Abstract
Uterine fibroids are the most common benign gynecologic tumors; in pregnancy, degeneration may occur but is uncommon, whereas spontaneous rupture is an exceedingly rare complication. We present the case of a 43-year-old woman, gravida 2 para 2, at 17 weeks and 6 days of gestation, who was admitted with progressive abdominal pain and distension. Imaging revealed a large degenerating intramural fibroid with hemoperitoneum and a focal contour defect, raising concern for rupture. Despite stable vital signs, the patient experienced spontaneous abortion during hospitalization. Persistent abdominal pain and imaging findings warranted exploratory laparotomy, which revealed approximately 2 L of hemoperitoneum and a ruptured, necrotic fibroid. A uterine-sparing myomectomy was performed, and histopathology confirmed infarct-type degeneration with extensive hemorrhage and necrosis. The postoperative course was uneventful, and the patient was discharged in good condition. This case underscores that fibroid rupture in pregnancy may evolve subacutely, with preserved hemodynamics, and should be considered in pregnant patients with ongoing abdominal pain and degenerating myomas. Prompt recognition and timely surgical intervention are critical for favorable outcomes and uterine preservation.
Introduction
Uterine fibroids, or leiomyomas, are benign smooth muscle tumors frequently encountered in women, with prevalence in population-based cohorts reported as high as 70% by midlife.1,2 These non-malignant growths are among the most frequently encountered gynecological tumors, with rates as high as 77% reported in reproductive-age women.3,4 Despite their high prevalence, the majority of nonpregnant women with fibroids remain asymptomatic.1,3 In symptomatic cases, fibroids may cause menorrhagia, pelvic discomfort, infertility, and other complications that can impact quality of life and reproductive outcomes.1,4 During pregnancy, the hormonal environment—particularly elevated levels of estrogen and progesterone—can lead to fibroid enlargement and a range of pregnancy-related complications.3,5 One of the better-known pregnancy-specific complications is red degeneration, a condition associated with fibroid ischemia and necrosis, often presenting as acute abdominal pain.5 -7 Fibroids are reported in approximately 10% of pregnancies, and while red degeneration is the most common complication, symptoms typically resolve with conservative management, and surgery is rarely indicated.5,8 In rare instances, fibroids may spontaneously rupture, leading to significant intra-abdominal bleeding and necessitating emergency surgical intervention.9,10 Such events have been reported in pregnant women without a prior history of uterine surgery, particularly during the second trimester or postpartum.11,12 Nevertheless, spontaneous fibroid rupture remains a rare but potentially life-threatening complication of pregnancy.9,10
Spontaneous rupture of a uterine fibroid during pregnancy is exceedingly rare, with only a handful of cases reported, particularly in women without known risk factors. Our case describes a spontaneous rupture of a large, degenerating fibroid in a patient at 17 weeks of gestation with a previously uneventful pregnancy. This report contributes to the limited literature on this life-threatening but rare condition and emphasizes the need for high clinical suspicion in similar presentations.9,11,13
Case Presentation
A 43-year-old woman, gravida 2, para 2 (previous twin pregnancy, ended at 36 weeks and 2 days; cesarean for Category III FHR tracing in labor), at 17 weeks and 6 days of gestation, presented to the emergency department with complaints of progressive abdominal pain and abdominal distension. She reported mild lower abdominal discomfort beginning approximately 1 week prior, which had intensified over the past 2 days. The pain was constant, moderate to severe in intensity, and was associated with abdominal fullness and constipation for 5 days. She had self-medicated with milk of magnesia (MoM) syrup without relief. There was no history of trauma, fever, urinary complaints, vaginal bleeding, or loss of fluid.
Her past medical history was notable for well-controlled hypothyroidism treated with levothyroxine 100 μg daily. She had a known history of uterine fibroids diagnosed prior to conception, which had not caused symptoms during her previous pregnancy or earlier in the current gestation. There was no history of assisted reproductive techniques, uterine surgery other than her previous cesarean section, or other known risk factors for uterine rupture or myoma-related complications.
Initial examination revealed a hemodynamically stable patient: blood pressure 110/70 mmHg, heart rate 88 bpm, respiratory rate 18/min, temperature 36.8°C. Routine labs were likewise reassuring: hemoglobin 8.6 g/dL, white blood cells 12.3 × 10⁹/L, and platelets 137 × 10⁹/L, with coagulation and metabolic parameters within normal limits (INR 1.0, aPTT 28 s, lactate 1.4 mmol/L, creatinine .6 mg/dL). Abdominal examination showed marked distension, diffuse tenderness, and reduced bowel sounds, but no guarding or rebound. Fetal heart rate was detected at 148 bpm via Doppler. A bedside obstetric ultrasound revealed a live fetus with cephalic presentation, an estimated fetal weight of 198 ± 31 g, normal amniotic fluid index (AFI), and posteriorly located placenta. Multiple uterine fibroids were noted, with the dominant lesion measuring approximately 150 mm × 110 mm in the right lateral uterine wall.
