Abstract
Malignant hyperthermia (MH) is an uncommon, autosomal dominant disorder of skeletal muscle, triggered by inhalational anaesthetics or depolarizing muscle relaxants. Masseter muscle rigidity (MMR) can be regarded as potentially a preceding sign for an MH reaction. Susceptibility to MH can be determined by the in vitro contracture test (IVCT) or DNA analysis where a familial variant is known. Our aims were to review patients with MMR, where IVCT and DNA analysis had been undertaken, to determine if DNA analysis could be used as an initial screening tool for MH susceptibility, and, by reviewing standard monitored variables (SMVs), to determine if any clinical characteristics could be used to differentiate between MMR patients who are MH susceptible (MHS) and those who are not. Patients with MMR were identified from the Palmerston North Hospital MH Reactions Database. IVCT and DNA analysis results were documented. DNA testing was performed retrospectively in the majority of patients as many patients had presented before DNA analysis was available. Forty-one patients were analysed. Fourteen were DNA positive/IVCT positive and six DNA positive only (48% in total), seven were IVCT positive/DNA negative and 14 were IVCT normal. Increased creatine kinase (>18,000 units/L) was consistent with MH susceptibility. Severity of MMR was not linked to MH susceptibility. This study confirmed that DNA analysis can be used as a first-line test for MH susceptibility in patients presenting with MMR (consistent with European MH Group recommendations). Creatine kinase was the only SMV that was significantly different between MHS and MH normal individuals.
Introduction
Malignant hyperthermia (MH) is an uncommon autosomal dominant pharmacogenetic disorder of skeletal muscle, triggered by inhalational agents and depolarizing muscle relaxants.1 Characterized by an excessive release of calcium in skeletal muscle, MH is the result of a mutated ryanodine receptor gene in the majority of patients.2,3 Clinical symptoms include hypercarbia, tachycardia, rapidly increasing hyperpyrexia and muscle rigidity.4
Masseter muscle rigidity (MMR) is an indicator of MH susceptibility.5–8 Defined as transient inability or extreme difficulty in distracting the mandible from the maxilla,9 MMR poses a significant risk to patients, as airway management becomes compromised.10 MMR is transient, may be accompanied by other signs of MH susceptibility and can occur following a depolarizing muscle relaxant, e.g. suxamethonium,11,12 or other drugs.13–16 Therefore, MMR should be considered a heterogeneous condition and, as such, not all MMR may indicate MH susceptibility.
In vitro contracture testing (IVCT) or the caffeine-halothane contracture test (CHCT) are regarded as the gold standard of MH susceptibility testing, but DNA analysis can also be used.1,17 The linkage of MMR with other signs of MH, IVCT and DNA analysis is important, as contracture testing may give false positive test results in patients with other muscle disorders.18 On the other hand, a positive DNA result, where the variant has been classified as pathogenic for MH, will confirm MH susceptibility as the cause of MMR.
A variant diagnostic for MH must co-segregate with MH susceptibility, be absent in the general population, be conserved across species, have chemical properties consistent with the pathophysiology of MH and demonstrate abnormal Ca2+ release from intracellular stores using functional analysis.1
The aims of this study were twofold. Firstly, to review patients referred for investigation with MMR, where IVCT and DNA analysis had been undertaken, to determine the utility of DNA testing. Secondly, to analyse standard monitored variables (SMVs) to determine if any characteristics associated with MMR make the diagnosis of MH susceptibility more likely.
Materials and methods
Ethical approvals
Ethical approval was obtained from the MidCentral Health Clinical Board, research register number 2016.04.003. DNA analysis approval was obtained from the Central Region Human Ethics committee, Ministry of Health, Wellington code MWH 03/04/018.
Establishing MH susceptibility status
Palmerston North is the only referral testing centre for suspected cases of MH in New Zealand. A database, established in 1991, of all known New Zealand MH reactions is held by the Palmerston North Malignant Hyperthermia Unit. Patients who are referred for testing are entered into this database and their MH susceptibility status documented, wherever possible. The initial presenting sign is also indicated and therefore the database can be searched to identify patients referred due to MMR. Clinical records of these patients were then manually reviewed. All patients with MMR were referred as a suspected MH reaction.
