Abstract
Introduction:
Malignant hyperthermia (MH) is a fatal hypermetabolic reaction of skeletal muscle, triggered by exposure to volatile anesthetic agents or depolarizing muscle relaxants. It typically exhibits hypercarbia, muscle rigidity, tachycardia, and hyperthermia. Diagnosis is often confirmed through a muscle biopsy for the in vitro contracture test or by identifying pathogenic genetic variants.
Case Presentation:
We report 2 cases of suspected MH. The first case involved a 4-year-old female (20 kg) undergoing adenotonsillectomy, and the second involved a 13-year-old female (56 kg) who underwent pedicle screw fixation surgery. Both patients had unremarkable medical histories. During maintenance of general anesthesia with sevoflurane, they developed clinical signs highly suggestive of MH—10 minutes after exposure in the first case and 120 minutes after exposure in the second case. Both cases were managed with dantrolene and supportive care. In the first case, dantrolene was administered 4 hours after the initial signs, by which time significant rhabdomyolysis had already developed. In the second case, early administration within 10 minutes was associated with a much milder degree of rhabdomyolysis.
Conclusion:
Early recognition of symptoms and accurate differentiation of MH from similar conditions are essential for favorable outcomes. Prompt administration of dantrolene at the first sign of an MH reaction is critical for effective management.
Introduction
Malignant hyperthermia (MH) is a disorder marked by excessive metabolic activity in skeletal muscle, usually provoked by volatile anesthetics or depolarizing muscle relaxants, and occasionally triggered by extreme physical exertion or heat. 1 Common clinical manifestations include elevated end-tidal CO₂ (hypercarbia), generalized muscle rigidity, tachycardia, and an abnormal rise in body temperature (hyperthermia). The pathophysiology of MH involves a disruption in excitation-contraction coupling, leading to increased calcium ions (Ca²⁺) mobilized from the sarcoplasmic reticulum via the ryanodine receptor 1 (RyR1) channel in skeletal muscle upon exposure to triggering agents. 1 MH is estimated to occur in about 1 in 10 000 to 1 in 250 000 anesthetic procedures. 2 MH reactions are more commonly observed in males than females, with the highest rates seen in younger populations.
Genetic variants in RYR1, CACNA1S, and STAC3 are implicated in MH susceptibility, with most cases involving RYR1 variants encoding the ryanodine receptor—RyR1. A smaller proportion of families susceptible to MH carry a variant in the CACNA1S gene, which encodes the α-1S component of the voltage-gated Ca²⁺ channel Cav1.1 in the T-tubules, commonly referred to as the dihydropyridine receptor. 3
In fulminant MH reactions, muscle cell breakdown (rhabdomyolysis) leads to hyperkalemia and acidosis, which can cause life-threatening arrhythmias and even cardiac arrest in the early stages. As the condition progresses, elevated serum creatine kinase (CK) levels and myoglobinuria may result in acute renal failure. Other complications, including central nervous system impairment, pulmonary edema, and disseminated intravascular coagulation, may also develop. Alongside supportive treatments to address these critical disturbances, preventing further muscle damage from sustained contractions is essential for reducing long-term complications. 4
Dantrolene binds to the RYR1 receptor on the sarcoplasmic reticulum in muscle cells, preventing the uncontrolled release of calcium ions into the cytoplasm and thereby reducing the risk of sustained muscle contractions in MH. 5 It has become a key component in the treatment of MH, with MH-related mortality having decreased from approximately 80% to less than 5% since the introduction of dantrolene.6,7
Confirmatory testing for MH typically involves a muscle biopsy for contracture tests or genetic study. 8 However, these tests are often performed only after an MH event has occurred and may be difficult to access in developing countries. Recognizing the initial clinical signs of MH and administering dantrolene promptly are top priorities for managing an MH crisis.6,8 Unfortunately, access to dantrolene remains limited in our country, which may delay its administration when MH occurs. Such delays can potentially compromise patient outcomes.
