Abstract
There are few reports on sepsis caused by infection with Nocardia in people with normal immune function, and there is no report on bronchial tumor caused by Nocardia. This paper describes a case of Nocardia farcinica pneumonia with sepsis and a bronchial neoplasm in a healthy patient.
Keywords
Introduction
Pulmonary Nocardia is a rare infectious disease. Nocardia is a genus of aerobic, filamentous, branched Gram-positive actinomycetes. It does not form spores, is weakly positive on acid-fast staining. It usually exists in soil and water. Nocardia infection is frequent in immunosuppressed individuals. However, it can also occur in individuals without obvious immune function limitation. Nocardia can also colonize the lower respiratory tract, especially in patients with basic lung disease. 1 More than 109 species of Nocardia have been identified, and about 54 species can infect humans, At present, the most common strains causing isolated nocardiosis include Nocardia brasiliensis, Nocardia asteroides and Nocardia farcinica. 2 This paper reports a case of Nocardia farcinica pulmonary disease complicated with sepsis and a bronchial neoplasm in a healthy patient.
The study was approved by our institutional review committee and the patient gave informed consent. The reporting of this study conforms to the CARE guidelines. 3
Case Report
A 59-year-old healthy man, who worked in the decoration and transportation sectors, suddenly developed a severe paroxysmal cough and expectoration on October 27, 2021. He presented to the hospital outpatient clinic, where chest tomography (CT) showed multiple patches and nodules, with some bronchiectasis in both lungs. The clinician suspected pneumonia and treated him with moxifloxacin tablets for three days, but his temperature remained elevated at up to 39.7°C. Therefore, he presented to the emergency department. Chest CT showed multiple infections of both lungs with bronchiectasis, and progression compared with the previous examination (10/27/2021) (Figure 1). Blood routine examination showed that leukocyte count increased to 18.1 × 109/L (normal range: 4.0–10.0 × 109/L); and blood C-reactive protein level (CRP) was 165.7 mg/L (normal range: 0–8.0 mg/L). Arterial blood gas indicates mild hypoxemia (PaO2, 8.7 kpa). He was admitted to hospital.

Chest CT performed on November 4, 2021 showing multiple spots and nodules in both lungs, suggesting infectious lesions compared (cf. the CT on October 27, 2021). There was emphysema in both upper lungs, calcified spots in the left coronary artery, and calcified lesions in the prostate.
On physical examination, his pulse was 94 per min, while the respiration rate was 20 per min, blood pressure was 102/68 mm Hg, and temperature was 40°C. The movement of both lungs was symmetrical, with normal percussion of both lungs, and reduced respiratory sounds in the right lower lung. The examinations of the heart, abdomen, limbs, skin, and nervous system were normal.
Laboratory examination showed mild abnormalities of coagulation function and liver function, and the CRP level was 185.9 mg/L (↑). Routine hematology revealed a leukocyte count of 21.7 × 109/L (↑),neutrophil percentage of 91.7% (↑), neutrophil count of 19.9 × 109/L (↑), erythrocyte sedimentation rate of 62 mm/h (↑), negative Cryptococcus haemolyticus capsule antigen test, GM [Galactomannan] antigen test result of 0.34 μg/L, “fungal G test” result of 56 ng/L, immunoglobulin E level of 1518 IU/mL (↑), and procalcitonin level of 0.65 μg/L (↑). Immunoglobulin, rheumatism, light chain, extractable nuclear antigen, and anti-neutrophil cytoplasmic antigen tests were all abnormal. Sputum culture and acid-fast bacilli staining of sputum was negative.