Patients were tested for MH susceptibility by IVCT according to the European MH Group (EMHG) protocol.1 Patients are defined as either MH susceptible (MHS, specifically MHShc), MH negative (MHN), or MHS to either halothane (MHSh) or caffeine (MHSc) alone. MHShc, MHSh and MHSc patients are all regarded as clinically susceptible to MH.1
DNA methods
DNA was extracted from whole blood using the Promega Wizard™ (Promega Corp., Madison, WI, USA) genomic DNA purification kit according to the manufacturer’s instructions. Familial variants were identified by kinetic polymerase chain reaction amplification of RYR1 (the gene most commonly associated with MH susceptibility3) sequences from genomic DNA using high-resolution amplicon melting for allele discrimination.19
Genomic DNA for patients where a familial RYR1 variant had not been identified was subjected to either targeted exon capture and next-generation sequencing as previously described,20 or paired-end exome sequencing. The exome sequencing was carried out by Macrogen, Seoul, South Korea. The libraries were prepared using SureSelectXT™ v6 with the TruSeq SBS kit v3 (Illumina) and analysed on an Illumina HiSeq 4000 sequencer with HCS v3.3 sequencing control software. The average coverage of raw data was 150× and the minimum coverage of each exon was 20×. The FastQ files were analysed at Massey University, Palmerston North, New Zealand using SureCall™ v4.0 (Agilent) software. All exons for RYR1, CACNA1S21 and STAC322,23 (the genes definitively associated with MH) were manually curated for coverage. Exons with less than the minimum coverage were amplified by polymerase chain reaction (PCR) and analysed by Sanger DNA sequencing with Big Dye v3 on an ABI 3730 instrument, which was carried out by the Massey Genome Service. Where DNA from a proband was not available, DNA from a sibling was used instead.
Standard monitored variables
Data obtained from patient records included age at event, gender, patient management following MMR, triggering agent, type of surgery and SMVs, i.e. blood pressure, maximal end-tidal carbon dioxide (ETCO2), maximum temperature, maximum heart rate and minimum peripheral capillary oxygen saturation (SpO2) measurement. Creatine kinase (CK), base deficit and serum potassium values were also recorded where available. The use of dantrolene and patient outcomes were also documented.
An attempt was made to grade the severity of MMR using information from the patient record. Grade 1 indicated patients in which direct laryngoscopy allowed an endotracheal tube to be placed; grade 2, placement of a supraglottic airway device only; and grade 3, unable to intubate or pass a supraglottic airway device.
Statistical analysis
Statistical analysis was performed using Minitab 17 Statistical Software (2010) (State College, PA: Minitab, Inc.; www.minitab.com).
Parametricity was determined using histograms, Q-Q plots and the Shapiro–Wilk test. All data was non-parametric and displayed as median ± interquartile range (IQR). The Kruskal–Wallis test was used to compare the groups. Results were considered statistically significant if p < 0.05.
Results
One hundred and ninety patients were referred with a suspected MH reaction during the study period (1974–2016). Of these, 46 patients (24%) were referred due to MMR with or without other signs of MH susceptibility. Three patients declined testing or have been lost to follow-up, one patient died of causes unrelated to MH with no MH investigation of other family members and another is awaiting testing, giving a final total of 41 patients included in our analysis.
Patients were allocated on the basis of IVCT and DNA analysis into one of three groups: (a) a positive IVCT/RYR1 variant positive DNA group (MHS/DNA positive), (b) a positive IVCT/RYR1 variant negative group (MHS/DNA negative) and (c) an MHN group. For ease of analysis, DNA positive patients with no IVCT but with pathogenic variants (abnormal calcium release demonstrated) were included in group 1 as initial DNA testing became our policy only in recent years. Twenty patients were included in the MHS/positive DNA group, seven in the MHS/DNA negative testing group and 14 tested MHN (Table 1).
Malignant hyperthermia and RYR1 variant status for patients presenting with masseter muscle rigidity.
