Abstract
Chorea Hyperglycemia Basal Ganglia Syndrome (CHBG) is an uncommon neurological complication arising in diabetic patients with severe, non-ketotic hyperglycemia. This case report describes a 50-year-old woman presenting with new-onset, choreiform movements in her extremities. Initial workup revealed uncontrolled diabetes (plasma glucose 410 mg/dl, HbA1c 11.2%) with negative serum ketones. Brain MRI findings supported the diagnosis, demonstrating characteristic T1 hyperintensity in the right basal ganglia. Implementation of gradual glycemic control over 48 hours led to significant improvement of her symptoms. This case emphasizes the importance of considering CHBG in the differential diagnosis of movement disorders in patients with uncontrolled diabetes. Early recognition and prompt glycemic management can lead to complete resolution of symptoms, highlighting the crucial role of maintaining proper blood sugar control in diabetic patients.
Keywords
Introduction
Diabetes mellitus (DM) can manifest with a wide range of neurological complications, commonly including peripheral neuropathy and stroke. 1 Chorea Hyperglycemia Basal Ganglia Syndrome (CHBG), also known as diabetic striatopathy or non-ketotic hyperglycemic hemichorea (NHH), is a rare neurological complication arising in patients with non-ketotic hyperglycemia.2,3 However, in exceedingly rare instances, this syndrome may also manifest in patients experiencing ketotic hyperglycemia. Characterized by involuntary, dance-like movements (chorea) and abnormalities within the basal ganglia—a brain region crucial for motor control—CHBG poses a unique diagnostic and therapeutic challenge.2,3 While most diabetic neurological complications are well-documented, CHBG remains underreported in the medical literature.
This case report highlights the importance of recognizing CHBG as a potential cause of movement disorders in diabetic patients. Through this case presentation, we aim to contribute to a more comprehensive understanding of CHBG and its clinical significance.
Case Report
A 50-year-old female, without any prior diagnosis of diabetes, presented with a sudden onset of uncontrolled choreiform movements in her left upper and lower extremities over the past 10 days. These involuntary movements persisted even during sleep and were exacerbated by activity. She had previously consulted a local physician and was given trihexyphenidyl tablets, but experienced minimal improvement in her symptoms. The patient reported no family history of neurological conditions such as chorea or endocrine diseases. There was also no history of medication use, including hormonal therapy or drug abuse, that could contribute to her symptoms. No recent head trauma, vascular events, or symptoms suggestive of neuroinfections were reported. No significant past history or comorbidities such as hypertension, hyperlipidemia, or other endocrine or autoimmune disorders were identified.
Upon examination, she had an unstable gait and hypotonia in her left upper and lower limbs. She exhibited involuntary and irregular movements that were non-rhythmic or repetitive, with greater involvement of the left upper extremity than the lower limb. Muscle power was normal, and systemic examination revealed no remarkable findings. The involuntary choreiform movements significantly impaired her gait, making walking difficult and unsteady. She reported challenges with activities of daily living (ADLs) such as dressing, eating, and self-care due to the constant limb movements which significantly impacted her quality of life.
Investigations
Initial investigations revealed a plasma glucose level of 410 mg/dl, with an HbA1c of 11.2%. Serum ketone bodies were negative, and arterial blood gas analysis was normal. Liver and renal function tests returned normal results. Computed tomography (CT) of the brain revealed subtle hyperdensity in the right basal ganglia region. Magnetic resonance imaging (MRI) brain showed T1 hyperintensity in the head of the right caudate and putamen nucleus (Figure 1). Based on the clinical presentation of involuntary choreiform movements, hyperglycemia with negative ketones, and characteristic MRI findings, a diagnosis of Chorea hyperglycemia basal ganglia syndrome (CHBG) was made. Specifically, for Wilson’s disease, slit-lamp examination revealed no Kayser-Fleischer rings. Although serum ceruloplasmin and 24-hour urinary copper levels were not performed due to the acute presentation and absence of clinical suspicion, the patient lacked typical features such as hepatic dysfunction or psychiatric disturbances. Mitochondrial disorders (eg, Leigh syndrome) and other metabolic conditions were deemed unlikely due to the absence of developmental delay, family history, or lactic acidosis (normal arterial blood gas analysis). Collectively, these findings supported the diagnosis of CHBG.
