Abstract
Lung carcinoma currently represents 1 of the leading causes of death from cancer worldwide and regionally. The molecular identification of sensitive mutations of targeted treatment have changed the strategies of pharmacologic management in non-small cell lung carcinoma. However, mechanisms of resistance have been described, among them the change of histological type to small cell carcinoma. We present the case of a 46-year-old male patient, non-smoker, with a clinical history of a mass in the upper lobe of the right lung and an initial histological diagnosis of adenosquamous carcinoma of the lung, with the presence of mutations for epidermal growth factor receptor (EGFR) in exons 20 (S768I) and 21 (L858R). He received treatment with tyrosine kinase inhibitor (Erlotinib) with good clinical and radiological response. However, 1 year after the start of the medication, he consulted for a progressive onset of constitutional symptoms and respiratory symptoms, with radiographic worsening and new biopsy with a diagnosis of adenosquamous carcinoma with the adenocarcinoma component transformed to small cell carcinoma, with persistence of EGFR mutation. We describe the clinical, radiological, and laboratory characteristics as well as the outcome of this case. To conclude, among the mechanisms of resistance described to the treatment with tyrosine kinase inhibitors in patients with carcinomas with mutated EGFR, the transformation to small cell carcinoma besides being infrequent is particular, requiring a different diagnostic and therapeutic approach.
Introduction
Lung cancer currently occupies the first place in cancer mortality worldwide in both men and women. 1 In 2014, in Colombia, it ranked second in causes of cancer mortality in women, after breast cancer and third in men, after stomach and prostate cancer. 2
In recent years, multiple treatment strategies have been developed, mainly focused on the molecular aspects of lung cancer, among these the mutational status of epidermal growth factor receptor (EGFR) and its therapeutic approach with tyrosine kinase inhibitors. However, mechanisms of resistance have been described, for example, the transformation to small cell carcinoma, which occurs between 3% and 15% of patients and has been associated with rapid clinical deterioration, disease progression, and complete need for change in oncological management. 3
We present the case of a patient with an initial diagnosis of adenosquamous carcinoma of the lung with mutated EGFR, with progression of the disease and transformation to small cell carcinoma after treatment with Erlotinib.
Case Report