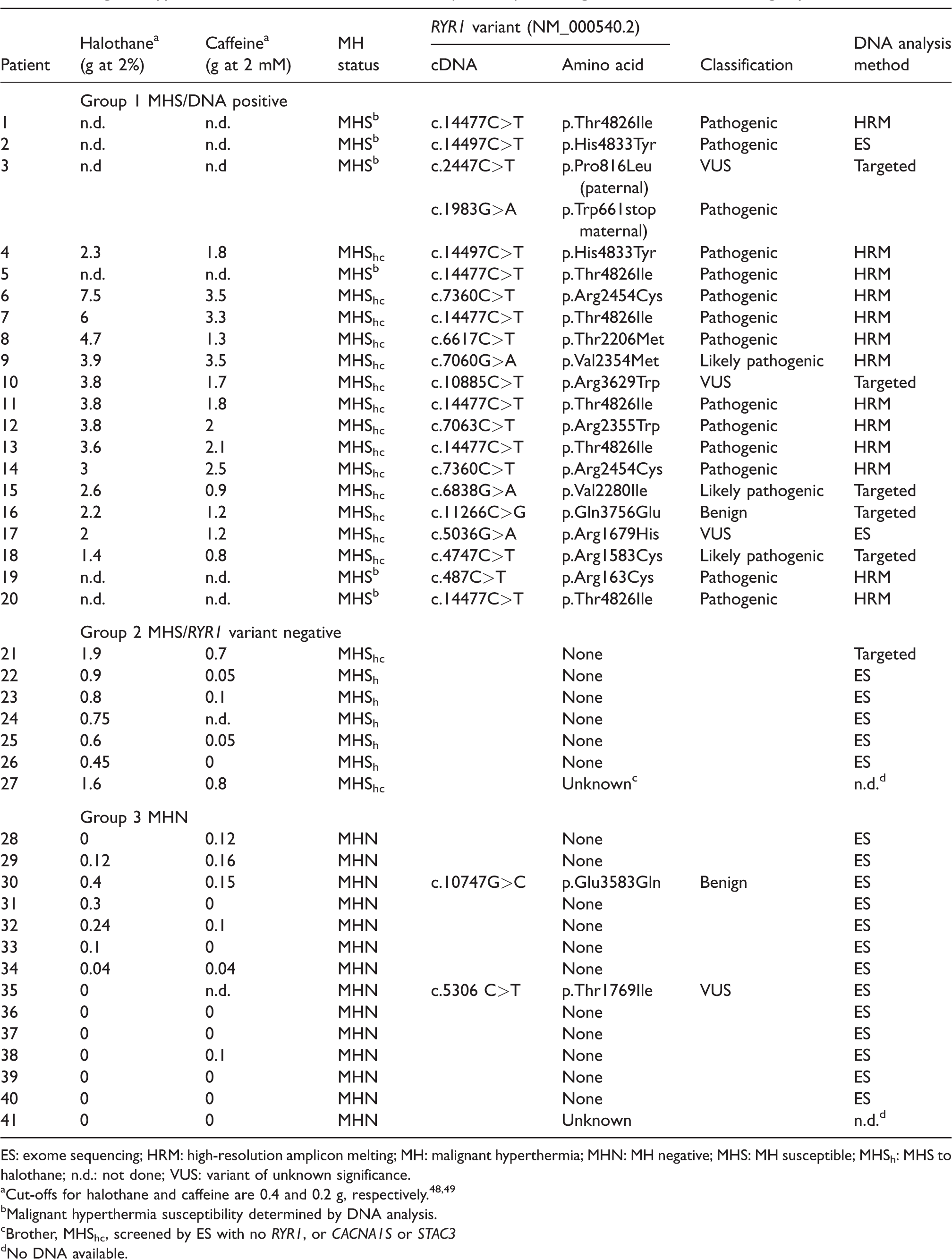
ES: exome sequencing; HRM: high-resolution amplicon melting; MH: malignant hyperthermia; MHN: MH negative; MHS: MH susceptible; MHSh: MHS to halothane; n.d.: not done; VUS: variant of unknown significance.
aCut-offs for halothane and caffeine are 0.4 and 0.2 g, respectively.48,49
bMalignant hyperthermia susceptibility determined by DNA analysis.
cBrother, MHShc, screened by ES with no RYR1, or CACNA1S or STAC3
dNo DNA available.
