Abstract
Introduction
Allergic fungal rhinosinusitis (AFRS) is characterized by refractory and high recurrence rate. Improper treatment may lead to repeated recurrence and even serious complications, including vision loss, blindness, and intracranial complications. However, AFRS is easy to be misdiagnosed clinically.
Objective
To ensure early diagnosis, the clinical presentations of patients with AFRS were studied.
Methods
Data from patients with sinusitis hospitalized in the First Affiliated Hospital of the University of Science and Technology of China (USTC) from January 2015 to October 2022 were collected. The patients were divided into three groups; group A patients with AFRS, group B patients suspected of AFRS, and group C patients with fungus ball sinusitis (FBS).We retrospectively analyzed the data using IBM SPSS 19.0 to perform the chi-square test and one-way ANOVA test.
Results
In total, 35 cases of AFRS, 91 cases of suspected AFRS, and 661 cases of FBS were rediagnosed. Compared with FBS patients, AFRS patients were younger, the total IgE, the percentage of eosinophils and basophils in peripheral blood were higher, and the proportion of patients with allergic rhinitis, asthma or hypo olfactory was higher. It had a higher recurrence rate. These results were also observed in the comparison between suspected AFRS patients and FBS patients, but no significant difference was found in the comparison between suspected AFRS patients and suspected AFRS patients.
Conclusions and Significance
AFRS may be misdiagnosed due to the low detection of fungi. To ensure early diagnosis, we recommend that patients with clinical, radiological, and laboratory features similar to those of AFRS but without evidence of fungal staining be treated according to the treatment criteria of AFRS.
Introduction
Allergic fungal rhinosinusitis (AFRS) is a noninvasive subtype of chronic rhinosinusitis (CRS) with nasal polyps (CRSwNP), which is characterized by the presence of eosinophilic mucin with noninvasive fungal hyphae within the sinuses and a type I hypersensitivity to fungi. The clinical symptoms of AFRS are similar to those of CRSwNP patients and mainly manifest as nasal congestion, purulent discharge, and hypoosmia. However, AFRS has some unique features, including radiology findings with differential densities of soft tissues in computed tomography (CT) scan and unilateral or discrete anatomical sinus involvement; endoscopic examination showed the growth of nasal polyp in the middle nasal canal or olfactory fissure, and there were many yellow−brown sticky secretions wrapped around it; the increase of total immunoglobulin E (IgE) and specific IgE in peripheral serum; and the discovery of a distinct mucinous material containing eosinophils, Charcot–Leyden crystals, and fungal hyphae was found in tissue resected from the sinuses.1,2 In 1993, Bent and Kuhn proposed the diagnostic criteria of AFRS based on a prospective study of 15 patients. 3 The main criteria are as follows: (1) nasal polyposis; (2) fungi in staining; (3) eosinophilic mucin without fungal invasion in sinus tissue; (4) type I hypersensitivity to fungi; and (5) characteristic radiology findings with differential densities of soft tissues in CT scan and unilateral or discrete anatomical sinus involvement. Minor criteria include bone erosion, Charcot–Leyden crystals, unilateral disease, peripheral eosinophilia, positive fungal culture, and the absence of immunodeficiency or diabetes. 4
AFRS usually develops in immunocompetent atopic individuals and presents at a younger age than other types of CRSwNP.5,6 Additionally, AFRS is much more common in geographic regions characterized by warm temperatures and high humidity, which are conducive to higher environmental fungal presence.7-10 In these regions, AFRS can account for 5%-10% of CRS cases and up to 32% of CRS patients undergoing sinus surgery.8,10 Sex predominance seems to vary among reports.10,11
Allergic fungal rhinosinusitis has a high recurrence rate. Although there are many kinds of treatment methods for this disease, including endoscopic sinus surgery, glucocorticoids, antifungal drugs, immunotherapy, and biological therapy, the current preferred treatment method is still endoscopic sinus surgery. 12 AFRS often requires two or more surgical treatments, but it is still difficult to achieve a satisfactory therapeutic effect. 13 Delays in misdiagnosis can lead to serious complications, including loss of smell, loss of vision, blindness, cranial nerve lesions, intracranial abscess, or cavernous sinus syndrome.14-17 In serious cases, fungi can cause invasive infection, resulting in patient death. 16 To ensure early diagnosis, a retrospective study was conducted to understand the clinical presentation of patients with AFRS.
