Abstract
Purpose:
Nintedanib is an approved treatment for idiopathic pulmonary fibrosis (IPF), which slows disease progression. Management of patients with IPF receiving nintedanib can be complicated by tolerability issues, comorbidities, and concomitant medications. We developed consensus recommendations on the management of dosing, adverse events and comorbidities in patients with IPF treated with nintedanib.
Methods:
A modified Delphi process using 3 questionnaires was used to survey 14 pulmonologists experienced in using nintedanib. Panelists rated their agreement with statements on a Likert scale from −5 (strongly disagree) to +5 (strongly agree). Consensus was predefined as a mean score of ⩽−2.5 or ⩾+2.5 with a standard deviation not crossing zero.
Results:
The panelists’ recommendations were largely aligned with clinical trial data, real-world evidence, and the prescribing information, and provided additional guidance regarding minimizing gastrointestinal effects, periodic monitoring for liver dysfunction, caution with respect to concomitant administration of cytochrome P450 3A4 and P-glycoprotein 1 inhibitors and inducers and anticoagulants, and management of comorbidities. The panelists unanimously agreed that adverse event management should be individualized, based on careful consideration of the risks and benefits of each possible intervention and discussion with the patient.
Conclusions:
These consensus recommendations provide additional guidance on the appropriate management of IPF with nintedanib, for use alongside evidence-based literature and the prescribing information.
Introduction
Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive, and ultimately fatal lung disease characterized by fibrosing interstitial pneumonia. 1 Current clinical practice guidelines for IPF issued by the American Thoracic Society (ATS), European Respiratory Society (ERS), Japanese Respiratory Society (JRS) and Latin American Thoracic Association (ALAT) provide conditional recommendations for use of the approved anti-fibrotic drugs nintedanib and pirfenidone. 2 Clinical trials of nintedanib, an intracellular inhibitor of tyrosine kinases, 3 have shown that nintedanib slows the rate of decline in forced vital capacity (FVC) in patients with IPF, with an adverse event (AE) profile characterized mainly by gastrointestinal (GI) events.4,5 In the Phase III INPULSIS trials, AEs, particularly diarrhea, were the most frequent reason for premature discontinuation of nintedanib. 5 The prescribing information for nintedanib provides broad recommendations for dosage adjustments (dose reduction from 150 to 100 mg bid and treatment interruption) and for the management of select AEs and drug interactions. 6 Adverse events specifically discussed in the prescribing information include liver enzyme elevations, drug-induced liver injury (DILI), GI disorders, arterial thromboembolic events, and gastrointestinal perforation. Additional AEs, including abdominal pain, decreased appetite, headache, weight loss, and hypertension, occurred in ⩾5% of patients treated with nintedanib in the INPULSIS trials. Management strategies for such AEs are not included in the prescribing information, and there is limited guidance in the literature on how they should be managed.7 -9 Considered together with the prescribing information, consensus recommendations for managing nintedanib dosing and adverse events, developed by clinicians with real-world experience, may help to provide a more standardized approach.
The Delphi process uses a sequence of structured questionnaires to identify and build consensus. 10 The authors initiated this Delphi study to develop consensus recommendations on the management of dosing, AEs, and comorbidities in patients with IPF treated with nintedanib. This manuscript builds on preliminary data presented at the 20th International Colloquium on Lung and Airway Fibrosis, Pacific Grove, CA, USA, 14–18 October 2018. 11
Methods
This study was conceived by Franck Rahaghi, Joao de Andrade, and Howard Lazarus, then of Boehringer Ingelheim Pharmaceuticals, Inc. (BIPI), who oversaw recruitment of the Delphi panel members and the conduct of the process. Panelists were pulmonologists based in the United States or Canada who had experience in using nintedanib in the treatment of IPF. One panelist chose not to be included as a co-author on this manuscript.
This study utilized a modified Delphi process with a sequence of 3 questionnaires (see Online Resource 1). Questionnaire 1 primarily consisted of open-ended questions to gather information on panelists’ practices and was developed based on clinical experience, a review of the literature, evidence from clinical trials, and the prescribing information for nintedanib. For AE management, panelists were asked about their strategies in 4 areas: non-pharmacological interventions, pharmacological interventions, dose reduction or treatment interruption, drug discontinuation or switching. These were carried through all 3 questionnaires. Panelists’ opinions and practices were converted to definite statements, consolidated as appropriate, and included in Questionnaire 2, along with additional open-ended questions as needed for clarification.
