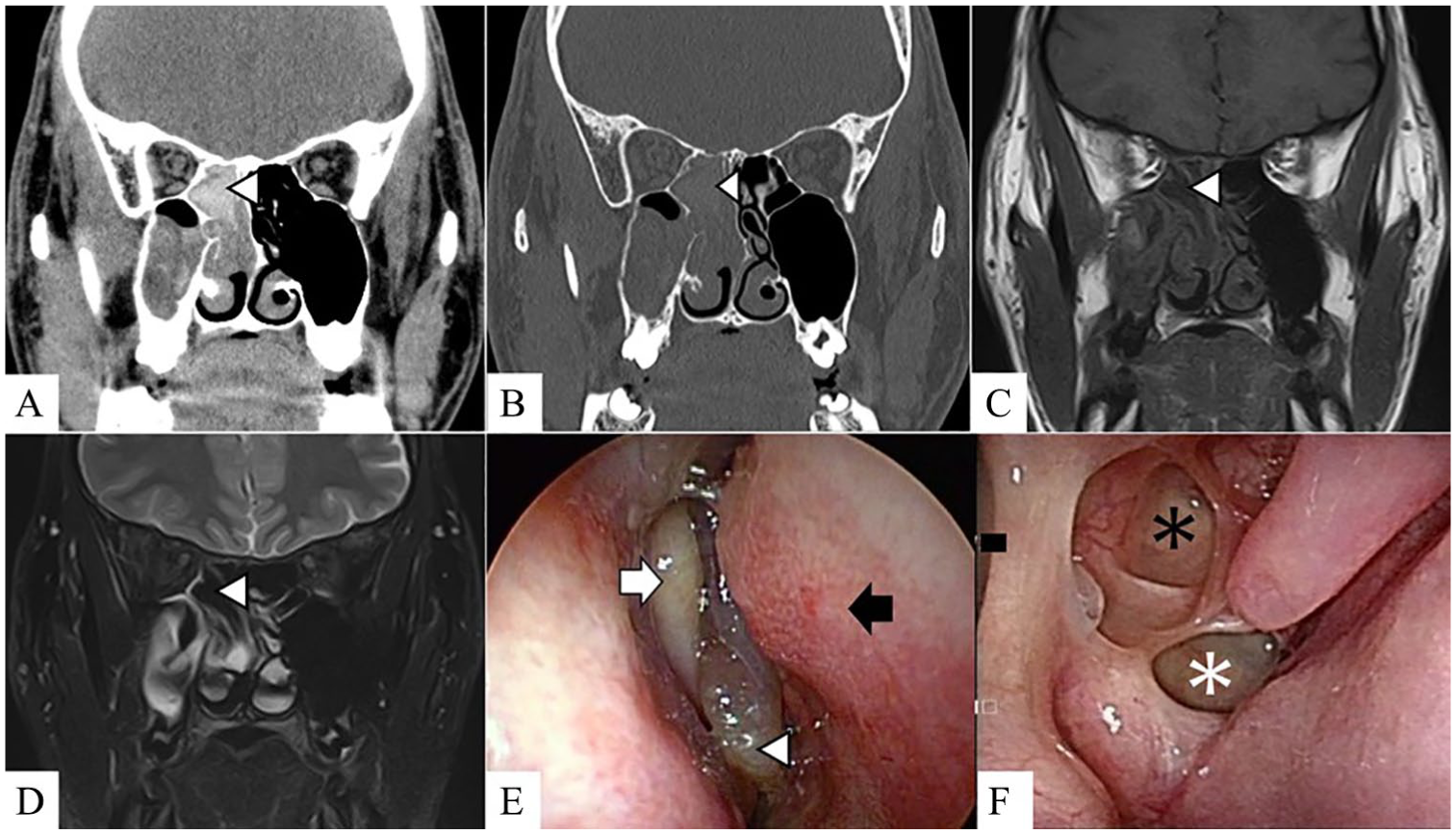
(A) Preoperative CT scan with soft-tissue window settings, (B) preoperative CT scan with bone window settings, (C) preoperative T1-weighted MRI imaging, (D) preoperative T2-weighted MRI imaging, (E) preoperative endoscopic findings in right nasal cavity, (F) postoperative endoscopic findings in right nasal cavity of case 4. (A-D) The white arrowheads indicate the viscous effusion suggesting eosinophilic mucin in the posterior ethmoid sinus on the right side. This region appeared (A, B) hyperdense on CT and (C) was hypointense on T1-weighted imaging and (D) no signal on T2-weighted imaging. (E) The white arrowhead shows a polyp in the right olfactory cleft. The polyp pushed the right middle turbinate (white arrow) laterally (black arrow: nasal septum). A year and a half after the surgery, there was no recurrence in (F) the nasal cavity (black asterisk: right posterior ethmoid sinus; white asterisk: right sphenoid sinus). CT indicates computed tomography; MRI, magnetic resonance imaging.
