Abstract
Infection by Aspergillus covers a broad clinical spectrum, including invasive pulmonary aspergillosis (IPA) and its disseminated extrapulmonary form, invasive aspergillosis (IA). It typically occurs in severely immunocompromised hosts, but it sometimes affects the immunocompetent population, especially patients with acute diseases being treated at the intensive care unit (ICU) and less often those with chronic conditions. In this article, we report the case of a 50-year-old male, with diabetes mellitus (DM) as the only risk factor, treated for IPA and IA with cardiac and central nervous system (CNS) involvement at a high complexity institution in Cali-Colombia. Clinical presentation and radiological findings are unspecific and require a high level of suspicion. To confirm the case, histological or cytological of the fungus is required; histopathological examination of lung tissue is the gold standard, but it is difficult to perform due to respiratory compromise and high risk of bleeding, so bronchoscopy and bronchoalveolar lavage (BAL) plays an essential role in the diagnostic process. A diagnostic algorithm that includes risk assessment, symptoms, imaging findings, and isolation in cultures is essential to allow the diagnosis and initiation of treatment promptly, which includes a combination of surgery and antifungal medications for long periods, even life-long treatment.
Introduction
Aspergillus is a ubiquitous, filamentous, saprophytic fungus, isolated from construction sites, soil, and hospital dust. 1 There are around 900 species of Aspergillus, classified in 18 groups, of which 12 cause diseases in humans. 2 Repeated inhalation of the spores is common but tends to trigger symptoms in susceptible individuals only, with either underlying lung disease or immune system dysfunction. 1 Infection by Aspergillus covers a broad clinical spectrum: (1) allergic bronchopulmonary aspergillosis (ABPA), which occurs in individuals with cystic fibrosis (CF) or asthma and is triggered by sensitization against A. fumigatus; 3 (2) chronic pulmonary aspergillosis (CPA), which affects immunocompetent individuals with a preexisting lung condition and has a wide range of clinical presentations (simple aspergilloma, Aspergillus nodules, chronic cavitary pulmonary aspergillosis [CCPA]), which progresses to fibrosing pulmonary aspergillosis and subacute invasive pulmonary aspergillosis (SAIA) 4 and invasive pulmonary aspergillosis (IPA). The latter typically occurs in severely immunocompromised hosts and is defined as the invasion of lung tissue by Aspergillus hyphae. 1 Nevertheless, it could present in the immunocompetent population; patients with malnutrition, diabetes mellitus (DM), or other underlying chronic diseases have a 27% higher risk of developing IPA than the general population. 5 Additionally, A. fumigates, which is responsible for the majority of cases of IPA, could invade the pulmonary vasculature and cause thrombosis, pulmonary infarctions, and posteriorly hematogenous dissemination in up to 25% of affected patients. 1
After hematogenous spread and involvement of organs outside the respiratory system occur, the disease is renamed as invasive aspergillosis (IA); an opportunistic infection with high morbidity and mortality rates exceeding 50%. 5 Aspergillus fumigatus is responsible for 68% to 72% of cases of IA, followed by A. flavus (10%-17%), A. terreus (3%-7%) and in a lower percentage A. niger, A. nidulans, and A. ustus. Invasive aspergillosis generally affects immunosuppressed patients, especially patients with hematologic malignancies and hematopoietic cell transplantation. 6 However, 5% to 7% occurs in critically ill individuals without immunocompromise, especially in the context of an acute illness managed at the intensive care unit (ICU). 5 Common risk factors are prolonged and severe neutropenia, chronic use of corticosteroids, immunosuppressants or biologic medications, and cancer.7,8 Other reported associated conditions are chronic granulomatous disease, chronic obstructive pulmonary disease (COPD), human immunodeficiency virus (HIV) infection, cytomegalovirus (CMV) infection or reactivation, liver disease, DM and occupational exposures in construction sites.1,9
Clinical manifestations of IPA are nonspecific and largely depend on the interaction between the pathogen and host factors. 10 This explains why the diagnosis of IPA represents a challenge for clinicians, mainly in immunocompetent hosts, in whom due to the low reproducibility of symptoms and low level of suspicion the performance of diagnostic tests is delayed.5,7,10 Moreover, it is essential to exclude other conditions that could explain the clinical presentation such as malignancy, infections by other microorganisms (Mycobacterium tuberculosis, non-tuberculous mycobacteria, histoplasmosis, etc.), connective tissue diseases, necrotizing pneumonia, and pulmonary infarcts.1,8 Early diagnosis of IPA is crucial to prevent the progression to a disseminated form of the disease and thus improve the chance of survival of patients.
