Abstract
Introduction:
Spontaneous splenic rupture (SSR) is an uncommon and potentially life-threatening cause of acute abdominal pain, often linked to underlying pathology. Because its presentation can mimic more common conditions, diagnosis is frequently delayed. We report a case from a resource-limited setting where SSR initially mimicked peptic ulcer disease, underscoring diagnostic challenges.
Case Presentation:
A 57-year-old Ethiopian woman with chronic reflux presented after 5 days of severe epigastric pain radiating to the left shoulder, with nausea and vomiting but no trauma or fever. Initial labs showed mild anemia. Abdominal examination revealed localized epigastric tenderness without peritonism. The initial working diagnosis was peptic ulcer disease; proton pump inhibitors were started, and endoscopy was unavailable. After 2 days, she developed hypotension and left upper quadrant pain. Bedside ultrasound (FAST) showed free intraperitoneal fluid, and contrast CT confirmed a ruptured spleen with hemoperitoneum.
Management and Outcome:
The patient underwent emergency laparotomy, which found a shattered spleen and ~3.5 L of intra-abdominal blood; a splenectomy was performed. She required an intraoperative transfusion and was stabilized. Histopathology confirmed a normal spleen with preserved architecture, supporting a diagnosis of idiopathic rupture possibly related to transient microvascular congestion. Postoperatively, she recovered uneventfully and was discharged on postoperative day 5. She received the vaccinations needed and was counseled about infection risk.
Conclusion:
This case illustrates that true spontaneous rupture can occur in a histologically normal spleen and may mimic peptic ulcer disease in presentation. Recognizing atypical features and ensuring timely imaging are crucial, especially in resource-limited settings where delayed diagnosis increases mortality.
Introduction
Spontaneous or atraumatic splenic rupture (SSR) is a rare condition in which the spleen ruptures without antecedent trauma.1,2 A systematic review by Renzulli et al 3 identified hundreds of atraumatic splenic ruptures and estimated that roughly 6% to 7% are truly idiopathic, with reported mortality varying by series but increasing when diagnosis is delayed or when underlying disease is present.1,4 Nearly all cases of SSR occur in the setting of splenic pathology. 3 Infections (especially malaria and viral mononucleosis), hematologic malignancies (eg, leukemia or lymphoma), and inflammatory or infiltrative diseases are the most common causes.3,5 True “idiopathic” rupture of a normal spleen is exceedingly uncommon (estimated 6%-7% of cases).1,6 Proposed mechanisms for SSR include rapid splenic enlargement with capsule tension (eg, from hyperplasia or infiltration) or intrasplenic vascular compromise.7,8 Infections such as Epstein-Barr virus (EBV) or malaria cause lymphocytic infiltration and splenomegaly, which can weaken the splenic capsule and precipitate rupture.7,9
Clinically, SSR typically presents as acute left upper quadrant pain, often with referred shoulder pain (Kehr’s sign) from diaphragmatic irritation.2,8 Nausea, vomiting, dizziness, or signs of hypovolemia may accompany the pain. 8 The presentation can mimic other causes of acute abdomen (eg, perforated ulcer or pancreatitis).1,8 Physical exam may be non-specific or reveal left-sided guarding if peritoneal irritation is present. 1 Laboratory tests are not diagnostic; anemia or leukocytosis may be seen, and markers such as Erythrocyte Sedimentation Rate (ESR) and C-reactive Protein (CRP) can be normal, as in our patient.
Definitive diagnosis requires imaging. A focused assessment with sonography in trauma (FAST) is a rapid bedside tool that may detect free fluid, but contrast-enhanced computed tomography (CT) is the gold standard for SSR, with sensitivities of ~85% to 95%.2,10 CT findings include splenic laceration or hematoma and intra-abdominal hemorrhage. In resource-constrained settings without immediate CT access, diagnosis may be delayed until hemodynamic collapse makes exploratory laparotomy necessary.7,11
Management of SSR depends on hemodynamic stability. Hemodynamically unstable patients require urgent laparotomy and usually splenectomy.2,10 In stable patients with contained hemorrhage, non-operative management (observation or splenic artery embolization) has been reported.12,13 However, many series report that the majority of SSR patients are managed surgically.2,10 After splenectomy, patients must receive appropriate immunizations and infection prophylaxis to prevent overwhelming post-splenectomy infection (OPSI).14,15
This case report presents a 57-year-old woman with SSR that initially mimicked peptic ulcer disease. It is unique in that it occurred in a normal spleen (idiopathic SSR) in a patient from a resource-limited setting, highlighting diagnostic challenges. We review the relevant literature on SSR epidemiology, pathophysiology, clinical features, and management.