Postoperative rhinorrhea and facial pain were significantly improved compared with before surgery (Figure 2). Postoperatively, recurrence was found in 1 patient (case 6), and the other 5 patients were recurrence-free. Case 6, who developed recurrence 6 months after the surgery, exhibited almost no subjective symptoms and refused further surgery. The patient is currently undergoing treatment with oral steroids, a steroid nasal spray, an oral antihistamine, antileukotriene, and inhaled steroid therapy because he has bronchial asthma. No patient underwent further surgery. Of the 5 patients who were recurrence-free, 4 underwent IgE testing both preoperatively and postoperatively; in these patients, postoperative total IgE levels decreased from the preoperative levels (Figure 3). In case 6, the patient with recurrence, the postoperative total IgE level was similar to the preoperative level. The postoperative eosinophil count decreased from the preoperative count in the 5 patients who were recurrence-free (Figure 4). In the patient who developed recurrence, the postoperative eosinophil count was similar to the preoperative count. The mean follow-up period was 43 months. Postoperative treatment consisted of nasal irrigation with saline and steroid nasal sprays in all cases; 5 patients (83.3%), excluding patient 2, took oral steroids. Postoperative oral corticosteroid intake in the patients gradually reduced the dose of prednisolone from 20 mg daily within 1 month after the operations. No fungus was identified from the eosinophilic mucin culture in any of the patients.
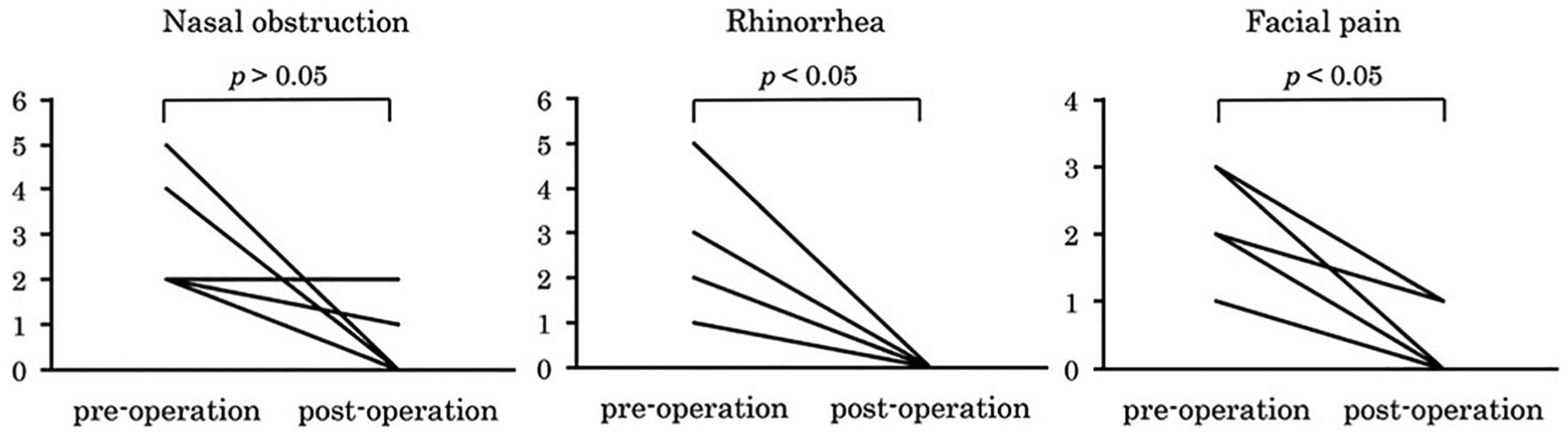
Changes between preoperative and postoperative symptoms (nasal obstruction, rhinorrhea, and facial pain) evaluated on a simple visual analogue scale from 0 (none) to 6 (extremely severe). Rhinorrhea and facial pain improved significantly after surgery.