Materials and Methods
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The protocols and experimental procedures in the present study were reviewed by the Ethics Committee of the First Affiliated Hospital of the University of Science and Technology of China (USTC), and ethical approval documents were obtained (2022−RE−012, January 14, 2022). Informed consent was not obtained since this was a retrospective study where there was no direct contact with the patients; however, patient-identifiable data were not recorded for the study. Three senior doctors participating in this study (Dr Tao Xu, Dr Yi-Cui Zhou, and Dr Qian Zhou) used the medical record information management system to search for discharge diagnoses with “sinusitis” as the keyword in chronological order, and searched for all types of sinusitis patients hospitalized in the First Affiliated Hospital of USTC from January 2015 to October 2022. We reread the patient’s surgical records and discharge records. The inclusion and exclusion criteria were as follows: patients with complete data, including previous medical history, complete surgical records, laboratory examination data, radiological imaging data, and pathological results; and exclusion criteria: (1) systemic antihistamines (including cold drugs) and/or corticosteroids were used within one week before hospitalization; (2) intranasal antihistamines and/or corticosteroids were used within 24 hours before hospitalization; and (3) incomplete records were excluded from the study. When the description of fungus ball, fungus filament or eosinophilic mucin appeared in the surgical records of patients who met the criteria, we reviewed the results of CT image, SPT, total serum IgE, fungal-specific IgE and histopathological sections, and rediagnosed the patient according to Bent−Kuhn diagnostic criteria. 3 We collected three groups of patients. Group A patients with AFRS, and evidence for rediagnosis: (1) nasal polyps under endoscopy; (2) CT image showed characteristic radiologic features of soft tissue density difference, unilateral or anatomically discrete sinus involvement; (3) fungal SPT positive or fungus-specific IgE increased; (4) pathological staining showed fungal infection; and (5) the eosinophilic mucin and noninvasive fungal hyphae are described in the records. Group B patients suspected of AFRS, and evidence for rediagnosis: (1) nasal polyps under endoscopy; (2) eosinophilic mucin without fungal invasion in sinus tissue; (3) characteristic radiology findings with differential densities of soft tissues in CT image; and (4) fungal-specific IgE was positive. Group C included patients with fungus ball sinusitis (FBS), and evidence for rediagnosis: (1) CT image shows unilateral or adjacent sinus disease, local density is uneven, and can show punctate calcification; (2) the operation record describes that there is a fungal mass filling the sinuses without mucin; and (3) fungal staining is positive in histopathological sections.
We collected the following data from the three groups: sex, age, number of preoperative recurrences, side and number of sinuses involved, bone erosion, peripheral blood eosinophil and basophil percentages, galactomannan (GM) test results, serum total IgE, pathological staining of fungi, and whether contained with allergic rhinitis, asthma, or olfactory hypothyroidism. We compared the differences in these items between groups A and B, groups A and C, and groups B and C.
Statistical Analysis
Statistical analysis was performed using IBM SPSS version 19.0 for Windows (IBM Corp., Armonk, NY, USA). The prevalence of AFRS was calculated as the percentage of patients diagnosed and treated with AFRS among all cases of CRS treated in the same period; the results are presented as a percentage with a 95% confidence interval. Descriptive statistics were used to present the demographic and clinical characteristics, as well as the imaging and pathological characteristics. Categorical variables are presented as frequencies and percentages, while continuous variables were first subjected to the Shapiro–Wilk test. If the data were normally distributed, the mean ± standard deviation was used, and if not, the median was used. The chi-square test was used for statistics of gender composition, GM test positive rate, and the proportion of allergic rhinitis, hypoosmia, and asthma in each group. One−way ANOVA was used to analyze the average age, peripheral blood eosinophils, basophils, number of recurrences and Lund–Mackay CT score among the 3 groups. If there were differences, the Mann–Whitney U test was used for further multiple comparisons, and the p values were subjected to Bonferroni correction. A P-value of <.05 was considered statistically significant at the 95% CI.