Thirteen patients from the MHS/positive DNA group had a confirmed pathogenic variant in RYR1. One patient (16) from this group had the common RYR1 variant NM_000540.2 (RYR1_v001): c.11266C>G, p.Gln3756Glu. This variant has a minor allele frequency of 0.03374,24 and is therefore unlikely to be pathogenic for MH. A rare variant of unknown significance was also identified in CACNA2D1 in this patient (Table 2). CACNA2D1 encodes two subunits of the dihydropyridine receptor.25 The functional role of this variant is unknown and a lack of familial DNA samples has precluded follow-up.
Rare variants of unknown significance identified by exome sequencing.

Six patients in the MHS/positive DNA group had RYR1 variants that have not yet been functionally characterized. These are listed in Table 1 as variants of unknown significance or that are likely pathogenic. A pathogenic familial variant was identified in six patients in this group prior to IVCT analysis. IVCT was therefore deemed unnecessary but these patients have been included in the MHS/DNA positive group. One deceased patient who died from an MH reaction has also been included in this group as immediate relatives tested MHShc with a pathogenic variant identified in the family.
There were seven patients (two MHShc and five MHSh) in the MHS/DNA negative group where variants in RYR1 had been excluded through DNA sequencing. Of 14 MHN patients, 12/14 did not have an RYR1 variant. One of these variants has been classified as benign. The other is a variant of unknown significance and DNA was not available for analysis in one patient. DNA analysis was therefore undertaken in 98% (40/41) of cases or in a sibling. One rare CACNA1S variant of unknown significance was identified in this group (Table 2). A rare variant in JSRP1 (encoding the protein JP-45 with a potential role in modulating RYR1-mediated calcium release)26 was also identified in the same patient (22) and a rare variant in STIM1 (encoding stromal interaction molecule 1, which has a role in store-operated calcium entry)27 was identified in an additional patient (24) in this group. Any role in MMR for any of these variants would be pure speculation in the absence of familial segregation or functional analysis.
MMR grading results are presented in Table 3 and indicate that severity did not correlate with MH susceptibility status. Patient age ranged from 1 to 67 years with a mean of 21 years. Fifty-two percent of the study population were female. Suxamethonium was the triggering agent for MMR in 39 of the 41 patients, while two patients in the MHN group did not receive suxamethonium. All patients received an inhalational agent with isoflurane, halothane, sevoflurane and others used in 40%, 30%, 25% and 5% of cases, respectively. Thirteen patients (31%) showed no other signs of an MH reaction apart from MMR (six MHN, six MHS/DNA positive and one MHS/DNA negative). Twenty-eight patients developed signs such as tachycardia, raised ETCO2 or raised temperature consistent with a clinical MH reaction. The time to onset of symptoms following MMR suggesting an MH reaction after the initial suxamethonium dose ranged from 5 to 60 mins (mean 20 min) in the MHS group and from 5 to 80 mins (mean 21 min) in the MHN group. The procedure was abandoned in six cases. Triggering agents were continued in 53% and 80% of cases in the MHS and MHN groups, respectively. Across all groups, 62% had their triggering anaesthetic continued. Seven patients received dantrolene (17%) and six were admitted to an intensive care unit following MMR, with the latter all testing MHS. Seven (17%) were managed successfully as day stay cases following an episode of MMR with no subsequent complications. Two patients developed generalized body rigidity and several patients complained of myalgia. Two patients died from a fulminant MH reaction following an initial episode of MMR. Both died before the first report, indicating that MMR was an early sign of MH susceptibility11. The RYR1 NM_000540.2 (RYR1_v001):c.487C>T, p.Arg163Cys variant was identified in autopsy tissue in one of these patients.
Grade of masseter muscle rigidity at induction of anaesthesia.

Description of the difficulty experienced in inserting an airway device.
