Abstract
BACKGROUND
COVID-19 placed a significant burden on the global healthcare system. Strain in critical care capacity has been associated with increased COVID-19-related ICU mortality. This study evaluates the impact of an early warning system and response team implemented on medical floors to safely triage and care for critically ill patients on the floor and preserve ICU capacity.
METHODS
We conducted a multicenter, retrospective cohort study, comparing outcomes between intervention and control hospitals within a US eight-hospital urban network. Patients hospitalized with COVID-19 pneumonia between April 13th, 2020 and June 19th, 2020 were included in the study, which was a time of a regional surge of COVID-19 admissions. An automated, electronic early warning protocol to identify patients with moderate-severe hypoxemia on the medical floors and implement early interventions was implemented at one of the eight hospitals (“the intervention hospital”).
RESULTS
Among 1024 patients, 403 (39%) were admitted to the intervention hospital and 621 (61%) were admitted to one of the control hospitals. Adjusted for potential confounders, patients at the intervention hospital were less likely to be admitted to the ICU (HR = 0.73, 95% CI 0.53, 1.000, P = .0499) compared to the control hospitals. Patients admitted from the floors to the ICU at the intervention hospital had shorter ICU stay (HR for ICU discharge: 1.74; 95% CI 1.21, 2.51, P = .003). There was no significant difference between intervention and control hospitals in need for mechanical ventilation (OR = 0.93; 95% CI 0.38, 2.31; P = .88) or hospital mortality (OR = 0.79; 95% CI 0.52, 1.18; P = .25).
CONCLUSION
A protocol to conserve ICU beds by implementing an early warning system with a dedicated response team to manage respiratory distress on the floors reduced ICU admission and was not associated with worse outcomes compared to hospitals that managed similar levels of respiratory distress in the ICU.
Background
Between January of 2020 and December 2021, coronavirus disease 2019 (COVID-19) infected more than 290 million people in over 216 countries, resulting in over 5.4 million deaths. 1 COVID-19 is primarily a pulmonary disease, with most hospitalized patients requiring supplemental oxygen. 2 Up to 14% of patients develop severe respiratory disease, 12% of hospitalized patients require ICU admission, and 60-70% of patients requiring ICU admission develop acute respiratory distress syndrome (ARDS). 3
The sheer volume of COVID-19 cases has placed significant strain on the healthcare system. In regions where the medical system is overwhelmed, standard of care for individuals seeking medical care may be affected, exacerbating negative health outcomes, including for those without COVID-19.4–6 Prior studies have also demonstrated poor outcomes compounded by shortages of intensive care facilities and/or access to mechanical ventilation equipment. 7 One study conducted in the United States has demonstrated that surges in hospital COVID-19 caseload remained detrimental to survival and potentially eroded benefits gained from emerging therapeutics. 8
At the onset of the pandemic, our hospital implemented an automated COVID-19 early electronic warning system with a dedicated response team protocol, called ‘CEWS’. The protocol was designed to preserve ICU capacity by identifying early signs of worsening hypoxemia and tachypnea, and responding with increased monitoring and treatment of patients on the medical floors. The CEWS represented a modification of an established, automated sepsis early warning system and 24/7 Sepsis Response Team that our hospital implemented in 2017, with improvement in sepsis-related patient outcomes. 9 Although this early warning system has shown benefit in sepsis, its value in patients with COVID-19 is unclear.
The aim of this study is to evaluate the impact of the CEWS protocol on ICU capacity while determining the safety of keeping patients with severe COVID-19 respiratory disease on the medical floors under augmented monitoring. Since several studies have shown benefit of automated early warning systems in sepsis,10–12 we hypothesize that the CEWS will decrease risk of ICU admission without worsening outcome of patients treated on the medical floor.