Panelists were asked to rate their agreement with each statement on an 11-point Likert scale ranging from −5 (strongly disagree) to +5 (strongly agree). Questionnaire 2 also asked panelists to rate the importance of specified items as follows: selected concomitant medications 0 to +5), laboratory tests (0 to +5), and comorbidities (−5 to +5). Questionnaire 3 contained the same statements and questions as Questionnaire 2 plus a few additional statements for clarification. A summary spreadsheet of the panelist’s own results and the panel’s aggregated results (mean and standard deviation [SD] of scores) from Questionnaire 2 was circulated along with Questionnaire 3. This was intended to help build consensus “for” or “against” statements for which consensus could potentially be reached. All responses were anonymized and weighed equally. For statements rated using a Likert scale, consensus was predefined as a mean Likert scale score ⩾+2.5 (consensus for) or ⩽–2.5 (consensus against) with a SD that did not cross zero. Questions asking for ratings of importance rather than agreement were not evaluated for consensus.
At the time that the questionnaires were completed, IPF was the only interstitial lung disease for which nintedanib was an approved treatment.
Results
The panel comprised 14 pulmonologists who practiced predominantly at academic centers. Their mean ± SD experience in treating patients with IPF was 17.5 ± 7.23 years; 43.6 ± 16.0% of their patients with IPF were prescribed nintedanib.
The final Delphi questionnaire consisted of 242 items, of which 224 were rated on a Likert scale and 18 were rated by importance. Of the Likert scale items, panelists reached consensus on 168 (75%) (167 with consensus for, 1 with consensus against). We describe below the items for which consensus was reached and selected items for which a lack of consensus was regarded as an important finding. Likert and importance scores are reported as mean ± SD. Online Resource 2 lists all the results of Questionnaire 3.
Nintedanib dosing and administration
Panelists reached consensus that nintedanib should be taken with food and swallowed whole with liquid (4.64 ± 0.74) and taken with a full meal wherever possible (4.00 ± 2.42). Missed doses should not be made up (4.71 ± 0.47) and the maximum recommended dose (150 mg bid) should not be exceeded (4.93 ± 0.27). If the patient is intolerant or experiences an AE, use of the lower 100 mg bid dosage (4.21 ± 1.19) or treatment interruption (4.36 ± 0.74) should be considered. Initiating treatment with 100 mg bid should be considered for patients <65 kg (2.64 ± 1.82) or who are Child-Pugh Class A (3.29 ± 1.86). The most important factors that influenced panelists’ dosing strategies were tolerability/side effects (4.93 ± 0.27), in particular GI toxicity (4.79 ± 0.43) (Figure 1).

Agreement/disagreement with statements on nintedanib dosing and administration.
Hepatic effects
Liver enzymes should be monitored monthly for the first 3 months and every 3 months thereafter (3.50 ± 2.38) (Figure 2). Treatment interruption should be considered for liver enzyme elevations of 3 to 5 times the upper limit of normal (ULN) (4.14 ± 1.35), with the length of the interruption dependent on the magnitude of the elevation (3.93 ± 1.86) and/or the time needed for normalization (4.21 ± 1.42), typically 1–2 weeks (3.29 ± 2.13). After liver enzymes have returned to their baseline values, nintedanib should be reintroduced at 100 mg bid and titrated up to the full dose (3.93 ± 1.14). Nintedanib should be interrupted immediately if serious liver injury with hyperbilirubinemia or jaundice occurs (5.00 ± 0.00).

Agreement/disagreement with statements on monitoring and management of hepatic effects of nintedanib.
Drug-drug interactions
Patients receiving strong cytochrome P450 3A4 (CYP3A4) and/or P-glycoprotein 1 (P-gp) inhibitors should be monitored closely (4.36 ± 1.74) (Figure 3). Examples of CYP3A4 and P-gp inhibitors and inducers are provided in Online Resource 3. For instances in which drug-drug interactions may occur, panelists reached consensus on management strategies including avoiding concomitant use with nintedanib if possible (4.29 ± 1.90), initiating nintedanib at 100 mg bid (4.29 ± 1.07), and monitoring liver function more frequently (3.64 ± 2.31). The panel reached consensus on 2 strategies for management of AEs due to concomitant use of these agents: (1) interrupt nintedanib until the AE resolves then resume at a lower dose (4.29 ± 1.14), or (2) reduce the dose of nintedanib, then interrupt treatment if needed, followed by discontinuation if needed (3.29 ± 2.46). Either strategy may be appropriate depending on the severity of the AE and the reliability of patient follow-up (3.93 ± 2.27).
