Abstract
Background:
Drug-induced acute pancreatitis is a rare condition occurs at an incidence rate of 0.1–1.4% and represents a diagnostic challenge. TNFα inhibitors specially adalimumab is very rarely reported in the literature, primarily through case reports, as a potential cause of acute pancreatitis.
Case presentation:
Our case report presents a case of a 23-year-old patient followed for Crohn disease in whom a diagnosis of acute pancreatitis induced by adalimumab was made. This diagnosis was confirmed after the elimination of other possible etiologies, and notably by the recurrence of pancreatitis after Adalimumab rechallenge. The occurrence of acute pancreatitis induced by TNFα inhibitors exposes to the risk of pancreatitis with other drugs in this class, hence the need to switch to another therapeutic class, which was Ustekinumab in our case.
Conclusion:
Acute pancreatitis is an unusual complication of treatment with Adalimumab. Through our experience, based on solid scientific data, we want to draw the attention of clinicians to the reality of this complication. It should be considered in any patient on TNFα inhibitors who presents with acute pancreatitis without an obvious cause.
Introduction
Drug-induced acute pancreatitis (DIAP) is a rare condition occurs at an incidence rate of 0.1 to 1.4% and represents a diagnostic challenge to the clinician. 1 The diagnosis requires ruling out common etiologies of acute pancreatitis. 2 Database of the World Health Organization has listed more than 500 drugs that could cause AP as an adverse effect. 3 Only two drugs, azathioprine and didanosine, have robust evidence from retrospective cohorts and randomized trials, while the true extent of DIAP remains unclear. 4 Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome is a rare condition resulting from a complex interaction between an individual’s genetic predisposition, alteration in drug metabolism and possible re-activation of latent viral infections. Acute pancreatitis is the second most frequent manifestation of gastrointestinal involvement of DRESS syndrome. 5 TNFα inhibitors have been rarely described as a cause of acute pancreatitis in case reports.
Case Report
A 23-year-old patient with no significant past medical history was diagnosed 1 year ago with colonic Crohn’s disease (CD), without anorectal lesions, classified as A2L2B1 according to the Montreal classification. The disease was corticosteroid-resistant and required Adalimumab-based therapy, starting with the initial dose and then maintenance at 40 mg every 2 weeks. Due to a primary non-response to Adalimumab, the treatment was optimized to 40 mg every 10 days. The patient’s condition showed clinical improvement, along with a significant decrease in fecal calprotectin levels.
Six weeks later, the patient presented with epigastric pain and a lipase level of 734 IU/L, more than three times the normal value, leading to a diagnosis of acute pancreatitis without signs of severity. An abdominal CT scan (Figure 1) performed on day 3 confirmed benign edematous acute pancreatitis, classified as stage C according to the Balthazar grading system, with a CTSI (Computed Tomography Severity Index) of 2. During the etiological investigation, the patient denied any alcohol or tobacco use. A fasting hepatobiliary ultrasound performed by an experienced radiologist within the first 24 hours of diagnosis, as well as an MRCP (performed 1 week later), showed no gallstones and no signs of autoimmune pancreatitis. Liver function tests and a cholestasis panel, including transaminases, alkaline phosphatase, GGT, and bilirubin, were normal. Calcium and triglyceride levels were also normal, and the serum IgG4 level was 0.8 g/L (NR: 0.04-0.87 g/L). The patient’s body mass index was 22 kg/m². Management involved temporary cessation of food intake with hydration and pain management, without discontinuing Adalimumab.

Computed tomography axial images showing edematous pancreatitis, particularly in the head. Note the infiltration of peripancreatic fat (arrows).
One month later, the patient experienced epigastric pain similar to the initial episode, with a lipase level of 546 IU/L, confirming a second episode of acute pancreatitis. The etiological workup was repeated, including biliary ultrasound, liver function tests, calcium levels, triglycerides, and IgG4 levels, all of which were normal. The decision was made to discontinue Adalimumab treatment, and a biliary endoscopic ultrasound was performed, which showed no signs of auto-immune pancreatitis but suggested the presence of gallbladder microlithiasis (Figure 2). This led to the performance of a cholecystectomy on the same day as the endoscopic ultrasound. The patient was discharged with permission to resume Adalimumab injections two weeks after the cholecystectomy.