Results
Demographic and Clinical Characteristics of the Study Subjects (n = 787).
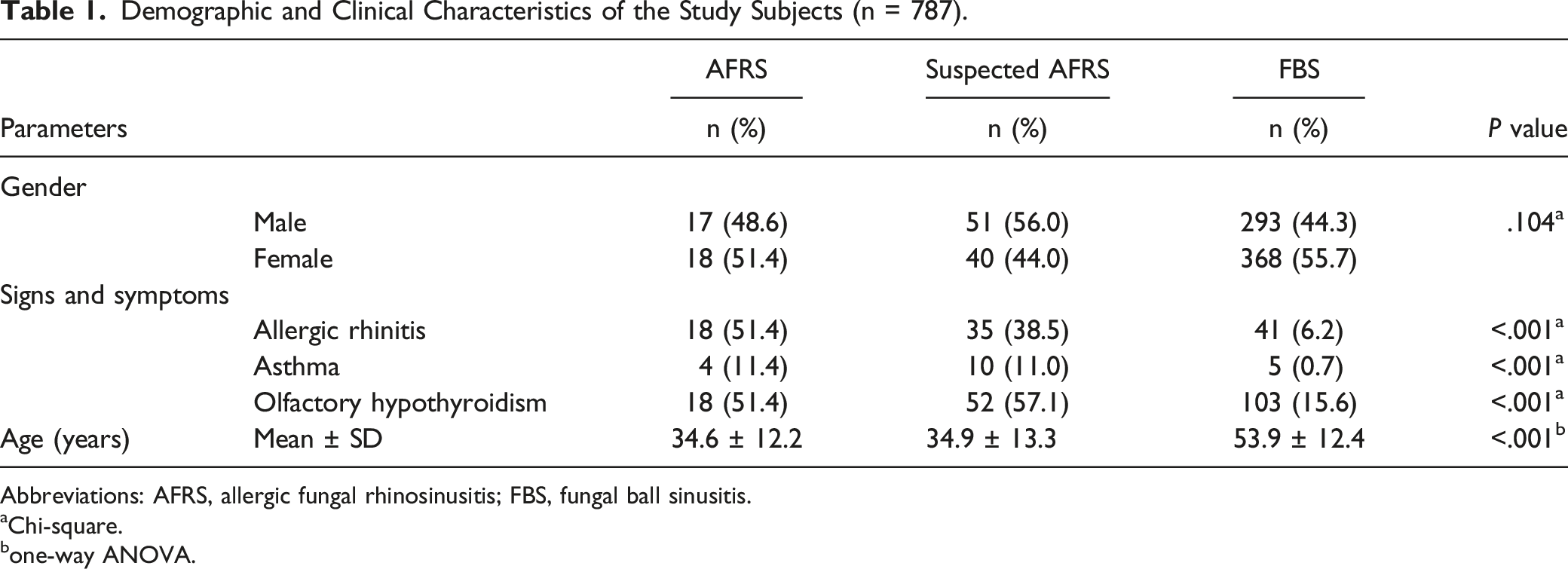
Abbreviations: AFRS, allergic fungal rhinosinusitis; FBS, fungal ball sinusitis.
aChi-square.
bone-way ANOVA.
Computed Tomography Scan Findings of the Study Subjects.