We report 2 suspected MH cases without confirmatory diagnostic testing. Both were successfully managed with dantrolene, administered at different times following MH onset, and exhibited varying degrees of rhabdomyolysis and other complications at the same medical center.
The patients’ parents provided Written informed consent to publish details of the patients’ medical history.
Case Presentation 1
A 4-year-old female (20 kg) was scheduled for adenotonsillectomy to treat hypertrophic tonsillitis and adenoiditis under general anesthesia. A pre-anesthetic assessment revealed normal findings, with no allergies, no significant social history, no muscle disorders, and a family history unclear regarding anesthesia complications.
Pre-anesthetic vital signs included a heart rate (HR) of 104 bpm, blood pressure (BP) of 95/55 mmHg, and a forehead temperature of 36.7°C (measured with an infrared thermometer). Induction of general anesthesia was performed with propofol (4 mg/kg, 80 mg), fentanyl (2 µg/kg, 40 µg), and rocuronium (0.6 mg/kg, 12 mg). After intubation, anesthesia was maintained with sevoflurane at an inspiratory concentration of 2%. Intraoperatively, ventilation was managed using Pressure Regulated Volume Control mode, with 160 mL tidal volume (TV), 16 bpm respiratory rate (RR), 1:2 inspiratory-to-expiratory (I:E) ratio, and 50% FiO₂. HR ranged from 90 to 120 bpm, and BP ranged from 85/45 to 110/60 mmHg. The initial rectal temperature (measured via probe sensor) was 37°C, and end-tidal carbon dioxide (EtCO₂) levels were 34 to 36 mmHg.
After 10 minutes of anesthesia, EtCO₂ levels rose above 55 mmHg. Following confirmation of no issues with the breathing system, ventilation was assisted (RR: 22 bpm, TV: 200 mL) to maintain EtCO₂ within the normal range. Despite these efforts, EtCO₂ reached 101 mmHg, accompanied by a rapid temperature increase (from 37.5°C to a peak of 39.9°C within 5 minutes) and progressive tachycardia with a HR of 150 to 180 bpm, and a BP of 145/90 mmHg Masseter muscle rigidity with restricted mouth opening and generalized tonic rigidity with limb extension were observed. Laboratory test revealed pH 7.20, BE -14.7 mEq/L, pCO₂ 68 mmHg, pO₂ 387 mmHg, K+ 4.96 mmol/L, CK (creatine kinase) 478 U/L. Suspecting MH, an emergency was declared, and dantrolene was requested from another center.
Sevoflurane was discontinued, and the patient was switched to propofol for maintenance anesthesia. An ice pack was applied, and cold normal saline was administered intravenously. The bladder and stomach were irrigated with cold saline via a Foley catheter and GI tube. Acetaminophen (15 mg/kg) and methylprednisolone (1 mg/kg) were given. Acidosis was corrected with bicarbonate, hyperventilation, and diuresis. K+ and hemodynamics were closely monitored. After surgery, the patient was moved to the ICU while sedated and intubated.
Four hours after the first signs of MH, an initial intravenous dose of dantrolene (Revonto™; US WorldMeds, Italy) 2.5 mg/kg (50 mg) was administered. Within 30 minutes, her temperature dropped to 37.8°C, and EtCO₂ was 35 mmHg. Dantrolene was continued at 1 mg/kg every 6 hours for 2 days. On postoperative day 2, the patient was extubated with stable hemodynamic and respiratory status. Lab values and symptoms gradually resolved, with a peak CK level of 10 458 UI/L (Figure 1) and GOT/GPT levels of 430/103 UI/L (Table 1). She was transferred to the postoperative ward and discharged home after 7 days, once CK levels had normalized.

Changes in potassium (K⁺) and creatine kinase (CK) levels over time after the onset of malignant hyperthermia (MH).
Laboratory tests after presenting malignant hyperthermia in 2 cases.
