Abstract
Antithyroid drug-induced agranulocytosis is a rare but potentially life-threatening complication, particularly in patients with limited therapeutic options. This case illustrates the complexity of managing uncontrolled thyrotoxicosis in the context of a patient with decompensated heart failure, chronic obstructive pulmonary disease (COPD), and adverse reactions to both methimazole and propylthiouracil. A 54-year-old male patient with a 2-year history of Graves’ disease presented with worsening dyspnea and tachycardia in the setting of thyrotoxicosis with a TSH of 0.0007 mIU/L. He had previously developed agranulocytosis (ANC 180 cells/µL) subsequent to treatment with methimazole, and subsequently developed febrile agranulocytosis (ANC 190 cells/µL) upon initiation of propylthiouracil, thereby exhausting all antithyroid drug options. A bridging therapy of Lugol’s iodine and granulocyte colony-stimulating factor (G-CSF) was utilized, which enabled successful total thyroidectomy. Right heart catheterization confirmed the patient’s suitability for surgery despite a left ventricular ejection fraction of 35%. This case highlights the importance of interdisciplinary collaboration in managing complex endocrine-cardiac-hematology presentations.
Introduction
Graves’ disease is the most common cause of hyperthyroidism, accounting for 60% to 80% of all hyperthyroidism cases worldwide. 1 The standard therapeutic options include antithyroid medications (thionamides), radioactive iodine (RAI) ablation, and thyroidectomy, with the choice of treatment depending on patient-specific factors such as disease severity, comorbidities, and patient preference. 2 In patients with significant cardiovascular comorbidities, such as systolic heart failure, pharmacological options become limited, as both RAI and surgery carry heightened perioperative risks in hemodynamically unstable patients. 3 Thionamides remain first-line therapy, yet they carry a risk of life-threatening agranulocytosis, an uncommon yet serious hematologic adverse event, occurring in approximately 0.2% to 0.5% of treated patients. 4 Cross-reactivity between methimazole and propylthiouracil with respect to agranulocytosis has been reported, with the risk of developing this complication with the second agent estimated to be as high as 50% after its occurrence with the first. 5 Sequential agranulocytosis from both thionamides is exceedingly rare and creates a therapeutic dilemma, as it eliminates all medical options for thyroid hormone control. We report a rare case of sequential antithyroid drug-induced agranulocytosis in a patient with decompensated heart failure, successfully managed through multidisciplinary strategies, culminating in definitive surgical therapy.
Case Presentation
A 54-year-old male with a 2-year history of Graves’ disease presented to the emergency department with worsening dyspnea and tachycardia. His past medical history was significant for: heart failure with reduced ejection fraction (HFrEF), nonvalvular atrial fibrillation, left bundle branch block, lower extremity venous insufficiency and a history of smoking.
On presentation, the patient was found to be in decompensated heart failure with rapid atrial fibrillation (Figure 1). One year prior, he had been deemed inoperable for thyroidectomy due to globally dilated cardiomyopathy, characterized by a left ventricular ejection fraction of 35% and a markedly enlarged right ventricle (diameter 70 mm) with preserved TAPSE (Figure 2). At that time, while thyrotoxic, he had received a single dose of radioactive iodine (RAI), followed by the initiation of methimazole in an attempt to control persistent thyrotoxicosis.