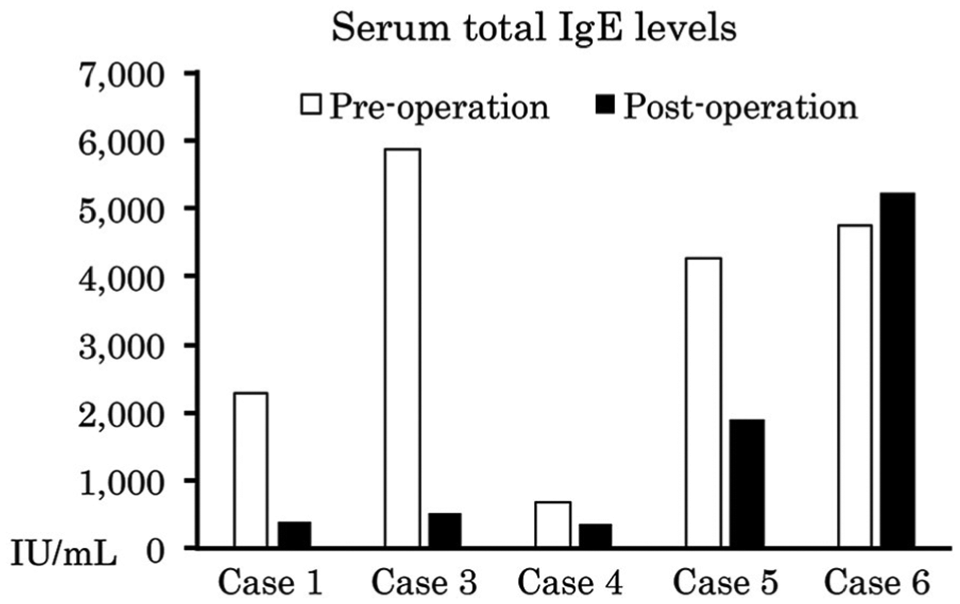
Preoperative and postoperative serum total IgE levels in each patient. The postoperative levels decreased after surgery in 4 cases. In contrast, the preoperative and postoperative levels were similar in the patient who developed recurrence (case 6).
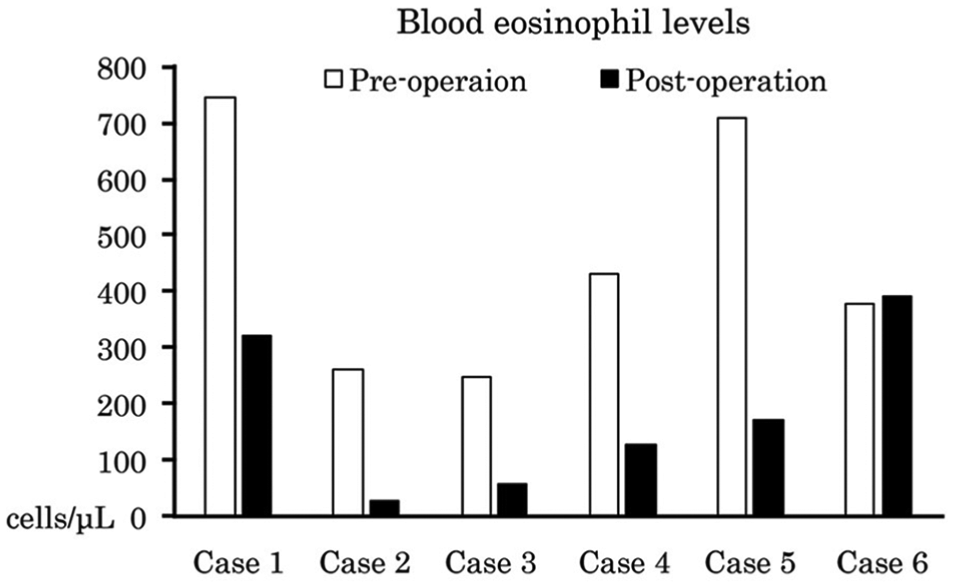
Preoperative and postoperative blood eosinophil levels. The postoperative eosinophil counts decreased after surgery in the 5 patients without recurrence. The preoperative and postoperative eosinophil counts were similar in the patient who developed recurrence (case 6).