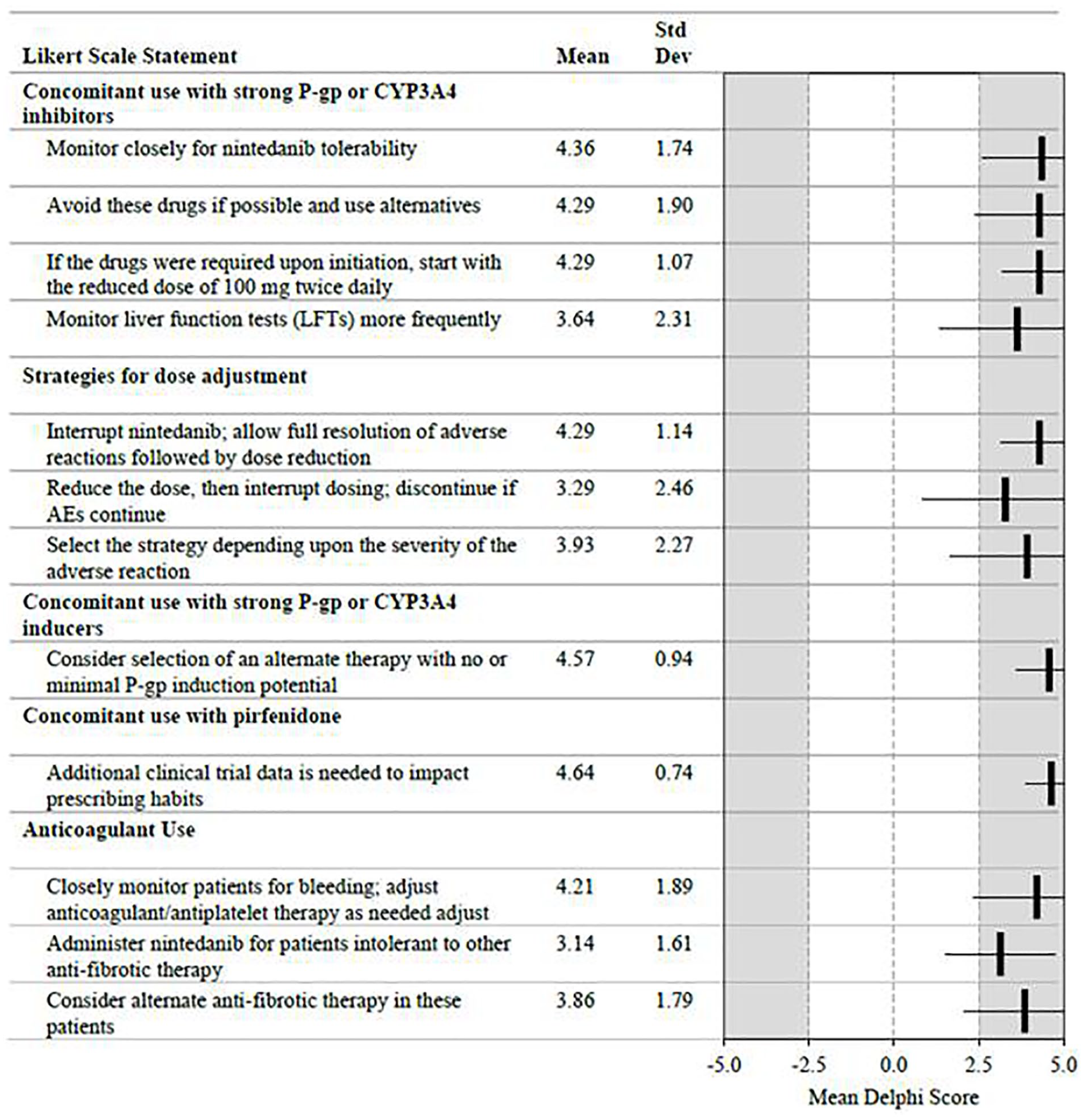
Agreement/disagreement with statements on drug-drug interactions and anticoagulant use.