Methods
We performed a multicenter, retrospective cohort study involving eight hospitals in a single healthcare system in the Mid-Atlantic region of the United States during the time period between April 13 and June 19, 2020. This period corresponded with the first peak of the COVID-19 pandemic in the United States, during Phase 1 COVID-19 restrictions, representing a regional surge of COVID-19 cases. Because all hospitals are part of the same network, common COVID-19 care protocols were created by a multi-disciplinary team of providers from all hospitals and used across all centers.
Although the COVID-19 early warning electronic monitoring and alert was available at all eight hospitals, only one of the hospitals, a 960-bed tertiary care hospital, implemented the treatment response and subsequent interventions as outlined in Figure 1. This was the only hospital to implement the intervention because it already had a sepsis early warning and response team in place, which was easily transitioned to the CEWS. Three of the seven control hospitals were academic tertiary-care hospitals and four were community hospitals. Although the seven hospitals did not implement the CEWS treatment protocol, data on the electronic alerts generated by the CEWS were available in the electronic medical record (EMR) of each hospital for analysis. Eligible patients were above 18 years of age with COVID-19 disease admitted to the medical floor with SARS-CoV-2 positivity on nasopharyngeal PCR. Exclusion criteria were patients admitted directly from the Emergency Department to the ICU, and thus not monitored with the CEWS, patients with Do Not Intubate and Do Not Resuscitate status, patients transferred between the control and intervention hospital, patients with missing SOFA score values within 48 h of admission or patients transferred between hospitals.
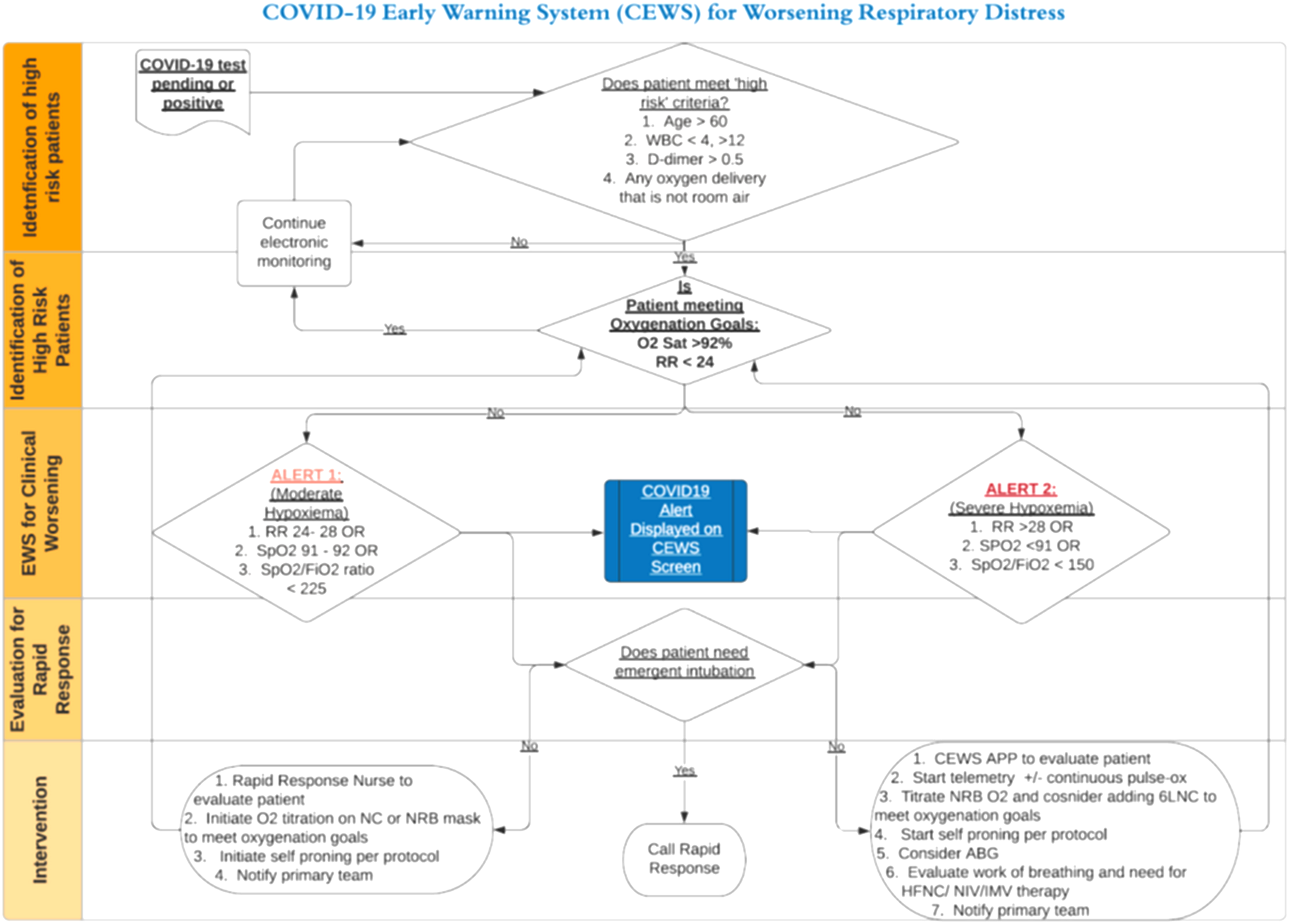
COVID-19 early warning system (CEWS) workflow.
The Intervention
The CEWS protocol was developed by a multidisciplinary team of physicians, nurses, advance practice providers, and programmed into the EHR by the hospital system's Chief Technology Officer. Prior to the COVID-19 pandemic, all eight hospitals had a protocol requiring all patients receiving supplemental oxygen via a non-rebreather mask, non-invasive positive pressure ventilation or high flow nasal cannula