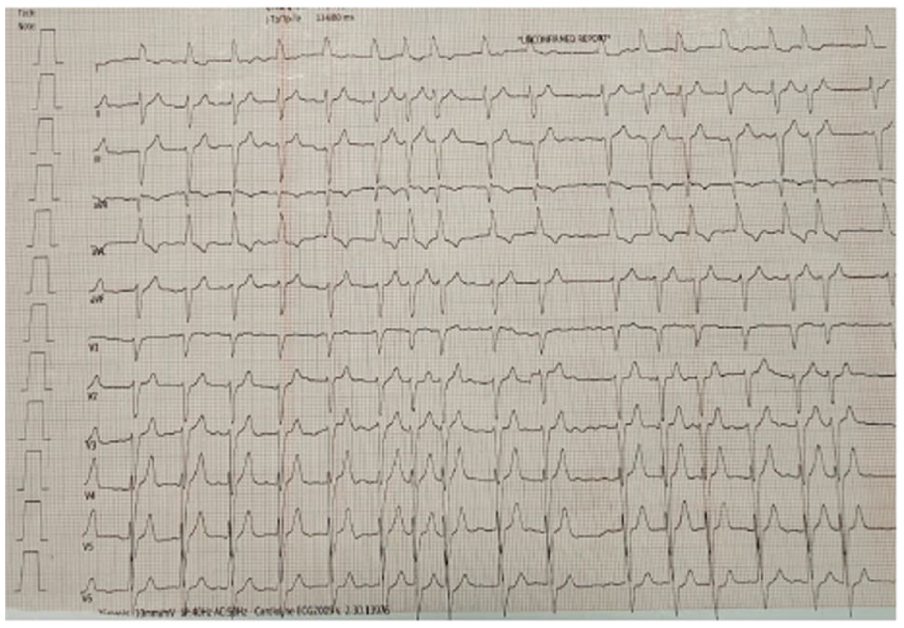
ECG upon admission showing rapid ventricular conducted atrial fibrillation.

Increased diameter of right ventricle measured at its base.
Diagnostic Assessment and Hospital Course
On admission, laboratory evaluation revealed overt thyrotoxicosis with a TSH of 0.0007 mIU/L (reference range: 0.4-4.0 mIU/L). Within days of methimazole treatment initiation 1 year prior, the patient had developed grade 4 neutropenia (absolute neutrophil count [ANC] 180 cells/µL; grade 4 defined as ANC <500 cells/µL), consistent with drug-induced agranulocytosis. Therefore, methimazole, not well tolerated by the patient due to the side effects, was discontinued. In the following months the patient remained stable, likely reflecting the delayed effect of RAI.
His medications at the current admission included edoxaban, propranolol, sacubitril/valsartan, furosemide, spironolactone and empagliflozin. Due to decompensated heart failure, he was admitted to the cardiology department, where propranolol was discontinued given low cardiac output. A respiratory consultation revealed COPD Stage C, and inhaled corticosteroids were initiated. The patient was stabilized on bisoprolol 15 mg/day, and high-dose propylthiouracil (900 mg/day) was introduced to control hyperthyroidism with the added benefit of reducing peripheral conversion of T4 to T3.
Although his heart failure clinical situation improved initially, by day 2 the patient developed leukopenia with grade 3 neutropenia (ANC 730 cells/µL; <1000 cells/µL).
Treatment
The propylthiouracil dose was reduced to 450 mg/day, but by day 3, he experienced febrile agranulocytosis (ANC 190 cells/µL; <500 cells/µL with fever). C-reactive protein (CRP) was elevated at 2.73 mg/dL (reference range: 0-0.5 mg/dL) and erythrocyte sedimentation rate (ESR) was 63 mm/h, confirming a systemic inflammatory response. Propylthiouracil was immediately discontinued, and the patient received 3 doses of filgrastim 5 µg/kg/daily (Neupogen).
Bridging therapy with Lugol’s iodine solution was initiated, resulting in a prompt decline in thyroid hormone levels and a rise in TSH within 3 days. A right heart catheterization performed on day 6 showed borderline right-sided pressures but no absolute contraindications to general anesthesia and surgery (Figure 3). Given the transient nature of iodine’s suppressive effect (Wolff-Chaikoff effect) and the patient’s now-stable cardiopulmonary status, he proceeded to a total thyroidectomy.