The panel also arrived at consensus that concomitant administration of CYP3A4 or P-gp inducers with nintedanib should be avoided, and alternative medications considered. However, concomitant use is a rare event that has not impacted clinical practice (3.29 ± 1.59). Additional data are needed on co-administration of nintedanib with pirfenidone (4.64 ± 0.74).
For patients receiving concomitant antiplatelet therapy or full anticoagulation therapy, panelists reached consensus that close monitoring with anticoagulant dose adjustments as needed is appropriate (4.21 ± 1.89), and that nintedanib can be administered to patients who are intolerant to other anti-fibrotic therapies and require anticoagulation (3.14 ± 1.61).
Arterial thromboembolic events
The panel achieved consensus that patients at higher risk (active angina, recent percutaneous coronary intervention, acute coronary syndrome, acute myocardial infarction, stroke, or severe/symptomatic coronary artery disease or peripheral vascular disease), should be treated with caution (4.29 ± 0.99), and that an alternative anti-fibrotic agent could potentially be considered (4.14 ± 1.51). Overall, the decision to use nintedanib should be individualized based on risk, benefit, and patient preference (4.21 ± 1.31). Consensus scores are summarized in Figure 4.

Agreement/disagreement with statements on arterial thromboembolic events and gastrointestinal perforation.
GI perforation
Patients with a history of recent abdominal surgery or diverticular disease, or who are receiving concomitant corticosteroids or nonsteroidal anti-inflammatory drugs (NSAIDs) should be monitored carefully (4.29 ± 1.44). Nintedanib should be held perioperatively for elective surgery (3.71 ± 1.33). In addition, nintedanib should be discontinued if GI perforation is present (4.93 ± 0.27) and should not be used in patients with a known risk of GI perforation (3.79 ± 1.12). An alternative anti-fibrotic should be considered (4.29 ± 1.20) (Figure 4).
Special populations
For geriatric patients, the panelists reached consensus that no dose adjustment is needed (3.43 ± 2.03), and that patients should be treated regardless of age, with more caution in patients over 80 years (4.50 ± 0.65). For patients over 75 years, liver function should be monitored monthly for at least the first 6 months (2.93 ± 2.79). For older and smaller patients, 100 mg bid should be used if dictated by side effects (3.86 ± 1.61). There was consensus that no nintedanib dose adjustment is needed for patients with mild to moderate renal impairment (4.36 ± 0.84). The panelists unanimously agreed that smoking cessation should be encouraged before and during therapy with nintedanib (5.00 ± 0.00).
AE management
Recommendations for AE management are summarized in Online Resource 4.
Mild to moderate diarrhea
Panelists reached consensus that mild to moderate diarrhea should be managed by taking nintedanib with food (4.71 ± 0.73), large meals with increased fiber (3.36 ± 1.74), and adequate hydration (4.14 ± 1.29). Therapies include loperamide (4.69 ± 0.63) and diphenoxylate/atropine (3.38 ± 2.14). If needed, the dose should be interrupted and then reduced if the patient feels the symptoms are significant (4.57 ± 0.65). This is a patient-specific decision (3.79 ± 1.72). If problematic diarrhea persists despite loperamide and dose reduction, discontinuation (4.00 ± 1.36) or changing to an alternative anti-fibrotic agent (3.50 ± 2.35) may be considered after conferring with the patient. For most patients, panelists suggested following the management algorithm recommended in the product label (3.79 ± 1.25). With regard to the 4–6 stools vs the over 6 stools strategy, panelists noted that fewer stools (3–4) may still be considered clinically significant by the patient (3.50 ± 1.51). Generally, therapy should be titrated according to patient preference unless there is evidence of dehydration (3.86 ± 1.41). Panelists reached consensus on the following dietary guidelines: use of a BRAT diet (bananas, rice, applesauce, toast) (3.43 ± 1.91), avoidance of dates, prunes, and other dried fruits with high pectin content (3.36 ± 1.50), and increased fiber intake (3.14 ± 1.56).