A 46-year-old male non-smoker, consulted for 4 months of evolution of dry cough, night sweats, and weight loss, with no relevant medical history. The physical examination revealed a decrease in respiratory sounds in the right hemithorax. A chest X-ray showed an opacity of the right upper lobe (RUL). In the thoracic computed tomography (CT) scan, a mass of 10.5 × 8.8 cm was observed that affected almost all of the RUL, with multiple adenomegalies in the mediastinum, subcarinal, and in the right pulmonary hilum (Figure 1A and B). Fibrobronchoscopy showed a tumor-like lesion in RUL, with endobronchial infiltration and severe extrinsic compression of that bronchus. The biopsy reported a mixed malignant lesion constituted by large hyperchromatic cells, organized in a solid and pseudo glandular pattern with a mixed immunohistochemical profile with focal positivity for TTF-1, Napsin A, and p40, compatible for diagnosis of primary adenosquamous carcinoma of the lung (Figure 2A and B). The genetic profile reported positive mutations for EGFR in exon 20 (S768I) and exon 21 (L858R), without rearrangements for the ALK gene. It was initially classified as stage IIIA. The treatment provided was chemotherapy with Cisplatin, Etoposide, and radiotherapy. During the second cycle of treatment, he presented progressive dyspnea, edema in the lower limbs, and syncope. He entered the intensive care unit where the presence of pericardial effusion was documented, which was drained obtaining a cytology with the presence of malignant cells compatible with metastatic adenocarcinoma (Figure 2C). At that time, given the presence of the EGFR mutation, the treatment was changed to a tyrosine kinase inhibitor, Erlotinib 150 mg/d with good clinical and radiological response. The thoracic CT-scan taken 6 months after the start of the medication showed a decrease in the size of the tumor, reporting a poorly defined 2 cm lesion of irregular contours in the RUL and horizontal fissure of nodular aspect with normal mediastinum (Figure 1C and D). One year after the start of the medication, he consulted for progressive constitutional symptoms, dyspnea and cough. The chest X-ray showed a lesion of 5 × 3 cm in the RUL with compression of the bronchial lumen and mediastinal involvement Figure 3. It was decided to suspend the tyrosine kinase inhibitor and initiate chemotherapy with Carboplatin, a dose calculated with AUC 7 (area under the curve), Pemetrexed 500 mg/m2 and Bevacizumab 7.5 mg/kg/dose every 21 days. He consulted after the sixth cycle with a history of increased cough, dyspnea, and fever. A thoracic CT-scan was performed which revealed a greater progression of the lesion in the RUL with vascular and right bronchus involvement as well as pleural effusion (Figure 3B and C). The brain magnetic resonance imaging (MRI) showed multiple supra and infratentorial metastatic lesions with an ependymal nodule inside the right lateral ventricle (Figure 3D). Biopsies taken by fibrobronchoscopy reported involvement by a mixed malignant neoplasia composed of small hyperchromatic cells and large eosinophilic cells. Immunohistochemistry was positive for CD56, TTF-1, and Synaptophysin in the small cells and for p40 in the large cell component. These findings lead to a diagnosis of adenosquamous carcinoma with transformation of the adenocarcinoma component to small cell carcinoma and persistence of the squamous component (Figure 2D-F). The genetic profile of this