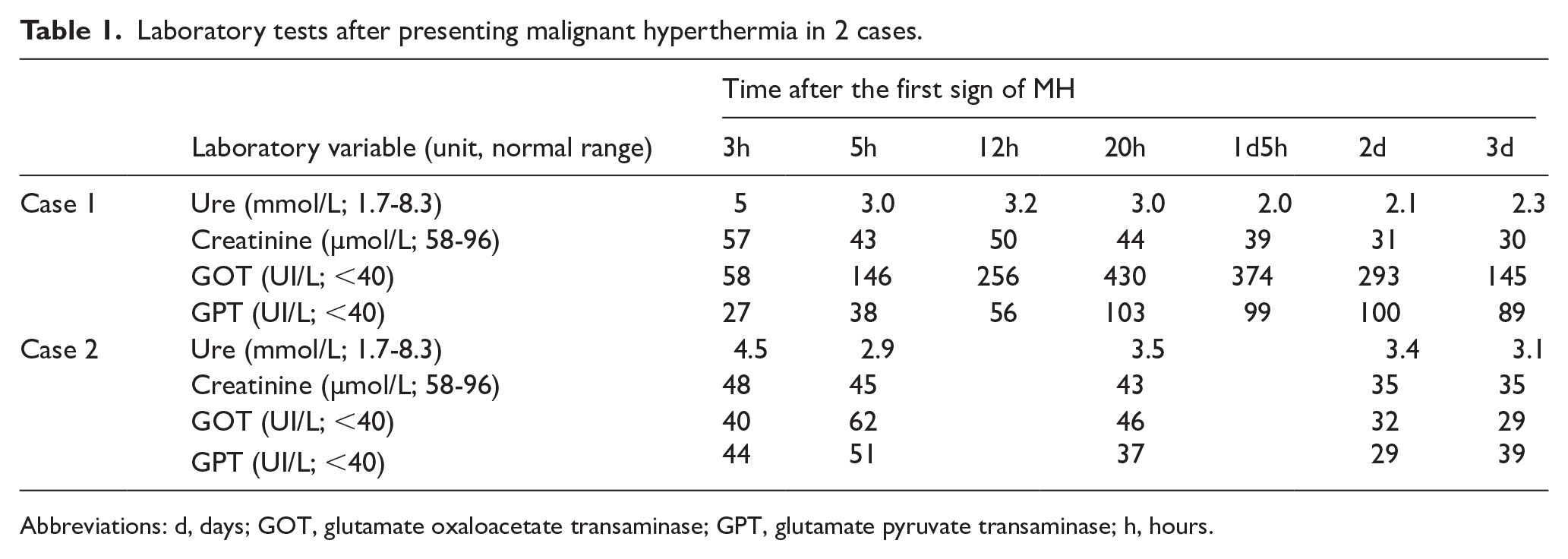
Abbreviations: d, days; GOT, glutamate oxaloacetate transaminase; GPT, glutamate pyruvate transaminase; h, hours.
Her total clinical MH raw score was 74, corresponding to an MH rank of 6 with an “almost certain” likelihood.
Genetic analysis of the RYR1 gene identified a heterozygous variant in exon 87 with a C-to-T nucleotide substitution at position 11 947 (c.11947C>T), resulting in a missense variant that leads to an amino acid change from arginine to cysteine (p.R3983C). This variant was present in the patient but absent in her parents and other relatives (Figure 2). No other relevant variants were identified. According to ClinVar (ID: 650932), this variant is classified as a variant of uncertain significance (VUS), with one submission reporting it as pathogenic, one as likely pathogenic, and one as of uncertain significance. It is not reported in large population databases such as gnomAD, suggesting it is a rare variant.

Sanger sequencing results. Electropherogram showing Sanger sequencing of exon 87 in the RYR1 gene. The nucleotide at position 11947 shows a C-to-T substitution (c.11947C>T), indicating a heterozygous missense variant resulting in an amino acid change from arginine to cysteine (p.R3983C). This variant was identified in the patient but was absent in her parents and other relatives. According to ClinVar (ID: 650932), this variant is classified as a variant of uncertain significance, with one report as pathogenic, one as likely pathogenic, and one as uncertain significance.