In this article, we present the case of a 50-year-old male patient with type 2 DM who developed IPA, followed by dissemination to the cardiac and central nervous system (CNS), treated at a high complexity institution in Cali-Colombia.
Case Report
A 50-year-old male patient with a history of arterial hypertension, type 2 DM, and heart failure, diagnosed 8 months before hospitalization. He consulted in another institution for 1 week of functional class deterioration, progressive edema in the lower limbs, orthopnea, hematemesis, and melaena. He underwent an upper gastrointestinal (GI) endoscopy which revealed a prepyloric ulcer with active bleeding at the edges, requiring sclerotherapy. He was then referred to our institution; at admission, he was without dyspnea at rest or nasal flutter, arterial tension (AT) 159/66 mmHg, heart rate 90 beats per minute, respiratory rate 20 breaths per minute, and SaO2 89% without supplemental oxygen. There was no evidence of ulcers in the nasal or oral cavity, no jugular engorgement, heart sounds were rhythmic without murmurs, no gallop or pericardial rub, decreased breath sounds at the base of the right lung, but no rales or wheezing. A painful and enlarged liver was found, but no ascites or collateral circulation reported, associated with grade III edema in the lower limbs. Neurological examination revealed no lateralization but time and space disorientation. The laboratory results can be seen in Table 1. Transthoracic echocardiogram reported a left ventricular ejection fraction (LVEF) of 40%, without any further alterations, diagnosing a decompensated chronic heart failure (profile B) with concomitant hypertension.
Laboratory Results.

Abbreviations: WBC, white blood cells; TSH, thyroid stimulating hormone; ALT, alanine aminotransferase; AST, aspartate aminotransferase; LDH, lactate dehydrogenase.
The patient was then transferred to the ICU. Management with vasodilators, diuretics, and non-invasive mechanical ventilation was initiated. A chest x-ray showed pulmonary congestion and a rounded, thick-walled image at the right pulmonary base (Figure 1). A chest computerized tomography (CT) scan showed a cavitated image in the lateral segment of the middle lobe with a nodule inside, in addition to varicose bronchiectasis and centrilobular nodules in the lower lobe (Figures 2 and 3). The patient was followed with bronchoscopy and bronchoalveolar lavage (BAL), in which transbronchial biopsies were taken, with results reporting generalized purulent endobronchitis. Bacilloscopic and polymerase chain reaction (GeneXpert) for tuberculosis were negative, as well as tests for syphilis, HIV, C, and B hepatitis. Direct studies for fungi detected hyaline septate hyphae, the galactomannan test for Aspergillosis was positive at 4.73 (negative value < 0.5), and cultures were positive for A. fumigatus. Cytology was negative for malignancy, but the biopsy showed chronic inflammation and necrosis associated with fungal structures suggestive of Aspergillus (Figures 4-6). A diagnosis of IPA was made and management with liposomal amphotericin B was initiated.