to be admitted to an Intermediate Care Unit (IMC) or Intensive Care Unit (ICU). At the intervention hospital, this policy was amended with integration of the CEWS protocol to allow patients with significant hypoxemia to receive higher fraction of inhaled oxygen (FiO2) via non-rebreather mask (NRM) at 15 liters/minute, with or without additional low-flow nasal canula support, on the medical floors. The CEWS protocol specified monitoring vital signs of these patients every 4 h and triggered subsequent treatment protocol per algorithm in Figure 1. At all hospitals, patients requiring NIV or HFNC were transferred to ICU.
The electronic component of the CEWS protocol flagged patients every time new vital signs or new labs were entered into the EMR. The alerts were displayed on a dedicated dashboard monitored 24/7 by the CEWS response team. The CEWS protocol algorithm was designed to identify high-risk patients, classify severity of respiratory distress as moderate or severe, increase bedside monitoring, allow for oxygen titration, and evaluate for transfer to a higher level of care as follows: (Figure 1)
The initial evaluation involved automated screening of age, WBC, D-Dimer and need for supplemental oxygen to identify patients with COVID-19 at “high risk” for deterioration based on available data at the time.13–15 Patients who met any ‘high risk’ criteria were evaluated for high respiratory rate (RR) or low oxygen saturation (SpO2), electronically triggering a “Moderate” or “Severe” hypoxemia alert. This was based on evidence suggesting that oxygen saturation and respiratory rate are predictors of progression to intubation.16,17
In conjunction with the CEWS protocol, an education campaign was implemented for floor nurses to respond to the CEWS electronic alerts and titrate oxygen to non-rebreather masks with the option to add nasal canula support and implement self-proning techniques as tolerated.
Standardization of Care Across All Hospitals
COVID-19 treatment guidelines and protocols were developed by a system-wide committee of physicians, pharmacists, respiratory therapists, and nursing leaders from all 8 hospitals. Treatment protocols included admission criteria, guidance on oxygen titration, pharmacotherapy, and ICU protocols for management of ARDS. The committee met weekly to develop and update guidelines as new evidence emerged.