The patient took moxifloxacin tablets orally in the outpatient department, the anti-inflammatory treatment effect was poor, and the pulmonary infection developed rapidly along the pulmonary vessels. We suspected the infection of special pathogens and blood transmission. After repeated inquiries, he recalled that he had transported moldy paper floors 15 days before the onset of the disease. While empirically treated with antibiotics (piperacillin tazobactam combined with moxifloxacin), peripheral blood was sent for quantitative macrogenomic analysis(Q-mNGS). Quantitative macro genome detection is to obtain the microbial species and abundance information contained in biological samples by high-throughput sequencing of nucleic acids extracted from biological samples and comparative analysis using bioinformatics. Q-mNGS based on Illumina sequencing platform and PCR free database building technology.Three days later, the result of blood quantitative macrogenomic analysis was Nocardia farcinica. Then he was started on sulfamethoxazole 1.92 gram orally q12h. As chest CT showed that the lumen of the basal segment of the right lower lobe was narrow (Figure 2), with the consent of the patient, tracheoscopy was performed. Under endotracheal endoscopy, a new mass was accidentally found to block the bronchial orifice of the basal segment of the right lower lobe (Figure 5a). Neobiopathology showed chronic active inflammation of mucosa with erosion and squamous metaplasia (Figure 3). The quantitative macrogenomic analysis of bronchoalveolar lavage fluid also showed Nocardia farcinica. One week later, his temperature was normal and his general condition improved. Blood leukocytes and CRP returned to normal, and liver function and coagulation function returned to normal. However, immunoglobulin E 1342iu / ml was rechecked after treatment. He was discharged from the hospital on November 15 and was diagnosed with nocardiosis and nocardiosis bacteremia. Although the pathology showed chronic inflammation in the basal segment of the right lower lung, we still suspected lung cancer clinically. The follow-up CT 2 weeks after discharge showed that the infected lesions were significantly absorbed and subsided (Figure 4). Reexamination of tracheoscopy showed that the new organisms in the basal segment of the right lower lobe disappeared (Figure 5b). Therefore, we speculate that the new bronchial organisms are caused by Nocardia infection. The patient was still taking sulfamethoxazole tablets and was tracked as an outpatient.
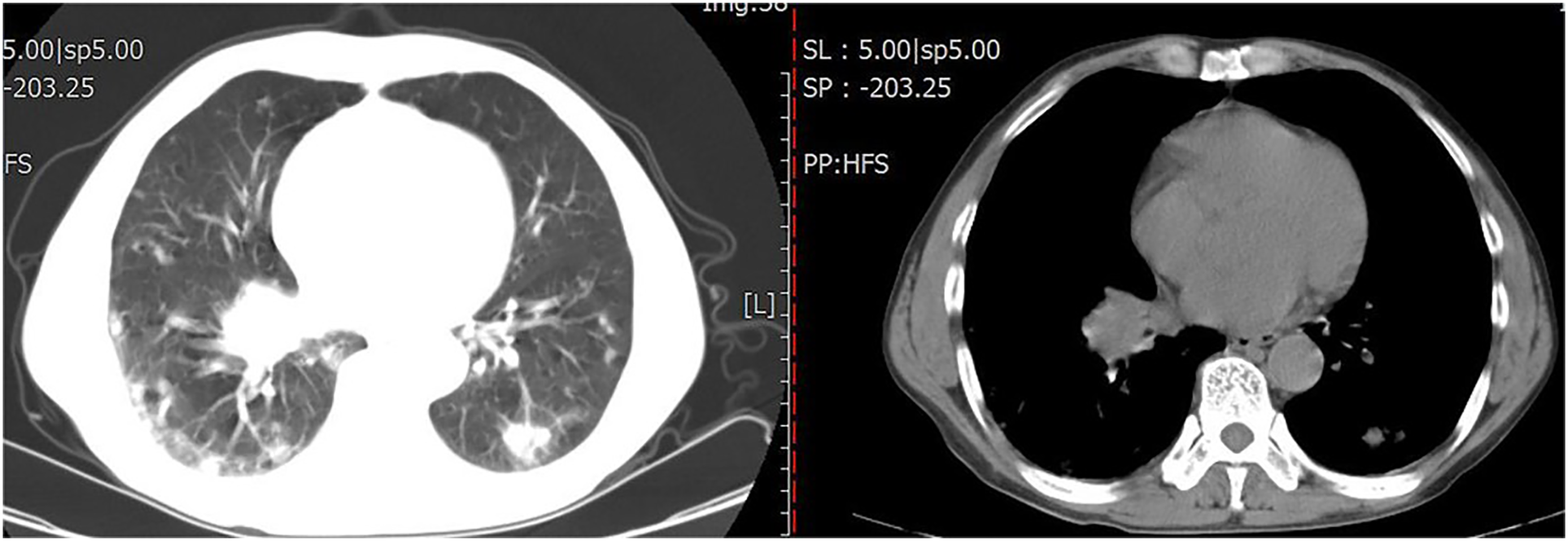
Chest CT performed on November 4, 2021 showing that the orifice of the basal segment of the lower lobe of the right lung was narrow.