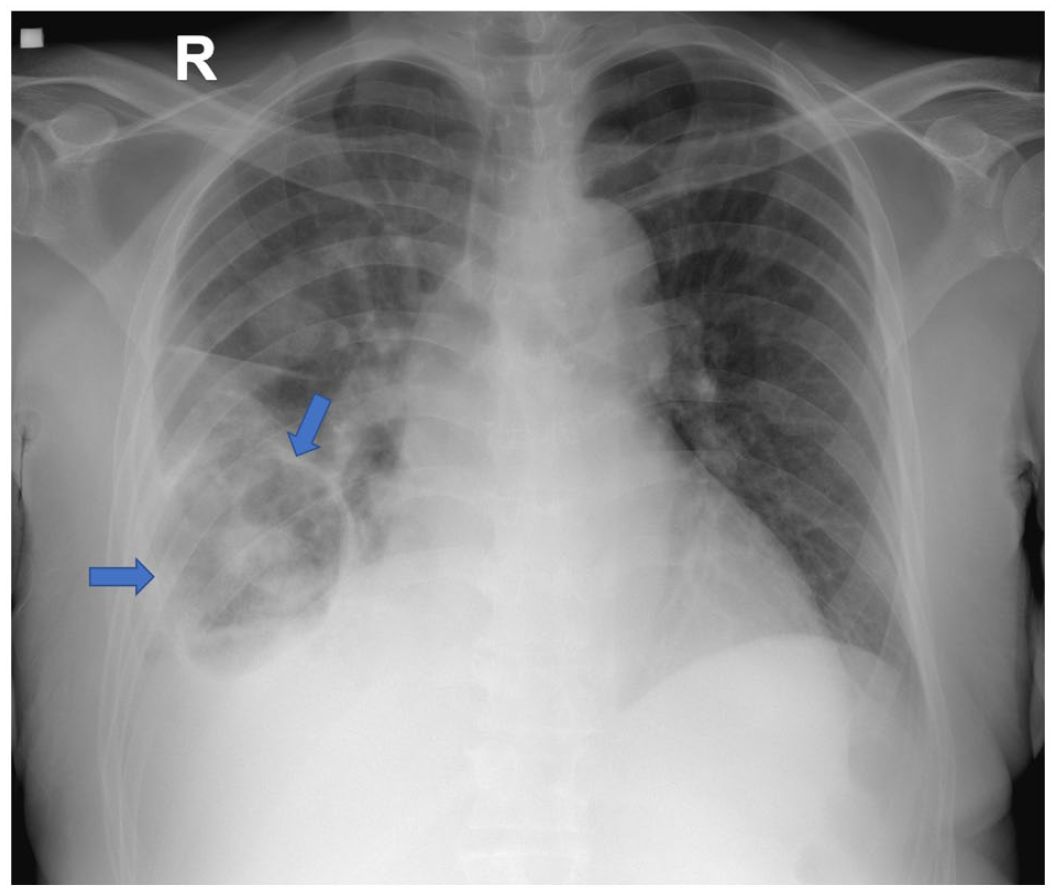
Chest x-ray showing a magnified cardiac silhouette due to anteroposterior projection, prominent aortic arch with elongation of the descendent aorta. Unremarkable pulmonary hila, central trachea, and in the right pulmonary base a cystic lesion, with thick walls, air-fluid level inside (blue arrows), alongside alveolar opacities without consolidation. Obliteration of right costophrenic angle due to pleural effusion.

Chest CT scan. (A-D) Sequence of images in the mediastinal window. (E-H) Sequence of images in lung window. There is a loss of the normal architecture of the middle and inferior right lobes, with consolidation in the posterior basal segment of the inferior right lobe, alongside varicose bronchiectasis (blue arrows). There is ipsilateral pleural effusion (red arrow), with air bubbles and anterior laminar pneumothorax (yellow arrow). The lateral segment of the middle lobe is replaced by thick-walled cavitation (green arrows).
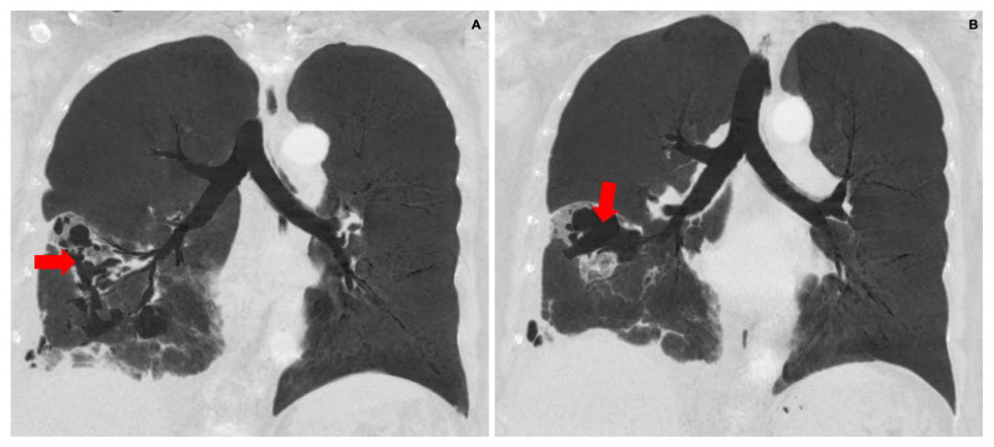
Chest CT scan, sagittal reconstruction. (A, B) A thick-walled cavitated lesion is observed, alongside varicose bronchiectasis (red arrows) and right pleural effusion.