Review of prior imaging revealed a progressive increase in fibroid size. A sonographic examination performed 2 months earlier (at 15 weeks 5 days) showed a large subserosal and intramural hypoechoic fibroid in the uterine fundus, measuring 153 mm × 104 mm × 95 mm. Another ultrasound conducted 1 month prior (at 16 weeks 4 days) showed continued fibroid growth with dimensions of 155 mm × 110 mm × 93 mm in the uterine body and fundus. A follow-up ultrasound during this admission, on hospital day 3 (approximately 18 weeks 2 days), described a large intramural fibroid with extreme heterogeneity and cystic/necrotic areas in the right lateral uterus extending from fundus to cervix, measuring 125 mm × 85 mm × 145 mm. These findings were suggestive of red degeneration.5,7
Given the persistent pain, an abdominopelvic MRI was performed on the day of admission. It revealed a large fibroid occupying the right lateral fundal region with a 42 mm × 45 mm contour defect in its lateral aspect (Figure 1). Extensive hemoperitoneum with heterogeneous signal intensity was observed in the peritoneal cavity, consistent with peripheral hematoma formation. These findings raised concern for fibroid rupture. 7
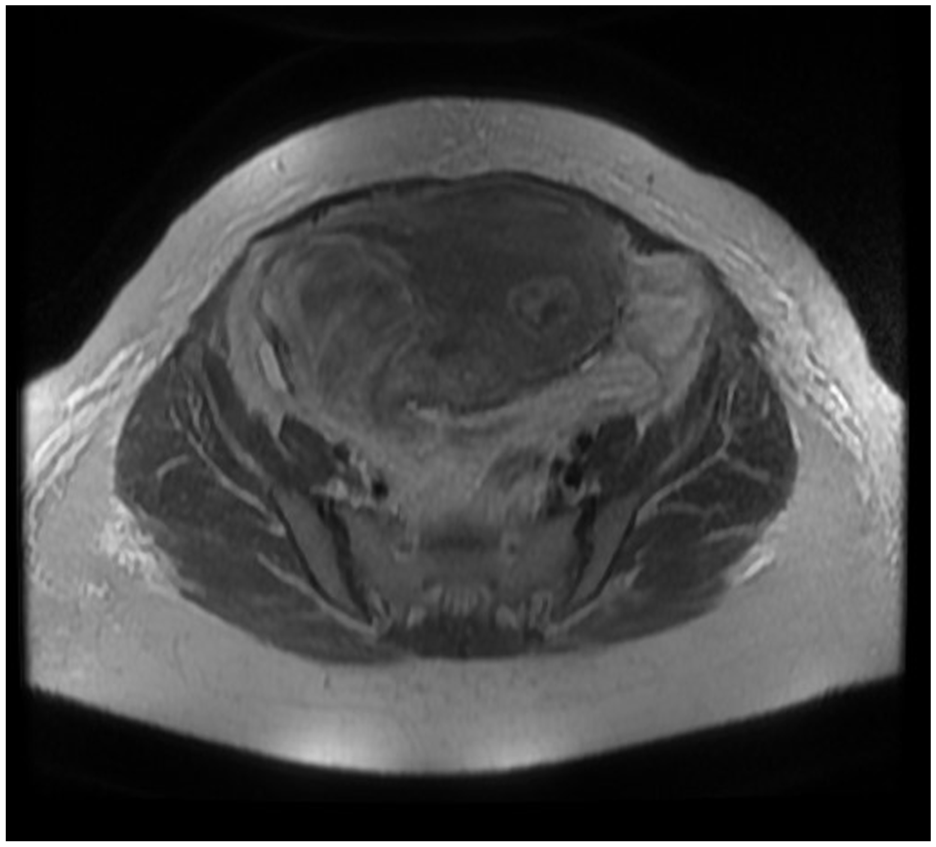
A large fibroid occupying the right lateral fundal region with a 42 mm × 45 mm contour defect in its lateral aspect.