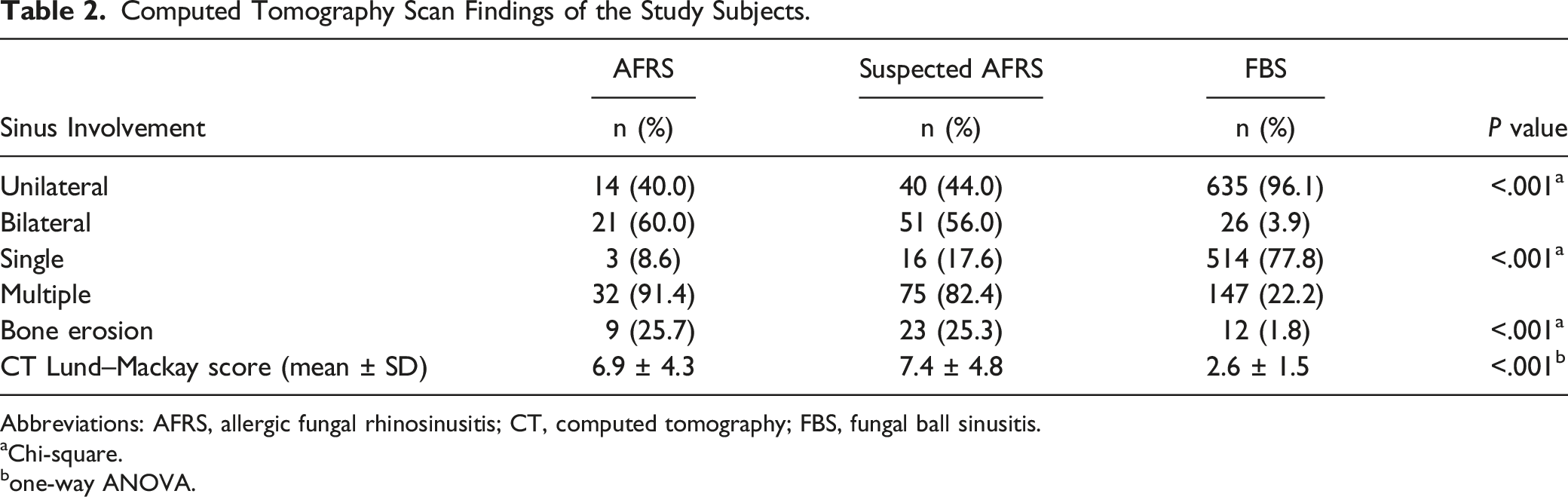
Abbreviations: AFRS, allergic fungal rhinosinusitis; CT, computed tomography; FBS, fungal ball sinusitis.
aChi-square.
bone-way ANOVA.
Comparison of Laboratory Findings Between the Study Groups.
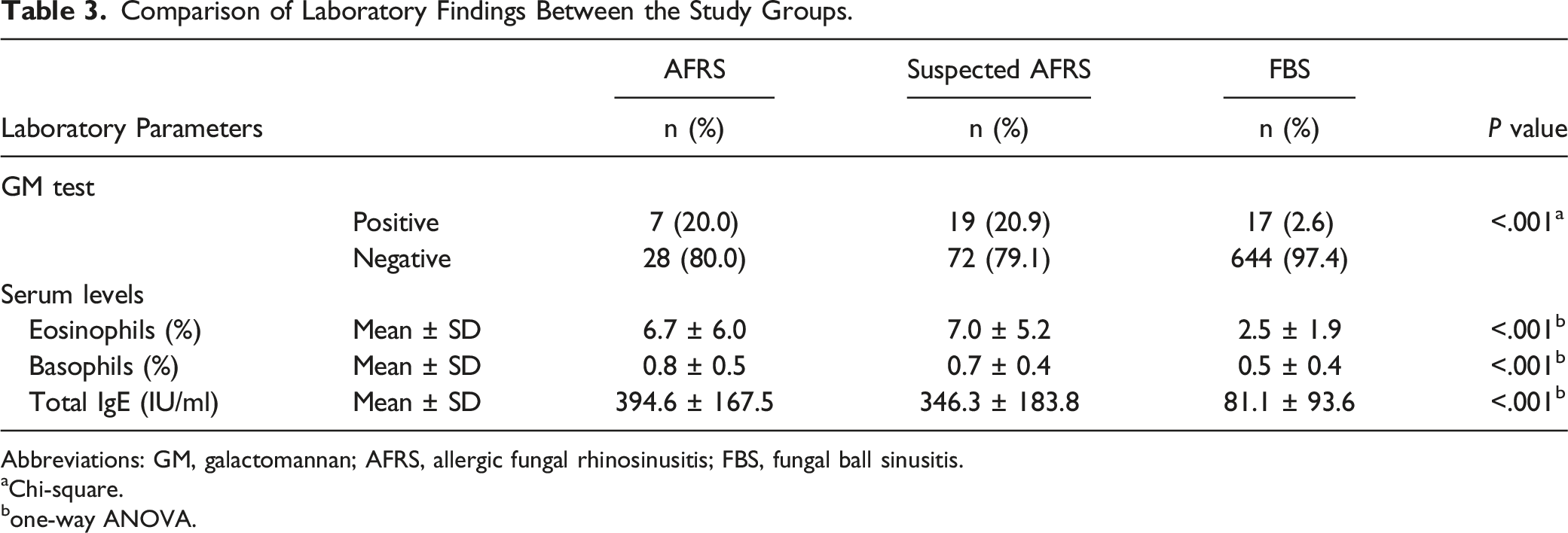
Abbreviations: GM, galactomannan; AFRS, allergic fungal rhinosinusitis; FBS, fungal ball sinusitis.