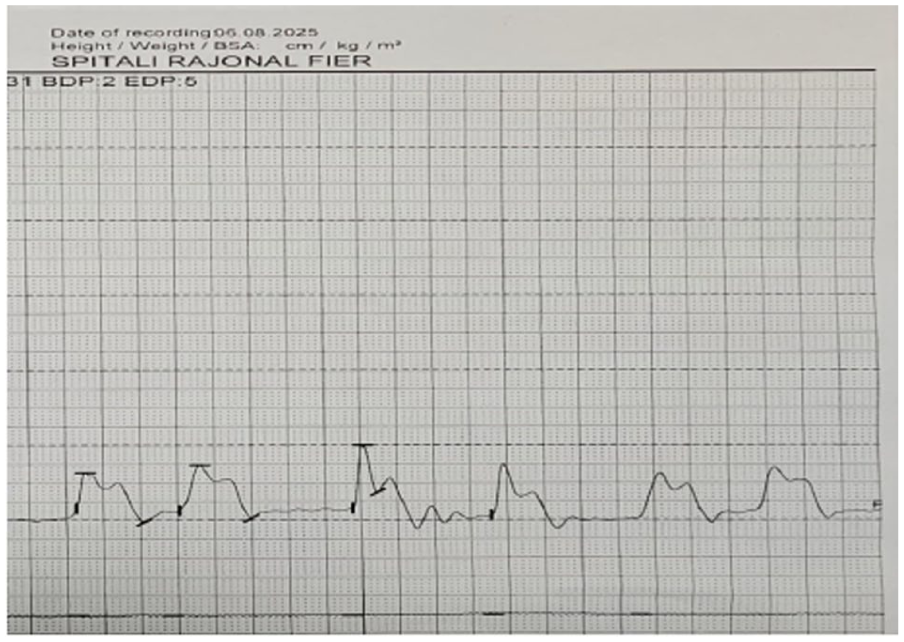
Invasive pressure tracing of the right ventricle.
Outcome and Follow-Up
The procedure was uneventful. Postoperatively, the patient remained stable (Figure 4) and was discharged on day 5 with arrangements for outpatient endocrine, cardiology and pneumology follow-up.

ECG upon discharge showing Atrial Fibrillation with controlled ventricular response.
Discussion
The management of hyperthyroidism in patients with severe cardiac comorbidities presents a complex clinical challenge. This case is particularly noteworthy, as our patient with Graves’ disease, decompensated heart failure, atrial fibrillation, and COPD developed agranulocytosis sequentially from both methimazole and propylthiouracil, exhausting conventional medical options and necessitating a carefully orchestrated definitive therapy.
Dilated cardiomyopathy in the context of thyrotoxicosis has a prevalence of 6% and only 1% of thyrotoxicosis cases are complicated with severe LV dysfunction. For thyrotoxic dilated cardiomyopathy complicated with LV dysfunction, evidence-based pharmacologic measures valid for heart failure should always be administered alongside specific thyroid therapies such as thionamides (methimazole or propylthiouracil). 1 In hyperthyroidism states there is a predictable increase in heart rate and contraction force, resulting in undue strain on the myocardium, significantly increasing the risk of atrial fibrillation and heart failure. 2
In our patient, we substituted propranolol with bisoprolol, considering the poor ejection fraction of the left ventricle and taking into consideration the effect of propylthiouracil in preventing the peripheral conversion of fT4 into fT3. 3
Agranulocytosis, a severe form of neutropenia (absolute neutrophil count <500 cells/µL), is the most feared idiosyncratic reaction to thionamide therapy, occurring in 0.2% to 0.5% of patients. 4 The risk appears to be dose-dependent, particularly for methimazole, and is highest within the first 3 months of treatment.5,6 The underlying mechanism is thought to be immune-mediated, involving drug-dependent antibodies against neutrophils or direct bone marrow toxicity.