During admission, the patient’s abdominal pain persisted and gradually worsened, though she did not report any significant change in severity to the clinical staff. On hospital day three, she experienced lower abdominal cramping and vaginal spotting, followed by spontaneous abortion (pregnancy loss) due to preterm labor. The fetus was expelled; however, due to incomplete expulsion of placental tissue, she underwent uterine evacuation via curettage with forceps. Notably, her abdominal discomfort became temporarily milder following the pregnancy loss. She remained clinically stable throughout, with no signs of excessive bleeding or infection.
On hospital day 4, due to persistent abdominal pain even after pregnancy loss, the patient underwent emergency laparotomy. Although she remained hemodynamically stable without any deterioration in vital signs, surgical intervention was deemed necessary based on clinical findings and imaging. In the 12 hours preceding surgery, she was afebrile (36.6°C-36.9°C) with blood pressure 110-118/68-76 mmHg, pulse 88-96 bpm, respiratory rate 16 to 20/min, and oxygen saturation 98% to 100% on room air; bowel sounds were normoactive, she had passed flatus, and she reported a bowel movement earlier that day. Intraoperatively, approximately 2 L of gelatinous hemoperitoneum mixed with blood and necrotic debris were noted (Figure 2). A large ruptured intramural fibroid with areas of necrosis and cystic degeneration was found protruding from the right lateral uterine wall (Figure 3). A myomectomy was performed. Inspection of the uterus confirmed an intact serosal surface elsewhere with no evidence of uterine rupture. No additional clinically significant fibroids requiring intervention were identified, and no adnexal pathology was seen. Hemostasis was achieved.

Gelatinous hemoperitoneum mixed with blood and necrotic debris.

Ruptured intramural fibroid with areas of necrosis and cystic degeneration.
Histopathological evaluation of the excised fibroid confirmed an infarct-type degeneration with widespread hemorrhage and necrosis. Inflammatory infiltration and hyaline degeneration were also present, consistent with a degenerating leiomyoma. 4 Additionally, pathology from the uterine contents following intra-admission pregnancy loss confirmed chorionic villi consistent with retained products of conception, without evidence of gestational trophoblastic disease (Figures 4 and 5).

Intra-abdominal specimen submitted for frozen section and permanent slides, demonstrating leiomyoma with red degeneration, extensive ischemic necrosis, and associated inflammation.

Histopathological section of uterine leiomyoma showing red (carneous) degeneration with extensive ischemic necrosis and associated inflammatory infiltrates (H&E stain, ×100).
Following surgery, the patient was transferred to the recovery unit in stable condition and later to the obstetric ward. Her postoperative course was uneventful. She remained afebrile and hemodynamically stable, with no signs of infection or excessive bleeding. Postoperatively, serial labs remained stable: on postoperative day 1 hemoglobin was 8.9 g/dL, white blood cells 9.7 × 10⁹/L, and platelets 161 × 10⁹/L; no transfusion was required. Postoperative management included intravenous fluids and analgesics. Vital signs remained within normal range throughout hospitalization. The patient reported improvement in abdominal pain and tolerated oral intake without difficulty. She was discharged in good general condition on the third postoperative day, with instructions for routine follow-up and postoperative care.
Discussion
Spontaneous rupture of a degenerating uterine fibroid in pregnancy is rare but clinically important. Whereas many published cases highlight hyperacute, exsanguinating hemorrhage, our mid–second-trimester case combined progressive abdominal pain, MRI evidence of a focal contour defect with hemoperitoneum, spontaneous abortion prior to surgery, and intraoperative confirmation of a ruptured intramural leiomyoma with gelatinous intraperitoneal blood and necrotic debris—yet the patient remained hemodynamically stable. This phenotype emphasizes that rupture may evolve subacutely from degenerative processes rather than present solely as hyperacute hemorrhage.9,10,12
In pregnant patients with acute abdomen, key differentials include uterine rupture, placental abruption, adnexal torsion or cyst rupture, bowel obstruction/ileus, and fibroid complications (degeneration, capsular disruption, or rupture).5,6,14 -18 Whereas true uterine rupture typically coincides with maternal instability and a gross serosal breach, serial ultrasound in our patient demonstrated an enlarging, markedly heterogeneous intramural lesion with cystic/necrotic areas; MRI showed a focal contour defect and hemoperitoneum. These findings, coupled with preserved maternal vitals and subsequent operative inspection of an intact uterine serosa apart from the fibroid defect, supported fibroid rupture over true uterine rupture. As others report for unstable presentations, point-of-care ultrasound can rapidly detect free fluid, while in stable patients—such as ours—MRI refines characterization of degeneration and suspected focal rupture.5,7,11