Nausea
For nausea, panelists reached consensus on reviewing reflux guidelines (3.50 ± 1.79), the patient’s diet (4.07 ± 1.38), and taking nintedanib with large meals rather than a snack (3.86 ± 1.66). Therapies that achieved consensus included ondansetron (3.43 ± 1.34) and proton pump inhibitors (PPIs) (2.71 ± 2.23). Dose modifications should be patient-specific and patient-driven (3.71 ± 1.38), with the patient providing guidance between dose reduction and treatment interruption (3.64 ± 1.78). In the absence of patient preference, it was agreed that clinicians should start with a dose reduction and interrupt therapy if dose reduction is ineffective (2.71 ± 2.13). If nausea persists after nintedanib interruption and reintroduction with an antiemetic or PPI, consider switching to an alternative anti-fibrotic (4.00 ± 1.04).
Vomiting
For non-pharmacological management, panelists reached consensus on taking nintedanib with large meals (3.14 ± 1.88), appropriate dietary selection and timing (3.50 ± 1.91), and adequate hydration (3.86 ± 1.70). Consensus was reached on management with ondansetron (4.21 ± 0.89). For persistent vomiting, both treatment interruption followed by dose reduction to 100 mg bid (4.21 ± 1.12) and dose reduction (3.14 ± 1.66) reached consensus. If treatment interruption and dose reduction do not resolve the vomiting, consider switching to another anti-fibrotic (4.21 ± 1.12).
Abdominal pain
The panel reached consensus that proactive non-pharmacological management of abdominal pain should include taking nintedanib with large meals (4.14 ± 1.17), reducing dairy and increasing fiber consideration (3.36 ± 1.69), and consideration of the BRAT diet (3.07 ± 1.98). Therapies include PPIs (3.36 ± 1.69) and H2-receptor antagonists (2.71 ± 1.59). Panelists reached consensus on interrupting therapy followed by restarting at 100 mg bid (4.29 ± 1.38) and on considering switching to an alternative anti-fibrotic drug if pain persists after a dose reduction (3.71 ± 1.54) or treatment interruption (4.07 ± 1.14).
Headache
Consensus was reached on the use of acetaminophen (4.36 ± 1.15) or low-dose NSAIDs (2.93 ± 1.94) to manage headache. Nintedanib dose modifications should be patient-specific and patient-driven (4.36 ± 1.39). Panelists recommended dose modifications including treatment interruptions and/or dose reductions but did not reach consensus on which should be tried first. If the headache is severe or persists despite dose interruption and reduction, consider switching to an alternative anti-fibrotic drug (4.50 ± 0.76). The experts recommended that alternative etiologies for headache should be ruled out before considering switching medications, although this option was not addressed in the Delphi questionnaire.
Decreased appetite
The panelists reached consensus that decreased appetite can be managed with quality food/protein intake (4.07 ± 1.14), high-protein nutritional drinks (3.86 ± 1.29) and more frequent meals (3.57 ± 1.60). No consensus on pharmacological interventions was reached. Dose modification should be patient-driven and should be initiated for weight loss >10 lbs (>4.5 kg) (4.07 ± 1.49). The initial dose modification can be a reduction (3.71 ± 1.20) or interruption (3.36 ± 1.82). Consider switching to an alternative anti-fibrotic drug if the appetite decrease persists (3.64 ± 1.74) or significant weight loss occurs (3.86 ± 1.29).
Weight loss
The panel’s consensus for management of weight loss was similar to that for decreased appetite. Non-pharmacologic interventions included increasing calories and dietary supplements (4.43 ± 0.76), eating 3 meals a day with small snacks between meals (4.43 ± 0.65), and use of high-protein nutritional drinks (4.07 ± 1.44). There was no consensus on pharmacological therapies. For persistent weight loss, consider treatment interruption (4.14 ± 1.10) or dose reduction (3.50 ± 1.02). If weight loss persists, discontinue nintedanib (4.00 ± 1.57) or switch to an alternative anti-fibrotic drug (4.07 ± 1.21).
Liver enzyme elevation
The panel reached consensus that patients with Child-Pugh A liver impairment should be treated with nintedanib 100 mg bid, approximately 12 hours apart with food, as recommended in the product label (4.07 ± 1.14). For these patients, liver function should be monitored monthly (4.43 ± 1.02). Use of nintedanib in patients with Child-Pugh B and C is not recommended (3.71 ± 1.90).