MHN: Malignant hyperthermia negative; MHS: malignant hyperthermia susceptible; SGA: supraglottic airway device.
Surgical procedures were obstetric and gynaecological, ear, nose and throat, general surgery, dental and others, making up 32%, 31%, 23%, 8% and 6% of cases, respectively.
SMVs and CK are outlined in Table 4. None of the four SMVs considered in the three subgroups reached statistical significance (p > 0.05). However, there is a trend towards higher heart rate, temperature and ETCO2 in the MHS/DNA positive group. MH Clinical Grading Scale (MHCGS) analysis was undertaken in all patients.28 Scores ranked from 6 to 3 in the MHS/DNA positive group, 4 to 3 in the MHS/DNA negative group and all MHN patients ranked 3 (somewhat unlikely risk of MH). The median CK value in the MHS groups was found to be higher than in the MHN group with median values of 18,013 units/L (MHS/DNA positive), 5080 units/L (MHS/DNA negative) and 180 units/L (MHN), respectively. This was significant with a p-value of 0.004. All patients with a CK of > 18,000 units/L were MHS/DNA positive. A similar analysis of the relationship between DNA positive and negative subjects, and airway insertion difficulty/degree of MMR, failed to show a significant difference (p > 0.05) (Table 5). Several patients had low SpO2 values, but these were caused by minor airway problems or hypoventilation, and were corrected promptly9. Minor arrhythmias, viz bigeminy and ventricular ectopics, were recorded in two patients. Arterial acid–base analysis was undertaken in 12 patients. Six patients were MHShc with a base excess of –7 to –13 mmol/L. Four of these patients tested DNA positive. All other measurements were < 7 mmol/L.4 Acid–base analysis was undertaken in only one MHN patient with a normal result. Electrolyte data were poorly recorded across all groups and therefore have not been included. Other parameters did not differ between groups.
Standard monitored variables.
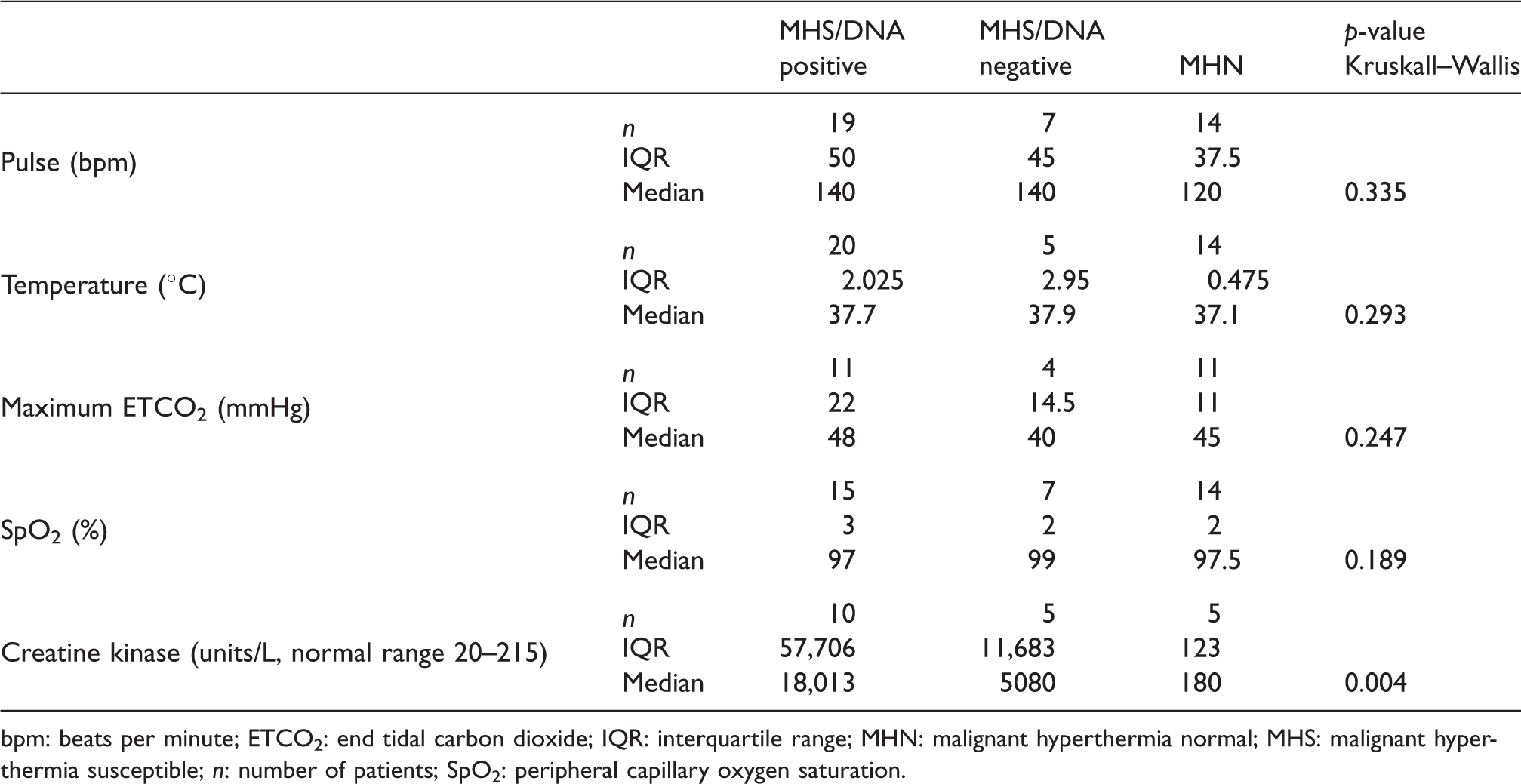
bpm: beats per minute; ETCO2: end tidal carbon dioxide; IQR: interquartile range; MHN: malignant hyperthermia normal; MHS: malignant hyperthermia susceptible; n: number of patients; SpO2: peripheral capillary oxygen saturation.
Supraglottic airway insertion.
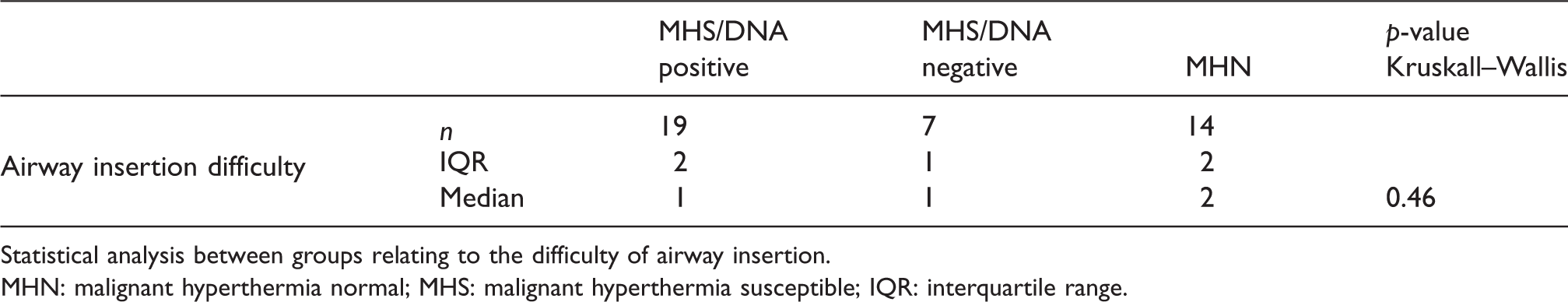
Statistical analysis between groups relating to the difficulty of airway insertion.
MHN: malignant hyperthermia normal; MHS: malignant hyperthermia susceptible; IQR: interquartile range.