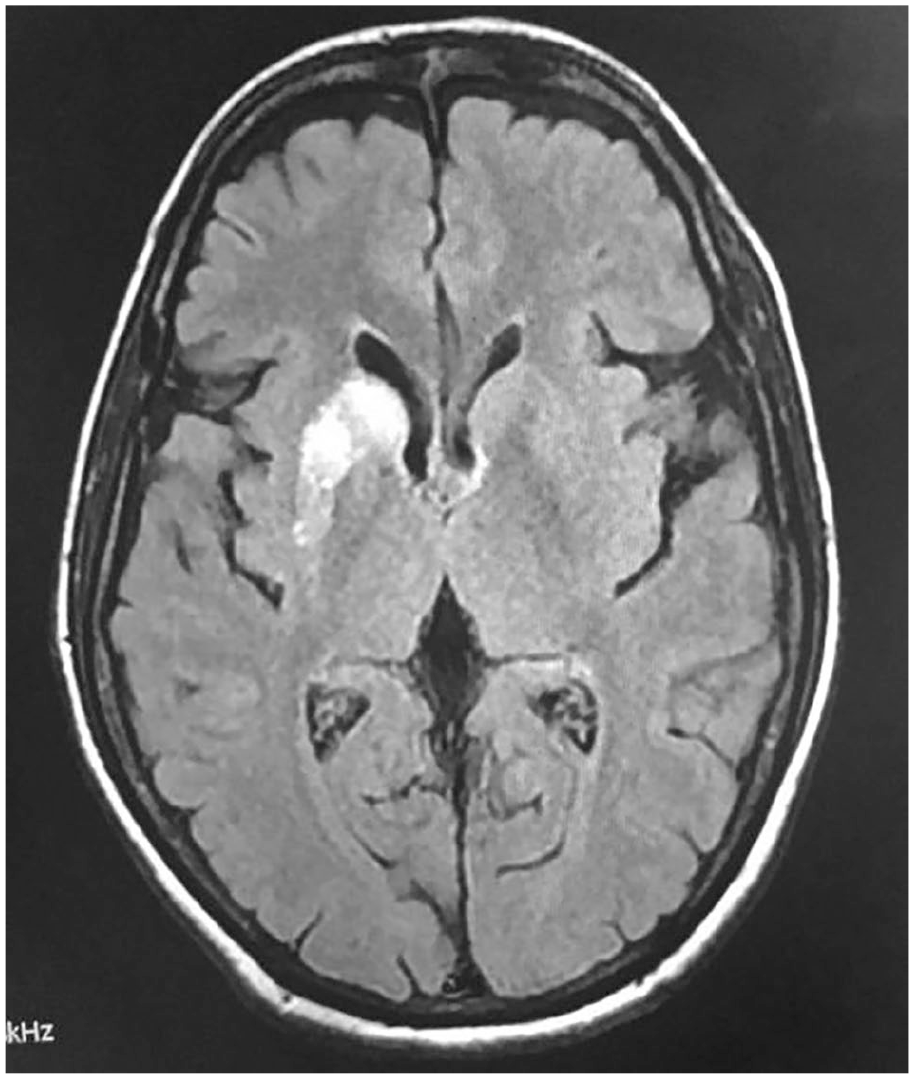
MRI brain showed T1 hyperintensity in the head of right caudate and putamen nucleus.
Management
Blood glucose was brought under control over 48 hours with close monitoring for hypoglycemia and electrolyte disturbances. Supportive care included hydration and electrolyte replacement as needed. Serum osmolality was estimated at 306 mOsm/kg, calculated using the formula: 2 × [Na+] + [glucose]/18 + [BUN]/2.8. This mildly elevated osmolality indicated a hyperosmolar state contributing to the neurological presentation. The diabetes management involved gradual glycemic control using a subcutaneous basal-bolus insulin regimen, aiming for reductions in plasma glucose levels of 50 to 75 mg/dl per hour to avoid rapid osmolar shifts. Patient was advised to continue the medications for control of diabetes and a follow-up after 4 months of discharge revealed a drop in her HbA1c level to 6.9%, and no recurrence of choreiform movements further. In our case, the management approach included only glycemic control without the addition of antipsychotics such as haloperidol. This decision was based on the absence of severe or refractory symptoms, allowing us to focus on targeted metabolic correction. Although literature supports the use of haloperidol in some cases, the patient’s response to glycemic control alone negated its necessity in this case. A repeat/follow-up MRI was not performed; however, it is noted in the literature that hyperintensities in CHBG typically resolve with glycemic control. If vascular or structural lesions were suspected, follow-up imaging would be critical to monitor changes in the basal ganglia.