Case Presentation
A 57-year-old female with a history of gastroesophageal reflux disease presented to the emergency department with severe epigastric abdominal pain of 5 days’ duration (Day 1-5) radiating to the left shoulder. The pain was associated with nausea and multiple episodes of vomiting but without fever, trauma, or changes in bowel habits. She had no prior history of abdominal surgery or known hematologic disease.
On initial evaluation (Day 5), she appeared uncomfortable but was hemodynamically stable, with mild tachycardia (heart rate [HR]: 103 and blood pressure [BP]: 115/75). Abdominal examination revealed epigastric tenderness without significant guarding or rebound tenderness. Laboratory investigations showed mild anemia, with a hemoglobin of 10.5 g/dL, and white blood cell count of 8000/µL (within the normal range). Liver function test (LFT), renal function test (RFT), erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and serum electrolytes were all normal. In view of her history of chronic epigastric pain and the absence of obvious risk factors, a provisional diagnosis of peptic ulcer disease was made. She was admitted for conservative management with was started on omeprazole 40 mg IV once daily, which was continued until definitive management. Upper gastrointestinal endoscopy was not available in the facility at the time. And, the anemia was initially attributed to chronic gastrointestinal blood loss based on her history of chronic epigastric symptoms; however, no prior endoscopic evaluation or fecal occult blood testing was available to localize the source.
Over the next 48 hours (Day 5-7), the patient’s condition progressively worsened. She developed abdominal distension, dizziness, and mild hypotension, with her blood pressure dropping to 90/60 mmHg. Repeat examination revealed localized tenderness in the left upper quadrant. Focused assessment with sonography in trauma (FAST) at the bedside demonstrated free intraperitoneal fluid in Morrison’s pouch (right upper quadrant) and in the pelvis (positive RUQ and pelvic views); trace fluid was also seen in the left upper quadrant. These findings were interpreted as consistent with intraperitoneal hemorrhage. This was followed by a contrast-enhanced abdominal CT, which showed a heterogeneous spleen with loss of normal architecture and capsular irregularity, and a large subcapsular/perisplenic hematoma with extensive hemoperitoneum. No focal arterial blush was evident on the portal-phase images. The appearance is most consistent with a shattered spleen. Radiologic and operative findings indicate an American Association for the Surgery of Trauma (AAST) grade V splenic injury (Figure 1).

Axial contrast-enhanced CT of the upper abdomen. The image demonstrates heterogeneous splenic parenchyma with capsular irregularity, a large subcapsular/perisplenic hematoma and extensive intraperitoneal hyperdense fluid layering in the left upper quadrant and pelvis consistent with hemoperitoneum. No unequivocal focal arterial blush is visible on this portal-phase image.
The patient was immediately prepared for surgery and underwent an emergency laparotomy. Intraoperative findings included a shattered spleen with active bleeding and a large associated hematoma (Figure 2A and B). Approximately 3.5 L of blood were aspirated from the peritoneal cavity. A splenectomy was performed, and a drain was left in situ. She was transfused intraoperatively and stabilized. Histopathology revealed preserved red and white pulp with intact trabeculae and capsule, and no evidence of neoplasm, infarction, or inflammatory infiltrate. Postoperative recovery was uneventful, and she was discharged in stable condition on the fifth postoperative day.
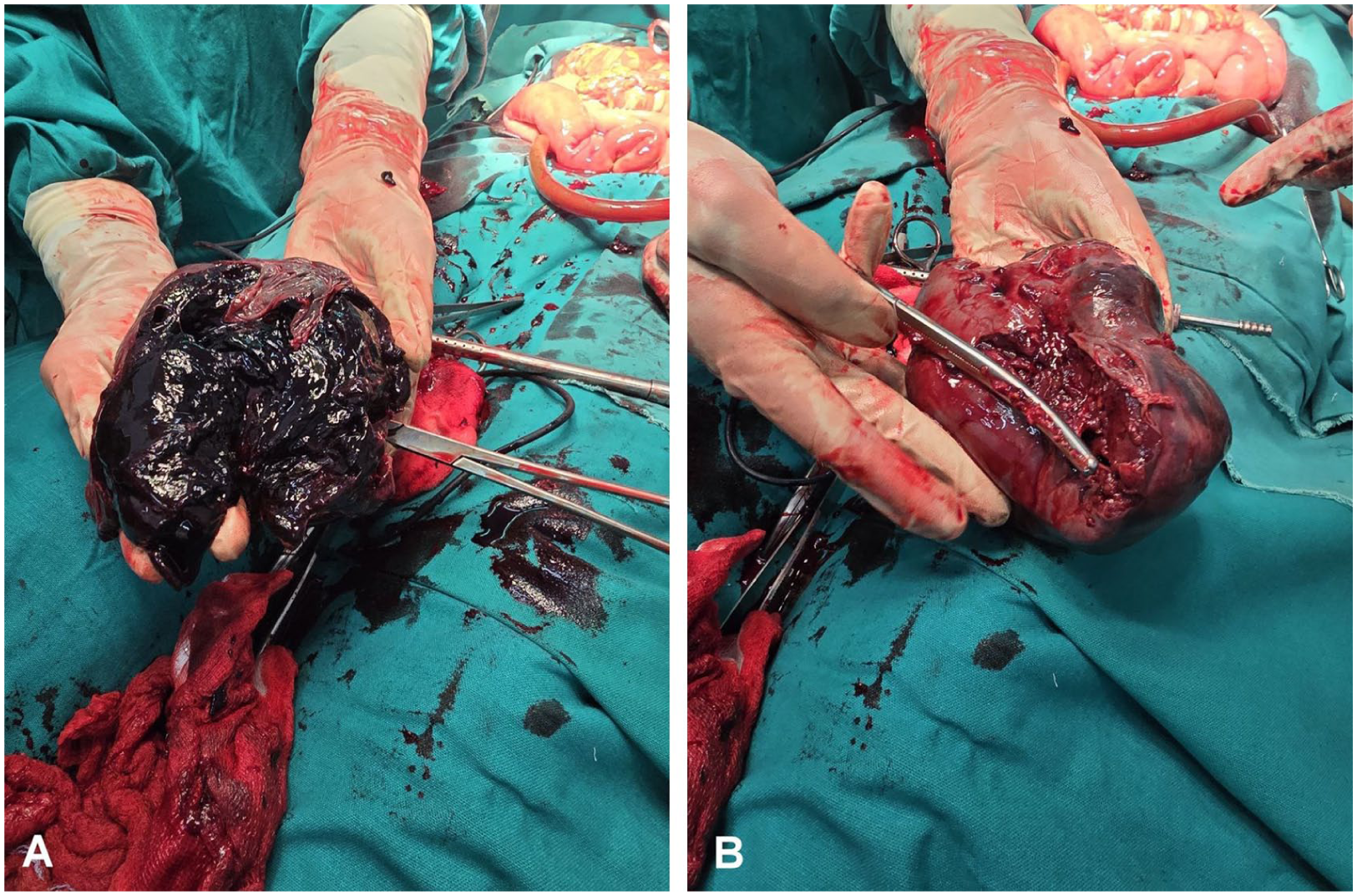