Discussion
Allergic fungal rhinosinusitis is 1 form of CRS caused by a type I/type III allergic reaction. The T-cell response to a fungus induces a strong local allergic inflammatory reaction resulting in the accumulation of gelatinous eosinophilic mucin containing fungal hyphae. The presence of fungus in the mucin pathology must be demonstrated for a definitive diagnosis by showing sparse fungal hyphae with degranulating eosinophils.
The number of AFRS cases reported in Japan is small compared with the reports in studies from Western countries. There are 2 possible reasons for this difference. One is a possibility of underdiagnosis. Usually, a biopsy specimen obtained from a nasal polyp and/or paranasal sinus mucosa in a CRS patient is submitted for pathologic examination, and mucin is not used for histologic assessment. Consequently, some patients with AFRS may not be correctly diagnosed because of low awareness of this condition. The possibility of AFRS must always be considered before surgery for patients in whom AFRS cannot be ruled out, and prior measurement of antifungal IgE antibodies may help to increase the definitive diagnosis rate. The other reason is regional differences. Significant regional differences have been reported in the United States. In the northern states, 0% to 4% of patients undergoing ESS have AFRS. In contrast, in the hot and humid climate of the southern states including the lower portion of the Mississippi River region, 10% to 23% of such patients have AFRS. 9 In India, where the climate is even hotter and more humid, the rate of AFRS has ranged from 8.2% 10 to 51%. 11
Regarding the incidence of AFRS in Japan, Nakatani et al 12 reported that AFRS was present in 3.2% of CRS patients who required surgery, and Matsuwaki et al 13 reported that it was present in 3.9%. The work reported by Nakatani et al, Matsuwaki et al, and our hospital was done in regions where the average annual temperature is around 16°C and humidity is around 65%. In contrast, Kawabori et al 14 did not identify any AFRS patients. This is probably because their study was performed in Hokkaido, a colder region with an average annual temperature of 9.1°C and humidity of 67%. 15 These findings suggest that climate-associated regional differences in prevalence may also be present in Japan. In addition, a recent review reported that AFRS is more common in people of African descent 2 ; thus, racial differences may also contribute to the lower prevalence of AFRS in Japan.
In our study of 429 cases, there were 3 cases in which we were unable to identify any fungi on histopathologic examination; however, these 3 patients met other diagnostic criteria for AFRS. The patients exhibited an AFRS-like syndrome in which fungi could not be identified in mucin despite the presence of a systemic fungal allergy. This condition is considered to be extremely rare.16,17 The presence of fungi in tiny samples was difficult to determine possibly because the count of fungi present in the eosinophilic mucin was so small. No recurrence of CRS has been observed in these 3 patients after ESS; however, we continue to follow these 3 cases as potential AFRS patients.