Data Collection
All data, including vital signs, physiologic parameters, demographic information and outcomes, were collected electronically and were automatically populated into a dedicated COVID-19 data mart and stored in a Microsoft SQL server across all 8 hospitals. The data definitions and data entry requirements were similar at all hospitals.
Sequential Organ Failure Assessment (SOFA) scores were calculated using the worst values within the first 48 h of admission. ICU Admission SOFA Score was calculated using the worst daily SOFA score during the patient's stay on the medical floor based on established guidelines for randomized controlled trials. 18 A program was written to import components of the SOFA Score into the data mart including Glasgow Coma Scale, mean arterial pressure, vasopressor dose, PaO2/FiO2 ratio, urine output, and laboratory organ function score values. The worst daily values were used to calculate the daily SOFA score. If a component of the SOFA score was missing, the SOFA score for the patient was considered incomplete.
Outcome Measures
The primary outcome was ICU admission. Secondary outcomes included rate of intubation, hospital length of stay (LOS), ICU LOS, and mortality. These measures were selected to determine if the protocol was effective at conserving ICU resources while still ensuring patient safety.
Statistical Analysis
For categorical variables, we used Chi-square test or Fisher exact test for univariable analysis, and logistic regression with generalized estimating equation for multivariable analysis to account for clustering of patients within each hospital. Patient level covariates were pre-specified based on prior literature showing worse outcomes in these groups.19–21 For continuous variables, we used t-test or Wilcoxon rank-sum test for univariable analysis. For time-to-event data, because of competing risks, we plotted cumulative incidence curves and analyzed the data using Gray's test for univariable analysis and proportional sub distribution hazards models for multivariable analysis.22,23 All analyses were done using SAS version 9.4 (Cary, NC).
The study was approved by the Georgetown University-MedStar Health IRB and met the regulatory requirements for approval for a HIPAA waiver of authorization under IRB ID: Study00003145. Only de-identified data was used by external collaborators.
Results
During the study period, 2601 patients with COVID-19 disease were admitted to the study hospitals. Of these 2601 patients, 391 were excluded from the study because they were directly admitted from the Emergency Department to the ICU. An additional 233 patients were excluded because they were transferred between the control and intervention hospitals. One hundred twenty-seven patients were excluded because they had a DNR/DNI status, and 826 patients were excluded because SOFA score values within 48 h of admission were incomplete. For the final analysis, 1024 patients were included into the study (Figure 2).

Patient enrollment.
Of the 1024 patients, 621 (61%) were admitted to the intervention hospital and 403 (39%) were admitted to one of the control hospitals. Age and gender distributions between patients at the control and intervention hospitals were not significantly different. There were fewer white patients (21.6% vs 3.7%) and more patients of other race at the intervention hospitals (41.9% vs 23.0%), which was statistically significant (P < .0001). There were a higher percentage of patients of Hispanic ethnicity at the intervention hospital (32.3% vs 18.4%, P < .0001). There were more obese patients (BMI > 30), at the control hospitals (51.4% vs 45.4%, P = .02). Comorbidities associated with COVID-19 outcomes, including diabetes, hypertension, or heart disease, did not show a statistically significant difference between the control and intervention groups. Differences in SOFA score within 48 h of admission and the highest daily SOFA score prior to ICU admission were not statistically significant between the two groups (Table 1).
Patient characteristics and baseline severity of illness.

Multi-variable analysis, adjusting for admission SOFA score, age, gender, race, obesity, diabetes, hypertension and heart disease. Patients at the intervention hospital were less likely to be admitted to the ICU at any point in time (HR = 0.73, 95% CI 0.53, 1.000, P = .0499) compared to the control hospitals. (Table 2). There was no significant difference between the groups in univariate analysis (P = .60). We next performed a sensitivity analysis using a cohort of patients for whom the highest daily SOFA score prior to ICU admission was available. The results remained consistent in that patients at the intervention hospital had a lower risk of ICU admission (HR = 0.68, 95% CI 0.49, 0.94, P = .02).
