Abstract
Pure ground-glass and part-solid lung nodules are becoming increasingly appreciated as harbingers of malignancy. However, occasionally diagnostic biopsies can demonstrate unexpected findings that may lead to a misleading diagnosis and clinical confusion. Here we present the case of a 70-year-old Taiwanese man with a light smoking history (10-20 pack years), emphysema, and childhood-treated tuberculosis who presented with a part-solid nodule that was diagnosed as a very rare case of adenocarcinoma of the lung with desquamative interstitial pneumonia (DIP)-like microscopic morphology. DIP is a non-neoplastic interstitial lung disease which presents as a diffuse and bilateral process and primarily occurs in smokers. When the morphologic pattern of DIP presents as a focal process, usually around bronchioles, it is labeled respiratory bronchiolitis. (RB)— a very common entity and almost universal in patients with significant smoking history. We describe this case to help educate and guide pathologists on the existence of this very rare entity and to consider performing lung cancer immunohistochemical markers whenever faced with this unexpected histological pattern in a pulmonary nodule.
Keywords
Introduction
DIP-like adenocarcinoma of the lung is exceedingly rare, with only 11 documented cases in the literature (Table 1). Although the histology mimics DIP under the microscope, the radiographic appearance is usually that of a lung nodule or mass and does not resemble the classic diffuse interstitial lung disease that is associated with smoking. However, the overlapping histologic features of DIP-like adenocarcinoma with smoking-related entities DIP and RB may lead to misdiagnosis or delays in treatment. In 2014, Raparia et al. reported 7 such cases of lung adenocarcinoma that mimicked DIP, noting their deceptive histologic presentation and the risk of underdiagnosis when misinterpreted as benign disease. 