Intraoperative and gross specimen views of the shattered spleen: (A) intraoperative photograph during emergency laparotomy showing the spleen with multiple deep lacerations, adherent perisplenic clot and areas of active bleeding from the splenic surface; (B) Gross specimen photograph taken immediately after resection demonstrating capsular disruption, multiple deep lacerations and a dark subcapsular hematoma.
At follow-up 3 months after surgery, the patient remained well, with no complications or readmissions. She had been counseled about her condition, educated on the risks of overwhelming post-splenectomy infection (OPSI), and received appropriate vaccinations against Streptococcus pneumoniae, Haemophilus influenzae type b, and Neisseria meningitidis.
Discussion
This case underscores several key clinical lessons. First, idiopathic SSR, rupture of a histologically normal spleen without trauma, is extremely rare.1,3 Most SSR cases involve pathology (malignancy, infection, etc.).3,5 The absence of any splenic pathology on histology (normal white and red pulp, intact follicles, and negative infectious/malignant studies) fulfilled the criteria for true spontaneous rupture.1,8 We describe this case as idiopathic SSR based on the absence of trauma, normal splenic histology, and the available laboratory data; however, because specific infectious and hematologic investigations were not performed, occult causes cannot be absolutely excluded. In the literature, idiopathic SSR comprises only about 6% to 7% of atraumatic cases.1,6 A systematic review by Renzulli et al 3 (1980-2008) identified 845 reported atraumatic splenic ruptures and classified ~7% as truly idiopathic, with age, splenomegaly, and underlying malignancy associated with worse outcomes. Spontaneous rupture of a normal spleen must be distinguished from occult pathology; in our patient, extensive testing found no cause, underscoring how rare idiopathic SSR is Liu et al., 2 Kaniappan et al. 5 Recent literature proposes several mechanisms for idiopathic splenic rupture, including microvascular occlusion with focal infarction, transient congestion raising intrasplenic pressure, and rupture of small subcapsular hematomas.3,16 A 2025 report by Medbal et al 17 and a 2024 review of cases with normal spleens both describe microdamage and pressure-related failure of the splenic capsule as plausible explanations. 1 In infectious mononucleosis, rapid splenic enlargement and capsular thinning may produce a similar pathophysiology. 13
Second, SSR can easily be mistaken for more common conditions. Our patient’s predominant epigastric pain and anemia led to an initial working diagnosis of peptic ulcer disease. Nonspecific symptoms and physical findings are common in SSR.2,8 Nonspecific symptoms and physical findings are common in SSR. Although the patient reported left-shoulder radiation of pain, no objective Kehr’s or Balance’s signs were elicited on initial physical examination. This dissociation between patient-reported referred shoulder pain and the absence of an elicited Kehr’s sign likely contributed to the initial diagnostic uncertainty. In fact, delayed diagnosis of SSR is frequent; 1 review noted that left shoulder pain and peritonism are not always seen, contributing to misdiagnosis. 18 SSR can mimic peptic ulcer perforation, acute pancreatitis, or myocardial ischemia because all may present with epigastric pain, nausea, and hemodynamic change.1,3 But Targeted bedside tests (plain X-ray, serum amylase/lipase, ECG/troponin) can quickly distinguish common mimics of SSR and help triage patients for urgent imaging or transfer.2,8 Diagnostic delay can be fatal: reported preoperative mortality is 8% to 12%.3,4 In our patient, deterioration (new hypotension, quadrant tenderness) prompted a repeat exam and imaging. SSR should be considered whenever unexplained hypotension or anemia develops in acute abdomen without trauma.5,8