Comorbidities
The panelists reached consensus that comorbidity management should be patient-specific, depending on the severity and impact of the comorbidity and the likelihood that it is related to IPF (4.57 ± 0.94). In general, sicker patients should receive closer and more frequent monitoring and assessment (4.07 ± 1.38). Comorbidity management may affect nintedanib dosing (4.14 ± 1.03). Panelists rated the importance of key comorbidities, shown in Figure 5. Ischemic heart disease, cardiac failure, and pulmonary hypertension were rated as the most important comorbidities. The panel’s recommendations related to management of specific comorbidities are reported in Online Resource 5.

Ratings of the importance of comorbidities in the management of IPF with nintedanib.
Management of acute exacerbations
The panel reached consensus that acute exacerbations of IPF should be treated with steroids and weaned quickly (3.93 ± 1.33). Patients with exacerbations should be evaluated for heart failure/fluid overload and treated if present (4.64 ± 0.63). The duration of steroid therapy and the use of empiric antibiotics were not assessed.
Treatment algorithm
Results of this Delphi panel are summarized in a generic algorithm that describes management of AEs in patients requiring ongoing therapy for chronic disorders (see Online Resource 4).
Discussion
Nintedanib slows the progression of IPF4,5 and is an approved treatment for IPF. However, effective management of patients receiving nintedanib can be challenging due to tolerability issues, comorbidities, and the need for concomitant medications. This Delphi process used clinical expertise to facilitate development of consensus recommendations for management of dosing, AEs, and comorbidities in patients receiving nintedanib for IPF.
With respect to dosing modifications for nintedanib, for a variety of clinical scenarios, panelists agreed on considering temporary interruption followed by re-introduction, dose reduction, switching to an alternative antifibrotic therapy, or discontinuation. Several of the consensus recommendations, including use of a lower dose for patients who weigh <65 kg, or in instances in which drug-drug interactions may occur, extend beyond the prescribing information for nintedanib. Such recommendations reflect the real-world experience of the panelists and should be considered alongside evidence-based literature and the prescribing information for nintedanib.
In the Phase II TOMORROW trial, nintedanib 100 mg bid reduced the rate of decline in FVC over 52 weeks in patients with IPF compared with placebo, but the difference between the groups was not statistically significant. 4 Dose reductions and treatment interruptions used to manage AEs during the INPULSIS trials 12 and their open-label extension, INPULSIS-ON 13 were not associated with faster FVC decline, suggesting that the efficacy of nintedanib in reducing disease progression is sustained in patients who require dose adjustments to manage adverse events. In this study, the panelists noted that in clinical practice, dose reduction can be used if patients have a history of medication or gastrointestinal intolerance and/or are nervous about potential side effects.
Although the panelists agreed that no dose adjustment is needed for geriatric patients, some experts noted that they use 100 mg bid for geriatric patients, particularly if the patient is frail or has low weight. Some experts noted that they start nintedanib at the full dose in geriatric patients and then reduce the dose if it is not tolerated. The panelists unanimously agreed that AE management should be individualized based on careful consideration of the risks and benefits of each possible intervention and discussion with the patient.
The panelists agreed that patients should be monitored for liver enzyme elevations. Nintedanib should be interrupted if liver function enzymes become elevated to 3–5× ULN. Patients with Child-Pugh A liver impairment should receive 100 mg bid with monthly monitoring, in alignment with the prescribing information. Drug-induced jaundice in patients with an alanine aminotransferase (ALT) ⩾3× ULN may portend a poor prognosis, requiring a hepatology consultation and immediate discontinuation of nintedanib. 14
For drug-drug interactions, the need for additional data on co-administration of nintedanib with pirfenidone, and the need for close monitoring of patients on anticoagulation therapy for bleeding events, the recommendations of the panelists were in alignment with the prescribing information. The panel agreed that nintedanib is not recommended for use in patients with a known risk of GI perforation including known peptic ulcer disease, ischemic colitis, inflammatory bowel disease, diverticulitis, colon cancer, recent intestinal surgery, or heavy NSAID use. This represents the panelists’ expert consensus, based on clinical experience, and should be considered together with the prescribing information. This study also took into consideration management of patients with a wide range of cardiovascular diseases. The panelists did not recommend withholding nintedanib in patients with stable cardiac disease and suggested assessing the risks versus benefits and patient’s preferences in order to make the best decision.