aChi-square.
bone-way ANOVA.
Comparison of Endoscopic Sinus Surgery Performed Between the Study Groups.
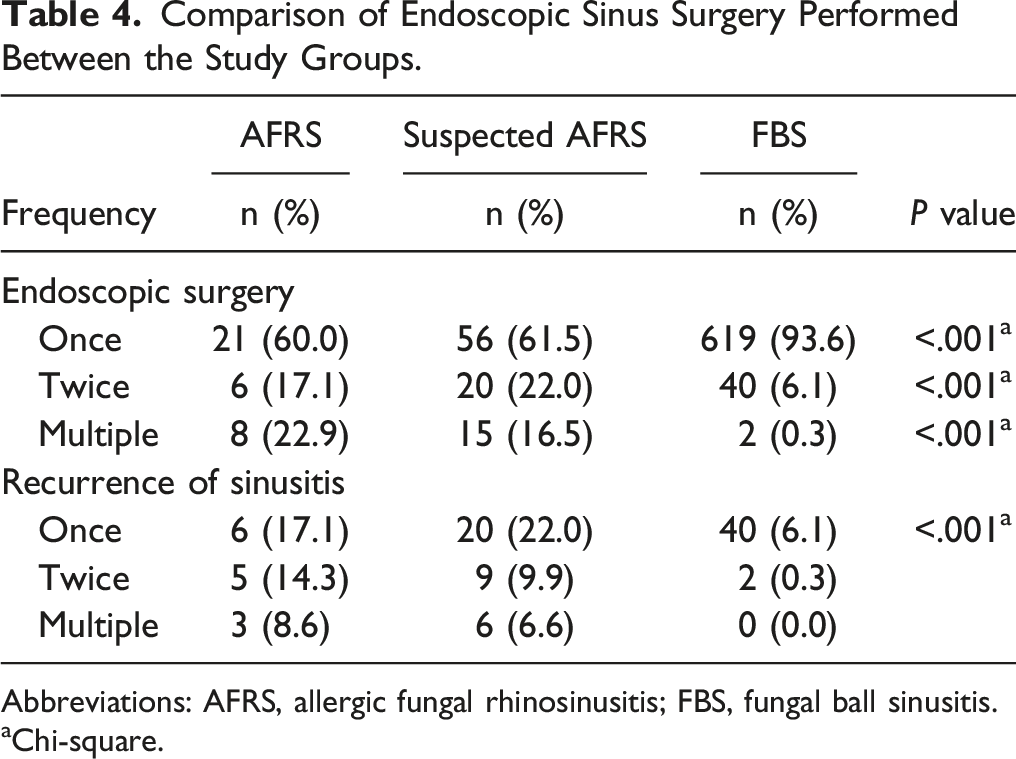
Abbreviations: AFRS, allergic fungal rhinosinusitis; FBS, fungal ball sinusitis.
aChi-square.