Discussion
DNA analysis
A primary aim of this study was to analyse DNA data to demonstrate that DNA analysis can be used as the initial diagnostic tool in patients presenting with MMR, with or without other signs of MH susceptibility present. Importantly, our study identified RYR1 variants in 19 patients, 46% of the total cohort. Of these, 17 variants are classified as pathogenic or likely pathogenic using the American College of Medical Genetics and Genomics guidelines for the interpretation of sequence variants.29 Reclassification of those that are likely pathogenic and variants of unknown significance to pathogenicity must await functional testing, as recommended by the European Malignant Hyperthermia Group (EMHG).1 It is notable that there is a general trend for pathogenic variants to be present in patients exhibiting stronger contractures in the IVCT (Table 1 and Supplemental Figure 1). Exome sequencing is now accessible and relatively inexpensive, and can be used as a first screening tool for identification of potential genetic variants in RYR1, CACNA1S or STAC3, and other future implicated genes. Given the paucity of variants that have been shown officially to be pathogenic for MH susceptibility,30 the potential for a definitive diagnosis from DNA screening is currently limited. Even in our small study, one MHShc patient carries an RYR1 variant that is unlikely to be pathogenic for MH and an MHN patient carries a novel RYR1 variant of unknown significance.31 Exome sequencing will become more effective as more genes and variants are definitively shown to be pathogenic for MH. Indeed, our sequencing exercise did highlight rare variants of unknown significance in other genes associated with the Ca2+ release channel or skeletal muscle Ca2+ homeostasis (Table 2). Although IVCT is regarded as the ‘gold standard’ of MH susceptibility investigation, it does have morbidity with pain initially, scarring, and potential wound infection, haematoma and nerve damage. The specificity of the test is only 93.5% (sensitivity 99%),32 which indicates some false positive test results.17,32 Conversely, DNA analysis requires only a blood specimen with minimum morbidity. If a pathogenic variant is identified, a diagnosis of MH susceptibility can be made without further testing.
In this group of patients, 7/41 were MHS with no pathogenic variant identified (MHS/DNA negative group). This could be due to the existence of a variant in a gene yet to be linked to MH susceptibility (and consequently MMR), or an unidentified muscle disorder exists that results in a false positive IVCT result and is the true cause of MMR. Indeed, five patients out of seven returned MHSh results for the IVCT, with most exhibiting lower contractures to halothane than the MHS/DNA positive group (Supplemental Figure 1). The exome datasets for this group, as well as the MHN group, represent a valuable data source, which could be tapped at a later date when new genes associated with MH susceptibility or other muscle disorders are identified.
SMVs
With the caveat of the small sample size, there was no significant difference or any clear trends in any of the measured SMVs, with the exception of CK, between the three groups. A previous report suggested that elevated CK in the context of MMR significantly increases the probability of MH susceptibility.7 This correlation is in accordance with our study. Patients with CK > 18,000 units/L were MHS with identification of a pathogenic variant, but this is only proposed as a guideline for MH susceptibility. Suxamethonium can cause an increase in CK levels due to fasciculation, although the degree of elevation is less than that seen with MMR.33,34 MHCGS ranking did not distinguish between MHShc and non-susceptible individuals.
Masseter muscle rigidity
Masseter muscle rigidity is normally reported to last for several minutes, although prolonged cases have been described previously.12,35 In two patients in this series, the rigidity lasted for 8 mins and longer than 15 mins, the latter requiring mask ventilation to complete the anaesthesia. Both patients were MHS, DNA positive (unpublished data, N. Pollock).
Perhaps the most unexpected finding relates to the relationship between MMR grade and MH susceptibility. Three categories of jaw ‘stiffness’ have been described previously and our classification has similar descriptions.36 Current referral guidelines place a higher priority on more severe MMR, and the probability of MH has been reported to increase with the degree of spasm.37 There was no correlation between MMR grade and MH susceptibility status in our cohort.
Differential diagnosis
A major reason for DNA analysis is to exclude the association of abnormal IVCT with MMR because of an underlying myopathy.18 One patient (28) in the MHN group had a co-existing neuromuscular disorder and another (patient 17) in the MHS group had a family history of myotonic dystrophy. Whether either of these co-existing disorders has a role in MMR cannot be determined at the present time. The list of differential diagnoses for MMR does include other muscle disorders as well as inadequate dose of neuromuscular blocking agent, inadequate level of anaesthesia and temporomandibular joint dysfunction.10 Each should be considered before making a diagnosis of MH. There is a strong association of MH susceptibility with uncommon muscle disorders including central core disease,38 King–Denborough syndrome,39 multiminicore disease,34,40 centronuclear myopathy41 and congenital fibre type disproportion myopathy.42 None of the patients in our study has any indication of suffering from any of these disorders.