Third, this case highlights the impact of resource limitations on care. Endoscopy was not immediately available, and initial reliance on clinical diagnosis delayed definitive imaging. In settings lacking routine CT access, point-of-care ultrasound (FAST) can expedite diagnosis of hemoperitoneum.2,10 Indeed, Free fluid was identified on FAST, which triggered the CT scan. However, in many low-resource hospitals, even ultrasound may be limited, further hampering early detection.10,11 We advocate high suspicion and early transfer for imaging in such contexts to avoid late collapse.
Regarding management, urgent surgery saved our patient’s life. In SSR with hemodynamic instability, splenectomy is generally recommended.2,10 In a recent series, over 80% of atraumatic ruptures were managed by splenectomy. 2 Our patient lost ~3.5 L of blood and required massive transfusion; delayed intervention would likely have been fatal. Some stable SSR cases can be managed nonoperatively (observation or angioembolization),12,13 but this approach is controversial and often reserved for clearly stable patients. In resource-limited settings without ready Interventional Radiology, splenectomy remains the mainstay. Notably, histology revealed no underlying disease, so further therapy for pathology was not needed.
Postoperative recovery was uneventful, consistent with reports that timely splenectomy yields good outcomes.2,5 After splenectomy, preventing OPSI is crucial. Overwhelming post-splenectomy infection is a rare but highly lethal syndrome.14,15 We ensured pneumococcal, meningococcal, and H. influenzae type b vaccines were given, following standard guidelines.19,20 The literature strongly supports vaccinations and patient education to reduce OPSI risk.14,15
Finally, the outcome in our patient aligns with published data. In atraumatic rupture, mortality depends on the promptness of care and the underlying etiology.2,5 Timely surgical intervention resulted in rapid stabilization, and the patient remained well at 3 months. By contrast, SSR associated with malignancy or massive splenomegaly carries a worse prognosis.3,21 Our patient had none of these risk factors, which likely contributed to her good recovery.
Conclusion
Spontaneous splenic rupture is an uncommon but potentially life-threatening cause of acute abdomen that can mimic peptic ulcer disease. Clinicians should maintain a high index of suspicion and promptly escalate imaging, starting with point-of-care ultrasound where available and proceeding to contrast CT, to identify hemoperitoneum or splenic injury when unexplained pain, anemia, or hemodynamic deterioration occur. Hemodynamically unstable patients usually require urgent surgical intervention (commonly splenectomy). Post-splenectomy vaccination and patient education are essential to reduce the risk of overwhelming post-splenectomy infection.
Footnotes
Acknowledgements
We thank the patients and their families for agreeing to give their consent to publish their clinical records.
Ethical Considerations
Institutional ethical approval was not required for a single-patient case report; written informed consent for publication was obtained from the patient.
Consent for Publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Author Contributions
Dereje G. Andargie: Conceptualization, Investigation, Data curation, Writing – Original Draft. Biruk T. Mengistie: Conceptualization, Methodology, Supervision, Writing – Review & Editing. Chernet T. Mengistie: Visualization, Writing – Review & Editing. Zelalem L. Adigeh: Data curation, Writing – Review & Editing. Bel Ami: Supervision, Project administration.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data supporting the findings of this report are available within the manuscript.