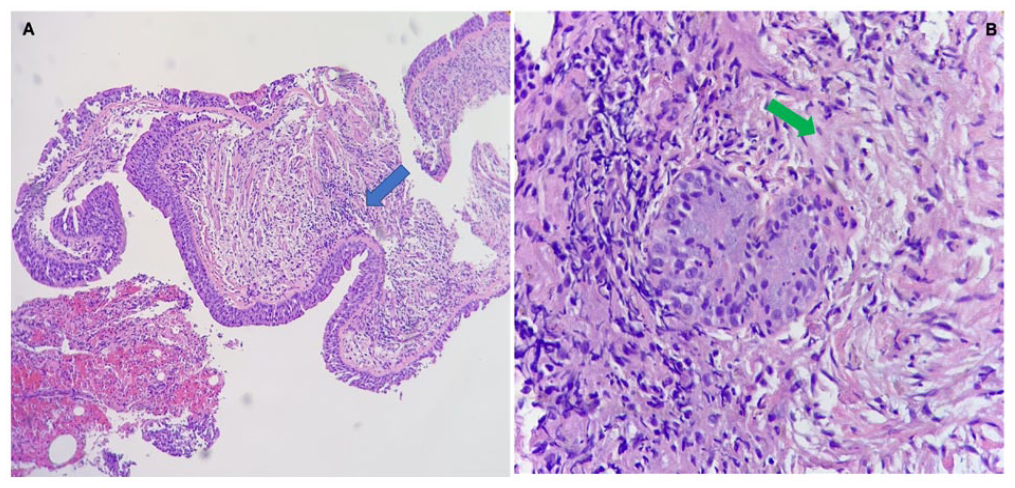
Transbronchial biopsy. (A) H&E staining showing a fragment of the respiratory epithelium with a dense subepithelial chronic inflammation (blue arrow). (B) Collagenous stroma (green arrow) with dense subepithelial chronic inflammation.
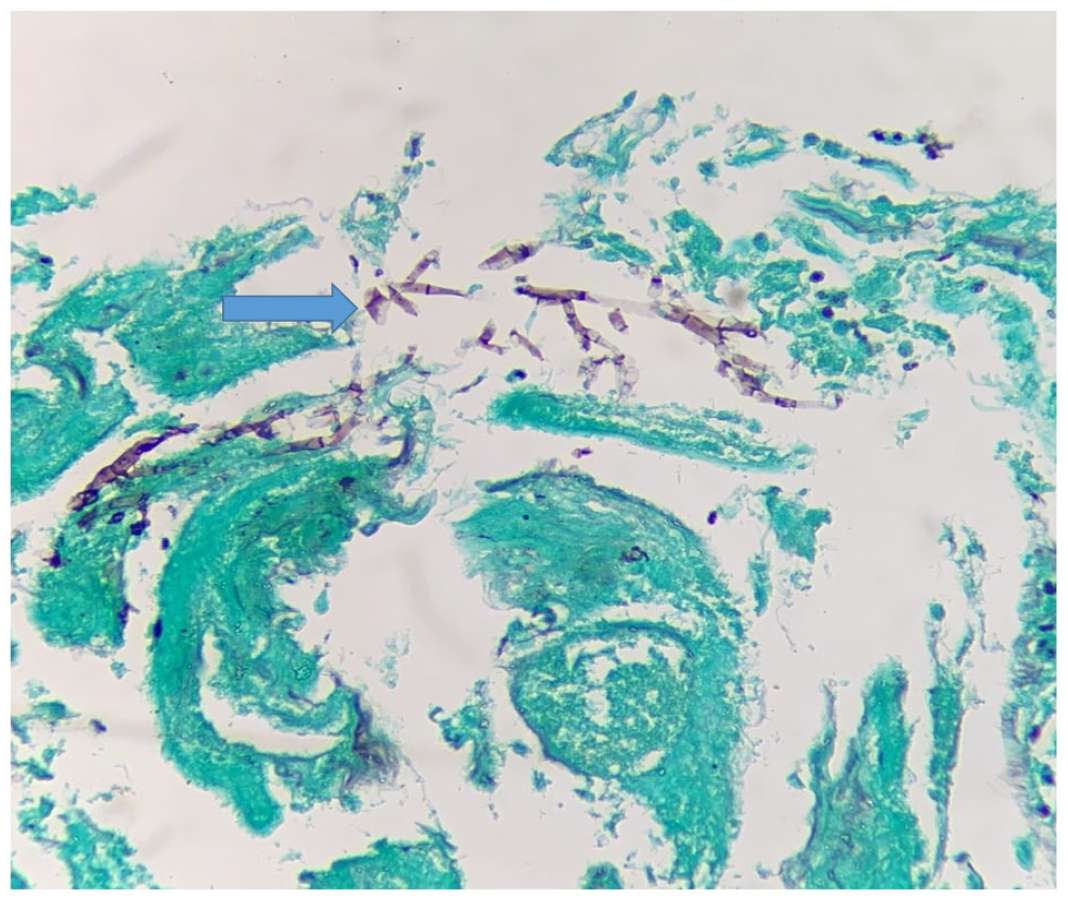
Transbronchial biopsy with Gomori Methenamine-Silver staining, where hyaline hypha is observed within the stroma, at a 45-degree angle (blue arrow).