Implications for clinical practice
Our results are based on patients who were referred with MMR for investigation of MH susceptibility from the different centres around the country. It is possible that not all patients with MMR were referred, thus, our results may be subject to bias. However, 65% of suspected MH reactions presenting with MMR tested MHS, a level comparable with previous studies. Likewise, our referral rate for this subset of patients (24%) is also consistent with referral rates at other centres of 25–29%,7,43 and a positive diagnosis in 65% of cases indicates that referrals are appropriate. Therefore, we suggest that a diagnosis of MH should be considered in all cases of MMR. Appropriate clinical actions should be commenced and the patient referred for postoperative MH susceptibility testing, initially by DNA analysis and then IVCT if a pathogenic variant is not identified according to the current guidelines.1 Preoperative assessment of all patients is important, and should include demonstration of appropriate mouth opening and a detailed family history. Two patients in the late 1970s in New Zealand died from MH after exhibiting MMR, and one of these deaths may have been avoided if a detailed family history had been obtained. This is supported in another study where three deaths occurred, but in subsequent family interrogation a family history of MH was revealed.44 At Palmerston North hospital, all patients presenting with MMR are referred for MH investigation.
Our policy in MHN patients with negative DNA analysis is for triggering agents to be administered in subsequent anaesthetics.31 However, unless uncommon variants associated with muscle disorders have been excluded, caution should be exercised with suxamethonium. Three MHN patients have had subsequent anaesthesia following an episode of MMR. All have received volatiles and one suxamethonium, with no indication of MH susceptibility (unpublished data: A. Woodhead and N. Pollock).
The EMHG guidelines suggest that after recognition of clinical signs of MH (such as MMR) appropriate management steps should be taken. These include cessation of all triggering agents and, if possible, termination of the procedure.45 This policy is supported by the North American Malignant Hyperthermia Registry (personal communication, B. Brandom). Brandom has proposed the following method of procedure if the anaesthetist considers that the MMR is likely to be pathological: initially check the muscle tone in the extremities, assess minute ventilation, measure arterial blood gases and serum potassium, measure plasma or urinary myoglobin, and terminate the procedure if possible. The anaesthetic team should ensure that specimens are taken for CK analysis immediately and at 24 hours.46 If a case of MMR is considered significant, the patient should be referred for appropriate testing postoperatively, which includes DNA analysis and IVCT47 if subsequently indicated.
In summary, no significant differences were found in any of the SMVs (except CK, which is not definitive for MH) between the three groups, so these cannot be used to assess the likelihood of MH susceptibility being the cause of the MMR. IVCT remains the gold standard of MH susceptibility testing, but DNA analysis is now recommended by the EMHG as the initial investigative step.1 Based on these recommendations and the results of this study, our policy is now to initially screen MMR patients and other patients with suspected reactions (where a familial variant has not been identified) by exome sequencing followed by IVCT if this proves inconclusive.
Supplemental Material
Supplemental material for Masseter muscle rigidity and the role of DNA analysis to confirm malignant hyperthermia susceptibility
Supplemental Material for Masseter muscle rigidity and the role of DNA analysis to confirm malignant hyperthermia susceptibility by Kate Hudig, Neil Pollock, Terasa Bulger, Roslyn G Machon, Andrew Woodhead, Anja H Schiemann and Kathryn M Stowell in Anaesthesia and Intensive Care
Footnotes
Acknowledgements
The authors wish to thank Prof. Patrick Morel (School of Agriculture and Environment, Massey University, Palmerston North, New Zealand) and Greig Russell (MidCentral District Health Board) for the statistical analysis. Dr. Anja Schiemann was supported by grant numbers 12/022 and 15/011 from the Australian and New Zealand College of Anaesthetists. All other support was provided solely from hospital or institutional funds.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