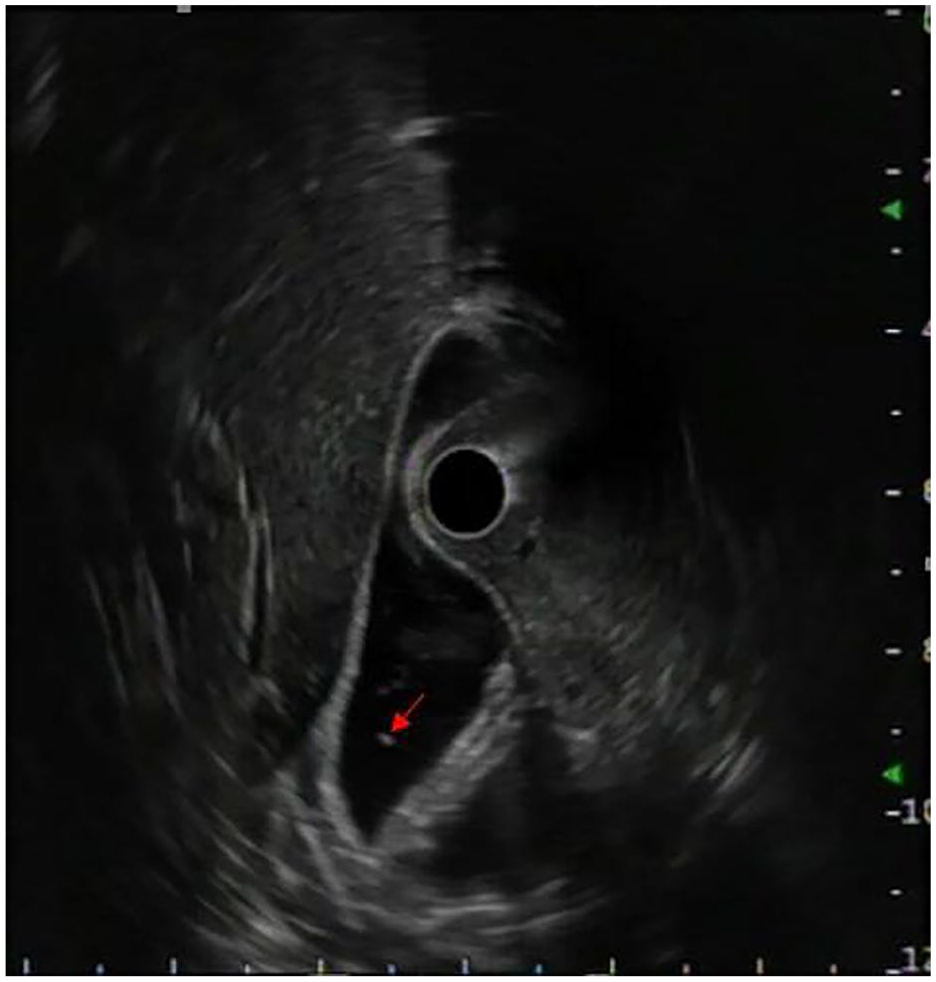
Radial endoscopic ultrasound via bulbar station showing gallbladder microlithiasis (arrow).
Three days after Adalimumab rechallenge, the patient experienced a recurrence of acute pancreatitis, indicated by abdominal pain and an elevated lipase level of 952 IU/L. An abdominal ultrasound, liver function tests, calcium level, and triglycerides were all normal, pointing to Adalimumab as the cause and leading to the permanent discontinuation of TNFα inhibitors and a therapeutic switch to Ustekinumab. It is important to note that gallbladder microlithiasis is not typically considered a cause of pancreatitis, but rather a result of stasis related to the patient’s fasting for several days due to fear of recurrent pain. In our patient, the hypothesis of biliary pancreatitis was definitively ruled out based on several factors: (1) liver function tests were consistently normal, (2) the acute pancreatitis occurred several days after the cholecystectomy, which was performed on the same day as an endoscopic ultrasound that ruled out the presence of bile duct stones, (3) the recurrence of pancreatitis immediately after Adalimumab reintroduction, and (4) the complete resolution of symptoms and absence of recurrence several months after discontinuing Adalimumab.
The evolution was marked by the disappearance of pain, clinical improvement, and no recurrence of pancreatitis episodes during 5 months of follow-up.
Discussion
The incidence of acute pancreatitis (AP) continues to rise, and 20% to 30% of cases are labeled as idiopathic in etiology. It is important to establish the etiology of AP to decrease morbidity and mortality related to an AP episode, especially in patients with recurrent AP. 4 Drug-induced acute pancreatitis is a rare condition occurs at an incidence rate of 0.1% to 1.4% 1 and represents a diagnostic challenge to the clinician; this is because our current DIAP knowledge is very limited as the vast majority of the evidence is from case reports. There are more than 120 drugs that have been implicated in causing AP with different mechanism of injury reported in the literature. 4 In addition, the World Health Organization database has listed more than 500 drugs that could cause AP as an adverse effect. 3
The diagnosis of DIAP first requires a diagnosis of acute pancreatitis. The next step requires ruling out more common etiologies such as gallstone pancreatitis, ethanol abuse, hypercalcemia, hypertriglyceridemia, and trauma. Lipase, triglyceride level, calcium level, and liver function tests should be ordered. Abdominal and endoscopic ultrasounds should be performed to evaluate for gallstones and other obstructive possibilities such as tumors of the pancreas head. 2
Following a second episode of AP with no identifiable cause, in patients fit for surgery, AGA guidelines suggests performing a cholecystectomy to reduce the risk of recurrent episodes of AP. 6 In our case, after ruling out others differential diagnoses, a cholecystectomy was performed due to the suspicion of tiny gallstones. Despite this, the patient experienced a recurrence of acute pancreatitis, suggesting a non-biliary cause.