A critical aspect highlighted by this case is the cross-reactivity between methimazole and propylthiouracil. While they are structurally different (imidazole vs thiouracil derivative), the risk of developing agranulocytosis with the second drug after experiencing it with the first is estimated to be as high as 50%. 5 Therefore, once agranulocytosis occurs with 1 ATD, the other is generally considered contraindicated. Our patient’s experience confirms the danger of this approach, reinforcing that definitive therapy, either RAI or surgery, is required. Management of the acute episode involves immediate cessation of the offending drug, supportive care, broad-spectrum antibiotics for febrile patients, and often, the administration of G-CSF to accelerate neutrophil recovery. 4
Right heart failure (RHF), defined as a clinical syndrome resulting from altered right heart structure and/or function leading to suboptimal blood flow delivery to the pulmonary circulation and/or elevated venous pressures, is strongly associated with poor postoperative outcomes. 7 Given our patient’s comorbidities (HFrEF, COPD and lower extremity venous insufficiency) and his echocardiographic features, it was mandatory to obtain invasive right heart pressures 8 as a measure of better anesthetic preparation. 9
With ATDs contraindicated, the choice lay between RAI and thyroidectomy. Although RAI had been administered previously, its effect was incomplete, and a repeat dose carried the risk of transiently worsening the thyrotoxicosis, which could precipitate a catastrophic cardiac event in a patient with an ejection fraction of 35%. 10 Thus, total thyroidectomy was by default the definitive treatment of choice. However, surgery in a thyrotoxic patient with severe cardiorespiratory compromise is fraught with risk, including thyroid storm and perioperative mortality. A multidisciplinary approach was essential for pre-operative optimization. The management of thyrotoxic dilated cardiomyopathy, which can complicate up to 1% of hyperthyroidism cases with severe LV dysfunction, requires both aggressive heart failure treatment and rapid control of thyroid hormones. 1 The switch to bisoprolol and the use of guideline-directed medical therapy were key cardiac interventions.
Limitations
This case report has several limitations inherent to the single-case study design. First, the diagnosis of drug-induced agranulocytosis was based on the temporal relationship between antithyroid drug administration and neutrophil decline; anti-neutrophil antibody testing was not performed, which would have provided more definitive evidence of an immune-mediated mechanism. Second, bone marrow biopsy was not obtained to formally exclude other causes of neutropenia, although the clinical presentation, timing, and rapid recovery with G-CSF were highly consistent with drug-induced agranulocytosis. Third, long-term follow-up data beyond the immediate postoperative period are limited; ongoing monitoring of thyroid replacement adequacy, cardiac function recovery, and potential late surgical complications would strengthen the conclusions. Finally, as a single-center experience in a resource-limited setting, the generalizability of the management strategy to other clinical contexts should be interpreted with caution.
Learning Points
• Multidisciplinary Coordination is Vital in Complex Comorbidities: Successful management required close collaboration between endocrinology, cardiology, hematology, anesthesiology, and surgery teams to stabilize a high-risk patient and achieve definitive therapy.
• Dual Antithyroid Drug Agranulocytosis is Rare but Critical: Sequential agranulocytosis from both methimazole and propylthiouracil is extremely uncommon but underscores the need for early detection and prompt discontinuation of the offending drugs.
• Definitive Treatment is Often Unavoidable: Once agranulocytosis occurs from thionamide therapy, repeat RAI or total thyroidectomy becomes the only viable option. Preoperative stabilization and endocrine-cardiac coordination are key to ensuring survival and long-term disease control.
• Bridging Therapy Can Enable Safe Surgical Intervention: Short-term use of Lugol’s iodine and granulocyte colony-stimulating factor (G-CSF) can bridge patients safely to thyroidectomy when medical therapy is contraindicated due to drug toxicity.
• Comprehensive Cardiac Assessment Guides Surgical Timing: In patients with severe heart failure, invasive hemodynamic evaluation (right heart catheterization) is essential to determine operability and minimize perioperative risk.
Footnotes
Consent to Participate
Signed informed consent obtained directly from the patient.
Author Contributions
Amarildo Voci was involved in admitting the patient, diagnosing, treatment and manuscript of the case. He was the following physician of the case. He coordinated the work done on the patient and the writing of the manuscript. Liri Seraj was involved in diagnosing and treatment of the patient. She was involved in writing the manuscript, and submitting the final manuscript. Asfloral Haxhiu and Mario Shehu were involved in the surgical treatment of the patient. They wrote part of the discussion and abstract. All authors reviewed and approved the final draft.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Original data generated and analyzed during this study are included in this published article.