1 Since then, only a handful of additional cases have been described.2-4 However, the true prevalence of this disease may be underreported because DIP-like lung adenocarcinoma is not at the forefront of most chest pathologists’ radar. Indeed, our case was initially missed upon review of ample bronchoscopic forceps samples, and was only caught later after surgical lung biopsy confirmed DIP-like adenocarcinoma histopathology. This case adds to the limited existing literature and will hopefully help educate pulmonologists and pathologists alike on the need to definitively rule-out lung cancer whenever faced with this unexpected histological pattern in a solitary pulmonary nodule.
Summary of Patient Demographics, Tumor Location, Origin, DIP-Like Histologic Percentage, and IHC Markers From Previously Published Cases.
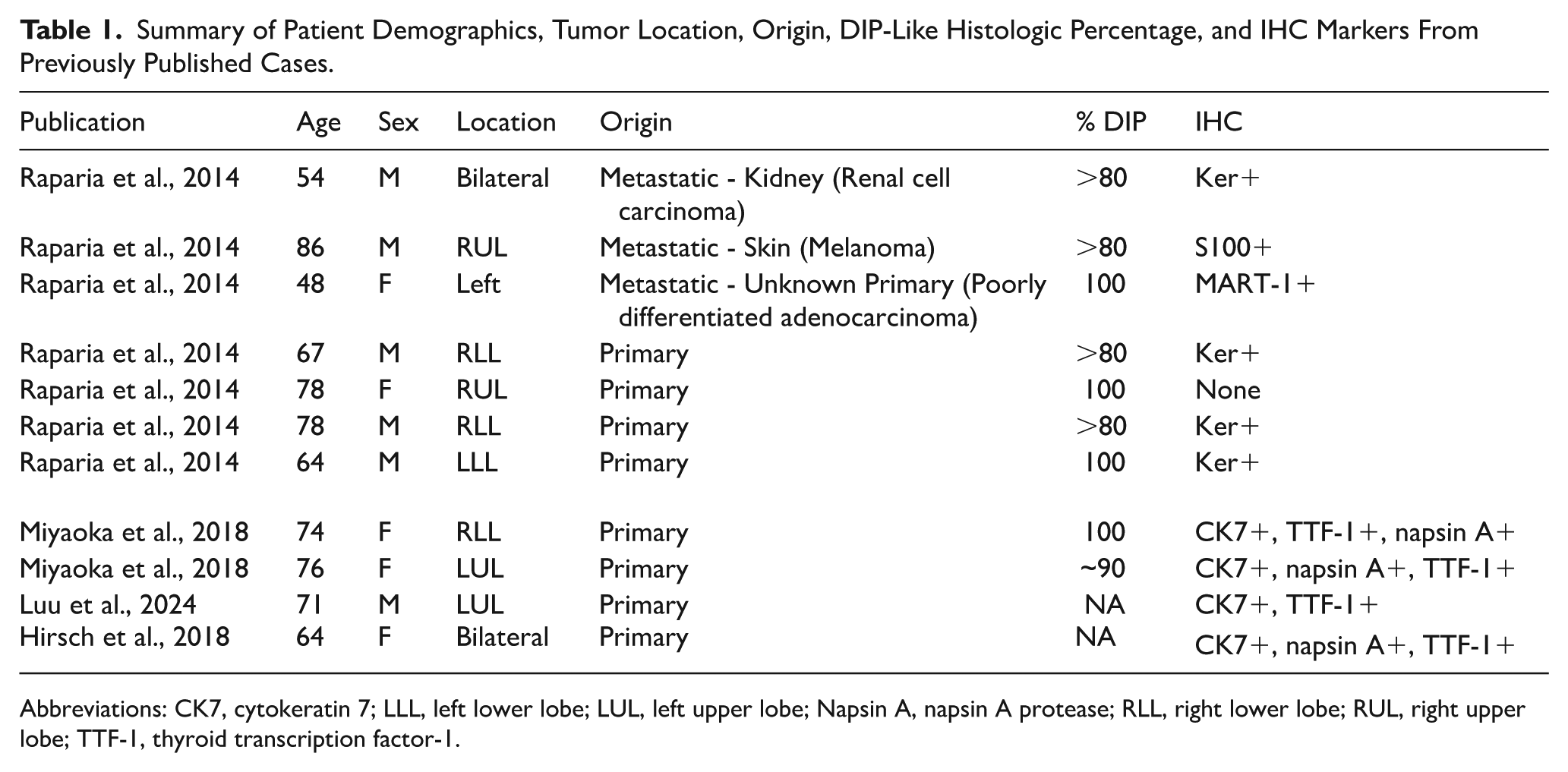
Abbreviations: CK7, cytokeratin 7; LLL, left lower lobe; LUL, left upper lobe; Napsin A, napsin A protease; RLL, right lower lobe; RUL, right upper lobe; TTF-1, thyroid transcription factor-1.
Patient Information
A 70-year-old Taiwanese man with a light smoking history (10-20 pack years), emphysema, and childhood-treated tuberculosis presented to his primary care provider with 1 episode of hemoptysis characterized by 1 to 2 cc of blood-tinged sputum. The patient denied any symptoms of fevers, night sweats, weight loss, or productive cough. He had no chest pain or shortness of breath. Review of systems was otherwise negative. The patient’s routine medications included those for hypertension, glaucoma, and gout, but no anticoagulants. He denied any drug allergies. Family history was significant for hypertension in his mother, but no known cancers otherwise. In terms of environmental exposures, he was a car salesman prior to immigrating from Taiwan in 1981. Afterward, he worked in a warehouse in downtown Los Angeles assembling artificial flowers which exposed him to chemicals used in the management of tree trunks, heavy second-hand smoke from co-workers, and air pollution from neighboring buildings and vehicle exhaust. His house was built in 1949 and may have contained asbestos; his wife did most of the Chinese-style cooking, but with adequate ventilation.