Multi-variable analysis of ICU admission based on Pre-specified variables.

*1024 patients had admission SOFA scores.
Secondary outcomes were also analyzed with adjustment for admission SOFA score, age, gender, race, obesity, diabetes, hypertension, and heart disease. Patients managed with CEWS had increased hospital LOS compared to the patients managed on the medical floor at control hospitals (hazard ratio for hospital discharge: 0.73; 95% CI 0.64, 0.83; P < .0001). The sensitivity analysis based on patients with highest available SOFA score before ICU admission provided similar results (HR = 0.70, 95% CI 0.62, 0.80, P < .0001). The 194 patients that required ICU admission had a shorter ICU stay (HR 1.74; 95% CI 1.21, 2.51; P = .003) if they were managed with the CEWS on the floors. The sensitivity analysis based on patients with highest available SOFA score before ICU admission provided similar results (HR = 1.72, 95% CI 1.19, 2.50, P = 0.004). There was no significant difference in need for mechanical ventilation between the intervention and control hospitals (OR = 0.93; 95% CI 0.38, 2.31; P = .88). Similarly, there was no significant difference in hospital mortality between intervention and control hospitals (OR = 0.79; 95% CI 0.52, 1.18; P = .25) (Table 3).
Comparison of secondary outcomes at intervention hospital and control hospitals.

* n = 1024.
** n = 194.
*** HR for hospital discharge.
**** HR for discharge from ICU.
Discussion
In this study, we evaluated the impact of a protocol using an electronic early warning system and a dedicated response team, with the goal of early identifying and managing COVID-19 patients with worsening respiratory distress on the medical floors during a COVID-19 surge, when ICU capacity was strained. These patients would have otherwise been transferred to higher levels of care in a non-pandemic setting. After adjusting for confounding variables, the protocol resulted in a significantly lower risk of ICU admission, allowing patients with severe hypoxemia to be managed on the medical floors with a non-rebreather mask and nasal cannula without negatively affecting rates of intubation or hospital mortality.
The correlation between ICU capacity and COVID-19 outcomes has been well been documented. A large study of US Veterans Administrations Hospitals found that strain on critical care capacity was associated with increased COVID-19 ICU mortality despite augmented ICU capacity during the pandemic. 24 Another study has found an association between the number of ICU beds/100 000 population and COVID-19 mortality. 25 The CEWS protocol proved to be a feasible strategy without compromising patient safety. In addition, oxygen support with a non-rebreather mask with the addition of nasal canula provided an inexpensive and easily accessible means of support compared to the resources utilized as a result of ICU admission.
While several studies in non-COVID-19 patients have shown that delaying ICU admission is associated with an increase in mortality,26–28 pairing the electronic early warning system with a dedicated response team may have ensured the safety of this protocol. 29 The CEWS protocol created a secondary triage to identify patients who required escalation to a higher level of care while safely keeping those that could be optimized on higher levels of oxygen support on the floors. We also found that ICU length of stay was shorter at the hospital that implemented the CEWS protocol thus creating additional critical care capacity. The shorter ICU stay was likely a combination of early identification of deteriorating patients, as well as ability to transfer patients who were not worsening out of the ICU and to the floors on high levels of oxygen on non-rebreather support.
The combination of an early warning system and a dedicated response team has been shown to improve response times and early escalation, leading to improved patient outcomes for sepsis and rapid response.9,10,30 In our study, we add to that body of literature in the setting of a pandemic where such an approach safely extended the availability of critical ICU resources.
In designing the study, we elected to compare outcomes across hospitals instead of doing a before and after study, as care protocols were frequently being updated during the first wave of the pandemic and standard of care changed during this time. In addition, we wanted to capture the impact of the protocol during similar phases of the COVID-19 surge and impact on capacity.