Case Presentation 2
A 13-year-old female patient (56 kg, 140 cm) was admitted for pedicle screw fixation surgery to correct congenital scoliosis. Her pre-anesthetic assessment showed no abnormalities, and her family history of MH was unclear.
The anesthesia procedure (maintained with sevoflurane) and ventilator settings (adjusted for body weight) were similar to the previous case, with an initial EtCO₂ range of 32 to 36 mmHg. The surgery and anesthesia proceeded without any incidents.
After 120 minutes of anesthesia, the patient’s heart rate was 140 bpm, blood pressure 145/70 mmHg, EtCO₂ 60 mmHg, and body temperature 37.6°C, showing an upward trend. Ten minutes later, the heart rate increased to 160 bpm, blood pressure to 160/90 mmHg, EtCO₂ to 94 mmHg, and body temperature rose to 40.1°C. Generalized tonic rigidity with limb extension was also observed. Arterial blood gas showed respiratory acidosis (pH 7.13, BE -16.9 mEq/L, pCO₂ 61 mmHg, pO₂ 236 mmHg, Hct 37%). MH was suspected.
Fortunately, after the previous case, the remaining dantrolene was still stored at our hospital. Therefore, in this case, dantrolene was utilized about 10 minutes after the first signs of MH, with an initial dose of 2.5 mg/kg, along with the same supportive interventions as in the previous case. The patient was moved to the ICU, where the condition gradually improved. Dantrolene was repeated at 1 mg/kg every 6 hours for the next 12 hours. The peak CK level was 1417 UI/L (Figure 1), with no subsequent abnormalities in renal or liver function (Table 1). She was transferred to the general ward in stable condition and discharged home after 10 days.
Her total clinical MH raw score was 64, corresponding to an MH rank of 6 with an “almost certain” likelihood. Genetic testing was not performed for her or her family members due to undisclosed reasons.
In both cases, a follow-up visit was conducted 3 weeks post-discharge. The patient had made a full recovery with no residual muscle weakness or renal dysfunction. He was counseled extensively on avoidance of triggering agents such as volatile anesthetics and succinylcholine. Written documentation was provided for inclusion in his medical records. The patient was advised to wear a medical alert bracelet indicating MH susceptibility and to notify all future healthcare providers.
In both cases, a follow-up visit was conducted 3 weeks after discharge. The patients had made a full recovery, with no residual muscle weakness or renal dysfunction. They were extensively counseled on the avoidance of triggering agents, such as volatile anesthetics and succinylcholine. Written documentation was provided for inclusion in their medical records. The patients were also advised to wear medical alert bracelets indicating MH susceptibility and to inform all future healthcare providers of their condition.
Discussion
Malignant hyperthermia is a potentially fatal condition associated with a hypermetabolic reaction, which can benefit from early treatment with dantrolene. The timing of the first dantrolene dose after initial MH signs is critical to patient outcomes. Riazi et al reported that each 10-minute delay in giving dantrolene significantly raises the risk of complications, with a delay of more than 50 minutes resulting in a 100% complication rate. 9 In Case 1, dantrolene was administered 4 hours after the initial signs of MH due to its unavailability at our institution, by which time the patient’s CK level had peaked at 10 485 U/L. In contrast, in Case 2, dantrolene was administered within 10 minutes of MH onset, and the peak CK level was significantly lower at 1417 U/L. Notably, despite the higher potential for muscle injury associated with pedicle screw fixation in Case 2 compared to adenotonsillectomy in Case 1, the treatment delay in Case 1 resulted in more severe muscle damage. Elevated liver enzymes were also observed in Case 1, whereas Case 2 showed no such elevation, even though both cases received similar supportive measures. Fortunately, neither case resulted in long-term, life-threatening complications. Additionally, studies show that CK levels in MH patients treated with dantrolene are significantly lower than in those untreated. 10 Thus, administering dantrolene at the earliest sign of MH is essential to limit muscle damage and prevent further complications.