Clinical Findings
The patient’s physical exam upon presentation was unremarkable. His labs were normal except for an elevated WBC count at 17,000/µL with neutrophilic predominance. Acid-Fast Bacilli cultures (AFB) were found to be negative. Fungal serologies for Pneumocystis jirovecii, Aspergillus fumigatus, Aspergillus terreus, and pan-Aspergillus antibodies were all negative. However, the Aspergillus galactomannan antigen assay was elevated at 1:150. An autoimmune panel was not performed. His primary care provider ordered a chest computed tomography (CT) scan to further work up the hemoptysis.
Diagnostic Assessment
The initial chest CT showed a 2.7 cm× 2.2 cm part-solid nodule in the right upper lobe (RUL) with a peripheral ground-glass appearance and no significant adenopathy (Figure 1). A follow-up PET/CT scan showed slight hypermetabolic uptake in the primary lesion, but negative everywhere else (Figure 2). Given his history of previous tuberculosis, differentiating between infection, inflammation, and malignancy was difficult.
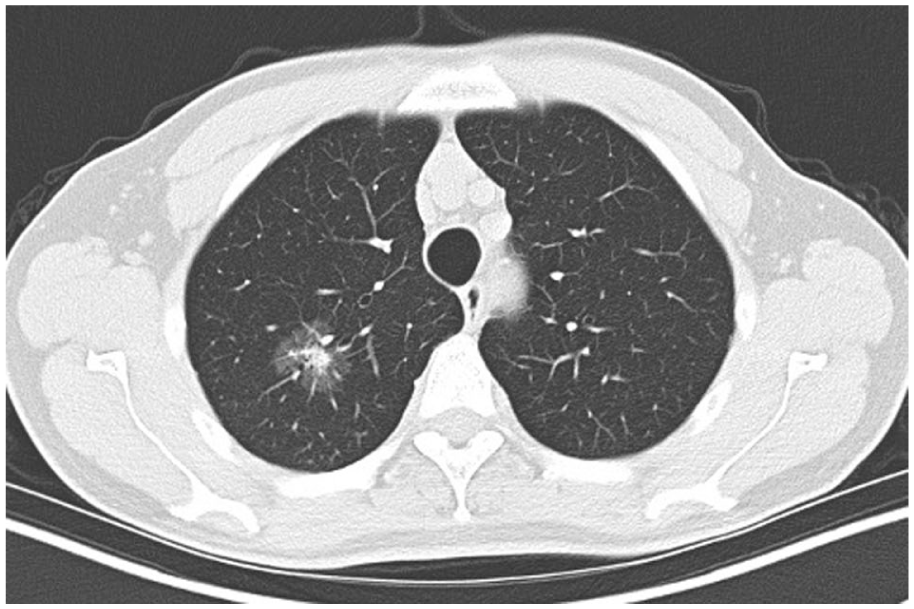
CT scan of 2.7 cm× 2.2 cm RUL part-solid lesion; no significant adenopathy was reported.

PET scan of RUL part-solid lesion which showed barely-discernible hypermetabolic activity in the primary lesion and no activity in the lymph nodes.
Based on these findings, the Mayo Nodule Risk Score for pretest probability of lung cancer was only 10.5%. However, because this fell within the “intermediate risk” category, the patient underwent a robotic bronchoscopy with endobronchial ultrasound (EBUS) staging. Unfortunately, the procedure was deemed non-diagnostic. The bronchoalveolar lavage (BAL) cultures (bacterial, fungal, AFB, and viral) were all negative, and the cytology was unrevealing except for peripheral blood elements, histiocytes, and bronchial epithelial cells. Generous forceps biopsies only showed crushed alveolar tissue with atypical pneumocyte proliferation, which was suspicious but not diagnostic for malignancy. Systematic staging of the lymph nodes was negative for cancer.