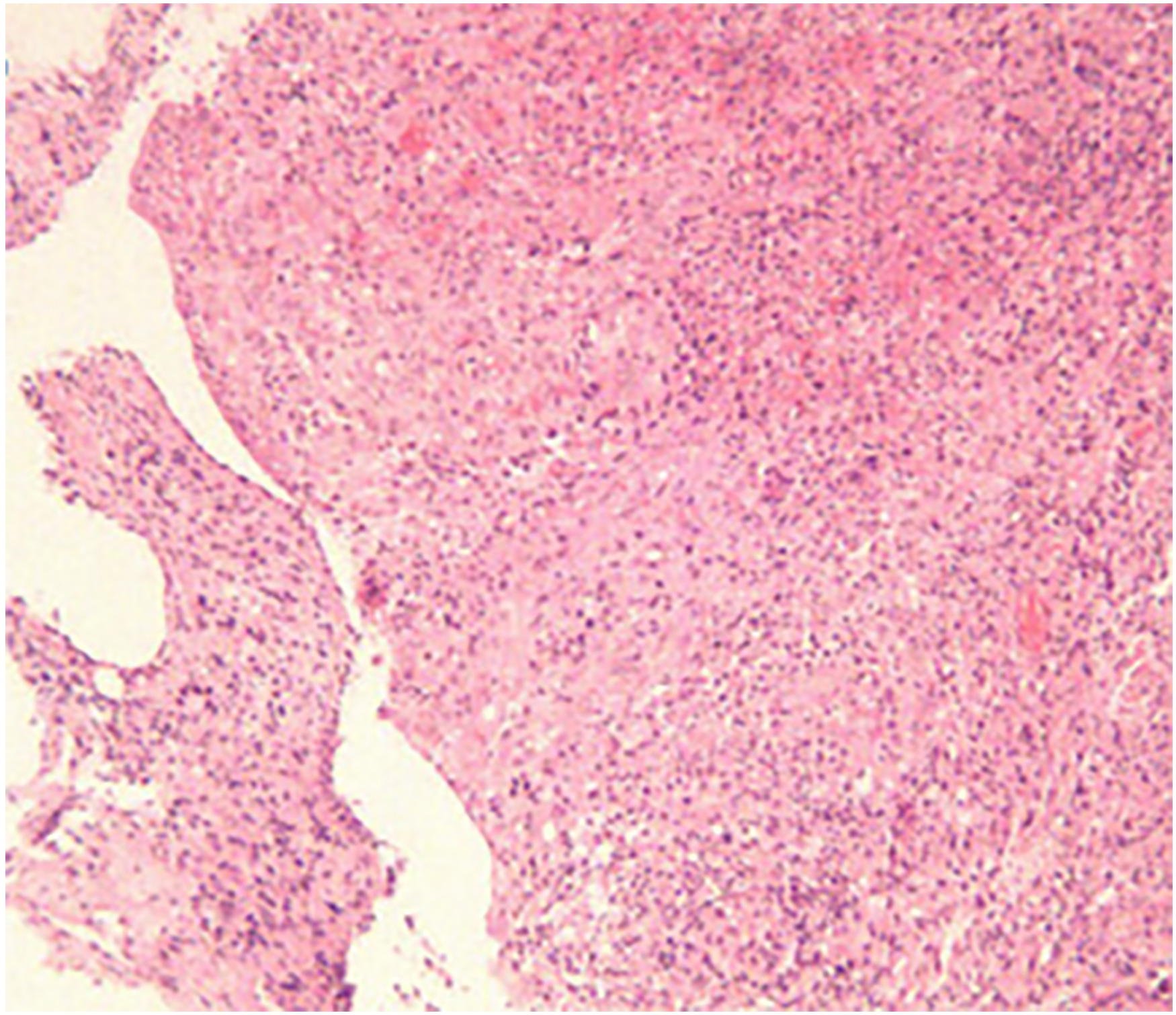
Chronic active mucosal inflammation, with erosion and squamous metaplasia of bronchial vegetative growth in the basal segment of the right lower lobe.

Chest CT performed on November 30, 2021 showing that the lung lesions had improved markedly compared with November 4.

New organisms in the orifice of the basal subsegment in the right lower lobe, blocking the orifice during bronchoscopy (November 9, 2021)(see blue arrow).

Tracheoscopy performed on December 21, 2021 showed that the new organisms in the orifice of the basal subsegment in the lower lobe of the right lung had disappeared.
Discussion
Nocardiosis farcinica infection is rare and its epidemiology is unclear. Nocardiosis can affect many organs, with the lungs accounting for more than 70% of all infections.4–7 A retrospective analysis of 52 Nocardiosis farcinica cases revealed that the most frequently involved organ was the lung/pleura (43%), followed by the brain (30%) and wounds (15%). 8 Nocardia infection is frequent in immunosuppressed individuals with acquired immunodeficiency syndrome, or malignancies, as well as in those undergoing corticosteroid therapy, immunosuppressive therapy, or transplantation, and in those with diabetes, connective tissue diseases, or underlying lung diseases. Infection of immunocompetent patients is rare.4,9 However, it can also occur in individuals without obvious immune function limitation. In fact, according to the Centers for Disease Control (CDC), a third of patients with nocardiosis have no known underlying disease. Some scholars summarized and analyzed 400 cases of Nocardia and found that there were undiagnosed primary immunodeficiency diseases in these healthy individuals, such as Hypogammaglobulinemia or chronic granulomatous disease, high IgE syndrome, idiopathic CD4 + T lymphopenia, anti GM-CSF autoantibody. 2 Our patient was healthy, with no underlying diseases or history of long-term hormone or immunosuppressant use. However, his immunoglobulin E increased significantly. This may be related to his individual susceptibility and blood seeding after inhalation of Nocardiosis farcinica.