Discussion
AFRS develops in immunocompetent patients, whose occurrence is influenced by climate, geography, and several identified host factors.9,18-20 Some studies have shown that AFRS mostly occurs in men aged 21–33 years, which is significantly younger than chronic sinusitis without nasal polyps (CRSsNP) and CRSwNP.21,22 However, there was a lack of multicenter research on the epidemiology of AFRS in China. In this study, the average age of patients with AFRS was 34.6 years, which were significantly lower than the 53.9 years in patients with FBS (P < .001). It can also occur in women and children, and the youngest patient with AFRS was 13 years old. In addition, it had been reported that AFRS in children is more invasive and prone to significant complications. 23
AFRS mainly affects the unilateral paranasal sinuses, and 30.8% of cases are delayed on the contralateral side. 24 However, Salamah et al reported that AFRS involved all sinuses in 69.6% of patients and was bilateral in >53.5% of infected sinuses, of which the maxillary and ethmoid sinuses were most frequently involved. In that study, CT imaging showed sinus expansion (35.3%–51.2%), remodeling (20.6%–37.2%), and wall thinning (41.2%–58.1%). 25 In our study, we also found that patients with AFRS had more bilateral sinus involvement, with an incidence of 60.0%, and the involvement of two or more sinuses was as high as 91.4%, of which most patients had maxillary sinus involvement (34, 97.1%), followed by the ethmoid sinus (27, 77.1%), frontal sinus (18, 51.4%), and sphenoid sinus (19, 54.3%) involvement (see Table 4). We analyzed the possible reasons for this result because AFRS patients may not be identified until a long time after onset, and this delay leads to the progression of the disease. As a result, we observed more bilateral and multiple sinus invasions. AFRS is a noninvasive fungal sinusitis, but 20%∼58% patients will suffer from bone erosion caused by pressure atrophy and inflammatory mediators induced by accumulated fungal debris. 26 Bone erosion was also found in our 3 groups, among which group A (9, 25.7%) and group B (23, 25.3%) were more common than group C (12, 1.8%). Bone erosion in AFRS is a mostly reversible process. The study found that the paranasal sinus mucosa and periosteum were usually intact. Complete bone regeneration occurs in more than two-thirds of patients in a short period of time. This may suggest that bone erosion can regenerate after the compression of allergic mucin and inflammatory stimulation are relieved. Additionally, the rate of bone regeneration is not affected by the sex or age of the patients, and there is no difference in the bone regeneration rate between pediatric and adult patients. 26
AFRS is a type 2 immune response, characterized by antifungal IgE sensitivity, eosinophil-rich mucus (i.e., allergic mucin) and characteristic CT and magnetic resonance imaging findings in the paranasal sinuses. 27 Generally, AFRS has a high relapse rate, often requiring repeated or multiple operations, after which it is still difficult to resolve satisfactorily. Younis and Ahmed retrospectively analyzed 117 patients diagnosed with AFRS or EMCRS over a 5-year period. Twenty-six of 117 (22%) study patients underwent revision surgery. Within the 2-year follow-up, 5 of the 26 (19%) required additional revision surgery. 13 In our study, the proportion of patients with 2 or more surgical revisions was 40.0% in AFRS patients, which was much higher than the 6.4% in patients with FBS. This high recurrence rate often indicates that the disease has not been well controlled. These patients often have uncontrollable symptoms of allergic rhinitis or asthma and may have a decreased or even lost sense of smell. We also found that 51.4% of the patients in group A had a decreased sense of smell, 51.4% had allergic rhinitis and 11.4% had obvious asthma symptoms, which was significantly higher than these proportions in group C (P < .001, see Table 4). Therefore, in the treatment of AFRS, it is important to control complications. Meanwhile, to avoid reoperation of AFRS patients as much as possible, more attention should be given to patient management after surgery. Of course, it is important to accurately identify this disease.
Brescia et al. reported that eosinophils and basophils in peripheral blood increased significantly in AFRS, and statistical analysis found significantly higher levels of blood eosinophils and basophils in patients with AFRS who relapsed than in those who did not. 28 Our statistical analysis also showed that the proportion of eosinophils and basophils in the peripheral blood of patients in group A was significantly higher than that of patients in group C (P < .001), and was positively correlated with the number of recurrences, and this situation also occured in group B patients. In addition, we found that there was no significant difference between group A and group B patients in terms of age, the positive rate of a galactomannan test, total serum IgE, and the proportions of allergic rhinitis, asthma, or olfactory decline, which may suggest that there were some similarities between group A and group B patients. Unfortunately, according to the Bent−Kuhn diagnostic criteria, 91 patients in group B could not be diagnosed with AFRS due to no fungal etiological evidence.