Despite the low PET avidity and non-diagnostic bronchoscopy, there was still a high suspicion for lung cancer given the patient’s risk factors and nodule characteristics. The patient’s old age, smoking history, history of emphysema, and history of tuberculosis plus the nodule’s right upper lobe location, significant size (2.7 cm) and part-solid appearance with no calcification pushed the overall risk for lung cancer higher on clinician gestalt. For this reason, the patient underwent surgery with full resection of the lesion for definitive diagnosis. He underwent a right upper segmentectomy with resection of regional lymph nodes including right level 4, level 7, level 9, anterior level 10, level 10, level 11, and anterior level 12 lymph nodes.
Histologic sections of the nodule revealed sheets of discohesive cells filling the alveolar spaces very similar to alveolar macrophages. This pattern of alveolar filling cells is characteristic of the pattern seen in DIP; however, careful review of the alveolar-filling cells revealed mild pleomorphism and irregular nuclear contours suspicious of a neoplastic process. Moreover, the radiographic appearance and presentation were not compatible with an interstitial lung disease. Subsequent immunohistochemical (IHC) staining for TTF-1 and pan-keratin revealed strong positivity and confirmed the nodule was an adenocarcinoma (Figure 3). Immunohistochemical analysis of the biopsy demonstrated positive staining for AE1/AE3 and Napsin A. Ki-67 showed mild increase, while p63 showed rare positive cells. Neuroendocrine markers, chromogranin A, synaptophysin, and CK5/6, were found to be negative. The final diagnosis was poorly differentiated adenocarcinoma of the lung with 100% DIP-like histologic pattern (Figures 4 and 5).

TTF-1 immunohistochemistry stain reveals positive expression in cells of interest, consistent with lung adenocarcinoma. Note many cells appear to be discohesive and “floating” in the alveolar space. (TTF-1 immunohistochemistry stain; original magnification 600×).

An example of true DIP (from different patient case, courtesy WDW). The low power view (A) 100× reveals discohesive sheets of histiocytes filling the alveolar spaces. The surrounding alveolar walls demonstrate fibrosis and architectural remodeling, consistent with smoking-related emphysematous changes. At higher power (B) 400×, the histiocytes are seen to contain fine golden brown pigment characteristic of DIP and smoking-related changes.(Hematoxylin & eosin stain; original magnification 400×).

DIP-Like Adenocarcinoma (from this patient case). The low power view (C) 100× reveals alveolar spaces occluded by discohesive cells including a few giant cells (arrow). At higher power (D) 400×, the cells show prominent nuclear atypia with pleomorphism and irregular nuclear contours, characteristic of carcinoma. There is no intracellular pigment present.(Hematoxylin & eosin stain; original magnification 400×).
Next generation sequencing testing of the specimen was sent which showed a SOS1 R552 mutation. No other mutations such as EGFR, ALK, ROS1, KRAS, PD-L1 were discovered. The tumor proportion score was 0%, and tumor mutation burden was 4 mut/Mb (Table 2).
Timeline of Events.

Timeline
Follow-Up and Outcomes
Our patient’s DIP-like lung adenocarcinoma was categorized as stage IA3 (pT1cN0). The surgical margins were clear, and 15 lymph nodes sampled during surgery were found to be negative for metastatic tumor. Medical oncology confirmed that he did not require adjuvant systemic therapy for this rare form of lung cancer. The patient had a slight post-operative complication of a surgical wound infection, but this was treated and his 3-month follow up CT showed no recurrence (Figure 6). He remains cancer-free and is doing well.