Discussion
Chorea Hyperglycemia Basal Ganglia Syndrome (CHBG) is a rare neurological disorder primarily observed in elderly Asian women with type 2 diabetes mellitus.1,2 Initially, the predominant occurrence of CHBG in the Asian population led to speculation about genetic predisposition or inadequate diabetes control systems in underdeveloped countries.2,3 However, reports of CHBGS in Caucasian and Hispanic populations have challenged these hypotheses. 4 Additionally, this gender predominance may be due to underdiagnosis in males rather than actual gender-based differences. Contributing variables have also been proposed to be changes in dopamine receptors or Gamma Amino Butyric Acid (GABA) in postmenopausal women after estrogenic changes. 5 Although the exact pathophysiological mechanisms are still unknown, the “ominous octet” model that has been suggested includes the following, accumulation of gemistocytes, as a result of ischemic events, followed by petechial hemorrhage, deposition of methemoglobin, mineral deposition, cytotoxic edema, myelinolysis, gliosis, and tissue atrophy.3,6
The characteristic clinical presentation often involves sudden or insidious onset of hemichorea or hemiballism, predominantly affecting the limbs. CHBGS is characterized by high signal on T1-weighted MRI, which is likely due to accumulation of lipid-laden macrophages, confined to the striatum. 6 With this clinical presentation, NHH and MRI findings, a diagnosis of diabetic CHBGS was made. Other conditions that can cause above similar movement disorders and hyperintense MRI signals in the basal ganglia include carbon monoxide poisoning, methanol intoxication, cyanide intoxication, and hyperammonemia from chronic cirrhosis. 3
However, given the absence of risk factors and the lack of characteristic clinical features typically seen in these conditions (such as coma in carbon monoxide poisoning), these differential diagnoses were effectively ruled out. Nevertheless, other potential etiologies—including mitochondrial encephalopathy, Wilson’s disease, and Leigh syndrome should also be considered in the diagnostic evaluation. Still, these were deemed unlikely in this patient due to the acute presentation, MRI characteristics, and resolution of symptoms with glycemic control. 7 Also, not all patients with high HbA1c develop diabetic striatopathy (DS), indicating multifactorial causes. 8 Proposed mechanisms include basal ganglia susceptibility to hyperglycemia-induced energy metabolism disruption, microvascular ischemia, and GABA depletion causing excitatory-inhibitory imbalance. Genetic predispositions and oxidative stress may also contribute. These factors highlight the need for further research to identify at-risk individuals. 8
Gradually improving blood glucose levels over several days appears to be effective, leading to a reduction in choreiform movements without causing hypoglycemia. The course of diabetic striatopathy typically involves resolution of involuntary movements within days to months following correction of hyperglycemia. Certain individuals may experience only partial improvement needing anti-chorea drugs along with strict glycemic control. 3 Prolonged movement abnormalities are rare but have been observed in cases with specific characteristics, such as persistent basal ganglia abnormalities on imaging or abnormal dopamine transporter (DAT) scans, indicating underlying dopaminergic dysfunction.7,8 A review of previous case reports reveals that such cases often occur in older individuals or those with additional vascular or neurodegenerative predispositions. Comprehensive follow-up, including repeat imaging and DAT scans when necessary, can help identify such atypical presentations. 7
Conclusion
Our case emphasizes the importance of considering CHBG in the differential diagnosis of movement disorders in patients with uncontrolled diabetes. Early recognition and prompt glycemic management can lead to complete resolution of symptoms, highlighting the crucial role of maintaining proper blood sugar control in diabetic patients.
Footnotes
Acknowledgements
Special thanks to Squad Medicine and Research (SMR) for their guidance and help in publication.
Ethical Considerations
In our university, Ethics approval was not required for case reports and case series.
Consent to Participate
Written informed consent was obtained from the patient for the research and the publication.
Author Contributions
Gundraju S, Menda J, Suvvari TK, and Pindi AK—Idea, conceptualization, resources, formal analysis, data collection, supervision, writing draft, approved final draft. Davala PK—visualization, resources, writing draft and approved final draft. Singh T—Supervision, formal analysis resources, writing draft and revision of draft, approved final draft. Thomas V—Project Administration, resources, formal analysis, writing draft and revision of draft, approved final draft.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