In Case 1, the CK level peaked on the second day following the MH episode, which may reflect either recrudescence or delayed rhabdomyolysis. Recrudescence has been reported in up to 25% of cases, with risk factors including muscular build, MH clinical score ⩾35, significant temperature rise, and delayed onset after induction (>150 minutes). 11 However, in this case, MH symptoms appeared within 10 minutes of induction, with no subsequent signs of recurrence such as rising temperature, tachycardia, hypercarbia, or muscle rigidity. Dantrolene continued for the next 2 days. Therefore, the delayed CK peak is more likely due to delayed rhabdomyolysis or subclinical muscle injury rather than recrudescence.
The product data sheet for dantrolene recommends an initial dose of 1 mg/kg with a maximum cumulative dose of 10 mg/kg. However, recent Western guidelines and reviews suggest a higher initial dose of 2.5 mg/kg with no cumulative ceiling, titrated based on tachycardia and hypercarbia.6,12 This recommendation is based on the average dose (2.4 mg/kg) required to achieve a plasma concentration sufficient to suppress twitch responses in in vitro skeletal muscle preparations. 13 Although pharmacokinetic differences in drug metabolism between Western and Asian populations have been suggested—particularly involving cytochrome P450 enzymes such as CYP3A4, with Asian individuals potentially exhibiting lower CYP3A4 activity compared to Western populations, 14 resulting in slower metabolism, longer half-life, and higher plasma concentrations of dantrolene—there is currently limited specific evidence or guidance available for adjusting dantrolene dosing in Asian populations. Dantrolene maintenance dosing for post-reaction management varies among guidelines.6,8,12,15 The Malignant Hyperthermia Association of the United States (MHAUS) suggests administering bolus doses of 1 mg/kg every 4 to 6 hours until hypermetabolism resolves and clinical signs of MH are controlled, with stabilization of the cardiac and respiratory systems. 15 In contrast, the European Malignant Hyperthermia Group (EMHG) recommends a 2.5 mg/kg bolus only if there is recrudescence of the MH crisis. 12 This discrepancy may reflect concerns regarding the potential side effects of dantrolene. 16 In both cases, we used an initial dose of 2.5 mg/kg and repeated 1 mg/kg every 6 hours until clinical signs stabilized. In Case 1, symptoms gradually improved after 48 hours of dantrolene use, while in Case 2, symptoms improved more rapidly, requiring only 12 hours of treatment. To the best of our knowledge, no reports have yet explored the relationship between the timing of the first dose and the duration of subsequent doses of dantrolene. However, early administration of dantrolene is known to reduce the incidence of complications and may help minimize the total amount of the drug needed.
In addition to the prompt administration of dantrolene, supportive treatment for malignant hyperthermia includes discontinuation of triggering agents, active cooling, correction of metabolic acidosis and hyperkalemia, management of arrhythmias, and maintenance of adequate urine output to prevent renal injury—all of which are crucial for managing this life-threatening condition. These measures were applied in both cases in accordance with current clinical guidelines.4,6,8 Although corticosteroids are not included in standard treatment protocols, intravenous methylprednisolone was administered as adjunctive therapy in these cases, based on the hypothesis that it might help mitigate systemic inflammation and potential airway edema following the hypermetabolic crisis.
The “gold standard” for diagnosing MH is currently the in vitro contracture test, which evaluates muscle fiber contraction in response to halothane or caffeine. Genetic testing for MH is increasingly considered a viable primary diagnostic approach, although a negative result does not exclude susceptibility and may still require confirmation through biopsy-based tests. 17 Clinically, treatment should not be postponed until MH is fully diagnosed. Therefore, MH is often recognized using the Clinical Grading Scale, 18 which is widely utilized to evaluate the severity of adverse anesthetic reactions. In both cases, the patients were ranked as “very likely MH” (muscular rigidity, respiratory acidosis, rapid increase in temperature, inappropriate sinus tachycardia) at the time of onset, and “almost certain” after evidence of muscle breakdown (elevated CK levels) was obtained, which required prompt treatment.