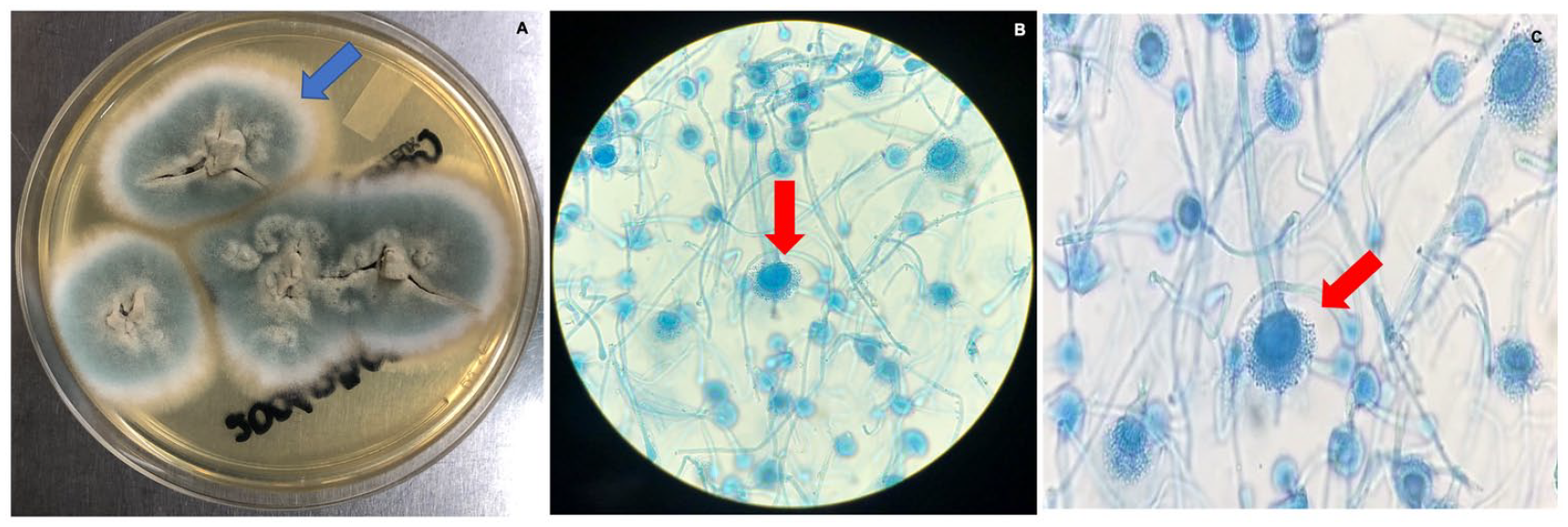
(A) Macroscopic aspect of the Aspergillus fumigatus colony in Sabouraud agar, showing a fast-growing colony, flat, velvety, initially green, which then turned into a grayish color with a white halo (blue arrow). (B, C) Microscopic characteristics, were typic columnar heads are seen, with short conidiophores, smooth walls, and cone-shaped terminal vesicles supporting a single row of phialides in the upper two-thirds of the vesicle. The conidia have a globose shape, with rough walls and a greenish color (red arrows).