Two forms of autoimmune pancreatitis (AIP) are described, AIP-1 and AIP-2, that differ in symptoms, high serum IgG4 level in AIP-1 and presence of extra pancreatic manifestations more frequently in AIP-2. AIP is rare among individuals with inflammatory bowel disease (IBD) but occurs more frequently in this group compared to the general population, with a prevalence of approximately 0.4% versus 0.1%. 7 The link between AIP and ulcerative colitis(UC) appears to be stronger than its association with CD. 8
The diagnosis of AIP according to the International Consensus Diagnostic Criteria is based on radiological findings, extra pancreatic involvement (sclerosing cholangitis, sclerosing dacryoadenitis/ sialoadenitis, retroperitoneal fibrosis. . .), serology (IgG4), pancreatic histology, and favorable response to steroid therapy. 9 The main radiological features of AIP and the diagnostic sensitivity of CT and MRI are: diffuse pancreatic enlargement (50%-70% on CT, 70% on MRI), focal pseudo mass (80% on MRI and less frequent on CT), diffuse or focal duct narrowing (66% on MRI), hypoattenuation in the arterial phase (58% on CT), late enhancement (low sensitivity on CT, 75% on MRI ), rim sign (rare on CT, 25% on MRI). 10 EUS in AIP allows the detection of both parenchymal and ductal features similar to those identified by CT and MRI. Additionally, it enables the visualization of parenchymal lobularity and a hyperechoic pancreatic duct margin, which are significantly associated with early-stage AIP. However, the greatest advantage of EUS remains the ability to perform tissue sampling for histological confirmation to differentiate with a pancreatic neoplastic mass. 11 IgG4 is the most reliable serum marker for diagnosing type 1 AIP, with a sensitivity of 86% and a specificity of 96% when levels are ⩾ 135 mg/dL. 12 IgG4 sensitibity is less than 10% in AIP-2. 7
In our case report, the radiological, biological, and extra pancreatic features of AIP were not observed as well as the resolution of pancreatitis without steroids rules out the possibility of AIP.
After ruling out common etiologies if the pancreatitis resolves after discontinuation of the drug, suspicion for DIAP increases. This connection proves difficult to establish, however, as the resolution of disease may be linked coincidentally with cessation of the inciting agent. A firm diagnosis can be reasonably established with a rechallenge of the offending drug that results in the recurrence of pancreatitis symptoms. Rechallenge should be conducted cautiously given the risk of severe reccurence. 2
TNFα inhibitors have exceptionally been described as a cause of acute pancreatitis. In our case report, we ruled out common etiologies, including alcohol consumption, tobacco use, biliary causes, autoimmune conditions, and metabolic factors. The resolution of acute pancreatitis upon cessation of Adalimumab treatment, the recurrence of pancreatitis following the reintroduction of Adalimumab, and the absence of further episodes after the definitive discontinuation of Adalimumab and the switch to Ustekinumab, provide strong evidence that Adalimumab was the cause of the acute pancreatitis. Based on these data, our case corresponds to classification 3a of the Evidence-based classification for drug-induced acute pancreatitis–revised of 2023. 13
The identification of TNFα inhibitors-induced acute pancreatitis in an IBD patient warrants consideration to swap to another therapeutic class. This is because switching from one TNFα inhibitors to another exposes the patient to the risk of recurrent acute pancreatitis. 14
Our case report is similar to that presented by Werlang et al 14 , which describes a case of acute pancreatitis induced by TNFα inhibitor (Infliximab) in a patient treated for UC. In their report, common etiologies of pancreatitis were excluded, with recurrent of pancreatitis upon switching to Adalimumab. Definitive resolution of the pancreatitis was achieved after discontinuation of this therapeutic class. 14
Several potential mechanisms have been proposed to underlie drug-induced pancreatitis, encompassing pancreatic duct constriction, arteriolar thrombosis, direct and indirect toxic effects, as well as hypersensitivity reactions. 2 Negative effects of drugs, such as hypertriglyceridemia and chronic hypercalcemia, are also mechanisms for drug-induced acute pancreatitis, as these effects are risk factors for acute pancreatitis. Other possible mechanisms of action are localized angioedema effect in the pancreas and arteriolar thrombosis. 15
Sahu et al 16 reported that Adalimumab is likely leading to severe hypertriglyceridemia inducing acute pancreatitis, but this mechanism has been ruled out in our case report.
Conclusion
Our clinical case report with strong evidence that Adalimumab can lead to acute pancreatitis and should be listed in drugs drug classes associated with acute pancreatitis. This evidence was reinforced in our case by the recurrence of pancreatitis with re-challenge of the drug.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
WE performed the bibliographic research and drafted the manuscript. MB: Conception of the study and Correction of the manuscript. All authors read and approved the final manuscript.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