transformation reported EGFR with persistence of mutations in exon 20 (S768I) and exon 21 (L858R). Given the new findings of pathology, therapy was modified and changed to a third line of chemotherapy with Cisplatin 7.5 mg/m2 and Etoposide 120 mg/m2/for 3 days every 21 days receiving 4 cycles. Despite treatment, the patient presented an inadequate evolution with an increase in brain lesions and appearance of bone lesions confirmed by whole body bone scintigraphy. With the latest findings, palliative treatment was decided, and the patient died 2 years after diagnosis.
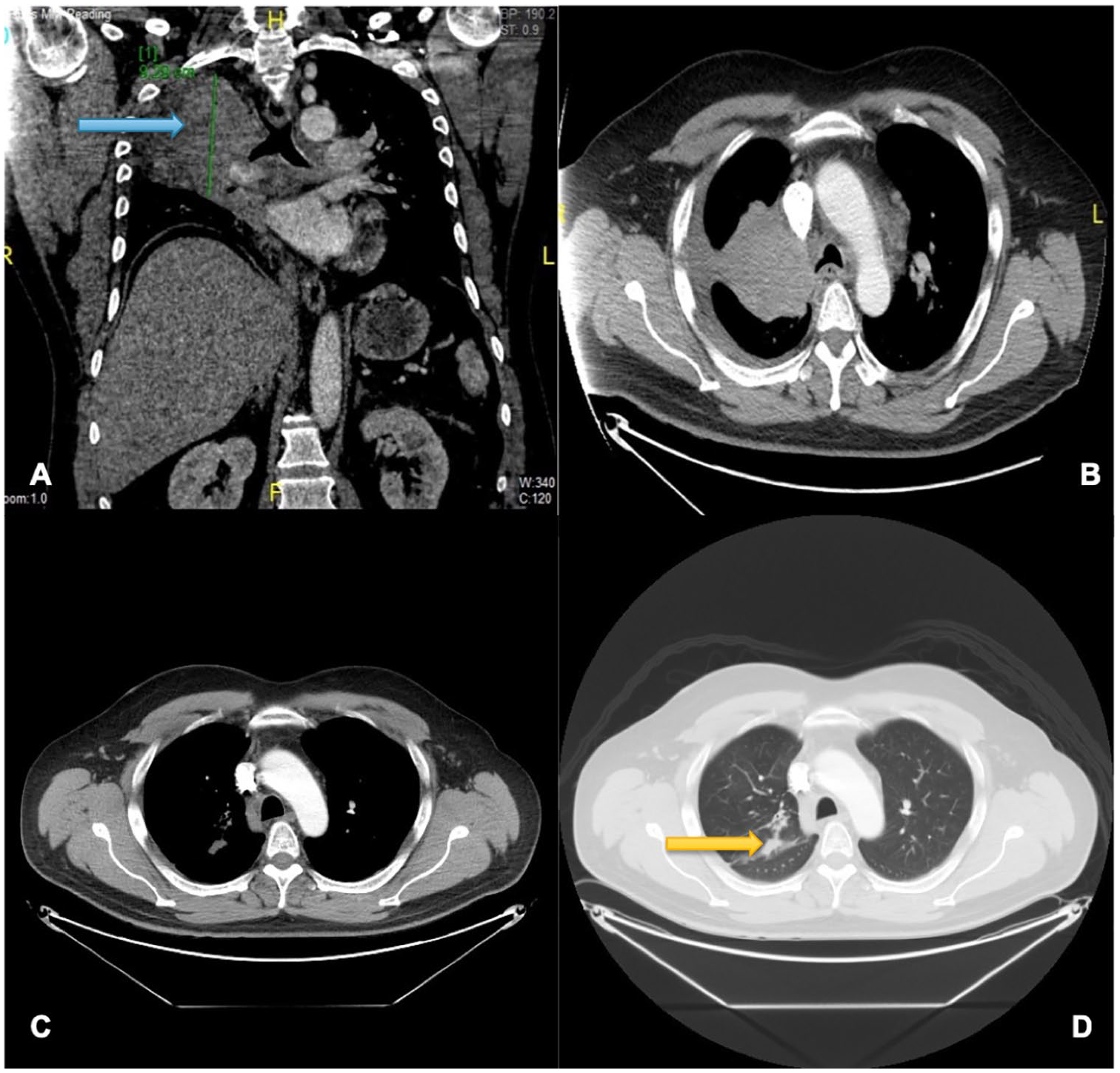
(A, B) Thoracic CT-scan showing, blue arrow: a large lesion in RUL, diaphragmatic elevation, decreased bronchial lumen, mediastinal involvement, and mild right pleural effusion. (C, D) Thoracic CT-scan showing a decrease of the RUL lesion. Yellow arrow: a 2-cm opacity of irregular nodular aspect persists and horizontal fissure with small nodules, without evident mediastinal involvement. CT indicates computed tomography; RUL, right upper lobe.

Sequential images of the histological evolution. (A, B) Primary tumor compatible with adenosquamous carcinoma. (A) Positivity for p40. (B) Positivity for TTF-1. (C) Adenocarcinoma in the cell block of the pericardial fluid cytology. H & E staining, blue arrow: cell block where it is appreciated pulmonary adenocarcinoma in pericardial effusion. Immunohistochemistry, yellow arrow: nuclear positivity for TTF-1. (D) Transformation to small cell carcinoma. H & E staining, blue arrow: component of squamous cell carcinoma. Yellow arrow: presence of small cell carcinoma transformed from the adenocarcinoma component. (E) Immunohistochemistry, blue arrow: positive p40 in squamous cell carcinoma. Yellow arrow: p40 negative in the small cell carcinoma component. (F) Immunohistochemistry, blue arrow: positivity for CD56 in the fragments of the transbronchial biopsy with representation of small cell carcinoma.

(A) Thoracic CT-scan with progression of the lesion of irregular aspect 5 × 3 × 3 cm in RUL with mediastinal involvement and satellite nodules. (B, C) Blue arrow: Thoracic CT-scan: large mass in RUL with involvement of structures of the pulmonary hilum, horizontal fissure, parietal and mediastinal pleura, pericardial sac with satellite nodules. (D) Yellow arrow: Brain MRI where there are multiple nodules of metastatic aspect. CT indicates computed tomography; RUL, right upper lobe.
Discussion/Conclusions
In this case, Carboplatin plus Etoposide was used in association with radiotherapy in the first phase of treatment as it was considered a locally advanced stage IIIA tumor. During the second cycle, a pericardial effusion appeared, and knowing the mutation, treatment with Erlotinib was initiated. A year later, after the evident progression of the disease, Carboplatin, Pemetrexed, and Bevacizumab were used. Finally, after finding the histological type of the small cell tumor, standard treatment of Carboplatin-Etoposide was started with the already known outcomes.