Abdomen ultrasonography reported normal-sized kidneys, with no ascites but a filling defect in the right hepatic vein that extended to the right atrium. A transesophageal echocardiogram was performed, describing a LVEF 39% and lesions suggesting vegetations of 9 mm x 11 mm in the aortic valve, 24 mm x 19 mm in the right atrium and 19 mm x 16 mm with a 26 mm filiform element in the crista terminalis. Due to the neurological manifestations at admission, a brain CT scan was performed, reporting a left frontal periventricular hypodensity. One week later, the patient presented with a left inferior monoparesis and nominal aphasia, so he was followed with a nuclear magnetic resonance of the brain finding multiple lesions along the corpus callosum, right cerebellar hemisphere, diffuse leptomeningeal enhancement and changes due to volume loss of the parenchyma (Figure 7). A lumbar puncture was unremarkable. Due to the characteristics of the lesions, an angio-invasive fungal parenchymal compromise was considered, despite having a normal spinal tap.

Cerebral nuclear magnetic resonance. (A-D) Multiple lesions with an increased signal intensity are observed, of rounded morphology, compromising the white batter of both cerebral hemispheres (yellow arrows). The knee of the corpus callosum and right cerebellar hemisphere are visualized. There is vasogenic edema, especially around the lesion located adjacent to the frontal horn of the left lateral ventricle. After the administration of contrast medium, enhancement of these lesions is shown, compared to the adjacent parenchyma.
Having pulmonary, cardiac, and CNS involvement, IA was diagnosed and posaconazole was added to amphotericin B. The possibility of aortic valve replacement and right lower lobe lobectomy was deferred, pending on the evolution of the patient once the antifungal management was initiated. The patient received 8 weeks of in-hospital management and despite the poor prognosis, clinical stability and regression of pulmonary, cardiac, and CNS lesions were achieved (Figures 8 and 9). The patient was then discharged with posaconazole to complete treatment for 1 year. At follow-up, he presented in good clinical condition. Control studies showed an echocardiogram with a notable regression in the size of the lesions, the largest being 11 mm in the right atrium, a chest CT scan with a decrease in centrilobular nodules and no further progression of lesions and a brain MRI with an improvement of lesions due to less enhancement and a decrease in size of edema.

(A) Echocardiographic image, showing 2, 8-mm lesions in the crista terminalis (red arrows), which have decreased in size compared to the initial echocardiography. (B) 3D image of the tricuspid valve, showing 2, 9-mm masses (black arrows), also smaller than seen in the initial exam.

Chest CT scan, lung window. (A, B) The cavitated lesion of the middle lobe has decreased in size, as well as the consolidation in the inferior right pulmonary lobe (blue arrows).
Discussion and Conclusions
Although IPA presents mainly in immunocompromised hosts, during the last decade an increased incidence of the disease has been reported in non-neutropenic patients (from 0.33% to 5.8%), 10 while prolonged neutropenia has been documented in only one-third of patients. 11 Initial manifestations of IPA are cough, dyspnea, and fever, as well as chest pain and hemoptysis. The latter 2 are related to angioinvasion followed by hematogenous spread, presenting in 25% of patients as a late manifestation, highlighting the importance of an early diagnosis to prevent progression of the disease to a disseminated form.1,6,9 The most commonly affected organs in IA are CNS, liver, and kidneys. Less frequent compromises are tracheobronchitis, rhinosinusitis, endophthalmitis, focal neurological deficits, seizures, and disseminated skin lesions with painful, erythematous papules, and central necrotic area.1,6
While A. fumigatus is responsible for the majority of IPA cases, non-fumigatus Aspergillus are responsible for up to one-third of cases. Moreover, an observational study describing the characteristics of culture-positive IPA in patients with hematologic malignancies, reported that those patients who developed IA more commonly had non-fumigatus IPA compared to those who did not develop the disseminated form. 12 From a pathophysiological point of view, IA is related to low levels of tumor necrosis factor (TNF) alpha, interferon (INF) gamma, interleukin (IL) 6, and IL 10. Also, an increase in the secretion of inhibitory cells, as well as lymphocytes CD4 and CD8, HLA DR, along with a decrease in dendritic cells and lower expression of the receptor of ligand binding in alveolar epithelial cells is observed.5,13 This is linked to an alteration in the immune status of the host as in the case of patients hospitalized in the ICU for severe acute syndromes. 5