Nocardiosis of the lung has no specific clinical manifestations. It can be acute, subacute, or chronic. Symptoms mainly depend on the underlying immune state. Fever, cough, expectoration, chest pain, night sweats, weight loss, and hemoptysis are common. 10 In some patients, infection can spread to other organs through the blood. The extrapulmonary system most commonly affected is the central nervous system (CNS), accounting for up to 44% of such cases, but this is uncommon in patients with normal immune function. 11 Imaging manifestations are diverse, and include consolidation, nodules, masses, and cavity formation, with chest wall involvement seen in a small number of patients. 12 A comparison of Nocardia infection between immunosuppressed and immunocompetent patients showed that common imaging manifestations of Nocardia in the immunocompetent lung included lobular neutral nodules and bronchiectasis. 10 Nocardia bacteremia is rare, and is most often seen in patients with low immune function or intravascular devices (91% of all cases). 13 The mortality can reach 50%. 14 Our patient showed acute onset, with cough and expectoration followed by fever. Chest CT showed patchy consolidation and multiple nodules, along with bacteremia. Unexpectedly, the patient had a growth in the bronchial mucosa of the dorsal segment of the right lower lobe. Although no tumor cells were seen on biopsy, we believe that the growth was bronchial lung cancer. Surprisingly, the polyps disappeared after 1 month of treatment. There has been no report on the mechanism by which Nocardia forms bronchial polyps. Unfortunately, our patient refused cranial magnetic resonance imaging while hospitalized, although there were no nervous system symptoms or signs; we were thus unable to determine whether there was central nervous cell infection.
Laboratory diagnosis of Nocardia is based on microscopy and culture. Nocardia grows slowly and is difficult to detect via hematoxylin eosin and acid-fast staining, while Gram and methylamine silver staining can be positive. On culture, Nocardia may take 5 days to grow. 15 Polymerase chain reaction and 16S rDNA sequencing can identify Nocardia more quickly and accurately than conventional phenotypic methods (including microscopy, culture, and biochemical characterization). 4 Some have suggested the application of molecular methods to identify suspected human nocardiosis using multipoint sequence analysis. 16 Our patient was diagnosed using high-throughput pathogen gene sequencing in blood and bronchoalveolar lavage fluid.
Nocardia is sensitive to most penicillins and cephalosporins, although the optimal treatment has not been determined. In the past 50 years, sulfonamides, sulfadiazine, and sulfamethoxazole have been the preferred drugs.11,17 However, Nocardia farcinica is usually sensitive to amikacin, imipenem, ciprofloxacin, and trimethoprim sulfamethoxazole, but resistant to ampicillin, third-generation cephalosporins (including cefotaxime and ceftriaxone), erythromycin, gentamicin, and tobramycin.8,15 For most forms of nocardiosis, combination therapy is initially recommended. There is no universal standard course of antibiotics. It is generally recommended that treatment of cases of pulmonary or multifocal (non-CNS) nocardiosis with normal immune function be applied for 6–12 months. Immunosuppressed patients and those with CNS disease should receive antibacterial treatment for at least 12 months.11,18 Abscesses and empyema may require surgical drainage and debridement. 12 Our patient had pulmonary nocardiosis, bacteremia and normal immune function. After making the diagnosis, we used only sulfamethoxazole tablets, and the infection improved quickly. We plan to treat the patient for 6 months.
Conclusion
This case report shows that healthy hosts can inhale Nocardia spores from the environment, leading to acute pulmonary nocardiosis, bacteremia and a bronchial polyp; this is easily confused with lung cancer.
Footnotes
Abbreviations
Acknowledgements
The authors thank the patient and his family for their participation.
Author contribution(s)
The study was designed by SXD, BYZ collected the data. XJD and QDL analyzed data; SXD wrote the paper. All authors read and approved the final manuscript.
DECLARATION OF CONFLICTING INTERESTS
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
FUNDING
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval / Patient consent
This study was approved by the Ethic Committee of the Dingli Clinical Institute of Wenzhou Medical University (Wenzhou Central Hospital). The authors have no ethical conflicts to disclose.