The definition and diagnostic criteria of AFRS are still under debate, and only minor progress has been made in the past two decades toward achieving a consensus. The most widely used are the Bent–Kuhn criteria. Based on the clinical experience of each researcher and the available literature, different lists of criteria are proposed. These include characteristic eosinophilic mucin containing hyphae, together with positive results from a fungal strain or culture in the absence of tissue invasion by fungi, in addition to other clinical and biological evidence suggesting an allergy, such as positive atopic history, nasal polyposis, absence of immunodeficiency, and elevation of total or specific IgE or a positive SPT for fungal antigens.4,25 Other studies have analyzed specific CT features or endoscopic diagnostic signs related to the presence of AFRS to help diagnose,29,30 but these diagnostic criteria are not consistently reported in the literature. The strict application of the aforementioned criteria may lead to missed diagnoses of AFRS cases. We have also tried to score according to the main and minor criteria in the Bunt−Kuhn standard recommended by EPOS, which is still in the exploratory stage.
On the basis of extensive reports, the prevalence of AFRS is 5%–10%,31,32 and the prevalence of AFRS is greatly influenced by geographical location and climatic conditions.8,18,19,33 In South America, the prevalence of AFRS can account for 10%–23% of CRS. 8 In Japan, the prevalence of AFRS accounts for 1.4%–3.9% of CRS and is significantly lower in the northern region than in the southern region. 19 In Serbia, where the average temperature is lower, the prevalence of AFRS represents 1.3% of CRS. 33 However, the AFRS patients in our center accounted for less than 1% of the total CRSsNP and CRSwNP collected, which made us worry about whether some AFRS patients were missed, especially those in group B, who had no significant difference from group A in the various data we collected. However, according to the Bent−Kuhn diagnostic criteria, the patients in group B could not be diagnosed as AFRS because of no evidence of fungi.
As this study is a retrospective case analysis, there may be limitations in the study, including: (1) it is difficult to control whether the submitted tissue samples are qualified and whether the pathogenic microorganism culture and fungal staining of tissues are standardized; (2) due to the lack of cooperation with the Pathology Department in advance, no more methods were used to detect the negative results of fungal staining in tissue sections, such as hexamine silver or periodic acid-Schiff staining, microwave EnVision immunohistochemistry, and PCR; (3) not all patients' nasal sinus secretions have been tested for fungal smears and cultured in the laboratory; and (4) negative SPT of the fungal allergen cannot completely exclude the diagnosis of fungus-associated allergic rhinosinusitis. Because the selected reagents may not cover all the fungal spectra in the region, there may be regional differences in antigenicity. These limitations may lead to missed diagnoses. In addition, the sample of our center is limited. Next, we may need to conduct large-scale prospective research with other medical centers to further demonstrate our results.
Conclusions
Combined with this study and related literature review and summary, in comparison to typical CRSwNP or FBS patients, AFRS patients tend to be younger, and express consistently high serum total IgE, eosinophils, and basophils in peripheral blood. Patients are often complicated by allergic rhinitis or asthma and are prone to symptoms of decreased smell. However, it is possible that some patients with AFRS were missed due to lack of evidence of fungal etiology, leading to failure to receive timely and standardized treatment. Therefore, we propose a suggestion: when we encounter cases with clinical symptoms, CT imaging characteristics, allergic mucin, and elevated elevation of fungal-specific IgE that meet the Bent–Kuhn diagnostic criteria, but there is no fungal etiological evidence, we can consider treating patients according to the principles of AFRS after excluding EMCR to reduce the rate of recurrence and the incidence of complications.
Footnotes
Author Contributions
Tao Xu: Conceptualization; statistical analysis; investigation; data curation; writing-original draft. Xiao-Tao Guo: Statistical analysis; investigation. Yi-Cui Zhou, Qian Zhou: Data curation. Yin-Feng Wang: Conceptualization; funding acquisition; review and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by Natural Science Foundation of Anhui Province (1808085MH294).
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