Traditionally, lung carcinoma has been divided into 2 histological types: non-small cell carcinoma (NSCLC), which represents 85% of cases, and small cell carcinoma that causes the remaining percentage. Within the NSCLC, there are the histological subtypes adenocarcinoma, squamous cell carcinoma, and large cell carcinoma. Current treatment strategies for lung carcinoma are based on differentiating these 2 histological types, as each respond to different treatment schemes.4-6 For example, in stage I and II NSCLC, the possibility of surgical management associated with neoadjuvant chemotherapy is considered, unlike small cell carcinoma, where surgery has no indication and its treatment is chemo and radiotherapy. In advanced stages of both histological types, chemotherapy remains the mainstay of treatment.5,6
In the last decade, a great emphasis has been manifested on the knowledge about the molecular determinants of lung cancer and the mutational profile of NSCLC. This is how, through sequencing techniques, numerous genomic events have been identified, such as activating mutations or genomic rearrangements in genes that encode protein kinases, including EGFR. 6
Epidermal growth factor receptor, also called ErbB1 or HER1, is part of the ErbB family, or transmembrane tyrosine kinase receptors found in the signal transduction pathways that regulate cell proliferation and apoptosis. 7 There are diverse mutations described in EGFR, which occur in up to 10% to 20% of NSCLC; most involve the tyrosine kinase domain and therefore, are candidates for treatment with tyrosine kinase inhibitors. Three inhibitors, Erlotinib, Gefitinib, and Afatinib, are widely used worldwide as first line of treatment for NSCLC with EGFR mutation. However, it has been described that, at 12 months after treatment, several of these carcinomas develop resistance. 6 Repetition of biopsy after treatment has shown the existence of mechanisms of acquired resistance to these drugs, including an uncommon one, in which a histological transformation of a NSCLC in small cell carcinoma is evidenced.8,9
The mechanism of resistance most often described in carcinomas with EGFR mutation is the acquisition of a second mutation in exon 20 (Thr790Met). This mutation increases the affinity for the ATP receptor and allows the EGFR signaling to continue in the presence of the tyrosine kinase inhibitor. This acquired mutation represents approximately 50% to 60% of the cases of resistance. There are other resistance mechanisms such as additional mutations in MET and HER2, which correspond to 15% to 20% of resistance.6,7
Several theories have been described that try to explain the transformed phenotype, 1 of these describes a pluripotential type II alveolar cell, with the ability to differentiate into adenocarcinoma or small cell carcinoma; this cell exposed to anti-EGFR treatment suffers histological transformation. 10 This hypothesis is favored by the finding, reported in almost all cases, and seen in our report, of persistence in the mutational state in the EGFR gene in the transformed phenotype, these mutations being extremely rare in primary small cell carcinoma. 11 In addition, losses of RB1 and TP53 or activation of PIK3CA has been reported at a mutational level, which are also considered critical for neuroendocrine differentiation after treatment with tyrosine kinase inhibitor. Another theory mentions the possibility that initially a mixed type lung carcinoma existed, with a minor component of small cell neuroendocrine carcinoma, which, after treatment, can persist and proliferate over the NSCLC component. 10
Among the risk factors that have been postulated that could predispose to this type of resistance mechanism are the longer treatment time with EGFR inhibitors, the female sex, and the history of cigarette smoking; however, studies regarding these factors are not conclusive. 11
As for the criteria which may lead to suspect a histological transformation is the rapid increase of neuroendocrine markers in blood, as well as the poor clinical response to the treatment established, making new biopsies fundamental, especially to focus pharmacologic management. 12 It is worth mentioning that in these cases, the liquid biopsy, another diagnostic option approved for the follow-up of patients with mutations in EGFR to monitor the appearance of resistance mutations, with a sensitivity for the Thr790Met mutation of up to 70%, would not be indicated, as the evaluation of the tissue is required to perform the histological diagnosis.13,14
In small cell carcinoma of the lung, the standard chemotherapeutic treatment is based on the combination of Cisplatin or Carboplatin plus Etoposide, with good response, both for the disease in early stages and for the advanced stage. However, recurrences are reported between 14 to 15 months for early stages and 6 months for advanced stages, with median survival of 15 and 8 months, respectively. In the cases of transformation, this same standard regimen is also administered, with reports of high response rates. 15
To conclude, among the mechanisms of resistance described to the treatment with tyrosine kinase inhibitors in patients with carcinomas with mutated EGFR, the transformation to small cell carcinoma besides being infrequent is particular, requiring a different diagnostic and therapeutic approach.
Footnotes
Acknowledgements
All the authors have approved the contents of this article and have agreed to the journal’s submission policies.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have significantly contributed to the article: LF-T contributed to the conception and design, literature review, manuscript writing and correction, and final approval of manuscript. LT contributed to the conception and design, literature review, manuscript writing and correction, and final approval of manuscript. MV contributed to conception and design, manuscript writing and correction, and final approval of manuscript. MA contributed to manuscript writing and correction and final approval of manuscript. JL contributed to manuscript writing and correction and final approval of manuscript. LFS contributed to conception and design, literature review, manuscript writing and correction, and final approval of manuscript.
Ethical Considerations
This report was prepared in accordance with the ethical standards of the institutional ethics committee and with the 1964 Helsinki Declaration. We have approval letter of Ethics Committee in biomedical research of the Fundación Valle del Lili to publish this manuscript, IRB/EC No. 162-2019.
Informed Consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