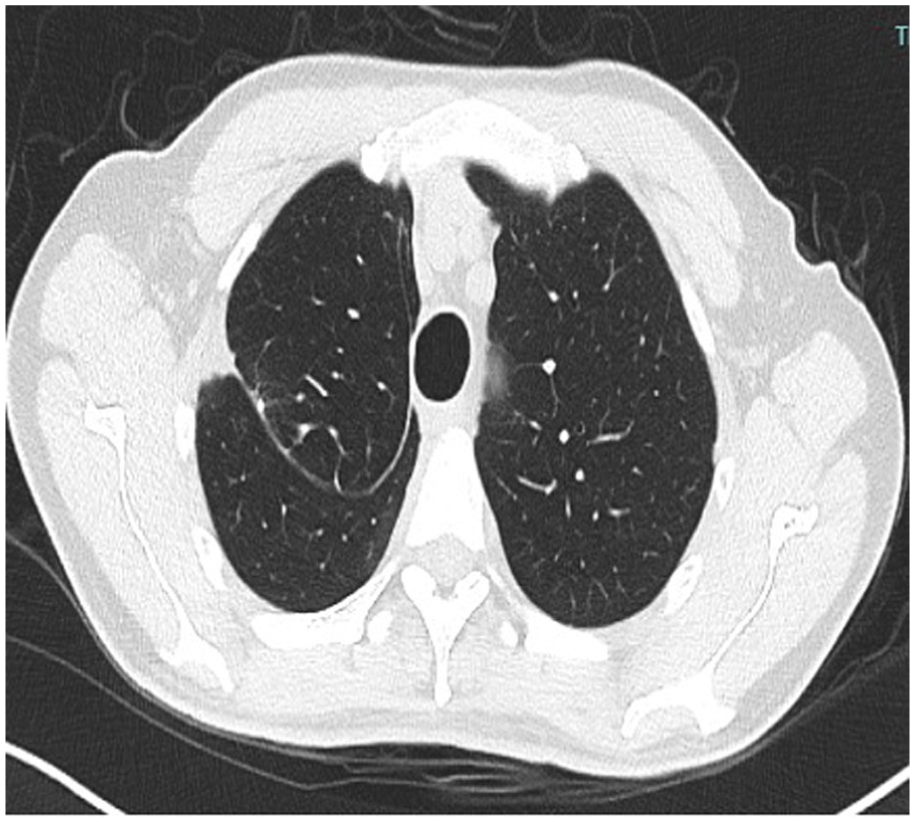
Three-month postoperative scan.
Discussion
Only 11 cases of DIP-like adenocarcinoma of the lung have been found in the literature. The true incidence may actually be higher if more clinicians are aware that lung cancer can look like DIP under the microscope. Unlike conventional lung adenocarcinomas, the cells that fill the alveoli in the DIP-variants appear superficially similar to benign alveolar macrophages, potentially leading to misdiagnosis, especially if the samples are from smaller bronchoscopy or CT-guided biopsies. Fortunately, for this patient, his risk factors for lung cancer raised enough suspicion that an aggressive workup ensued, culminating in a diagnostic surgical biopsy and resection. However, in patients with more nebulous clinical presentations, a cancer diagnosis may be easily missed if this entity is not under consideration and/or if exploratory IHC staining is not performed and carefully analyzed. We present this rare case to help educate pulmonologists and pathologists on the need to definitively rule-out cancer whenever faced with this seemingly benign histological pattern in a lung nodule.
Footnotes
Acknowledgements
The authors wish to thank the patient and his family for allowing his story to contribute to the advancement of medical knowledge. The authors would also like to thank the Ming Hsieh Institute for its generous support of this and other related research efforts.
Ethical Considerations
No ethics approval was necessary for this case report.
Consent to Participate
Written informed consent for this case report was obtained by his treating oncologist (RH) and documented in his electronic medical record.
Author Contributions
Natalia Brar, BS (candidate): Conducted literature review, organized clinical data, prepared the initial manuscript draft, and assisted with figure and table preparation. Robert Hsu, MD: Treating physician for the patient, clinical data acquisition, case interpretation, and manuscript review. William D. Wallace, MD: Reviewed and interpreted pathology findings, prepared and annotated histopathology figures, and manuscript revision. Ching-Fei Chang, MD: Supervised case report conception, coordinated multidisciplinary input, final manuscript revisions, and approved the submitted version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