Pregnancy-related hormonal milieu and relative ischemia predispose leiomyomas to infarct (red) degeneration with intra-lesional hemorrhage and necrosis. Whereas many reports emphasize brisk arterial/venous bleeding with shock, progressive degeneration can weaken the capsule, permitting seepage of blood and necrotic material into the peritoneal cavity—clinically producing severe pain and peritoneal irritation even without shock. Histopathology in this case (infarct-type degeneration with hemorrhage/necrosis and hyaline change) mirrors this mechanism and aligns with the gelatinous hemoperitoneum observed intraoperatively. Elevated inflammatory/tissue-turnover markers (eg, LDH; CA-125) can be compatible with active degeneration but are nonspecific in pregnancy.1,4,5,7
Whereas most published pregnancy-associated ruptures present intrapartum or postpartum with rapid hemodynamic compromise due to capsular or overlying vessel disruption—prompting emergent laparotomy9,10,12—our case occurred in the second trimester, with hemodynamic stability, MRI depiction of a focal defect, and partially organized/gelatinous hemoperitoneum. These features are more consistent with leakage from a necrotic intramural lesion than with acute vascular avulsion. Similarly, whereas rupture is often discovered intrapartum at cesarean or managed urgently postpartum, our sequence—pregnancy loss followed by persistent pain leading to uterine-sparing myomectomy—differs in timing and operative pathway.11,13
In unstable patients or when active hemorrhage is suspected, immediate laparotomy is indicated. By contrast, in stable presentations—like ours—careful observation with serial examinations and targeted imaging can clarify trajectory. Our patient underwent uterine evacuation for incomplete pregnancy loss yet continued to experience abdominal pain with imaging concern for ongoing peritoneal irritation from a ruptured degenerating fibroid; therefore, laparotomy and myomectomy were appropriate despite stable vitals. While conservative measures are sometimes reported for degenerated fibroids in pregnancy, suspected rupture with persistent symptoms generally favors surgery in order to control the peritoneal process and preserve the uterus when feasible.4,5,9
Following myomectomy, recovery was uneventful. Whereas outcomes in the literature range from uncomplicated recoveries to significant morbidity, case-based experiences suggest favorable maternal courses when recognition and operative management are timely; in keeping with that, our patient’s course was benign. Given uterine preservation (and prior cesarean), future pregnancy counseling should address individualized surveillance of the myomectomy bed, prompt evaluation of new abdominal pain, and delivery planning based on obstetric history.5,13
Strengths of this report include concordance across modalities—ultrasound and MRI pointing to degeneration with a focal defect, operative confirmation of an intramural rupture with gelatinous hemoperitoneum, and histologic proof of infarct-type degeneration. A limitation is unavoidable imprecision in timing of pathophysiologic transitions around the pregnancy loss event. Practically, as opposed to the assumption that stability rules out major intra-abdominal bleeding, clinicians should recognize that hemodynamic stability does not exclude clinically meaningful fibroid rupture in pregnancy; persistent or escalating pain with supportive imaging warrants timely surgical consideration, with uterine-sparing intent when appropriate.1,5
Conclusion
Second-trimester rupture of a degenerating intramural leiomyoma can evolve subacutely with organized, gelatinous hemoperitoneum and preserved hemodynamics. In pregnant patients with persistent abdominal pain and imaging features of myoma degeneration with a focal defect or hemoperitoneum, clinicians should maintain a high index of suspicion for fibroid rupture—even when uterine rupture is unlikely and even after pregnancy loss. When symptoms persist despite conservative measures, timely laparotomy with uterine-sparing myomectomy can definitively control the peritoneal process and preserve future reproductive potential. Prioritizing clinical trajectory over vital-sign stability may prevent delays in intervention and improve maternal outcomes.
Footnotes
Acknowledgements
The authors express their gratitude to Mazandaran University of Medical Sciences for its valuable support.
Consent for Publication
The patient provided written informed consent for both treatment and publication.
Author Contribution
BF contributed to the conception of the work, collection of clinical data, and drafting of the manuscript. KS was responsible for anesthetic management of the patient, interpretation of perioperative findings, and critical revision of the manuscript for important intellectual content. NM assisted in the acquisition of patient information, literature review, and manuscript preparation. AEJ contributed to the review of relevant literature, data analysis, and editing of the final draft. ME supervised the project, coordinated the writing process, and finalized the manuscript as the corresponding author. All authors read and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data used in this study can be obtained from the corresponding author upon reasonable request.