The Delphi process applied to develop these recommendations provided a systematic method for obtaining consensus that prevented dominance by individual panelists. However, the Delphi process has several limitations, including that it is not evidence-based, lacks generally accepted criteria for consensus, and may be biased by panel selection and questionnaire development. 15 In this study, the selection of panelists was limited to 14 US/Canada-based participants. As a result, the findings may not represent global perspectives or important perspectives from a larger and more representative population of US/Canada-based physicians. Other potential stakeholders, such as patients, pharmacists, and payers, were not included, which limited the diversity of perspectives.
Conclusions
IPF is an intrinsically progressive and ultimately fatal disease. Nintedanib slows the progression of IPF, but in order to maintain patients on nintedanib therapy, it is critical to effectively manage dosing, AEs, and comorbidities. Findings from this modified Delphi process provide pharmacologic, non-pharmacologic, and dose modification strategies for preventing or mitigating AEs in patients taking nintedanib, corroborating data from clinical trials and real-world experience, and complementing the prescribing information for nintedanib.
Supplemental Material
sj-docx-1-cra-10.1177_11795484211006050 – Supplemental material for Delphi Consensus Recommendations on Management of Dosing, Adverse Events, and Comorbidities in the Treatment of Idiopathic Pulmonary Fibrosis with Nintedanib
Supplemental material, sj-docx-1-cra-10.1177_11795484211006050 for Delphi Consensus Recommendations on Management of Dosing, Adverse Events, and Comorbidities in the Treatment of Idiopathic Pulmonary Fibrosis with Nintedanib by Franck Rahaghi, John A Belperio, John Fitzgerald, Mridu Gulati, Robert Hallowell, Kristin B Highland, Tristan J Huie, Hyun J Kim, Martin Kolb, Joseph A Lasky, Brian D Southern, Jeffrey J Swigris and Joao A de Andrade in Clinical Medicine Insights: Circulatory, Respiratory and Pulmonary Medicine
Footnotes
Acknowledgements
This Delphi study was funded by Boehringer Ingelheim Pharmaceuticals, Inc. (BIPI). The authors meet criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICMJE). The authors did not receive payment for development of this article. Writing support was provided by Edward K. Baldwin, PhD, of AXON Communications, and Melanie Stephens, PhD, from Fleishman-Hillard, which were contracted and funded by BIPI. Boehringer Ingelheim was given the opportunity to review the manuscript for medical and scientific accuracy as well as intellectual property considerations. The authors thank Dr. Howard M. Lazarus, then an employee of BIPI, for his assistance with this project.
Authors’ contributions
All authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship. All authors had access to the Delphi questionnaire analysis and data, and participated in the review, revision, and approval of this manuscript. The viewpoints expressed in this manuscript solely represent those of its authors. These viewpoints do not necessarily expressly reflect those of BI.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FFR reports personal fees from Boehringer Ingelheim (BI) and Genentech. JAB has no competing interests to disclose. JF has no competing interests to disclose. MG reports grants, non-financial support, and other support from BI; personal fees, non-financial support, and other support from the France Foundation; personal fees from Genentech; and grants, non-financial support, and other support from the Pulmonary Fibrosis Foundation. RH has no competing interests to disclose. KBH reports grants and personal fees from Actelion Pharmaceuticals, BI, and United Therapeutics; personal fees from Bayer; and grants from Genentech, Eiger Pharmaceuticals, and Reata Pharmaceuticals. TJH reports research support from BI and Promedior and writing support funded by BI. HJK has no competing interests to disclose. MK reports grants and personal fees from BI, Gilead, GlaxoSmithKline, Prometic, and Roche; and personal fees from AstraZeneca, Covance, Galapagos NV, Indalo, and Third Pole Inc. JAL reports personal fees from Biogen, BI, Galecto, Roche/Genentech, and Veracyte. BDS reports grants from Genentech and personal fees from BI. JJS has no competing interests to disclose. JAD reports personal fees from BI.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This Delphi study was funded by Boehringer Ingelheim Pharmaceuticals, Inc. (BIPI). The authors did not receive payment for development of this article.
Availability of data and material
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