Radiological findings of IPA are nonspecific; they range from early solitary nodular lesions surrounded by a frosted glass attenuation halo to large masses, consolidations, cavitation’s, air crescent sign indicating necrosis at the center of the nodule, diffuse bilateral infiltrates and pleural effusion.1,10 Although a high-resolution chest CT scan is an excellent choice for monitoring patients with aspergillosis, a CT pulmonary angiogram (CTPA) better allows the detection of angioinvasion and signs of vascular occlusion that are specific and key for the diagnosis of patients with IPA. 10 Currently, the use of immunoPET/MR imaging for the detection of A. fumigatus is being performed, permitting rapid and noninvasive detection of the fungus, differentiation from a bacterial process, or a general inflammatory response. 14
The isolation of the fungus in the cultures of the respiratory tract should always be evaluated in conjunction with the clinical presentation to differentiate between a true infection or colonization. 10 Isolation of Aspergillus in sputum is rarely indicative of IPA in immunocompetent patients, but it does weight in neutropenic and oncological patients. 1 The performance of the culture is low due to the slow growth of the fungus and poor sensitivity of 25% to 50%.10,15 Therefore, there are markers based on the detection of components of the fungus cell wall such as galactomannan, which in the case of invasive infections is expressed not only in the respiratory system but also in tissues and blood. 16 In non-neutropenic patients, serum galactomannan has a sensitivity and negative predictive value (NPV) of 50-58 and 87 to 88% respectively. 5 Sensitivity increases to 100% and specificity lie between 47 to 92% in samples from BAL.10,17 On the other hand, in immunocompromised patients, the sensitivity of galactomannan in BAL varies from 58 to 84.6%, specificity is around 90.7% and NPV is around 70-90%.1,5
The study of (1,3) B-D- glucan, part of the fungal wall of several fungi, is also proposed. However, as it is not specific to Aspergillus, it is useful to rule out the disease but does not confirm it. Polymerase chain reaction for Aspergillus is not globally standardized and therefore is not included in the diagnostic criteria.1,10 Histopathological examination of lung tissue is the gold standard for diagnosis, but it is difficult to perform due to respiratory compromise and high risk of bleeding, so BAL still plays an essential role in the diagnostic process. 1
Diagnostic criteria for IPA in immunocompromised individuals were described in 2008 by the European Organization for Research and Treatment of Cancer and Invasive Fungal Infections and the National Institute of Allergy and Mycotic Infectious Diseases Study Group (EORTC/MSG); 18 a proven case is defined by a histopathological or cytopathological examination of lung tissue with evidence of hyphae and associated tissue damage or a positive culture test for Aspergillus from a sample obtained from the lung, plus clinical or radiological proof of infection. A probable case is composed of a patient with risk factors for IPA and mycological evidence plus clinical/radiological criteria consistent with infection. Finally, a possible case is characterized by host factors and clinical criteria of infection but the absence of microbiological isolation of Aspergillus (Table 2).1,5,19 In 2020, Blot et al 20 described diagnostic criteria for patients in the ICU: patients must meet the same characteristics of the EORTC/MSG criteria or present 4 of the following: positive lower respiratory tract culture for Aspergillus, compatible signs and symptoms, abnormal imaging findings and a risk factor for IPA or a positive culture for Aspergillus in BAL without other bacterial growth and cytology with evidence of septate hyphae. If it does not meet more than 1 criterion for IPA, it is then considered colonization by Aspergillus. 5
Diagnostic Criteria for Invasive Pulmonary Aspergillosis in Immunocompromised Patients.

European Organization for Research and Treatment of Cancer and Invasive Fungal Infections and the National Institute of Allergy and Mycotic Infectious Diseases Study Group (EORTC/MSG).