Regarding patients with CRS for whom ESS is under consideration, CT is an essential imaging modality. Allergic fungal rhinosinusitis typically shows unilateral CT shadows but may be bilateral in some cases. In our patients, AFRS was bilateral in 16.7% of cases. Allergic fungal rhinosinusitis is typically found on CT as a hyperdense region due to the presence of iron, manganese, other heavy metals, or calcium components in eosinophilic mucin. The eosinophilic mucin of AFRS causes expansion of bony walls with consequent thinning and eventual erosion. The possible causes of bone erosion in AFRS are persistent pressure, hyperaemia, and inflammation of the surrounding mucosa. In addition, incipient infiltration by fungal elements and subsequent granulomatous reactions are also suggested.18,19 All 6 patients in our study exhibited hyperdense areas on CT, but none exhibited bone erosion and bony expansion. According to a prior study, bone erosion occurs in approximately 20% of cases of AFRS in Western countries. 20 In a review of Japan, bone erosion occurs in 52.6% of cases of AFRS. 12 The absence of bone erosion in our study might be due to the short time from onset to hospital visit.
The obstruction of sinus ostium, which may be accentuated by anatomic factors such as septal deviation or turbinate hypertrophy, results in blocked sinus drainage. This creates an ideal environment for proliferation of the fungus in the paranasal sinus. 21 A recent study reported that concha bullosa was more prevalent in patients with AFRS as compared with other forms of CRS with an accumulation of allergic mucin. 22 In this study, both a deviated nasal septum and concha bullosa were detected in only 1 case. The effects of the deviated nasal septum and concha bullosa might be limited during the onset and development of AFRS in Japan.
The high-protein components in sinonasal lesions that appeared hyperdense on CT can be detected as hyperintense signals on T1-weighted imaging and as hypointense/no signal regions on T2-weighted MRI imaging. In this study, these findings were present in all 5 patients who underwent MRI scanning. Magnetic resonance imaging is useful for distinguishing tumorous lesions and monitoring their spread into the cranium and is strongly recommended if AFRS is suspected. 23
According to Schubert and Goetz, 24 serum total IgE correlates with the disease status of AFRS and is useful as a postoperative monitoring biomarker to evaluate AFRS. An increase of ⩾10% in serum total IgE may indicate the recurrence of AFRS. Another study reported that the eosinophil count in peripheral blood may also reflect the disease status. 13 In this study, no postoperative recurrence was evident in the patients who had decreased IgE and eosinophil counts in peripheral blood after ESS.
Endoscopic sinus surgery is very important for AFRS therapy and diagnosis. It aims to completely remove allergic mucin and the causative pathogen, as well as to make a sufficient pathway for ventilation and drainage. 4 The postoperative nasal irrigation has an important role in the management of AFRS. All of the patients in this study were treated with nasal irrigation using saline only and steroid nasal sprays. Topical steroid irrigation is a basic tenet of AFRS management in the United States, but it is less common in Japan.
Postoperative medical treatment of AFRS is critical because AFRS is a frequently recurrent disease. 1 The recurrence rate ranges from 10% to nearly 100%.25,26 In this study, the recurrence was detected in 1 patient (16.7%). Sufficient surgery and postoperative steroid therapy may have beneficial effects. According to Schubert and Goetz, 24 patients who took oral steroids (tapering from prednisolone 0.5 mg/kg/d, for at least 2 months postoperatively) exhibited significantly greater improvement during 1 year of postoperative follow-up than patients who did not take steroids. A recent placebo-controlled, double-blinded, comparative clinical trial demonstrated that oral steroids after ESS have an additional effect. 27 However, long-term use of oral steroid may induce severe complications. Because AFRS is prone to recurrence, we continue to monitor our patients carefully, including the patient who already developed recurrence.
Conclusions
In this study, AFRS was found in 6 (1.4%) patients out of the 429 CRS patients who required surgery. Even though 5 of these 6 patients were recurrence-free, continued careful monitoring is required. The postoperative serum total IgE and blood eosinophil count were lower in patients without recurrence, so they may be useful biomarkers during postoperative follow-up.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI (Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan; Grant Number JP17K11329).
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SM and SK contributed to the design, implementation and presentation of the research, the writing of the manuscript and editing of the manuscript. SM, SK, TN, JM, MO, and KN contributed to the analysis of the results and the scientific discussion on the topic.