Although the R3983C mutation in the RYR1 gene is classified as a variant of uncertain significance for MH according to the American College of Medical Genetics and Genomics and the Association for Molecular Pathology (ACMG/AMP), some studies have reported this mutation as pathogenic in RYR1-related disorders,19,20 including stress-induced malignant hyperthermia. 20 Furthermore, approximately half of the individuals susceptible to MH lack pathogenic variants in the currently identified MH-related genes. 3 Thus, even with negative DNA test results, a predisposition to MH cannot be entirely ruled out. 21 Unfortunately, muscle biopsy testing was not conducted, so the MH diagnosis in these cases was based on clinical presentation. Additionally, the variant was identified only in the patient and was absent in her parents and other relatives. While de novo variants may occur, confirmation of biological parentage is necessary to establish this with certainty.
The differential diagnosis should also consider conditions such as thyrotoxicosis, pheochromocytoma, neuroleptic malignant syndrome, and serotonin syndrome. Thyroid storm was excluded due to normal preoperative thyroid function tests. Pheochromocytoma crisis shares symptoms with MH, such as hypertension and tachycardia, but does not involve muscle rigidity. Neuroleptic malignant syndrome, associated with dopamine antagonists, 22 typically responds to non-depolarizing muscle relaxants; however, in this case, rigidity persisted despite rocuronium use, and no antipsychotics were prescribed. Serotonin syndrome, triggered by drugs like selective serotonin reuptake inhibitors or monoamine oxidase inhibitors, 23 was also ruled out due to an absence of relevant medication history. While diagnosing these severe MH episodes can be challenging, dantrolene may temporarily alleviate critical symptoms caused by other conditions, 21 providing time for an accurate diagnosis.
In Vietnam, there are currently no available data on the incidence or mortality rates of MH. While genetic testing for MH susceptibility is technically available, in vitro contracture testing is not, limiting the ability to confirm diagnoses. In many developing countries, the true incidence of MH remains unclear due to underreporting, limited awareness, and restricted access to diagnostic resources. Although global estimates suggest an incidence ranging from 1 in 10 000 to 1 in 250 000 anesthetic procedures, 2 the actual rate may be higher in low-resource settings. Dantrolene should be stocked in at least one major medical center per region and made accessible to surrounding facilities in emergencies. Establishing national MH registries could further improve timely access to dantrolene and enhance patient outcomes. In Vietnam, access to dantrolene remains limited; however, regulatory revisions are currently underway to allow its official importation and distribution, which is expected to improve availability for suspected MH cases.
Conclusion
Although rare, MH requires prompt recognition, rapid initiation of treatment, and accurate differentiation from other conditions for effective management. Dantrolene should be administered without delay at the earliest suspicion of an acute MH episode, alongside supportive measures to address hyperkalemia, acidosis, and hemodynamic instability.
Footnotes
Acknowledgements
We thank the staff at St. Paul’s Hospital for their support with sample collection and assistance during the study, as well as Assoc. Prof. Le Dinh Tuan for revising the manuscript.
Ethical Considerations
This study was conducted in accordance with the Declaration of Helsinki.
Consent to Participate
The patients’ parents provided informed consent to publish details of the patients’ medical history.
Consent for Publication
The patient’s parents provided informed consent for publication of the medical details.
Author Contributions
Concept and study design: Nguyen Trung Kien, Tran Nguyen Nhat, Nguyen Dang Thu. Data acquisition: Tran Nguyen Nhat, Do Dinh Tung, Tran Quang Hai, Le Thi Nguyet. Data analysis and interpretation: Nguyen Trung Kien, Tran Nguyen Nhat, Nguyen Dang Thu. Writing—original draft: Nguyen Dang Thu, Nguyen Trung Kien, Tran Nguyen Nhat. Writing, review, and editing: Nguyen Trung Kien, Tran Nguyen Nhat, Do Dinh Tung, Tran Quang Hai, Le Thi Nguyet, Pham Quang Minh, Luu Quang Thuy, Cong Quyet Thang, Ngo Van Dinh, Vu The Anh, Nguyen Huu Tu, Nguyen Dang Thu. All contributing authors are anesthesiologists and have given their approval to the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The corresponding author can provide access to the data used in this case report upon reasonable request.