Cardiac involvement is rare and aggressive, occurring in immunocompromised individuals and solid organ transplant recipients. 21 Aspergillus is responsible for 20% to 30% of cases of fungal endocarditis, usually presenting with fever and embolisms involving CNS, lungs, or lower extremities.21,22 Blood cultures are occasionally negative, 22 being clinical suspicion of foremost importance in the context of the previously described symptoms and presence of large vegetations (>35 mm).21,22 The recommended treatment is voriconazole for long periods, even life-long, and surgical procedures are almost always required. Mortality rates range from 77% to 80%. 21
On the other hand, cerebral aspergillosis occurs frequently in immunocompromised patients but occasionally presents in immunocompetent hosts as well, with a mortality rate of around 100%. It occurs in the form of meningitis, thrombosis, increased intracranial pressure, and the formation of abscesses. 23 Space-occupying lesions are observed in immunocompetent patients, with an irregular thick wall indicating the hosts were trying to encapsulate the microorganism. 24 The diagnosis implies a high level of suspicion because symptoms are nonspecific. Spinal tap usually reveals pleocytosis with a slightly elevated protein level, positive galactomannan, and is rarely isolated in the cultures of the cerebrospinal fluid (CSF). Imaging findings on brain CT scans include edema and multiple areas of infarction with or without bleeding. Brain MRI is more sensitive in detecting multiple small intracerebral abscesses and single or multiple hemorrhagic infarcts. 23 The characteristic sign of Aspergillus infection is the low intensity at the periphery of the lesion, secondary to the presence of ferromagnetic elements such as iron, magnesium, and manganese.25,26 Surgery reduces the local load of the pathogen, but the clinical condition of the patient is determinant as well as the accessibility of the lesion. Additionally, antifungal treatment for 12 to 18 months is recommended. The prognosis after partial or complete surgical excision is good; 60% of immunocompetent patients have an adequate evolution after the procedure, with a relapse rate of 15% and a mortality rate of 25%. 23
The early instauration of treatment reduces mortality and disease progression. 1 Among the antifungals used in the management of IA is liposomal amphotericin B, which was considered the mainstay of treatment until the arrival of voriconazole and isavuconazole. 27 Voriconazole is currently the drug of choice, although being an inhibitor of CYP450, it has multiple drug interactions. However, its use is associated with decreased mortality in non-neutropenic patients.1,28 Isavuconazole offers an advantage as it has fewer CYP-mediated drug interactions. 10 Echinocandins are useful in those who do not tolerate first-line therapies and should not be used as monotherapy but in synergy with voriconazole for patients with severe and refractory IPA. 1 On the other hand, itraconazole and posaconazole are considered second-line drugs in cases of severe compromise. 9 Treatment duration depends on the evolution of each patient but the guidelines from the American Society for Infectious Diseases (ASID) recommend a minimum time of 6 to 12 weeks. 9
In patients with IPA, surgery is performed in patients with low surgical risk, who present an aspergilloma, persistent hemoptysis, or patients who do not respond to antifungal treatment.1,29 In cases were complete resection of the lesion is not achieved or when contamination of healthy tissue occurs during surgery, antifungal treatment must be continued to prevent empyema and disease recurrence. 29 Wedge resection and lobectomy are preferred over pneumonectomy, which should be avoided. 1 Mortality rates in the last decade have been decreasing from 89% to 90% to 40% to 50%, due in part to earlier detection and initiation of antifungal drugs. 6 Follow-up should be carried out every 3 to 6 months to evaluate clinical and radiological evolution, as well as the quality of life, C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), and levels of immunoglobulin G (IgG) against A. fumigatus, which can decrease without returning to normal levels. 1 Regarding the case of fungal endocarditis, it mandates the surgical replacement of the infected valve in almost all patients. 30
In conclusion, IPA and IA present a high morbidity and mortality rate. Although it mainly affects immunosuppressed individuals, immunocompetent people have also been found to be susceptible. The lack of specific clinical manifestations makes diagnosis quite challenging and early recognition of IPA with the subsequent prevention of progression to IA required a high level of suspicion, especially in immunocompetent patients. A diagnostic algorithm that includes risk assessment, symptoms, imaging findings, and isolation in cultures is essential to allow the diagnosis and initiation of treatment promptly.
Footnotes
Prior Presentation of Abstract Statement
This case was not presented as an abstract.
Availability of Data and Materials
Datasets used and / or analyzed during the current study are available from the corresponding author upon reasonable request.
Authors’ Contributions
All authors have read and approved the manuscript, and significantly contributed to this paper. L.F.T.: Conception and design, literature review, manuscript writing and correction, final approval of manuscript. I.E.: Literature review, manuscript writing and correction, final approval of manuscript. E.I.M.: Conception and design, literature review, manuscript writing and correction, final approval of manuscript. L.F.S.: Conception and design, literature review, manuscript writing and correction, final approval of manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This report was prepared in accordance with the ethical standards of the institutional ethics committee and with the 1964 Helsinki Declaration. We have approval letter of Ethics Committee in biomedical research IRB / EC No. 274-2020 of the Fundación Valle del Lili to publish this manuscript, registered in Act No. 22 of 2020.
Informed Consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