Given that the intervention was implemented at one hospital in a network of hospitals under the same system, and all hospitals shared COVID-19 care protocols and outcome databases, we were able to compare outcomes between intervention and control hospitals during the same surge level of the pandemic. However, there are several limitations of the study. It is a retrospective study and limited to a single hospital system in one area of the country, potentially limiting the generalizability of the results. In addition, we were limited by missing data for severity of illness analysis using SOFA score; however, a sensitivity analysis utilizing highest pre-ICU SOFA score did not significantly alter the results. Some patients may have been admitted to the ICU for COVID-19 complications other than respiratory distress alone, and this may not have been detected by the CEWS protocol. We were unable to capture the precise reason for ICU admission in the cohort, even though the large majority of ICU admissions have been for respiratory distress. Furthermore, despite efforts to control for confounding factors, residual confounding may be present due to potential between-hospital differences that are not accounted for in our multivariable analysis.
While isolated to a single geographic region, there may also have also been differences in case volumes and hospital capacity limits between control hospitals and the intervention hospital. Since the CEWS protocol consisted of several components, including an automated early warning system and dedicated treatment team, it is difficult to determine which components of this protocol improved outcomes at the intervention hospital. It is also possible that providing such close monitoring to critically ill patients on the medical floors may have diverted care from other patients and resulted in worse clinical outcomes for that population.
Implementing such a protocol and treatment team may be costly and cumbersome in facilities lacking similar pre-existing services. Although the CEWS protocol likely improved ICU capacity by reducing or delaying ICU admission for severe COVID-19 respiratory failure on the medical floors, direct measurements of ICU capacity could not be precisely determined. However, since the majority of the patients receiving a severe hypoxemia alert met criteria for admission to the ICU without the CEWS protocol, those admissions would have adversely affected ICU capacity. We also saw that patients monitored and treated with the CEWS protocol had overall longer hospital LOS but shorter ICU LOS compared to those who did not receive the protocol. These results are not surprising since longer hospital LOS in the intervention group is likely a result of increased severity of illness in this population of patients that would ordinarily be admitted to the ICU but were instead managed on the medical floors.
There has been a significant improvement in the strain on ICU capacity from COVID-19 after the advent of vaccines and with subsequent less virulent strains. 31 Thus, a protocol such as ours was less likely to be beneficial in subsequent waves of the pandemic. However, our strategy may be relevant for future emerging strains or pathogens that have the potential to result in similar capacity challenges.
In summary, a protocol designed to conserve ICU beds by implementing an early warning system and a dedicated treatment team allowed for the safe management of patients with moderate to severe COVID-19 ARDS on the medical floors during a regional surge in cases, resulting in a reduction in ICU admissions. With new strains of the virus causing surges of the ongoing pandemic around the world, implementation of similar strategies could conserve ICU capacity during times of capacity stress.
Footnotes
List of Abbreviations
Acknowledgments
Drs Seife Yohannes, Nitin Seam, Junfeng Sun, and Michael Keller participated in research design, analysis of results, writing and editing of manuscript.
Dr Sun Junfeng provided statistical analysis for the study.
Drs Joel McAlduff, Susanne Lara, and Ms. Janet Thorne participated in review and editing of the manuscript.
Dr Seife Yohannes participated in database creation and data organization/validation.The Authors would like to acknowledge the following additional individuals for their support:
Ms. Mary Kim, MedStar Health, for data analytics and data visualization support.
Mr Yidnekachew Ejamo, MedStar Health, for clinical data mart development and data validation.
Mr Gaurav Metha, MedStar Washington Hospital Center, for data analytics and visualization support.
Dr Seife Yohannes is the guarantor of the paper and takes full responsibility for the integrity of the work as a hole, from inception to published article.
Author Contribution(s)
Ethics Approval and Consent to Participate
The study was approved by the host hospital Institutional Review Board and granted approval for a HIPAA waiver of authorization. Collaborators at outside institutions only had access to anonymized data.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Funding
All authors contributed their time and no source of funding was used to conduct the study. Article Processing Charge was contributed by the National Institutes of Health. All artistic works and figures are owned by the authors.
